Abstract
Background
In Mexico, there is an ancestral custom about the knowledge of the medicinal use of plants. Bixa orellana L. is a plant species that represents one of the main crops used in traditional medicine in southeastern Mexico. Background studies on its biological properties have been conducted on accessions harvested in Conkal, Yucatan, Mexico.
Objectives
In this work, the idea was to perform a phytochemical analysis of methanolic extracts of B. orellana.
Materials and Methods
Phytochemical screening was performed on methanolic extracts of leaves and seeds from two accessions of B. orellana, as well as on their antioxidant and antiproliferative activities.
Results
The phytochemical screening of the methanolic extracts of the leaves revealed the presence of tannins, terpenes, and saponins. A higher total content of phenols and flavonoids in leaves was also found compared to seeds, which coincides with a higher antioxidant activity in leaves compared to seeds. Regarding antiproliferative activity, the seeds and leaves of accession 3 showed the best results.
Conclusion
Accession 3 has better bioactive properties and can be a candidate in the search for antiproliferative and antioxidant compounds; this is the first study that associates this plant with this activity and demonstrates the chemical differences that exist between the accessions of B. orellana.
Introduction
In Mexico, there is an ancestral custom about the knowledge of the medicinal use of plants. The information has been transmitted from generation to generation. They have been and are used to relieve pain and treat various diseases, which is why they represent the basis of current medicine. Therefore, there is scientific interest in isolating the different active compounds (from the bark, leaves, seeds, roots, fruits, and resins) that have demonstrated clinical efficacy to develop drugs for the treatment of various diseases.1–3
In the case of Bixa orellana L., commonly known as achiote, it belongs to the Bixaceae family and is native to southern tropical America. It is a multipurpose plant due to its great ability to adapt to various soils, even poor or acidic ones, so there are a large number of morphotypes. 4 Achiote is used in the traditional medicine of several countries in America, such as Brazil, Cuba, Peru, and Mexico. Its use lies in infusions of the seed, flowers, bark, root, and leaves; cooking the root is used as a diuretic, antidysenteric, and antidiabetic; cooking the bark serves to soothe muscle pain; and cooking the leaves is used as an antibiotic, laxative, healing, and against dandruff and hair loss. The seeds are used to treat bronchitis and hemorrhoids, so the therapeutic use of this species is varied depending on the portion and tissue of the plant to be used. 5 Information has been reported about the bioactive compounds present in B. orellana from plantations of other accessions that have shown that the seed, leaves, and other parts of the plant have metabolites of biological interest that can be useful to demonstrate interesting antioxidant and pharmacological activities. Among these biological compounds are, in addition to carotenoids, apocarotenoids (bixin), terpenes, terpenoids, sterols, aliphatic compounds, and phenolic compounds, among others. 6 These compounds have been related to antiproliferative effect, but this effect is not clear for annatto.
In addition, investigations have been carried out in different parts of achiote,7–10 where the activity of its bioactive components with antihyperglycemic, antimalarial, antiulcer activity, antimicrobial activity of pathogenic bacteria, antiproliferative in vitro through mechanisms involving apoptosis and cell disruption is reported, 11 information that gives this plant a scientific support for its use in traditional medicine,7–10 and taking as reference previous reports of studies of the same accessions.12, 13
B. orellana is a traditional resource used in popular medicine in Yucatan, Mexico, so it is convenient to investigate its chemical constituents and its biological activity for the treatment of diseases. Therefore, the objective of the study was to carry out a phytochemical screening and determine the antioxidant capacity, total phenolic compounds (TPC), total flavonoid content (TFLC), and antiproliferative activity of leaves and seeds of two accessions of B. orellana, representing the same collections located in the municipality of Conkal, Yucatan, Mexico, that have shown anti-inflammatory effects in previous investigations. 13
Materials and Methods
Biological Material
In this study, seeds and leaves from B. orellana accessions 9 and 11 were used, previously reported by Zarza-García et al.,12, 13 and renamed as accessions 2 and 3, respectively. These samples have the germplasm authenticity voucher geolocated at the Instituto Tecnológico de Conkal (Technological Institute of Conkal), Yucatan, of the Achiote Network of SINAREFI, México (Table 1).
Germplasm Bank, Instituto Tecnológico de Conkal, México.

Preparation of Extracts From Seeds and Leaves
A quantity of 10 g of dry powdered seeds or leaves of B. orellana (accessions 2 and 3) were weighed, 250 mL of methanol (Sigma-Aldrich, USA) was added, and extraction for 3 h was carried out in Soxhlet equipment (Pyrex®, Sigma-Aldrich, USA). Subsequently, the extracts were concentrated to a final volume of 30 mL using a vacuum rotary evaporator (Buchi, Switzerland, model R-215) at reduced pressure, 130 rpm, and constant temperature (40°C), and the extracts were stored in amber bottles at –70°C for later analyses.
Qualitative Analyses of the Extracts
The phytochemical analyses for the identification of chemical groups present in the extracts of B. orellana were carried out using the procedures described by Cuéllar et al., 14 which have been used by other authors in other species, such as Cnidoscolus chayamansa (Chaya), 15 Moringa oleifera Lam., 16 and Tagetes lucida Cav., popularly known as Pericón. 17 These techniques are based on sensitive, reproducible, and low-cost reactions.
Terpenes
Terpene determination was carried out using Lieberman–Buchard’s test. An aliquot of the extract (0.05 g) was taken, and 1 mL of chloroform (Sigma-Aldrich, USA) and 1 mL of acetic anhydride (Sigma-Aldrich, USA) were added and mixed. Afterwards, through the wall of the tube, two to three drops of concentrated sulfuric acid were added without stirring. The reaction was considered positive if the color ranged from pink to blue.
Flavonoids
The presence of flavonoids was determined by Shinoda’s test. Briefly, 2 mL of absolute ethanol (Sigma-Aldrich, USA) was added to the extract (0.05 g), then two drops of concentrated hydrochloric acid (Sigma-Aldrich, USA), and finally, a trace of magnesium was added; a red coloration indicates the presence of flavonoids.
Saponins
To determine the presence of saponins, Rosenthaler’s test was carried out, for which two drops of Rosenthaler’s reagent and two drops of concentrated sulfuric acid (Sigma-Aldrich, USA) were added to an aliquot of the methanolic extract (0.05 g). The formation of a violet coloration is considered positive for triterpene saponins.
Tannins
This determination was carried out with the reaction of ferric chloride; 1 mL of distilled water was added to an aliquot of the extract (0.05 g), then three drops of 2% (w/v) sodium chloride were added, and the mixture was heated to boiling for 1 min, allowed to cool, and filtered. A drop of 1% (w/v) ferric chloride was added to the filtrate; the appearance of a green color is interpreted as a positive reaction and indicates the presence of catechol derivatives, and a dark color indicates the presence of gallic acid.
Quinones
For the determination of quinones, a drop of concentrated sulfuric acid (Sigma-Aldrich, USA) was added to an aliquot of the extract (0.1 mL). The appearance of a red color indicates the presence of anthraquinones.
Coumarins
The determination of coumarins was carried out using 0.5 M potassium hydroxide (Sigma-Aldrich, USA), for which an aliquot of the extract (0.05 g) was dissolved in boiling water, a capillary tube was filled, and a drop was then placed on filter paper. Afterwards, a drop of 0.5 M potassium hydroxide was placed on the previously placed drop of the extract and observed in a UV lamp at 365 nm; the presence of fluorescence indicates a positive result for this metabolite.
Alkaloids
A volume of 10 mL of hydrochloric acid 10% (v/v) (Sigma-Aldrich, USA) was added to an aliquot of the extract (0.1 mL), it was heated until boiling for 5 min, cooled, and filtered using a funnel with Whatman 4 filter paper. Subsequently, the filtrate was divided into two test tubes for the following reactions. Wagner’s test: Two drops of Wagner’s reagent were added, and the formation of a brown precipitate indicated a positive response. Mayer’s test: Two to three drops of Mayer’s reagent were added, and a white precipitate indicated a positive response.
Cardiotonic Glycosides
For the determination of cardiotonic glycosides, the Keller-Kilian’s test was performed. For this test, an aliquot of the extract (0.1 mL) was placed into a test tube, it was dissolved in 1 mL of glacial acetic acid (Sigma-Aldrich, USA), and two drops of iron chloride were added. Afterwards, 2 mL of concentrated sulfuric acid (Sigma-Aldrich, USA) were added through the wall of the tube without stirring. The presence of a red ring in the meniscus of the liquid indicated a positive reaction, indicative of the presence of these metabolites.
Quantitative Analyses of the Extracts
DPPH Antioxidant Determination
The antioxidant activity was determined according to the study by Usia, 18 with slight modifications. The dry extracts from B. orellana were dissolved in absolute ethanol (5 mg mL–1) (Sigma-Aldrich, USA).
The extracts (100 µL) at different concentrations (0–400 µg mL–1) were mixed with 100 µL of 2,2-diphenyl-1-picrylhydrazyl (DPPH) solution (300 µM). The reaction mixture was incubated in the dark for 30 min at room temperature. Absorbance was measured at 515 nm using a microplate spectrophotometer (Thermo Scientific Multiskan GO). Vitamin C was used as a reference (70 µM, 12.6 µg mL–1). The results were expressed as IC50 and as a percentage of antioxidant activity based on the rate of degradation of DPPH compared to the control (1:1 mixture, ethanol:DPPH) according to the following formula:
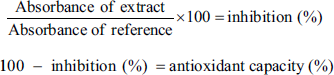
The IC50 was determined from the graph of the percentage of inhibition using a linear regression method (Microsoft Excel) and corresponds to the concentration at which 50% of the free radicals are neutralized.
Total Flavonoids
The TFLC was reported in terms of flavones and flavonols, which were measured by the colorimetric method based on the formation of a complex between the aluminum ion and the carbonyl and hydroxyl groups of the flavonoids. 19 To 10 µL of methanolic extract (5 mg mL–1), 130 µL of methanol (Sigma-Aldrich, USA) was added, followed by 10 µL of AlCl3 5% (w/v) (Sigma-Aldrich, USA) were added. The reaction mixture was incubated in the dark for 30 min, and the absorbance was measured at 412 nm in a microplate reader (Thermo Scientific Multiskan GO). The results were expressed as mg equivalent of the flavonol quercetin g–1 of dry extract (mg EQ g–1).
Total Phenolic Compounds
The total phenolic content of the extracts was measured by the method described by Ainsworth and Gillespie, 20 with slight modifications. 21 A volume of 10 µL (2.5 mg mL–1) of the extract were deposited in a 96-well microplate. Subsequently, 40 µL of Folin-Ciocalteu 0.2N reagent, 60 µL of sodium carbonate (Na2CO3), 7% (p/v) (Merck), and 90 µL of water were added to obtain a final volume of 200 µL per well. The plate was incubated in the dark at room temperature (25°C) for 1 h. Absorbance was measured at 750 nm in a microplate reader (Thermo Scientific Multiskan GO). The results were calculated using a gallic acid standard curve and expressed as mg gallic acid equivalents g–1 dry extract (mg EAG g–1).
Evaluation of the Antiproliferative Activity in Cancer Cell Lines
For the evaluation of the inhibition of cell proliferation, the cancer cell lines RAW 264.7 (murine macrophage transformed by the Abelson leukemia virus) and HeLa (adenocarcinoma of the cervix) were used, obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cell cultures were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 5% fetal bovine serum at 37°C in an atmosphere of 5% CO2 and 80%–90% relative humidity in an incubator Isoterm (Fisher Scientific, USA). To evaluate the antiproliferative effect of B. orellana extracts on cell lines, the standard method of tetrazolium salts (MTT) (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, MTT) 22 was used, with slight modifications. 23 The assays were performed using cell cultures in the logarithmic growth phase. Cell suspensions of 200,000 cells mL–1 were obtained and incubated in 96-well flat-bottom plates (10,000 cells per well, 50 µL) in the presence of different concentrations of the extracts (from 200 to 0 µg mL–1). The cell cultures were incubated for 24 h at a temperature of 37°C in an atmosphere of 5% CO2 and a relative humidity of 80%–90%. Subsequently, the extract was added, and the plates were left for 48 h. During the last 4 h of incubation, 10 µL of MTT (5 mg mL–1) were added to assess cell proliferation. The resulting formazan crystals were solubilized with acidic isopropanol and the absorbance of the colored solution was obtained at 570–655 nm in an ELISA reader. The results were expressed as IC50 values, which were calculated using a simple linear regression model (Microsoft Excel).
Statistical Analysis
All experiments were carried out in triplicate; the values represent the mean of three independent experiments and the standard deviation. To demonstrate the statistical difference, a two-tailed hypothesis test with α = 0.05 between the mean was carried out using the t-student test. Statdisk 13 for Windows 10 was used for the analyses.
Results
This study was carried out with the purpose of identifying the chemical groups in the extracts and to determine the biological properties of B. orellana L., which provide scientific support for its use in traditional Mayan medicine as well as for the propagation of these accessions.
Qualitative Analyses of the Extracts
Phytochemical Screening
The qualitative profile of secondary metabolites is the starting point for the screening of the structural diversity of compounds with beneficial properties for health that can be isolated for further studies. 24 In this study, an ideal scale was created to indicate the presence or absence of the phytochemicals.
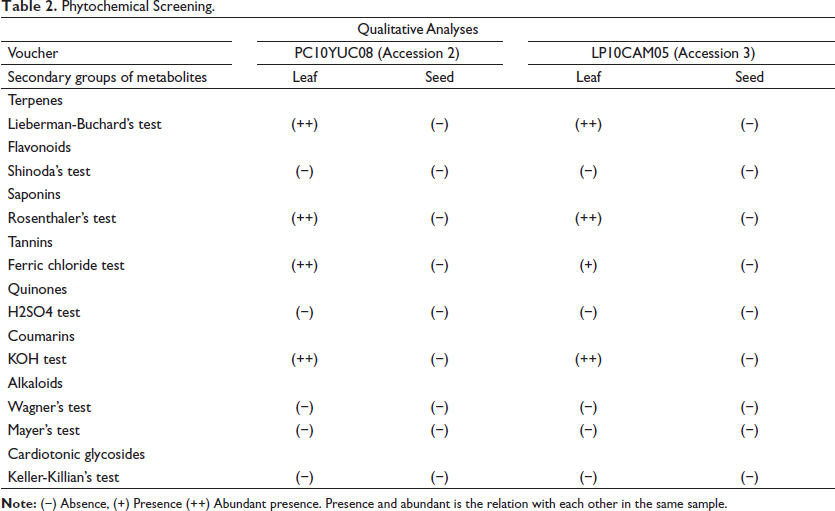
The results of the phytochemical screening of the methanolic extracts of the leaves (Table 2) show the presence of phenolic compounds such as tannins and coumarins, as well as terpenes and saponins, chemical groups associated with therapeutic principles,25, 26 and metabolites that, by the virtue of their structure, are chemically reactive. In the methanolic extracts of the seeds, none of the groups of metabolites evaluated were observed.
Phytochemical Screening.
Quantitative Analyses of the Extracts
DPPH Antioxidant Determination
The concentrations evaluated were between 400 and 3,125 µg mL–1, and Vitamin C (70 µM, 12.6 µg mL–1) was used as a positive control. The antioxidant activity of the methanolic extracts of leaves and seeds from accessions 2 and 3 is shown in Table 3.
Antioxidant Activity of the Accessions.
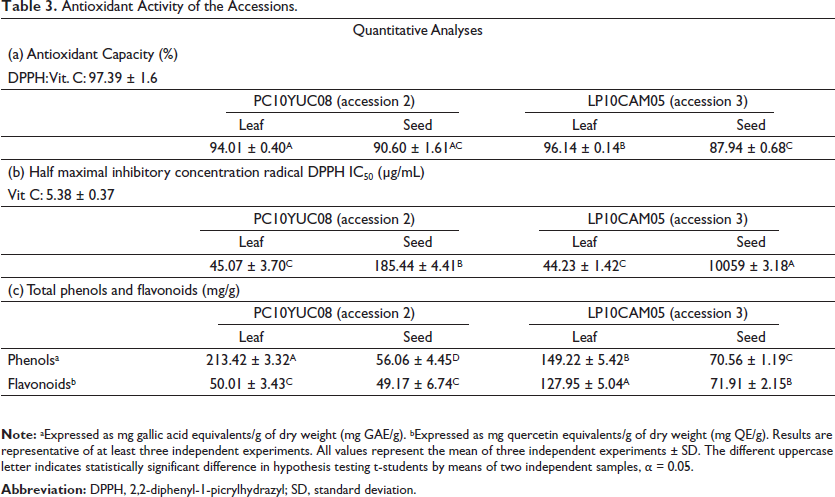
It is noteworthy that at the highest concentration tested (400 µg mL–1), the percentage of antioxidant activity of the methanolic extract of leaves 2 (L2), methanolic extract of leaves 3 (L3), and methanolic extract of seeds 2 (S2) was 94.01 ± 0.40%; 96.14 ± 0.14%, and 90.60 ± 1.61%, respectively, while for the methanolic extract of seed 3 (S3), the percentage of antioxidant activity was lower at this same concentration (87.94 ± 0.68). The positive control, Vitamin C, showed an antioxidant activity of 97.39 ± 1.6%.
Table 3 shows the half-maximal inhibitory concentration of the radical in the methanolic extracts (IC50). The concentrations tested for the methanolic extracts and for Vitamin C were 3.125–400 and 0–12.6 µg mL–1, respectively. The results show that the lowest IC50 values were obtained for the methanolic extracts of the leaves (L2 and L3), which indicates their higher antioxidant potential compared to the extracts of the seeds (S2 and S3).
Quantification of Total Phenols and Flavonoids
These results in Table 3 show that the TPC were observed in the following order: L2 > L3 > S3 ˃ S2, with L2 the extract that showed the highest value (˃200 mg GAE gdw–1). For the specific case of TFLC, the following order was observed: L3 > S3 > L2 = S2.
Evaluation of Antiproliferative Activity in Cancer Cell Lines
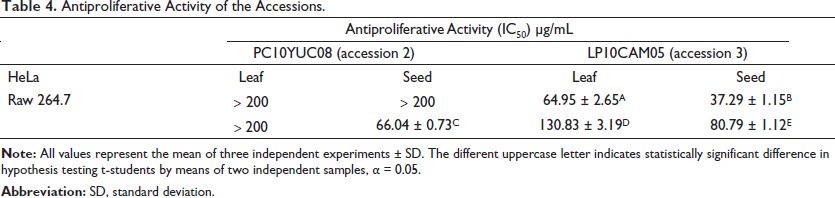
In vitro assays were performed using HeLa and RAW 267.4 cancer cell lines. The assayed concentrations were 200, 100, 50, 25, and 12.5 µg mL–1. In Table 4, the IC50 values are shown for each of the extracts evaluated in the aforementioned cell lines. The extracts corresponding to accession 3 (leaves and seeds) showed a higher antiproliferative activity for HeLa than the effect that was observed in accession 2 (leaves and seeds); likewise, the effect on cell proliferation of RAW 267.4 cells from the seeds of both accessions showed higher activity than the extracts corresponding to the leaves (L2 and L3), with L2 the extract that presented less effectiveness in the cell lines. The order of antiproliferative activity of B. orellana extracts in the HeLa cell line was S3 ˃ L3. No effects were observed in S2 and L2, with IC50 values of 37.29 ± 1.15, 64.95 ± 2.65, ˃200, ˃200 µg mL–1, respectively. The order of activity in RAW 267.4 cells was S2 ˃ S3 ˃ L3, with IC50 values of 66.04 ± 0.73, 80.79 ± 1.12 µg mL–1 and 130.83 ± 3.19 µg mL–1, respectively, while the value for L2 was ˃200 µg mL–1.
Antiproliferative Activity of the Accessions.
Discussion
Our results of phytochemical screening agree with those of a previous study reported by Zarza-García et al., 12 where the presence of saponins was observed in extracts of leaves from the same accessions; however, in the present study, no anthraquinones were observed, in contrast to the study mentioned above, where the presence of this metabolite was reported. On the contrary, in the present study, coumarins were observed, which was not the case in a previous study. 12 This could be due to the different extraction methods in these studies. 27 In other investigations of the phytochemical screening of other accessions of achiote, saponins and tannins were found; 28 in addition,7, 29–32 reported the presence of tannins, coumarins, and alkaloids in methanolic extracts of leaves from other accessions of B. orellana. These differences may be related to the biotic and abiotic conditions of the samples studied.
The analyzed metabolites were not found in the extracts of the seeds in the phytochemical screening; this differs from a qualitative study with other accessions of B. orellana, published by Prathima et al., 33 where the presence of alkaloids, flavonoids, saponins, and terpenoids is reported in methanolic extracts of seeds. However, in this same study, no cardiac glycosides or steroids were found.
The differences observed in the results of the phytochemical screening of our investigation on the chemical composition of the seeds and leaves analyzed, compared to other studies,10, 34 show the relationship that exists between the type of pretreatment and method of extraction to which the samples are subjected (maceration, infusion, etc.) and the content of secondary metabolites; however, these bioactive compounds can be vulnerable not only to temperature but also to other factors such as light, pH, oxygen, soil fertility, and climate, among others. Another important aspect to consider during the physico-chemical screening of medicinal plants is the kind of solvent used in the extraction, the time of collection of the material, the transport, its conservation method, the determination methods, and other factors that can affect the results of chemical determinations. 17
The results of the percentage of antioxidant activity show that the DPPH antiradical activity of the extracts was presented in the following order: L3 > L2 = S2 =S3, being the methanolic extract of the leaf of accession 3, the one that presented the highest DPPH antiradical activity (>95% inhibition). The antioxidant activity of the leaves of these same accessions (2 and 3) was reported by Zarza-García, 12 but the values were slightly lower than those of this study, probably due to differences in extraction method, 35 storage time and conditions, and other factors such as pH and soluble solids. 34 Regarding the seeds, the results previously published for these same accessions S2 and S3 13 showed a high concentration of bixin in both 36.91 ± 2.52 and 40.83 ± 1.27, respectively, which indicates that the antioxidant activity is related to this metabolite. 36 Regarding the antioxidant results, expressed as IC50, 37 reported that methanolic extracts of leaves from B. orellana (an ecotype from Bangladesh) showed an IC50 of 22.36 µg mL–1, values lower than those found in this study.
The antioxidant activity observed in the extracts of the seeds and leaves of B. orellana may be due to the presence of some phytochemicals, mainly flavonoids. 38
The interest in studying the content of phenolic compounds and flavonoids present in medicinal plants is related to the various biological activities of these secondary metabolites, which are considered bioactive agents. In recent years, scientific evidence has been reported that supports their properties and benefits, with a wide range of activities, such as antioxidant, anticancer, anti-inflammatory, and antimicrobial activity, among others, for its use and application in various medical and pharmacological treatments with an impact on health.27, 39–42
The high antioxidant activity found in achiote leaves (L2 and L3) may be related to various phytochemicals, mainly phenolics, according to the data shown in Table 2. It can be observed that the values of TPC TFLC of the extracts of B. orellana are correlated to their antioxidant activity, that is, the higher the content of total phenols, the lower the value of IC50. In another study with methanolic extracts from the leaves of these accessions, similar values of TPC were reported (158.77 ± 3.75 (L2) and 163.00 ± 8.41 mg GAE g–1 (L3)). 12 In the case of methanolic extracts from the seeds of these accessions S2 and S3, in a study carried out by Zarza-García et al., 13 differences in the TPC content were reported, with a higher content of these in S3 (9.65 ± 0.18), the same accession that shows higher antioxidant activity (2.45 ± 0.00 mM g–1 CAET) with respect to accession S2. The TPC content was 4.99 ± 0.14 GAE g–1, and the antioxidant activity was 2.43 ± 0.01 mM g–1 CAET. These results show that the antioxidant activity of the seeds is related to the bixin content.
It is important to note that Shinoda’s test resulted in a negative in all extracts. However, TFLC did reveal the presence of flavonoids. This could be due to the sensitivity of the Shinoda test or because of some interference that made it impossible to reveal flavonoids in this qualitative test.
Considering the criteria of the National Cancer Institute of the United States of America, an extract is considered to have high activity in tumor cells if the IC50 ranges from 31 to 60 µg mL-1 and low activity if it ranges from 61 to 99 µg mL–1. 43 The results from our study suggest that the methanolic extract of the seed of accession 3 (S3) possesses potential as a possible source of bioactive compounds with antiproliferative activity, followed by the extract of the leaves of accession 3 (L3) and by S2, with moderate and low potential, respectively.
In a study carried out with the seeds of other accessions of B. orellana, the antiproliferative activity in tumor cells and chemical profile was determined. 11 They demonstrated an important cytostatic effect, attributed to geranylgeraniol. Therefore, it is very important to determine the chemical profiles (identification and quantification of phenolic compounds and flavonoids) of the same accessions that are reported in this article to obtain more information about the phytochemicals that could be related to the antiproliferative effects in the carcinogenic cell lines evaluated. Natural products represent a significant resource in disease prevention, in addition to being important substrates in a healthy diet due to their content and the great variety in the wide range of bioactive compounds that exert a synergistic action for health; however, It is very important to expand studies for the identification and quantification of compounds such as phenolics, specifically, phenolic acids and flavonoids, which are recognized in the literature as having high antioxidant activity,38, 44 in addition to the identification of these compounds in the various plant tissues, since they can differ in composition and concentration in each tissue or organ of the plant. 45
Conclusion
The results of this study of two accessions of leaves and seeds of B. orellana showed functional activity related to antioxidant activity, due to the presence of a significant concentration of phenolic compounds. The phytochemical screening of the methanolic extracts of seed revealed the presence of phenolic compounds, such as terpenes and saponins, chemical groups associated with various principles that have been described by other authors as potential therapeutic bioactive for various diseases. The results of the antiproliferative activity of the two cancer cell lines showed an interesting cytostatic effect with low IC50 values; that is, the extracts of the seeds and leaves of accession 3 showed greater antiproliferative activity on the HeLa cell line. The seeds of both accessions (S2 and S3) showed higher activity on cell proliferation of RAW cell lines compared to what was observed in leaves (L2 and L3). The scientific contribution of this study is significant for B. orellana, cultivated in the Mexican southeast, providing sustenance for its use in traditional Mayan medicine, justifying the propagation of these cultivars in the Mayan zone, and contributing data to the integral use of cultivation.
Footnotes
Summary
Achiote is a medicinal plant used in several countries and for several diseases. Seeds of achiote have shown great antioxidant capacity in vitro.
The leaves of achiote also exhibit antioxidant capacity as well as antiproliferative effects.
Abbreviations
Acknowledgment
For the support received from the following institutions: SINAREFI-RED ACHIOTE, REMEFI, Instituto Tecnológico de Conkal, Yucatán, Mexico, and Universidad Autónoma del Carmen, Campeche, Mexico. To the University, Campus Cajeme, Department of Health Sciences, and the Department of Chemical-Biological Sciences of the Universidad de Sonora Centro, in addition to being part of the Sabbatical of PhD José A. Mendoza of the Universidad Autónoma de la Ciudad de México.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors are grateful to the Departamento de ciencias Químico-Biológicas y Agropecuarias and the División de Ciencias e Ingeniería of the Universidad de Sonora, Unidad Regional Norte (UNISON URN), for funding.
Statement of Informed Consent and Ethical Approval
Not applicable.
