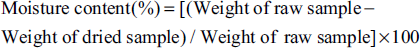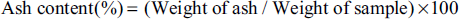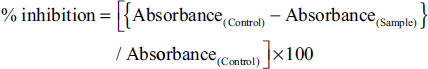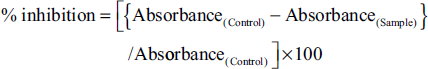Abstract
Background
Garcinia pedunculata (GP) Roxb. is a traditional medicinal plant used for treating gastrointestinal disorders. The fruit is always consumed in dried form in cuisines and as medicine.
Objectives
To study the effect of drying conditions (shade, oven, and traditional sun drying) on the nutritional profile of GP while assessing the choice of solvent extraction for the maximum phytocompounds extraction; their in vitro biological activities and chemical profiling.
Materials and Methods
The fruits were cut into thin 0.9 ± 0.3 cm slices and dried in the shade (25–30°C), sun (34–40°C), and oven (70–80°C). The extraction was done using methanol, hydromethanol, ethanol, hydroethanol, and water. HPLC and LC-ESI-MS/MS were used to identify the bioactive compounds.
Results
In this study, the impact of three drying conditions has been investigated and established that shade-dried (ShD) has a better-preserved nutritional profile. In terms of phytochemical extraction, comparatively higher amounts of phenols (394.00 ± 12.28 mg Gallic acid equivalent (GAE)/g of dried sample) and flavonoids (72.62 ± 2.06 mg Quercetin/g of dried sample) were recorded in the shade-dried hydromethanolic extract (ShDMH), which was directly in line with the better antioxidant activity (IC50 for 1,1-diphenyl-2-picrylhydrazyl (DPPH), 647.99 ± 7.66 µg/mL, 2,2’-azino-bis(3-ethylbenzothiazoline)6-sulfonic acid (ABTS), 762.02 ± 18.74 µg/mL) and enzyme-inhibitory potential (IC50 for α- glucosidase 1908.06 ± 13.26 µg/mL and for lipase 613.59 ± 7.57 µg/mL) of this extract.
Conclusion
The conclusive evidence from this study indicates that shade drying and extraction with hydromethanolic solvent preserves most of the nutritional components and has a higher extraction yield with well-conserved phytochemical composition. Also, these bioactive compounds such as hydroxycitric acid, GB-1a, garcinone A, 9-hydroxycalabaxanthone, chlorogenic acid, and garcinol are present in these extracts. This fruit, although beneficial is still underutilized, and this outcome is believed to be significantly valuable for the further aspects of GP fruit-based nutraceutical or phytopharmaceutical development.
Introduction
Modern medicine has a specific stratum for medicinal plants that are utilized as medicines after rigorous scientific experimentation. Traditional medicines have integrated their use in practice from the prehistoric era. Plants are rich sources of diverse bioactive molecules, including polyphenols, terpenes, alkaloids, and glycosides. According to the reports of the United Nations Food and Agriculture Organization in 2002, the number of medicinal plants used worldwide was more than 50,000. 1 The preservation of therapeutic potential during storage with extended shelf-life is of the highest importance for the pharmaceutical and industrial applications of plants. Drying seems to be a primary method taken up to meet these criteria of long-term preservation, employed for removing the excessive moisture content. It is important to follow regulations while drying medicinal plants, including lowering the moisture content to below 10%, preserving the bioactive composition while avoiding microbial degradation, and preventing loss of colour, flavour, and aroma.2–4 The quality of finished products suffers due to the loss of nutrients and bioactive molecules caused by extreme drying conditions under intense heat, increased oxygen levels, microwave or UV radiations, airspeed, and humidity. Although sun-drying is the most sought-after drying technique, prolonged exposure, and excessive dependency on external weather with non-uniform drying made the samples prone to fungal or bacterial contamination. These drawbacks made the shade drying technique preferable to sun drying.5–7 In addition to drying conditions, the solvent used, its polarity, concentration, time duration, and even the extraction temperature affect the yield and the type of phytochemicals extracted, undoubtedly affecting the antioxidant activity of the extract.8–10 The extensive demand and interest in bioactive molecules of natural origin in the pharmaceutical, chemical, and food industries have led to the development of targeted techniques ensuring maximum extraction yield. 11 An aqueous mixture of solvents like ethanol and methanol is preferable for maximum recovery of polyphenolic contents, especially from medicinal plants. In an era before the concept of pharmaceutical drugs and surgical treatment, the plant realm made a significant contribution to society’s ability to provide for its health benefits. 12 It is a fact that local populations are familiar with medicinal plants and other natural resources on which they depend largely for treating a wide range of illnesses. Moreover, it is not possible to extract each compound in a single solvent successfully; hence, it is essential to devise solvent systems based on the targeted type of constituent compounds.10, 13
Garcinia pedunculata (GP) Roxb. is commonly known as ‘Borthekera’ in Assam, India. It is an endemic medicinal plant of India’s North-Eastern Region (NER) and belongs to the Clusiaceae family. 14 It is an evergreen tree bearing an edible, highly acidic, globose fruit that has enormous medicinal properties and is widely used in culinary applications.15, 16 As per the ethnomedicinal practices of NER, the juice prepared from the dried pulps of G. pedunculata (GP) fruits has immense therapeutic importance in treating gastrointestinal disorders such as jaundice, indigestion, dysentery, and diarrhoea. However, consuming fresh pulps of GP fruit is not advised since it causes dysentery. Instead, after drying in the sun for about 15 days, the pulps are used to prepare Assamese delicacies and consumed. This dried preserved product is called ‘Suthi’ locally, and it is believed that the effectiveness of its medicinal properties increases over time.15, 17 From earlier published reports, it is evident that GP is a rich source of polyphenols (total phenolic content of 700.1 ± 10.74 mg catechin equivalent/100 g dried pulp), flavonoids (total flavonoid content 71.4 ± 0.84 quercetin equivalent (QE)/100g dried pulp) with profound antioxidant activity as reported by Mudoi and coworkers from its methanolic extract. 18 Several studies have confirmed its hypolipidemic, 19 neuroprotective, 20 nephroprotective, 21 and antidiabetic, 22 effects using its methanolic and aqueous extracts.
Prior research on the drying of GP Roxb. focused on the development of new techniques like free convection corrugated solar dryer and comparing it to sun drying, or examining the effect of various pretreatment with 10% fructose, 10% sucrose, and 2% NaCl on drying in a humidity chamber at temperature 45°C, 50°C, and 55°C, at constant relative humidity and air velocity or to draw a comparison between GP and Garcinia cowa, while drying both the fruits in the oven at 50°C, 70°C, 90°C, in solar cabinet dryer and sun-drying and then comparing their nutritional parameters.23–25
This study has been designed to address these points in shade drying, sun drying, and oven drying conditions, comparing any changes in their nutritional profile and extract yield, antioxidant, and enzyme inhibitory potential after extracting with different solvents. Then the chemical profiling of the extracts with comparatively higher activity in the three drying conditions was done using HPLC and UHPLC-MSMS to ascertain their bioactive components. It is believed that the outcome of the study will be significantly valuable for the further aspects of GP fruit-based nutraceutical or phytopharmaceutical development.
Materials and Methods
Chemicals and Reagents Used
Ascorbic acid, quercetin, gallic acid, 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2’-azino-bis (3-ethylbenzothiazoline) 6-sulfonic acid (ABTS), acarbose, 4-nitrophenyl butyrate, 4-nitrophenyl β-
Collection of Plant Samples and Identification
The GP fruits were collected from the Sarbhog town of Barpeta district of Assam (26° 19’ 22.55’’ North latitude and 91° 00’ 22.75’’ East latitude) from May to July 2019. The identification was made at the Herbarium center at the Department of Botany, Gauhati University, and the samples were submitted there for future reference under Ref. No. Herb. /GUBH/2022/002 and accession number GUBH19815.
Processing and Extraction
After being carefully washed, the ripe GP fruits were sliced into thin slices (0.9 ± 0.3 cm) and dried. The cut pieces were shade-dried (ShD) for 20 days at a temperature of 25–30°C in well-aerated chambers. Sun-dried (SD) samples were prepared under direct sunlight and recorded between 34°C and 40°C. The sliced pieces were kept in glass trays covered with muslin cloth for 15 days while being SD. Likewise, to obtain oven-dried (OD) samples, the cut pieces were kept in a hot air oven (Ikon Instruments, Delhi), and maintained at a temperature of 70–80°C for 4–5 days. These samples were then collected and placed in dry, airtight plastic bags for storage. About 10 g of these three dried samples were then extracted with 100 mL of five different solvents viz. hydromethanolic (90% methanol, MH), methanol (M), hydroethanolic (90% ethanol, EH), ethanol (E), and water (A) each (10:100 w/v) via cold maceration technique. Each extract was set for 7 days, with occasional shaking, and extracted thrice with fresh solvents every time. They were collected at the end of the extraction. The solvents were evaporated using a rotary evaporator (Buchi R210, Switzerland) and a lyophilizer (FreeZone Freeze Dryer, Labconco) to dry the extracts’ aqueous part. The % yield of each extract was then calculated and stored at 4°C–8°C in the dark for further experiments. The nutritional contents were estimated directly from the dried samples.
Proximate Analysis
Total carbohydrate, total protein, starch, and reducing sugar contents were determined in the proximate analysis.
26
The total ash and moisture content were determined by following the AOAC guidelines of 2005.
27
To determine the moisture content, 50 g of GP sliced fruit pulp was taken in a dried Petri dish, evenly distributed, and kept in a hot air oven at a temperature of 105 ± 2°C and cooled in a desiccator. The heating and cooling process was repeated till a constant weight of the sample was achieved. The moisture content was calculated as follows:
For ash content analysis, three dried samples (ShD, SD, and OD) were taken (2 g each) in clean sterile crucibles. These crucibles were heated on the Bunsen burner till the smoke emission ceased. Then, the burned-out samples were transferred to the muffle furnace (JSGW) for turning them to grey ash by heating at 500 ± 5°C. The ash content was calculated as follows:
For mineral content analysis, 100 mg of all three samples (ShD, SD, and OD) were digested with concentrated nitric acid (65%) while heating to obtain a clear solution, followed by the addition of a few drops of hydrogen peroxide. After cooling, the solution was filtered, and the final volume was made up of 100 mL of deionised distilled water. The mineral contents were then determined using atomic absorption spectrophotometer (SHIMADZU AAC-7000). 28 The total phenolic content was estimated spectrophotometrically using the Folin-Ciocalteu method and gallic acid as standard and the flavonoid content was estimated using the aluminium chloride method and Quercetin as standard. 29
HPLC Separation and Fraction Collection
The three different types of GP extract were analyzed using Waters Binary HPLC Pump (Waters, Milford, MA, USA) equipped with a manual injection valve with a loop of 20 µL and UV-visible detector controlled by Waters Breeze 2 Software. The extracts were prepared in methanol and passed through a C18 column with a gradient mobile phase of solvent A: water and solvent B: acetonitrile at a constant flow rate of 0.2 mL/min. The chromatogram was recorded as 254 nm.
Identification of Bioactive Compounds Through MS/MS
Exactive™ Plus-Orbitrap mass spectrometer (Thermo Scientific, Germany) coupled with UHPLC (Dionex Inc., Sunnyvale, CA, USA). The samples were dissolved with methanol and followed the previous method with a slight modification.30, 31 The extracts were passed through a Hypersil Gold C18 column (1.9) with a gradient mobile phase of solvent A: water and solvent B: acetonitrile at a constant flow rate of 0.2 mL/min. Using an electrospray ionisation (ESI) source, all MS/MS studies were carried out in both positive and negative modes. Xcalibur software was used for data collecting and processing. Mass spectra from m/z 100–1500 amu have a resolution of 70,000. The mass-to-charge ratio was compared to the literature and mass databases to characterize the extract contents.31, 32
Free Radical Scavenging Assay
The DPPH and ABTS radical scavenging assays were performed using the standard protocol with slight modifications.8, 33 To the 100 µL of different dilutions of extracts, 100 µL of DPPH or ABTS solution was added, and the absorbance was measured using the microplate reader (Multiskan, Thermo, Germany) at 517 nm and 734 nm, respectively, after half an hour of incubation of the reaction mixture at room temperature. Ascorbic acid was used as standard.
Ferric Reducing Antioxidant Power Assay
Ferric reducing antioxidant power assay (FRAP) assay was performed using the standard protocol with slight modifications. 34 To 100 µL of extract, 250 µL of the freshly prepared 0.2 M sodium phosphate buffer (pH 6.6) and 1% potassium ferricyanide were added, mixed thoroughly, and incubated at 50°C for 30 min. After incubation, 250 µL of 10% trichloroacetic acid solution was added, and then 250 µL of the reaction mixture was drawn out and mixed with 250 µL of double-distilled water. To this, 50 µL of freshly prepared 0.1% ferric chloride solution was added, and the absorbance was measured at 700 nm. The assay was performed in triplicates taking ascorbic acid as standard, and the increase in absorbance indicated the increase in reducing power.
Phosphomolybdenum Assay
Phosphomolybdate assay (PMD) was performed using the standard protocol with slight modifications. 35 About 150 µL of extracts in different concentrations were made from the 1 mg/mL stock solution, then mixed with 1500 µL of phosphomolybdate reagent (28 mM sodium phosphate, 0.6 M sulphuric acid, 4 mM ammonium molybdate) prepared freshly. This reaction mixture was mixed properly, then incubated at 95°C for 90 min and then the absorbance was measured at 695 nm, and ascorbic acid served as the standard compound.
α-Glucosidase Inhibition Potential
The assay was performed using the method of Inthongkaew et al.
36
with a few modifications. About 50 µL samples were plated on microtiter plates prepared in phosphate buffer solution (0.1 M, pH 6.8), mixed with 25 µL of enzyme solution (1U/mL), and set incubation at 37°C for 10 min, after which 25 µL of 4-Nitrophenyl β-
Pancreatic Lipase Inhibition Potential
The assay was performed following the method of Vangoori et al.,
37
with some modifications. To 100 µL of the sample, 700 µL of Tris buffer (10mg/mL) was added along with 200 µL of lipase enzyme, mixed properly, and incubated at 37°C for 15 min. Then, 5 µL of 10 mM p-nitrophenyl butyrate (dissolved in acetonitrile) substrate was added to this reaction mixture, and then incubated at 37°C for 30 min. Absorbance was measured at 405 nm with Orlistat as standard. The percentage of pancreatic lipase inhibitory activity was calculated as follows:
Statistical Analysis
All the results were expressed as mean ± standard deviation for triplicate experiments. The significance has been calculated using one-way ANOVA using the software GraphPad Prism Version 5.0 (San Diego, CA, USA), followed by Tukey’s post hoc test. The results of the same row with different superscripts were significantly different from each other in terms of drying conditions and extraction with the same solvent at p < 0.05.
Results
Impact of Drying on Proximate Composition
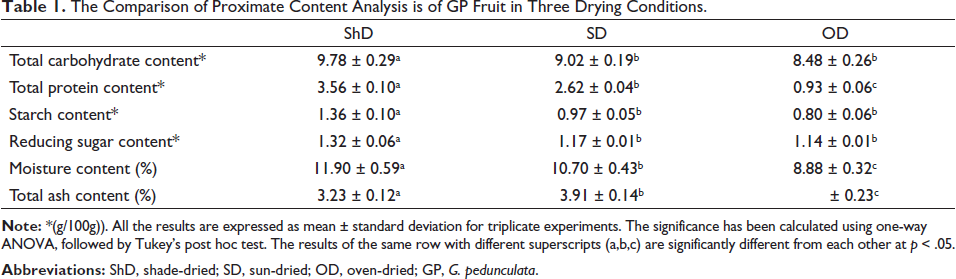
The dried fruits of GP under three drying conditions (ShD, SD, and OD) (Figure 1) were utilized for proximate content analysis. Comparing the total carbohydrate content, ShD showed the highest carbohydrate content (9.78 ± 0.29 g/100 g) as compared to the SD (9.02 ± 0.19 g/100 g) and OD (8.48 ± 0.26 g/100 g). In total protein content also, ShD had significantly higher protein content, 3.56 ± 0.10 g/ 100 g of dried sample, followed by SD (2.62 ± 0.04 g/100 g) and OD (0.93 ± 0.06 g/100 g) in decreasing order. Similar trends have been followed in starch and reducing sugar analysis.

In moisture content analysis, ShD (11.90 ± 0.59%) had significantly higher moisture content followed by SD (10.70 ± 0.43 %) and OD (8.88 ± 0.32%). ShD had significantly higher moisture content when compared to OD. The ash content analysis revealed a reverse trend as compared to moisture content as OD had the highest content with 4.43 ± 0.230%, followed by SD with 3.91 ± 0.141% and then ShD with 3.23 ± 0.120%. The detailed nutritional parameters are given in Table 1.
The Comparison of Proximate Content Analysis is of GP Fruit in Three Drying Conditions.
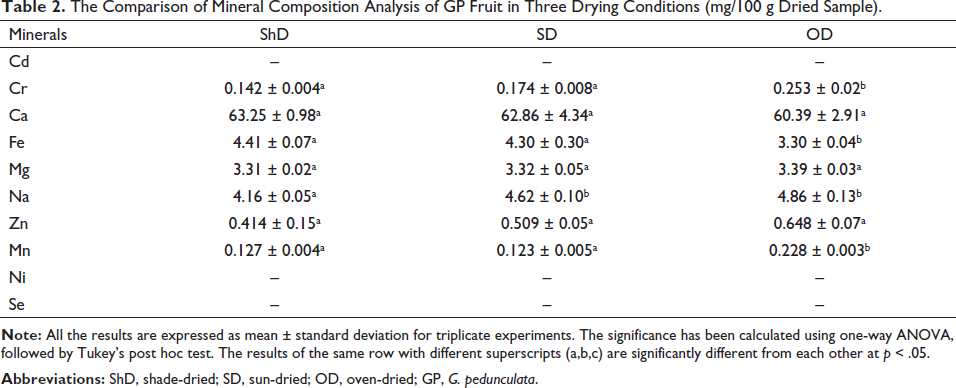
From the mineral content analysis, it was found that manganese (Mn), chromium (Cr), magnesium (Mg), zinc and sodium (Na) were present in higher quantities in OD whereas the iron (Fe) content of ShD was significantly higher than OD. The Cr, Mg, zinc, calcium (Ca), Fe, and Mn content did not vary significantly in ShD and SD, while Ca was lower in OD when compared to ShD but was not significantly different in the three drying conditions. The detailed mineral content analysis is presented in Table 2.
The Comparison of Mineral Composition Analysis of GP Fruit in Three Drying Conditions (mg/100 g Dried Sample).
Extractive Yield and Phytochemical Analysis
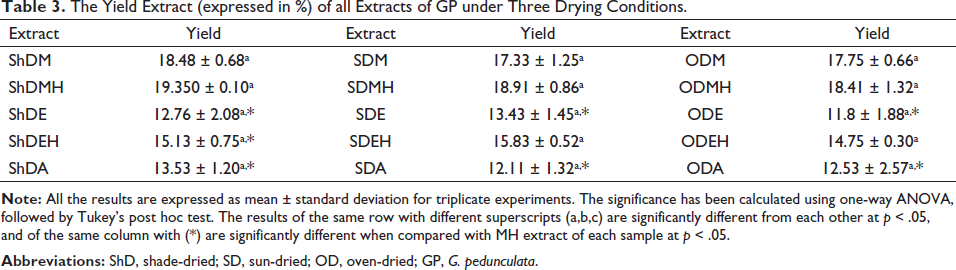
Comparing the three drying conditions, the yields of extracts with the same solvent are not significantly different, as shown in Table 3. Here, upon comparison, ShDMH was found to have a significantly higher yield when compared to shade-dried ethanolic extract (ShDE), shade-dried hydroethanolic extract (ShDEH), and shade-dried aqueous extract (ShDA). Similarly, sun-dried hydromethanolic extract (SDMH) had a significantly higher yield for sun-drying conditions than sun-dried ethanolic extract (SDE) and sun-dried aqueous extract (SDA). Oven-dried hydromethanolic extract (ODMH) had a higher yield for oven drying conditions, not so different from oven-dried methanolic extract (ODM) and oven-dried hydroethanolic extract (ODEH) while having a significantly higher yield than oven-dried ethanol extract (ODE) and oven-dried aqueous extract (ODA).
The Yield Extract (expressed in %) of all Extracts of GP under Three Drying Conditions.
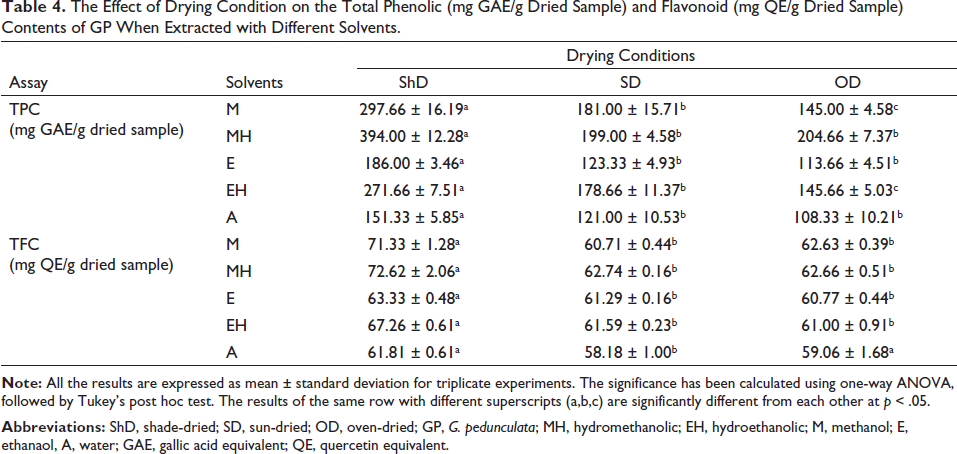
In phytochemical analysis, when compared in terms of extraction solvent and drying conditions, the phenolic content of hydromethanolic extracts under all drying conditions were found to be higher when compared to the methanolic, ethanolic, or aqueous extracts. In ShD conditions, the overall phenolic content was found to be significantly higher when compared to SD and OD conditions for each of the solvent extraction systems. In the flavonoid content analysis, ShDMH had been recorded to have the highest content, followed by shade-dried methanolic extract (ShDM) among all the samples. The exact values of phenolic and flavonoid content are given in Table 4.
The Effect of Drying Condition on the Total Phenolic (mg GAE/g Dried Sample) and Flavonoid (mg QE/g Dried Sample) Contents of GP When Extracted with Different Solvents.
HPLC/LCMS-MS Analysis
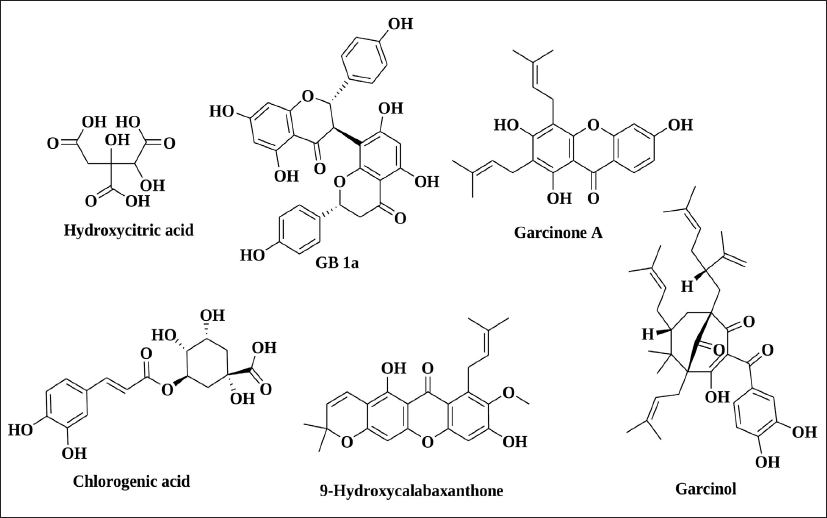
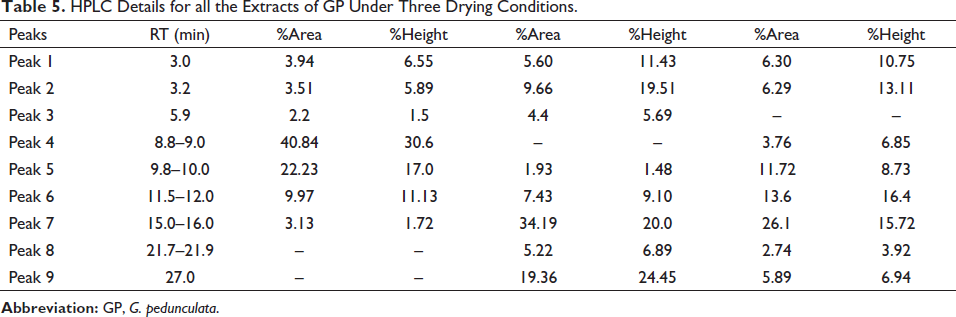
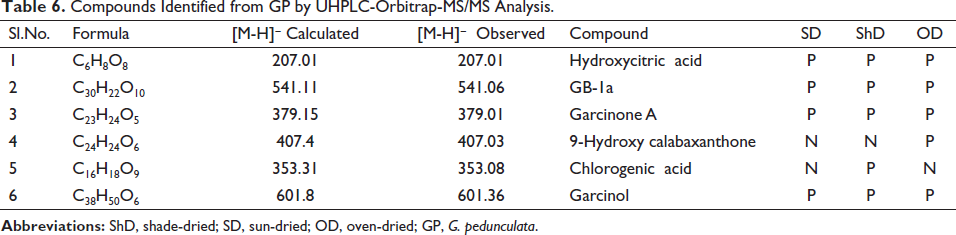
In preliminary investigation and identification of the GP bioactive compounds, the HPLC technique demonstrated linearity, precision, and accuracy in the concentration range, meeting regulatory criteria. Different wavelengths and sample run times were attempted before the concluded protocol. Nine peaks were obtained at the optimized wavelength of 254 nm. The comparisons of the HPLC chromatogram of these three extracts are shown in Figure 2. Qualitative screening of multiclass secondary metabolites present in the fruit extracts of GP was carried out using UHPLC-Orbitrap-MS/MS analysis. This UHPLC-ESI-MS/MS has well-equipped to differentiate the masses, even if they can be eluted. Together, HPLC profile and UHPLC-MS/MS confirmed that the compounds, hydroxycitric acid, GB-1a, Garcinone A, 9-hydroxycalabaxanthone, chlorogenic acid, and garcinol (Figure 3) were present in these extracts. All the compounds have shown the corresponding [M-H]− peak in the negative mode of ionization. The retention time, percentage area, and percentage height of the peaks are listed in Table 5. The compound with m/z 207.01 [M-H]− is identified as hydroxycitric acid. The compound, GB-1a has 541.06 [M-H]− and Garcinone A has 379.01 [M-H]−. Similarly, it has to be understood that the molecules such as 9-hydroxycalabaxanthone has 407.03 [M-H]−, chlorogenic acid has 353.08 [M-H]−, and Garcinol has 601.36 [M-H]− as shown in Table 6.
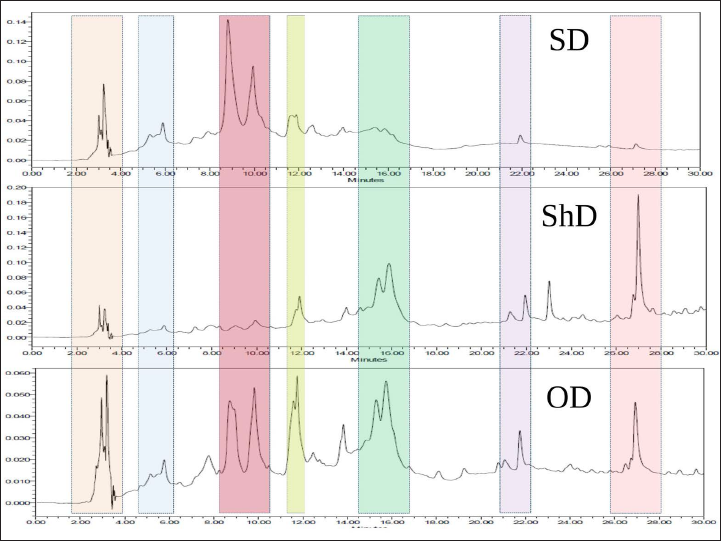
HPLC Details for all the Extracts of GP Under Three Drying Conditions.
Compounds Identified from GP by UHPLC-Orbitrap-MS/MS Analysis.
Antioxidant Properties of the Different Extracts
The antioxidant properties had been assessed using DPPH and ABTS free radical scavenging assays and the IC50 for all 15 extracts, as shown in Figure 4. In the DPPH assay, ShDMH had been found to have a significantly lower IC50, indicating the highest antioxidant activity among all the extracts. Also, in shade drying conditions, the IC50 of ShDMH was followed by ShDM, ShDEH, ShDE, and ShDA in increasing order. In sun drying conditions also, SDMH had been found to have the lowest IC50 amongst the five different extracts which followed the trend of SDMH <SDEH < SDM < SDE < SDA in increasing order, while in the oven drying conditions, the IC50 varies along with the increasing order of ODMH< ODM < ODEH < ODE < ODA. Also, all the hydromethanolic extracts had better activity in three drying conditions, and ShDMH had significantly lower IC50 when compared to SDMH and ODMH. In comparing a particular solvent extract among three drying conditions, it was found that ShD extract has better activity when compared with SD and OD conditions in their respective solvents.
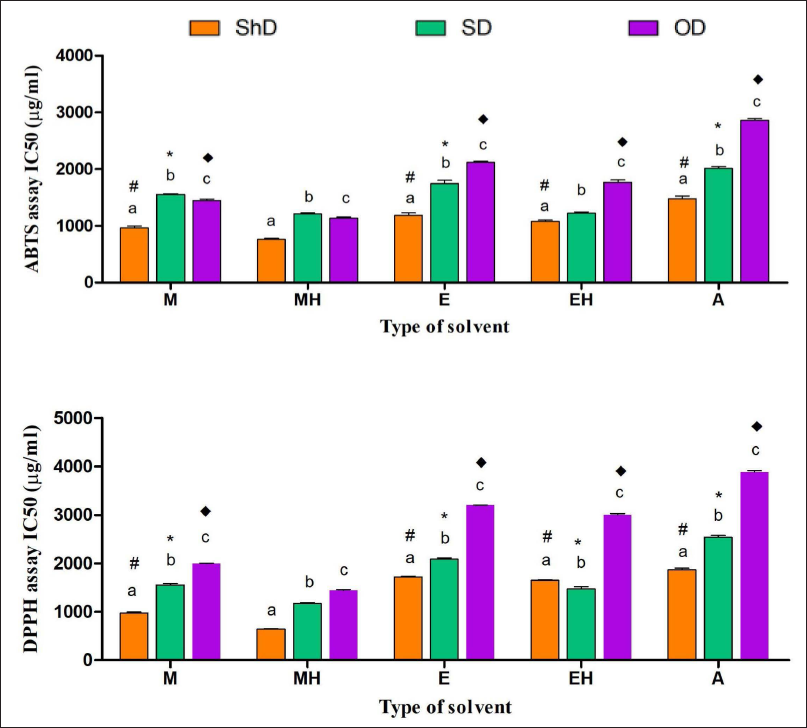
In the ABTS scavenging assay as well, ShDMH exhibited the best scavenging activity amongst all the extracts under consideration, while ODA had the least recorded activity. In shade drying conditions, the IC50 is seen to be increasing in order of ShDMH < ShDM < ShDEH < ShDE < ShDA. The values found are significantly different from each other within the same drying condition. In sun drying conditions, the IC50 is in the order of SDMH < SDEH < SDM < SDE < SDA, with the best activity recorded in SDMH and the least activity in SDA. In oven drying conditions, ODMH < ODM < ODEH < ODE < ODA order is followed by IC50 values, which showed that ODMH had better activity among the five extracts. In terms of drying conditions, ShD samples had significantly lower IC50 values in comparison with their respective solvent extracts in the two other drying conditions, while hydromethanolic extract showed higher activity amongst all the three drying conditions and their respective extracts with different solvents.
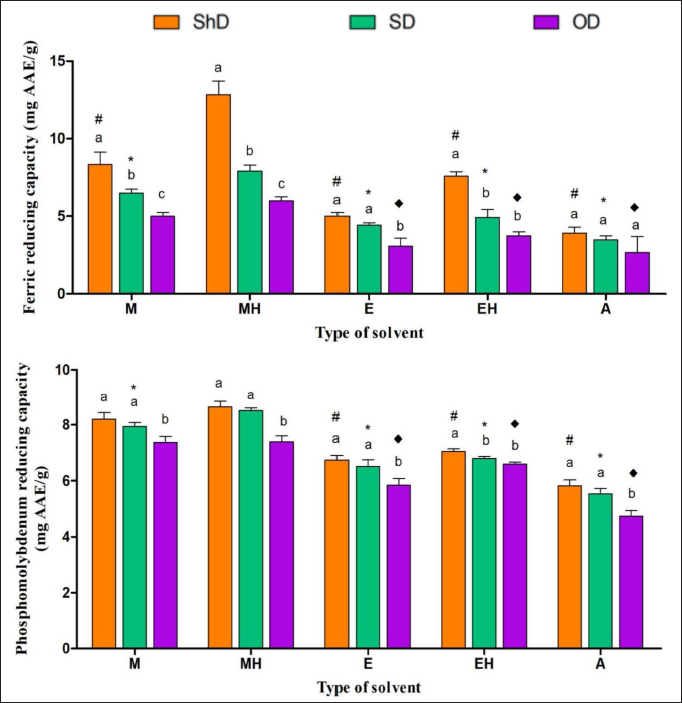
In FRAP and PMD assays also, ShDMH revealed the highest reduction capability among all the extracts. In a comparison drawn between the three drying conditions, the ShD samples showed better preserved, reducing activity when compared to SD and OD conditions. In between all the five extraction solvent systems in all three drying conditions, hydromethanolic extracts showed the maximum activity. Their comparative results are shown graphically in Figure 5.
Enzyme Inhibition Properties of Different Extracts
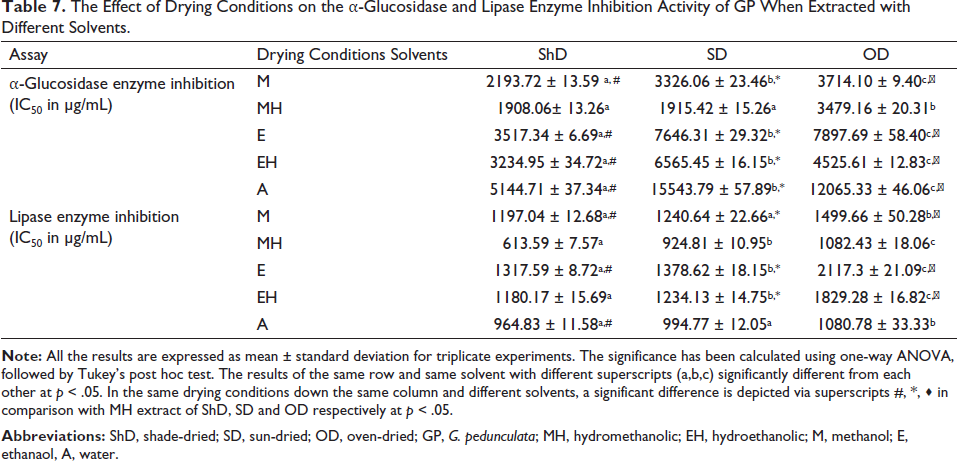
In the α-glucosidase inhibition assay, amongst all the extracts, ShDMH had the maximum enzyme-inhibiting activity compared to other extracts. While comparing respective solvents under different drying conditions, it has been found that ShDMH and SDMH had nearly the same activity. In contrast, ODMH had a much-reduced inhibitory effect, while sun-dried methanolic extract (SDM), ShDM, and ODM showed significantly different inhibitory capacities. In between the three drying conditions, the ShD samples had shown better activity when compared to their respective solvent extracts of the SD and OD samples. In lipase inhibition assay as well, ShDMH had been found to have the maximum activity, which is significantly higher than the SDMH and ODMH extracts. In a comparison of the drying conditions, ShD samples had shown much better lipase inhibiting activity than the other two drying conditions as well as for the solvent extraction system among the three drying conditions (Table 7).
The Effect of Drying Conditions on the α-Glucosidase and Lipase Enzyme Inhibition Activity of GP When Extracted with Different Solvents.
Discussion
Drying fruits and vegetables are often associated with reducing their weight, color, and size shrinkage, which increases their preservation time, and helps in easy packaging, handy storage, and transportation with less spoilage. Although, with drying conditions, the end product’s antioxidant capacity changes, along with colour, texture, physical shape, and nutritional and phytochemical composition. Hence, the appropriate drying condition must maintain the quality during preservation. 38 The raw fruit pulp of GP is off-whitish, which after drying is turned into deep brown to blackish brown, and the weight was reduced by almost 85 ± 3.3135% in shade drying, 88 ± 5.2489% in the sun drying, and 91 ± 6.8322% in the oven drying conditions as represented in Figure 1.
Traditionally, dried GP fruit is used in preparing culinary delicacies. These dried fruit pulps are also used in different herbal formulations for treating gastrointestinal disorders. Also, as per popular belief, the therapeutic potential of dried GP increases as its ages in storage. 17 The results of this study clearly established that shade-dried GP (ShD) had the highest preserved carbohydrates, proteins, and moisture while OD samples had the highest ash content. The significant reduction of carbohydrates and protein, as noted in OD, might be attributed to its exposure to very high drying temperatures, which is not the case with ShD and SD being dried at much lower temperatures. It is already reported that simple sugar such as fructose, glucose (carbohydrates), and proteins undergo degradation with exposure to higher drying temperatures, and the browning of the sample after drying might be because of high amounts of reducing sugar in them. Since the fruit of GP is highly acidic, it might be a reason for the fruit not undergoing severe degradation in its macronutrients even with a longer dehydration time. The loss of nutrients that happened might be attributed to the high temperature of the drying conditions, which induces non-enzymatic browning involving a complex degradation reaction amongst the amino groups of proteins or amino acids and the reducing sugar moieties in the fruit. This reaction, called the Maillard reaction, with the production of brown melanoidin pigments, is also responsible for changing the colour, taste, and lower protein nutritive value of the fruit after drying.25, 38, 39 The results of this study in terms of nutritional profiling are similar to the earlier studies published on Moringa oleferia leaves, Musa paradisiaca, Dioscorea srotundata, Colocasia esculenta, Garcinia cowa, Garcinia indica, Vernonia amygdalina (bitter leaf), and leaves of Mentha spicata.25, 40–44 This study also found that the fresh fruit has the highest moisture, and after drying, ShD has higher moisture content followed by SD and OD had the lowest. Also, OD had the highest ash content, and ShD had the lowest. Moreover, OD samples have higher mineral contents when compared to ShD or sun-dried which is mainly because of its reduced minimal moisture content and thus, concentrated dry matter. These results are in accord with the earlier published studies on Vernonia amygdalina (bitter leaf), leaves of Mentha spicata, and Hibiscus sabdariffa, where in oven drying conditions, due to the availability of uniform heating and high temperature the moisture loss is more as compared shade drying and sun drying. And the ash content is often inversely proportional to moisture content and with the loss of moisture, the minerals were more concentrated in the OD samples, as compared to the sun-dried and the ShD ones.43–47 In this study, Ca and Fe are found to be the most abundant minerals in all three samples similar to the previous report. 25 In comparison to the reports published by Parthasarathy and Nandakishore 2014, the Ca content reported here is much higher, while the Mg content is lower in quantity. 48 Also, the Mn, Zn, and Cd levels were much similar, while the Na and Fe were somewhat higher than the reports of Sarma et al., 2016. These differences in the minerals in the analyzed data for this study and the earlier reports might be because of the surrounding environmental factors, availability of nutrients, time, and collection of the fruit.49, 50
In this study, the trend in extract yield is in agreement with the previous studies, where the most efficient extraction of antioxidants and highest yield in the methanolic extract compared to acetone and water.51, 52 In yet another study, with methanolic, ethanolic, and hydroethanolic extracts of various medicinal plants in comparison, the hydroethanolic extracts were reported to have maximum yield. One of the crucial reasons behind the highest total phenolic and flavonoid content in ShDMH, followed by ShDM might be attributed to the higher polarity of aqueous methanol or ethanol compared to pure methanol and ethanol. 53
The phenolic and flavonoid contents in this study are found to be better preserved in the ShD conditions, and were much reduced in OD samples which are in line with earlier studies on black chokeberries, black rice, Dryopteris erythrosora, Pimpinella anisum seeds, and with rising in drying temperature, a loss to the phenolic compounds was observed, which might actually be due to their thermal degradation owning to their heat-sensitive nature, or they may even chemically interact with other biomolecules forming conjugated products which might be out of scope for the detection technique used. They may also be acted upon by oxidative enzymes which may reduce their levels in the end products. The phenolic and flavonoid content is also directly related to the antioxidant capacity of these extracts. This explains the better antioxidant activity in terms of radical scavenging and metal-reducing power which lies in direct correlation with the higher phenolic and flavonoid content of the ShD samples, as compared to the SD or OD.54–57 For the enzyme inhibitory potential as well, a similar trend was followed, and with the reduction in phenolics and flavonoids, the inhibitory potential also decreased.58–62 Hence, this study concludes that these dried fruit pulps would be of utmost benefit if the fruits were shade-dried and extracted with hydromethanolic solvent for better yield and preserved phytochemical extractions.
Summary
The fruit of GP Roxb. is a sour-tasting, acidic fruit consumed only in its dried form, especially in Assam, where it is an integral part of ethnic cuisine. In traditional practice, the fruit is sun-dried for at least 15 days, and this dried fruit is then stored over time as per the widespread belief that its medicinal properties enhance as the fruit ages. Keeping the importance of drying in mind for this fruit, we designed this study to ascertain the impact of drying techniques and different extraction solvents on the nutritional, antioxidant, and enzyme inhibitory activity of GP fruit. From this study, we have established that shade-dried samples exhibited better preserved nutritional properties, phenolics, and flavonoids compared to sun-dried and oven-dried conditions. The antioxidant and enzyme inhibitory potential were also higher in the shade-dried samples, especially in the hydromethanolic extracts compared to the other solvents. The significant finding from the HPLC analysis revealed the presence of garcinol, hydroxycitric acid, GB-1a, garcinone A, 9-hydroxycalabaxanthone, and chlorogenic acid from the hydromethanolic extracts of the GP fruit. These bioactive compounds are believed to be responsible for the potent antioxidant and enzyme inhibitory potential of the hydromethanolic extracts under the three drying conditions.
Footnotes
Abbreviations
Acknowledgment
The authors are thankful to the Director, IASST, Guwahati, and the Vice-Chancellor, Annamalai University for giving us the facility to carry out this work. We also thank IASST and Annamalai University for providing instrumentation facilities.
Authors’ Contributions
S.B. conducted all the experiments and analysis of results; D.C. (Trainee at IASST) helped in the experiments; P.P.S helped with the HPLC experiment; R.E. helped in analysing the HPLC, LCMS-MS results, identification of compounds; P.K.D. helped in the concept, and analysis of results; S.B., R.E., P.K.D. helped in the writing of the paper; R.D. and S.K. have overall conceptualized and supervised the project.
Declaration of Conflicting Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
S.K. is a recipient of the UGC-BSR Faculty fellowship, and DST PURSE Phase-II, has been provided to Elancheran Ramakrishnan by Annamalai University as Research Associate.