Abstract
Background
Tamsulosin is an α1-adrenoceptor antagonist used to treat lower urinary tract symptoms (LUTS) and benign prostatic hyperplasia (BPH). Palmijihwang-hwan (PMJHH) is a traditional herbal formula for treating prostatic disorders and is often used with tamsulosin for treating LUTS/BPH. The co-administration of herbal medicines and conventional drugs increases the potential for clinically considerable herb−drug interactions. Hence, it is important to evaluate the safety of such combinations of drugs. This study aimed to investigate the influence of PMJHH on the pharmacokinetics of tamsulosin in rats.
Materials and Methods
We developed and validated a simple and rapid method to determine the plasma levels of tamsulosin in rats using an ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) system. After oral administration of tamsulosin (2 mg/kg), either alone or in combination with PMJHH (930 mg/kg), to rats, blood samples were collected at various times and used for pharmacokinetic studies. The pharmacokinetic parameters were analyzed using a non-compartmental model.
Results
Compared to tamsulosin alone, co-administration of PMJHH had no significant effects on any pharmacokinetic parameters of tamsulosin, including maximal plasma concentration (Cmax), time to Cmax (Tmax), terminal elimination half-life (t1/2), AUC from time 0 to the last observation (AUC0−t), mean residence from time 0 to infinity (MRT0−∞), apparent oral clearance (Cl/F), and apparent volume of distribution during terminal elimination (Vz/F).
Conclusion
The pharmacokinetics of tamsulosin were not affected by the co-administration of PMJHH. Our findings, therefore, validate the safety of the combination of tamsulosin and PMJHH commonly used for treating LUTS/BPH.
Introduction
Benign prostatic hyperplasia (BPH), characterized by an increase in the size and muscle tone of the prostate, is common in men over the age of 50 years and causes lower urinary tract symptoms (LUTS). 1 The medicines, including alpha-blockers and 5-alpha reductase inhibitors, are usually preferred over surgical procedures to treat LUTS/BPH.1, 2 Tamsulosin is one of the most widely used therapeutic agents for LUTS suggestive of BPH. 3 A selective α1-adrenoceptor antagonist, tamsulosin, has an oral bioavailability close to 100% in the fasting state. It is commonly available in a modified-release (MR) formulation taken once a day. 2
Various approaches have been attempted for the effective treatment of BPH because of its complex and diverse etiologies. Herbal medicines with various active components are an alternative to conventional drugs for treating BPH. 4 According to World Health Organization, herbal medicines are used by almost 80% of the population as complementary and alternative medicines. 5 In the United States alone, approximately 40%–60% of adults with chronic diseases use dietary supplements, and about 20%–25% of patients use a dietary supplement in addition to prescription medications. 6
Palmijihwang-hwan (PMJHH; Hachimijio-gan in Japanese and Baweidìhuang-wan in Chinese), a herbal formula consisting of six herbal medicines (Table S1), has been used for the treatment of prostatic diseases such as prostatic hypertrophy and prostatic cancer. 7 Yoshimura et al. have demonstrated the effectiveness of PMJHH in relieving symptoms, including a significant improvement in the peak flow rate in patients with BPH. 8 We have previously reported that PMJHH inhibits prostaglandin E2-mediated and cyclooxygenase-2-mediated inflammation in RAW 264.7 macrophages. 9 Additionally, based on drug metabolism studies, we showed that PMJHH inhibits the activity of cytochrome P450 (CYP) 2D6 among seven major CYPs and three UDP-glucuronosyltransferases in human hepatic microsomes. 9
Herbal medicines and conventional drugs have been used simultaneously to treat various chronic diseases, 10 including BPH.1, 4 Several studies have compared the efficacy of conventional drugs administered alone vs. in combination with herbal in the treatment of BPH. Clinical studies have reported that the combination of Qian Lie Xin, a traditional Chinese medicine, and tamsulosin is more effective than tamsulosin alone for the treatment of BPH. 11 In addition, clinical investigations have shown that terazosin, when combined with Saxifraga stolonifera, a traditional Chinese medicine is superior in improving BPH symptoms compared to its use alone. 4 However, when herbal medicines and conventional drugs are combined, the pharmacokinetic interactions affect the drug’s blood concentration and pharmacological action. 6 The safety of the co-administration of herbal medicines and conventional drugs for treating BPH is unclear because there is insufficient information on the safety of α-blockers in men using drugs for erectile dysfunction and there are no absolute contraindications for their use in combination. 12 Absorption of the tamsulosin MR formulation commonly used for the treatment of LUTS/BPH is affected by concomitant food intake, 2 suggesting that its absorption could also be affected when used in combination with herbal medicines.
The influence of PMJHH on the pharmacokinetics of tamsulosin when used in combination for the treatment of LUTS/BPH has not yet been reported. In this study, we investigated the pharmacokinetics of tamsulosin and its potential interactions with PMJHH when used in combination. We developed a simple and rapid method to quantify rat plasma concentrations of tamsulosin using ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) and investigated the influence of PMJHH on tamsulosin pharmacokinetics in rats.
Materials and Methods
Chemicals and Reagents
Harnal®D, a MR formulation of tamsulosin hydrochloride, was obtained from Astellas Pharma Korea, Inc. (Seoul, Republic of Korea; Lot number: 19226R1). PMJHH extract granules were supplied by Hanpoong Pharm and Foods Co. Ltd. (Seoul, Republic of Korea; serial number: 18411). Tamsulosin, avanafil (internal standard, IS), and formic acid were supplied by Sigma-Aldrich (St. Louis, MO, USA). Reference standard compounds for high-performance liquid chromatography (HPLC) profiling analysis of PMJHH were purchased from commercial companies: 5-hydroxymethylfurfural (5-HMF; catalog number: W501808), cinnamic acid (catalog number: C80857), and paeonol (catalog number: H35803) from Merck KGaA (Darmstadt, Germany); loganin (catalog number: 125-03621) from Fujifilm Wako Pure Chemical Co. (Osaka, Japan); and morroniside (catalog number: BP0960) from Biopurify Phytochemicals (Chengdu, China) (Figure S1). Acetonitrile (ACN), methanol (MeOH), and water (LC-MS grade) were provided by Thermo Fisher Scientific (Waltham, MA, USA). ACS-grade reagents, acetic acid, and trifluoroacetic acid were products of Merck (Darmstadt, Germany).
HPLC Analysis of PMJHH
HPLC analysis of PMJHH was conducted according to a previously reported analytical protocol based on a Shimadzu Prominence LC-20A series HPLC system (Kyoto, Japan). 13 Briefly, all markers in each sample were separated on a Gemini C18 column (250 × 4.6 mm, 5 µm, Phenomenex, Torrance, CA, USA) maintained at 40°C. The mobile phase was flowed under gradient elution conditions in a distilled water–ACN system containing acetic acid (Table S2). Five marker compounds (5-HMF, morroniside, loganin, cinnamic acid, and paeonol) were separated within 35 min without interference from other peaks (Figure S2). The content of markers detected in PMJHH is presented in Table S3.
UPLC-MS/MS
UPLC-MS/MS analysis was performed according to a previously described method with some modifications. 14 UPLC was performed on a Thermo Scientific™ Dionex™ UltiMate™ 3000 system equipped with a binary pump, autosampler, solvent degasser, and automatic thermostatic column oven. The Hypersil GOLD™ C18 column (2.1 × 50 mm, 1.9 µm; Thermo Fisher Scientific) was connected with an ACQUITY UPLC BEH C18 VanGuard pre-column (130 Å, 1.7 µm, 2.1 × 5 mm; Waters Corp., Milford, MA, USA) was used for chromatographic separations and maintained at 40°C. The mobile phase consisted of 0.1% formic acid in water (solvent A) and ACN (solvent B) using gradient elution (Table S4). The flow rate was 0.4 mL/min, and 3 µL of each sample was injected into the column. The autosampler temperature was maintained at 5°C.
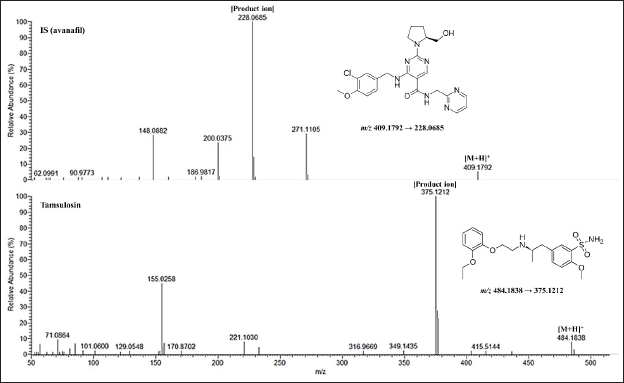
Mass spectrometry (MS)-based detection was performed using a Thermo Scientific™ Q-Exactive™ mass spectrometer (Thermo Fisher Scientific) equipped with a heated electrospray ionization source. Positive electrospray ionization in parallel reaction monitoring scan mode was used to collect data. The MS/MS conditions are summarized in Table S4. The m/z ratios for the precursor ion and product ion of tamsulosin were 409.1792 and 228.0685, respectively, while those for avanafil were 484.1838 and 375.1212, respectively. Data acquisition and quantification were performed using Xcalibur (Thermo Fisher Scientific) and TraceFinder software (Thermo Fisher Scientific).
Calibration of Standard and Quality Control Samples
Tamsulosin was prepared in MeOH and diluted to working concentrations of 0.313–10 ng/mL. The IS working solution (10 ng/mL) was diluted in ACN. Standard solutions for calibration were made by combining stock solution and rat blank plasma and spiking in a 1:1 ratio with ACN containing IS (final concentration of 5 ng/mL) to precipitate the plasma proteins. The mixture was vortexed for 10 min, followed by centrifugation at 15,000× g for 15 min at 4°C. The supernatant (100 µL) was filtered through a 0.2-µm syringe filter (Thermo Fisher Scientific) and injected into the LC-MS/MS system for analysis after being transferred to a fresh vial. The same procedure was followed to prepare quality control (QC) samples for method validation at concentrations of 0.313, 0.625, 2.5, and 10 ng/mL.
Method Validation
The method was validated in accordance with the Bioanalytical Method Validation Guidance for Industry, recommended by the Food and Drug Administration. 15
Selectivity
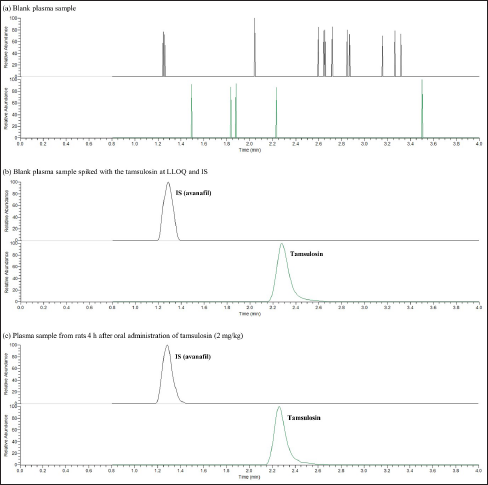
The selectivity of a method for the interference of endogenous substances and metabolites was evaluated by comparing the chromatograms of blank plasma, blank plasma spiked with tamsulosin and IS standards, and plasma samples collected after oral administration of tamsulosin.
Calibration and LLOQ
Six concentrations of the calibration standard (0.313, 0.625, 1.25, 2.5, 5, and 10 ng/mL) were used to create the calibration curve. The calibration curve (y = ax + b) was calculated using least-squares linear regression (y = ax + b), where x is the concentration of the standard solution, and y is the peak area ratio of tamsulosin and IS. The degree of linearity was assessed using the coefficient of determination (r 2 ). The lower limits of quantification (LLOQ), defined as the lowest concentration on the calibration curve, was determined based on a signal-to-noise ratio > 10. The accuracy (relative error, RE) and precision (relative standard deviation, RSD) of LLOQ were determined as follows: “RE = [(measured concentration – nominal concentration)/nominal concentration] × 100%; RSD = (standard deviation (SD)/mean of measured concentration) × 100%.” The accuracy and precision were less than 15% and were reliably detected and reproduced.
Accuracy and Precision
Four concentrations of QC samples (0.313, 0.625, 2.5, and 10 ng/mL) were used to evaluate the intra- and inter-day accuracy and precision. Measurements were taken on five consecutive days to determine the inter-day accuracy and precision. Accuracy and precision are expressed as RE and RSD, respectively.
Matrix Effect and Recovery
The matrix effect and recovery were evaluated using three sets of QC samples at concentrations of 0.313, 0.625, 2.5, and 10 ng/mL. The matrix effect was assessed by comparing the peak areas of analytes in blank plasma extracts spiked with standard solutions in the mobile phase. It was less than 15% at each concentration tested, which is within the acceptable range. The peak areas of the analytes in blank plasma spiked before and after extraction were compared to determine recovery.
Stability
The stability was evaluated by analyzing three sets of QC samples at 0.625, 2.5, and 10 ng/mL, exposing to different conditions.
Autosampler: Plasma samples were stored in an autosampler at 5°C for 24 h before analysis.
Freeze-thaw: Plasma samples were stored at −81°C for 24 h and thawed at 23 ± 3°C. The freeze-thaw cycle was repeated three times.
Short-term: Plasma samples were stored at 23 ± 3°C for 4 h before analysis.
Long-term: Plasma samples were stored at −81°C for 15 days before analysis.
Stability was defined as accuracy and precision of less than 15% at each concentration compared to the QC samples not exposed to these conditions.
Pharmacokinetic Study
Animals
A total of 12 male Sprague–Dawley rats (6-week-old, body weight 210–240 g) were obtained from Orient Bio Inc. (Seongnam, Republic of Korea) and acclimated for a week prior to the start of the experiment. The rats were kept in an environment with a temperature of 23±3°C, a relative humidity of 40%–60%, and a light/dark cycle of 12/12 h. Commercial rodent chow and water were freely available. This study was approved by the Institutional Animal Care and Use Committee of the Korea Institute of Oriental Medicine (approval number: 21−004). It was performed in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals. 16
Administration of Drugs and Sample Collection
Rats were fasted for 16 h with free access to water prior to the pharmacokinetic study. They were randomly divided into two groups of six rats each. Harnal®D (tamsulosin) and PMJHH extract granules were dissolved in distilled water. Group I was administered tamsulosin (2 mg/kg), while Group II received tamsulosin (2 mg/kg) combined with PMJHH (930 mg/kg), both orally. The dose of PMJHH extract granules (Hanpoong Pharm and Foods Co. Ltd.) is 9 g/day for a human (approximately 60 kg), which is converted to 930 mg/kg in rats based on body surface area. 17 At 1, 2, 3, 4, 5, 6, 7, 8, 10, 12, and 24 h after the drug administration, blood samples (250–300 µL) obtained via the jugular vein were collected in the K2-EDTA-coated blood collection tubes. Plasma was obtained from the blood samples by centrifugation at 6,000 × g for 10 min at 4°C and kept at −81°C until further analysis.
Plasma Sample Preparation
To precipitate the plasma proteins, a plasma sample (100 µL) was mixed with 100 µL of ACN containing IS (10 ng/mL). The samples were processed as described in Calibration of standard and quality control samples.
Data Analysis
The pharmacokinetic parameters, including area under the plasma concentration-time curve (AUC), maximal plasma concentration (Cmax), time to Cmax (Tmax), and (t1/2), were calculated by a non-compartmental model using the PKSolver program. 18 The data are expressed as mean ± SD. Statistical analysis was performed using the Student’s t-test (GraphPad Prism, Version 8.4.3, GraphPad Software, LLC, San Diego, CA, USA). Differences were considered statistically significant at p < 0.05.
Results
Optimization of UPLC-MS/MS Separation
To obtain suitable retention times, symmetrical peak shapes, and appropriate ionization of the analytes, we compared the effects of different mobile phase conditions, including ACN, MeOH, and water, in varying proportions. The use of ACN-water resulted in a shorter analysis time, improved peak shape, and reduced background. Furthermore, the addition of formic acid to the mobile phase enhanced the ionization efficiency and improved the response of the analytes. Finally, a gradient elution method with ACN and 0.1% formic acid in water was selected for good separation of the analytes and IS, symmetrical peaks, and high sensitivity. The mass spectra of tamsulosin and IS are shown in Figure 1. The retention times for IS and tamsulosin were 1.29 and 2.26 min, respectively.
Validation of the Method for Tamsulosin Measurement
The method for determining the concentration of tamsulosin in rat plasma was successfully validated. We could completely separate tamsulosin and IS under the established chromatographic conditions with no endogenous interference at their respective retention times (Figure 2). These results demonstrate the good selectivity of the analytical method. The tamsulosin calibration curve was linear over the concentration range of 0.313–10 ng/mL and had a regression equation of y = 0.2276x – 0.0056 with an r 2 of 0.9997. The LLOQ was 0.313 ng/mL, with a RE of –1.60 ± 2.80% and an RSD of 2.85%. The intra- and inter-day accuracy and precision of the QC samples are presented in Table 1. The RE values ranged from –5.40% to 0.68%, while the RSD values ranged from 0.21% to 5.13% (Table 1). The matrix effect of the analytes ranged from 88.24% to 106.86% in the QC samples at the concentrations tested. These results indicated that all the data were within the required limits, with no matrix effect for detecting tamsulosin and IS. The tamsulosin recovered from rat plasma ranged from 100.59% to 106.02% in QC samples at concentrations of 0.313 (LLOQ), 0.625, 2.5, and 10 ng/mL (Table 1). Therefore, this method satisfied the pharmacokinetic requirements of tamsulosin adequately.
Accuracy, Precision, Matrix Effect, and Recovery of Tamsulosin in Rat Plasma.
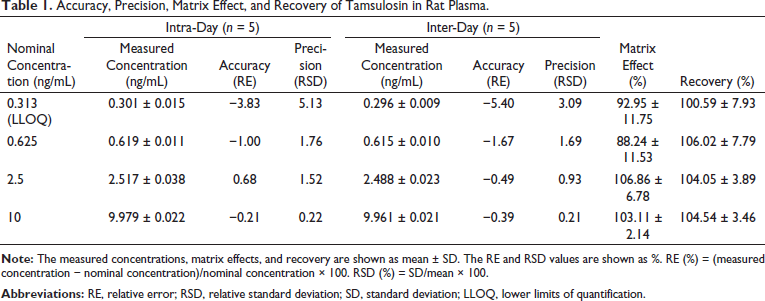
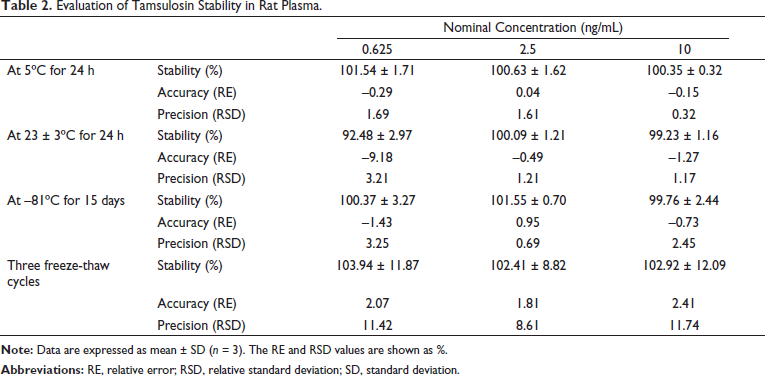
Table 2 shows the stability of tamsulosin in the biological samples under various storage conditions, including the autosampler (5°C) for 24 h, 23 ± 3°C for 24 h, –81°C for 15 days, and three freeze-thaw cycles. Its stability ranged from 92.48% to 103.94%.
Evaluation of Tamsulosin Stability in Rat Plasma.
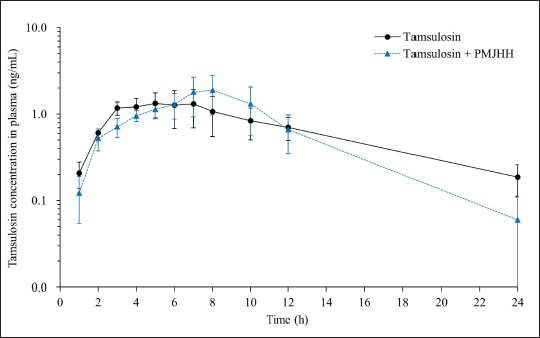
Pharmacokinetic Study
The newly developed UPLC-MS/MS method was used to determine the oral pharmacokinetic parameters of tamsulosin in rat plasma samples. Figure 3 shows changes in the plasma concentrations of tamsulosin over time after it was orally administered either alone (Group I) or in combination with PMJHH (Group II) in rats. Table 3 presents the pharmacokinetic parameters in these two groups of rats. The Cmax of tamsulosin alone (Group I) was 1.73 ± 1.26 ng/mL (Table 3), which increased to 2.61 ± 2.17 ng/mL in combination with PMJHH (Group II). However, the increase was not significant (p > 0.05). There were no significant differences in the Tmax, AUC for tamsulosin from time 0 to the last observation (AUC0-t), apparent oral clearance (Cl/F), or apparent volume of distribution during the terminal elimination (Vz/F) between the two groups. Tamsulosin in the presence of PMJHH showed a slight but non-significant decrease in the t1/2 and mean residence from time 0 to infinity (MRT0−∞), compared to tamsulosin alone.
Pharmacokinetic Parameters of Tamsulosin after Oral Administration alone or in Combination with PMJHH in Rats.

Discussion
Herbal medicines are widely used to improve health and prevent and treat various diseases worldwide. 10 The combination of herbs and drugs as an alternative to conventional therapy is increasing. Although herbal medicines may improve the efficacy or reduce the side effects of conventional drugs when taken together, they also have the potential to increase the risk of clinically relevant herb−drug interactions, resulting in increased drug toxicity or even death.10, 19, 20 Given that the use of herbal medicines is based on tradition and experience, there are no comprehensive federal regulations regarding their safety. Hence, regulatory authorities do not require rigorous preclinical and clinical evaluations of herbal medicines. 20 In addition, most patients do not inform their medical professionals that they are taking herbal medicines. 20 As a result, herb−drug interactions are not systematically monitored, and adverse reactions caused by these interactions may be underreported.
It is important to confirm the safety of herbal medicines when combined with synthetic drugs with similar pharmacological activities and metabolic pathways. 21 The safety of the combination can be assessed by determining the influence of herbal medicines in the presence of synthetic drugs. 21 In this study, we evaluated the effect of PMJHH on the pharmacokinetics of tamsulosin in rats to confirm the safety of this combination, which is commonly used for LUTS/BPH.
Tamsulosin is primarily metabolized by hepatic CYP3A4 and CYP2D6, resulting in the generation of five metabolites, which include an acidic metabolite, AM-1, and four basic metabolites, M-1 to M-4, which are excreted in the urine.2, 22 These five primary metabolites are subsequently partially converted to glucuronide- or sulfate-conjugated forms. 3 Herbal medicines containing multiple constituents can affect the pharmacokinetics of synthetic drugs by sharing drug-metabolizing enzymes and transporters. 23 Therefore, PMJHH containing various components such as 5-HMF, morroniside, loganin, paeonol, cinnamic acid, coumarin, cinnamaldehyde, and mesaconitine may affect the metabolism and pharmacokinetic parameters of tamsulosin when taken concomitantly. In particular, morroniside can induce the hepatic expression and activity of CYP3A in rats. 24 In our previous study, PMJHH was reported to inhibit the activity of human hepatic CYP2D6 with an IC50 value of 280.89 µg/mL, 9 suggesting that PMJHH may increase the plasma concentration of tamsulosin by inhibiting its metabolism. However, only previous reports are not sufficient to conclude whether PMJHH affect the pharmacokinetics of tamsulosin. Therefore, in this study, we investigated whether PMJHH affects the plasma concentrations of tamsulosin in vivo.
The most widely used approach for quantitating components in biological samples is LC-MS/MS. In this study, we optimized the rapid and accurate separation of the tamsulosin peak with no significant interference from endogenous substances. No significant differences were seen in any pharmacokinetic parameters (Cmax, Tmax, t1/2, AUC0-t, MRT0−∞, Cl/F, and Vz/F) of tamsulosin when administered alone vs. in combination with PMJHH. After oral administration with or without PMJHH in rats, tamsulosin levels were detected in the initial plasma samples collected (at 1 h). The Tmax and MRT0−∞ in both groups were 5.17–5.67 h and 17.23–20.08 h, respectively. These findings suggest that tamsulosin was rapidly absorbed in the gastrointestinal tract and remained in the plasma for approximately 20 h, regardless of PMJHH intake.
The increase in plasma concentration and AUC of drugs is likely to cause adverse effects. 20 No significant difference was seen in the Cmax and AUC of tamsulosin between the two groups, indicating that PMJHH neither altered tamsulosin’s absorption and clearance pathways nor affected the systemic exposure to tamsulosin. Therefore, an herb−drug interaction between these agents is unlikely, and dosage adjustment of tamsulosin may not be needed when combined with PMJHH.
Our findings have some implications for safe herb−drug combinations in clinical practice, but they have several limitations, as follows: First, this study evaluated the effect of PMJHH on the pharmacokinetics of tamsulosin only under the condition of single administration. It is also critical to derive a recommended dose that achieves the desired exposure after repeated administration. Second, no research on the pharmacokinetic interaction of tamsulosin with compounds present in PMJHH has been conducted. Studies on the pharmacokinetic interactions of compounds found in herbs and drugs may also be significant.
Conclusion
The UPLC-MS/MS-based method we have described in this study determines plasma levels of tamsulosin in rats accurately and precisely. This method was applied for the pharmacokinetic studies of tamsulosin in rats following its oral administration with and without PMJHH. Our findings suggest that PMJHH does not affect the pharmacokinetics of tamsulosin, thereby confirming the safety of this combination for the treatment of LUTS/BPH. Based on these findings, it is possible to plan a randomized clinical trial for patients in the future, and furthermore, it is expected that guidelines for the combination therapy of tamsulosin and PMJHH can be prepared.
Summary
We assessed the effects of PMJHH on the pharmacokinetics of tamsulosin in rats. We describe a UPLC-MS/MS method to determine plasma levels of tamsulosin in rats. The co-administration of PMJHH did not affect the pharmacokinetics of tamsulosin in rats. PMJHH in combination with tamsulosin is safe for treating LUTS/BPH.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Financial Support and Sponsorship
This research was supported by the grant “Construction of safety and efficacy for traditional herbal prescriptions of medicinal institutions (KSN2021310 and KSN2022310)” from the Korea Institute of Oriental Medicine (KIOM).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
