Abstract
Background
Cerebral ischemia is a condition of acute brain damage due to the depletion of oxygenated blood supply to cerebral tissues. Daidzein is an isoflavonoid predominantly present in soya, Pueraria species, and red cloves. Traditional Chinese medicine utilizes daidzein to alleviate diseases such as inflammation, hyperglycemia, gastric diseases, allergies and aches. The neuroprotective effect of daidzein on cerebral ischemic conditions and its mechanism of action was not yet elucidated.
Materials and Methods
24 healthy male adult Wistar rats were grouped into four and the control rats were sham-operated, cerebral ischemic-reperfusion induced rats subjected to middle cerebral artery occlusion (MCAO). Low- and high-dose daidzein rats were treated with 25 and 50 mg/kg daidzein respectively for 7 consecutive days before the induction of cerebral ischemic reperfusion. On completion of treatment, the rats were assessed for neurological deficit scoring and then euthanized for further analysis. The percentage of brain water content and cerebral infarct was evaluated. The ability of daidzein on preventing oxidative stress-induced damage was assessed by quantifying lipid peroxidation and antioxidant levels. The neuroprotective Daidzein was evaluated by measuring the acetylcholinesterase activity and brain ATP levels. The anti-inflammatory role of Daidzein was measured by quantifying the nitric oxide (NO) and inflammatory cytokines. Further, the anti-ischemic role of Daidzein was confirmed by estimating nuclear factor-kappa B (NF-κB) p65 and Caspase 3 levels.
Results
Daidzein treatment significantly prevented brain edema and cerebral infarction and neurological deficit in cerebral I/R injured rats. It also scavenged the free radicals and prevented the decline in antioxidant levels of ischemic rats. Daidzein decreased the acetylcholinesterase activity, NO, and inflammatory and significantly increased the brain ATP levels signifying its neuroprotective role in ischemic-induced rats. The reduction in the levels of NF-κB p65 and Caspase 3 confirms daidzein prevents neuroinflammation and neuronal apoptosis in ischemic rats.
Conclusion
Overall our analysis confirms daidzein is a potent neuroprotective drug which can effectively inhibit postischemic complications.
Introduction
Nutraceuticals are natural oral dietary supplements which possess pharmacological properties. Global usage of nutraceuticals had increased due to their therapeutic efficacy and safety. 1 Nutraceuticals have been reported to have promising effects on diseases such as atherosclerosis, 2 diabetes, 3 CVD, 4 gastrointestinal diseases, tumor, 5 renal, and neurological diseases. 6 Nutraceuticals acts as antioxidants and their intake improves the quality of life, increases life expectancy, delays aging, and also prevents chronic diseases. 7 Nutraceuticals are the selective combinations of phytochemicals such as flavonoids, carotenoids, polyphenols, phytoestrogens, terpenoids, and isoflavonoids. 8 Flavonoids which have been subjected to extensive pharmaceutical research are phytoestrogens, which are comprised of isoflavones, prenylflavonoids, and coumarins. 9
Daidzein is a phytoestrogen isoflavonoid present mainly in soyabeans, red cloves, and Pueraria sps. It belongs to the category of nonsteroidal estrogens which possess a broad spectrum of pharmacologic effects. 10 Diadzein possesses anti-hemolytic, anti-inflammatory, anti-obesity, and anticancer properties. It is a potent antioxidant used in traditional Chinese medicine to treat hyperlipidemia, hyperglycemia, allergies, skin diseases and arthritis. 11 Daidzein prevents neuronal cell death in oxygen–glucose-deprived conditions via activating the gamma receptors. It also prevented cultured neuronal cells from glutamate excitotoxicity. 12 In vitro primary neuronal cell cultures, daidzein significantly protected the neurons from neurotoxicity induced by β-amyloids. 13 Recent studies revealed that daidzein attenuated urinary bladder dysfunction in diabetic rats, 14 mitigated myocardial damage, 15 and normalized the gentamicin-induced nephrotoxicity. 16 Even though numerous neuroprotective roles of daidzein have been the ameliorative role of daidzein on cerebral ischemic-reperfusion induced post complications is not yet elucidated.
Globally around 15 million stroke incidences were reported every year with a high relapse rate. 17 Cerebral ischemia ensues due to the depletion of oxygenated blood supply to cerebral tissues. The ischemic condition followed by reperfusion cause severe brain tissue damage. 18 Inflammation is considered to be the primary mechanism of action for the induction of infract in brain tissues. Inflammation occurs due to the activation of resident immune cells and also by the recruitment of inflammatory cytokines due to the leukocyte activation enabling the blood–brain barrier disruption. 19 Nuclear factor-kappa B (NF-κB) is ubiquitously present in all the cells of brain tissue and disruption to NF-κB signaling causes excitotoxicity.20, 21 NF-κB signaling pathway plays a vital role in ischemic conditions via regulating the inflammation and apoptotic pathways. 22 The increase in NF-κB levels has been positively correlated with the severity of stroke. The increase in NF-κB p65 and p50 expression was reported in middle cerebral artery occlusion (MCAO)-induced rats suggesting the role of ischemic induction.23, 24 Hence we analyzed the role of daidzein in preventing cerebral ischemic reperfusion-induced neuroinflammation in MCAO rat model via inhibiting NF-κB signaling pathway.
Materials and Methods
Chemicals
Daidzein, HPLC grade, Bradford reagent, and triphenyl tetrazolium chloride were procured from Sigma Aldrich, Mybiosiurce, Abcam, and Elabscience, USA. Only analytical grade chemicals were procured and utilized for the study.
Animals and Ethics
Each procedure is carried out on animals as per the institutional ethical committee guidelines. Healthy male Wistar rats aged about 10 weeks old were utilized for the study. The rats were acclimatized for a period of 7 days on a 12 hr light/dark cycle at 22 ± 2°C with 55 ± 5% relative humidity before the initiation of the experiment. The rats were fed with a standard laboratory pellet diet and water ad libitum.
Animal Grouping
The healthy acclimatized rats were randomly grouped into four. Sham-operated control rats were not treated with any drugs. Cerebral I/R injured rats are subjected to cerebral ischemic-reperfusion insult and were not treated with any drugs. The other two groups of rats were treated with 25 mg/kg daidzein and 50 mg/kg bwt daidzein for seven successive days and subjected to cerebral ischemia-reperfusion insult.
Induction of Cerebral I/R Injury
Cerebral I/R injury was done in rats according to protocol of Long et al., 24 MCAO model; 350 mg/kg of 10% chloral hydrate were injected intraperitoneally into rats to get anaesthetized. The anesthetized rats were subjected to a midline neck slit and the carotid artery was carefully isolated from the vagus nerve. A nylon thread measuring about 4 cm long and 0.2 mm in width was inserted gently into the internal carotid artery. A 18–20 mm thread was inserted until it reaches the tiny anterior cerebral artery which blocks the middle cerebral artery blood flow for an hour. After an hour, the nylon thread was gently drawn back and the arterial stump was knotted. Subsequently suturing was performed on the skin and the subcutaneous tissue. The whole-body temperature and the arterial blood pressure of rats were observed carefully. Sham-operated rats were subjected to the same surgical procedure without performing occlusion. After 24 hr of reperfusion, the rats were subjected to neurological deficit scoring and then euthanized for further analysis.
Evaluation of Cerebral Edema
Brain water content in the rats was estimated after the cerebral I/R injury in Daidzein-treated and untreated rats. The isolated brain was weighted using a digital weighing machine and the total wet brain weight was noted. The brain was subjected to drying at 100°C for 24 hr in a hot air oven and the dried brain tissue was weighed again. The total water content in brain tissue was estimated using the formula
Assessing Cerebral Infract
The isolated brains were carefully rinsed with physiological saline and dried with filter paper, subsequently frozen. The frozen brain was subjected to coronal section of 2 mm thickness and the sections were stained with 2% triphenyl tetrazolium chloride solution for 30 min in dark at 37°C. The cerebral sections were incubated for a period of 12 hr with 4% paraformaldehyde and sections were assessed for infract with Image analysis System DT200 (Nanjing Tech, China). The infarct regions displayed a white color whereas the non-infract regions were red in color.
Scoring of Neurological Deficit
The rats were subjected to neurological deficit scoring after the reperfusion period according to the protocol of Long et al. 24 The rats were assessed according to the scoring point of Longa and graded from 0 to 4. 0—Healthy rats with no neurological impairment, 1—Mild deficit with contralateral body and forepaw stretching impairment, 2—Moderate deficit with circling toward the contralateral side, 3—Severe deficit the rat fall toward the contralateral side, and 4—Rats were lack spontaneous walking and were unconscious.
Preparation of Tissue Homogenate
The brain tissue homogenate was prepared for the biochemical analysis of protein, antioxidants, inflammatory cytokines, ATP levels, NF-κB, and caspase 3 activities. One hundred milligrams of brain tissue was homogenized with 0.1 mol/L of Tris-HCl buffer (pH 7.4) using a tissue homogenizer. The tissue was homogenized with an intermediate interval of 10 sec to avoid an increase in temperature. The homogenate was subjected to centrifugation at 15000 rpm for 10 min at 4°C, supernatant was collected for biochemical analysis.
Estimation of Total Protein
The total protein content in the tissue homogenate was estimated using the commercially available Bradford reagent procured from Sigma Aldrich, USA. The final product absorbance was measured at 595 nm using ELISA microplate reader.
Estimation of Acetylcholinesterase Activity
Acetylcholinesterase activity in cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats were quantified using the method of Ellman et al. 25 An artificial substrate acetythiocholine iodide was used to measure the acetylcholinesterase activity. The acetylcholine iodide is cleaved to thiocholine which further reacts with DTNB reagent leading to the form of reduced thionitrobenzoic acid. This yellow-colored thionitrobenzoic acid was measured at an absorbance of 412 nm using ELISA microplate reader.
Estimation of Lipid Peroxidation
The levels of lipid peroxidation in the cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats brain tissue were done using the protocol of Devasagayam and Tarachand 26 Two hundred microliters of tissue homogenate was mixed with a cocktail mixture consisting of 1 mL 0.15M Tris HCl buffer, 300 µL 10mM potassium dihydrogen phosphate, and 1.5 mL of thiobarbituric acid. The reaction mixture was incubated at 100°C for 20 min and then a 10% trichloroacetic acid stop solution was added. The end product of malondialdehyde forms a pink chromogen adduct with thiobarbituric acid which was measured at 532 nm using ELISA microplate reader.
Estimation of Antioxidants
Quantification of Superoxide Dismutase
The superoxide dismutase (SOD) activity in brain tissue of cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats were evaluated according to the protocol of Marklund and Marklund. 27 Five hundred microliters of tissue supernatant was mixed with 250 µL of absolute ethanol and 150 µL of chloroform. The mixture was placed on to mechanical shaker for a period of 15 min. The mixture was then subjected to centrifugation at 10,000 rpm for 10 min at 4°C. The supernatant was subjected to autooxidation by mixing with 2 mL Tris-HCl buffer and 500 µL of 2mM pyrogallol along with 1.5 mL distilled water. The absorbance of the assay mixture was measured at 470 nm using ELISA microplate reader.
Quantification of Catalase
The catalase enzyme activity was quantified using the method of Sinha 1972 28 in cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rat’s brain tissue. Assay mixture consisting of 400 µL distilled water, 500 µL 0.2M hydrogen peroxide, and 1 mL sodium phosphate buffer were added to 1 mL of tissue homogenate. Two milliliter of dichromate acetic acid reagent was added to the reaction mixture to arrest the reaction and the tubes were incubated in a boiling water bath for 10 min. The mixture was allowed to cool, and the final mixture was subjected to measurement of absorbance at 590 nm. The catalase activity in the samples was represented as U/mg protein.
Quantification of Glutathione Peroxidase
Glutathione peroxidase activity in cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats’ brain tissue was determined using the protocol of Rotruck et al. 29 Five hundred microliter of tissue extract was mixed with the assay mixture consisting of 500 µL sodium phosphate, 200 µL 4mM reduced glutathione, 100 µL 2.5mM hydrogen peroxide, and 100 µL 10mM sodium azide. The reaction mixture was made up to 2 mL with distilled and incubated at 37°C for 5 min. Five hundred microliters of 10% trichloroacetic acid stop solution was added. The mixture was subjected to 7500 rpm for 15 min and the supernatant was collected. To the supernatant 4 mL of 0.3 m disodium hydrogen phosphate and 1 mL DTNB reagent were added. The final mixture absorbance was measured at 412 nm.
Quantification of Glutathione
The levels of glutathione in brain tissue of cerebral I/R injured untreated and Daidzein-treated Cerebral I/R injured rats were quantified using the method of Vandeputte et al. 30 Twenty microliter of KPE buffer was used as blank consisting of 0.1 M potassium phosphate buffer and 5 mM EDTA disodium. To 20 µL of sample 120 µL of DTNB and GR reagent (1:1) ratio was added. After 30 sec, 60 µL of β-NADPH was added and the reaction mixture was measured at 412 nm.
Estimation of Brain ATP Levels
The levels of brain ATP were measured in cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats’ brain tissue using a commercially available ATP assay kit procured from Sigma Aldrich, USA. It is a highly sensitive method, the levels of phosphorylating glycerol are directly proportional to the levels of ATP. The final mixture was measured at 570 nm and the levels were expressed as pg/100mg wet tissue.
Estimation of Inflammatory Cytokines
The brain tissue homogenate of cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats were subjected to estimation of inflammatory cytokines TNF-α, IL-6, and IL-10 using ELISA kits purchased from Abcam, USA. The test was performed according to the instruction of the manufacturer’s protocol.
Estimation of Nitric Oxide
The nitric oxide (NO) levels in cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats’ brain tissue were estimated as total nitrates/nitrites using the commercially available NO estimation kit, Sigma Aldrich, USA. To 20 µL of sample 20 µL nitrate assay buffer, 10 µL of Griess reagent I and II were added. The reaction mixture was incubated at 37°C for 10min and the final absorbance was measured 540 nm.
Estimation of Brain NF-κB p65 Level
The brain tissue homogenate was prepared with phosphate-buffered saline using the freeze-thaw method. The cell membranes of brain tissue were ruptured by repeated freezing and thawing carried out for two cycles. The homogenate was further centrifuged at 7500 rpm for 10 min 4°C and the supernatant was collected for the further estimation of brain nuclear factor–κB p65 (NF-κB p65) level. NF-κB p65 levels were quantified using ELISA kit, MyBioSource, USA. The final absorbance was read at 450 nm using ELISA microplate reader.
Estimation of Caspase-3 Activity
The levels of caspase 3 in brain tissue homogenate of cerebral I/R injured untreated and Daidzein-treated cerebral I/R injured rats were estimated using colorimetric kit purchased from Elabscience, USA. The caspase-3 in the sample conjugates with p-nitroaniline which was measured at 405 nm.
Statistical Analysis
The results were analyzed with a one-way analysis of variance (ANOVA) followed by the post hoc Dunnett’s test using statistical software SPSS version 23, USA. The significance was p < 0.05. The results were expressed as mean ± standard deviation for three independent observations.
Results
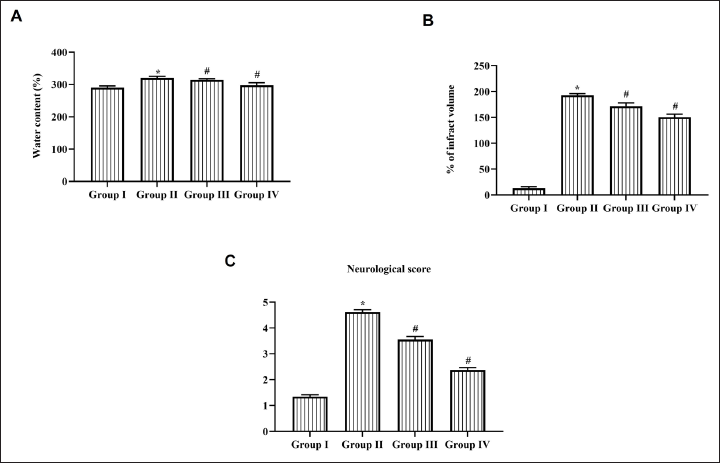
Daidzein Prevented the Cerebral Infarction in Cerebral I/R Injured Rats
Daidzein treatment considerably decreased the level of brain water content (Figure 1A). Cerebral I/R injured rats showed 180% infract whereas it decreased to 20% in 25 mg/kg Daidzein treatment and 30% in 50 mg/kg Daidzein-treated rats (Figure 1B). Daidzein treatment significantly decreased the neurological deficit scoring in cerebral I/R injured rats. Both 25 and 50 mg/kg Daidzein treatment showed scoring of moderate deficit, whereas cerebral I/R injured untreated rat showed unconsciousness which lacks spontaneous walking (Figure 1C).
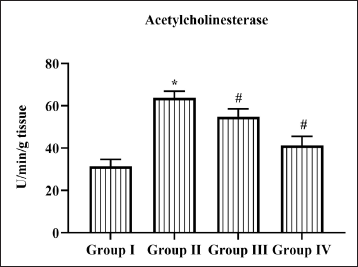
Daidzein Inhibited Acetylcholinesterase Activity in Cerebral I/R Injured Rats
Increased level of acetylcholinesterase was associated with cognitive impairment in ischemic patients. Cerebral I/R induction increased level of 65 ± 1.5 U/min/mg protein acetylcholinesterase activity compared to control rats which showed 30 ± 2 U/min/mg protein acetylcholinesterase activity. Pretreatment with Daidzein significantly inhibited the elevation of acetylcholinesterase activity to 54 ± 1.7 U/min/mg protein in 25 mg Daidzein-treated rats and to 36 ± 2.8 U/min/mg protein (Figure 2).
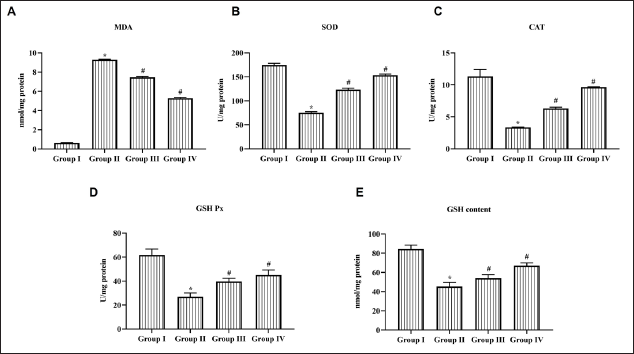
Daidzein Reduced Lipid Peroxidation and Increased Antioxidants in Cerebral I/R Injured Rats
Figure 3 represents the levels of lipid peroxidation and antioxidant in cerebral I/R injured rats. Ischemia followed by reperfusion increased the levels of malondialdehyde to 9.3 ± 0.3 nmol/mg protein, whereas sham-operated control showed only 0.7 nmol/mg protein of malondialdehyde. Daidzein treatment significantly decreased the malondialdehyde levels to 7.2 ± 0.4 nmol/mg protein and 5.8 ± 0.1 nmol/mg protein in 25 and 50 mg/kg treatment, respectively. Pretreatment with Daidzein inhibited the decline in antioxidants SOD, catalase, glutathione peroxidase, and glutathione in a dose-dependent manner. Cerebral I/R induction significantly decreased the level of antioxidants.
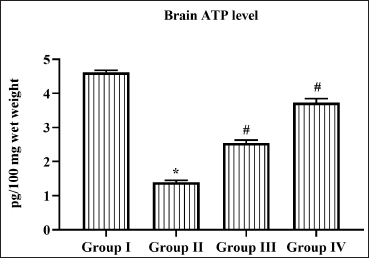
Daidzein Stimulated the Brain ATP Levels in Cerebral I/R Injured Rats
Cerebral ischemia followed by reperfusion impaired the mitochondrial synthesis of ATP in brain tissue. The ATP levels were significantly decreased to 1.3 ± 0.08 pg/100 mg wet tissue in untreated cerebral I/R injured rats. Both doses of Daidzein treatment prevented the decline in ATP levels in cerebral I/R injured rats; 2.3 ± 0.02 pg/100 mg wet tissue and 3.7 ± 0.04 pg/100 mg wet tissue ATP levels were observed in 25 and 50 mg/kg Daidzein-treated cerebral I/R injured rats (Figure 4).
Daidzein Decreased the Level of the Inflammatory Cytokine in Cerebral I/R Injured Rats
Figure 5 depicts the levels of proinflammatory cytokine TNF-α, IL-6, and anti-inflammatory cytokine IL-10 in cerebral I/R injured untreated and Daidzein pretreated rats. Cerebral I/R induction significantly increased the levels of TNF-α and IL-6 to 78 ± 6 pg/mL and 310 ± 10 mg/g protein, respectively. It also decreased IL-10 cytokine level to 280 ± 7 mg/g protein. Daidzein treatment significantly inhibited the synthesis of TNF-α and IL-6 in a dose-dependent manner. Both 25 and 50 mg/kg bwt Daidzein treatment increased the levels of IL-10 to 295 ± 3 mg/g protein and 312 ± 5 mg/g protein, respectively.
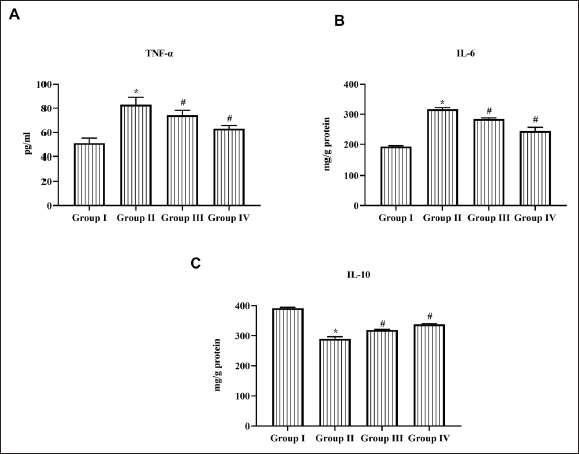
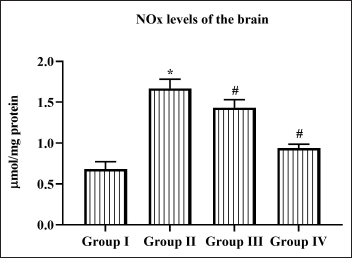
Daidzein Reduced the NO Level in Cerebral I/R Injured Rats
Figure 6 depicts the levels of NO in cerebral I/R injured untreated and Daidzein pretreated rats. NO levels were significantly increased to 1.7 ± 0.05 µmol/mg protein compared to sham control rats which exhibits 0.6 ± 0.01 µmol/mg protein of NO. Compared to 25 mg Daidzein pretreated rats, 50 mg Daidzein treated showed a decreased level of 0.8 ± 0.01 µmol/mg protein of NO.
Daidzein Decreased the Brain NF-κB p65 Level in Cerebral I/R Injured Rats
NF-κB is ubiquitously expressed in all the brain cells and performs various functions they act as a molecular switch in controlling innate inflammation. Cerebral I/R injured rats significantly increased the NF-κB p65 to 6.2 ± 0.02 pg/100 mg wet tissue compared to control (3.8 ± 0.03 pg/100 mg wet tissue); 50 mg/kg Daidzein treatment significantly decreased NF-κB p65 to 4.8 ± 0.01 pg/100 mg wet tissue compared to 25 mg/kg Daidzein treatment (5.7 ± 0.02 pg/100 mg wet tissue) (Figure 7A).
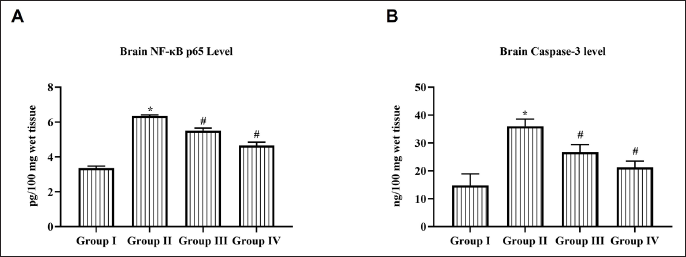
Daidzein Decreased the Caspase-3 Activity in Cerebral I/R Injured Rats
Caspase 3 is a proteolytic enzyme which plays a crucial role in the apoptotic signaling pathway. Figure 7B depicts the results of caspase 3 activity in cerebral I/R injured untreated and Daidzein-treated rats. Caspase 3 activity was significantly increased in cerebral I/R injured rats (37 ± 0.3 ng/100 mg wet tissue) compared to control (14 ± 0.2 ng/100 mg wet tissue). A significant reduction in caspase 3 activity was observed in both 25 mg/kg Daidzein treated (28 ± 0.4 ng/100 mg wet tissue) and 50 mg/kg Daidzein-treated rats (17 ± 0.2 ng/100 mg wet tissue).
Discussion
Epidemiological findings recommend isoflavones intake lowers the risk of cardiovascular diseases, cancer, osteoporosis, 31 neurological, 8 and various other age-related diseases.9, 32 It is proven to be an alternative drug to treat hormonal disorders in women. 33 Intake of isoflavone during childhood prevents breast cancer incidence in later years of life—Clark. 34 Daidzein is one such isoflavone found in soy products. Daidzein is found as glycoside, acetyglycoside, and aglycone conjugates. 35 In this study, we have analyzed the role of daidzein in cerebral ischemia reperfusion-induced mice.
Various mechanisms of action have been proposed for the induction of cerebral ischemia including oxidative stress, ATP depletion, mitochondrial dysfunction, altered calcium homeostasis, glutamate excitotoxicity, inflammation, and neuronal apoptosis.14, 36, 37 In this study, we have induced cerebral ischemic injury in rats using MCAO model which is a well-established model and also resembles the cerebral ischemic condition in humans. 38 Neuroinflammation and increased neuronal deficit scores were reported in the untreated MCAO injured rats.
The life-threatening complication of cerebral ischemia is cerebral edema which causes 5% mortality in stroke patients. 39 Cerebral edema occurs due to blood–brain barrier disruption causing infiltration and leading to increased intracranial pressure.40, 41 Therefore, treating cerebral edema is a critical step in preventing the complications induced by ischemic condition. Daidzein treatment significantly decreased the percentage of water content in the brain of MCAO injured thereby decreasing the cerebral infarct percentage and neuronal deficiency in rats.
Treating neurodegenerative diseases with acetylcholinesterase inhibitors has proven to render beneficial effects and it has been approved to treat diseases including Alzheimer’s and dementia.42, 43 Acetylcholinesterase inhibitors inhibit inflammatory cytokines protection thereby defending endothelial cells from ischemic insults.44–46 Daidzein acts as a potent acetylcholinesterase inhibitor which has significantly decreased the levels of acetylcholinesterase in MCAO injured rats. This may be the reason for the decreased cerebral infarct percentage in pretreated daidzein MCAO injured rats. The interrupted blood during severe ischemic conditions causes hypoglycemia and hypoxia condition in neuronal cells which leads to a decrease in ATP generation which in turn leads to neuronal apoptosis. 47 The marked reduction of ATP occurs during the initial 5 min after the induction of arterial occlusion. 48 Daidzein treatment significantly prevented mitochondrial dysfunction thereby increasing the ATP synthesis in MCAO injured rats.
Oxidative stress has been implicated as a prime factor in the induction of apoptosis during the ischemic condition and it significantly triggers apoptosis on reperfusion. Free radicals superoxide and NO generate peroxynitrite which plays a key role in ischemic-induced inflammation.49–51 Mice with SOD deficient gene depict larger cerebral infarct after ischemic insult compared to the wild-type mice. 52 Inhibition of NO synthase in cultured neuronal cells has prevented the induction of apoptosis. 53 Peroxynitrite disrupts the cell membrane and promotes lipid peroxidation in brain tissue.54, 55 Daidzein treatment elevated the SOD and catalase levels in MCAO injured thereby preventing lipid peroxidation. It also significantly decreased the levels of NO which is the key initiator of neuronal apoptosis. Daidzein increased the glutathione and glutathione peroxidase levels in MCAO injured rats, which are reported to decrease in both ischemic and reperfusion. 56 Overall antioxidant status of daidzein-treated MCAO injured rats confirms the daidzein potency of free radical scavenging and preventing cerebral infract.
NF-κB proteins consist of Rel protein dimers which include RelA – p65, p50, p52 subunits, Rel B, and RelC. RelA p65 and p50 heterodimers are ubiquitously expressed in both neuronal and glial cells. During the normal condition, inactive NF-κB dimers bounds with IκB and retains in the cytoplasm, whereas on neuronal activation, it translocate to the nucleus and contributes to neuronal apoptosis.57, 58 During ischemic conditions, the NF-κB p50 subunit deficit mice expressed decreased levels of cerebral infarct compared to wild mice. M1 microglial cells trigger the synthesis of IL-1β, TNF-α, and free radicals which exacerbate cerebral infarct, 59 whereas the M2 microglial cells activate the release of anti-inflammatory cytokines Il-10 and alleviate the ischemia inducing infract.60, 61 The synthesis of proinflammatory cytokines activates astrocytes there triggers secondary neurotoxicity and neuronal apoptosis. NF-κB inhibition has shown promising effects in preventing the synthesis of inflammatory cytokines and neuronal apoptosis. In this study, daidzein significantly inhibited the synthesis NF-κB p65 subunit which in turn reduced proinflammatory cytokine IL-6, TNF-α, and anti-inflammatory cytokine IL-10. It also decreased proteolytic enzyme executor Caspase-3 confirming the inhibition of neuronal apoptosis in MCAO-induced rats.
Conclusion
Nutraceuticals are potent and safe alternative drugs which have already attracted the global pharmaceutical market. In this study, we have evaluated the anti-ischemic role of phytoestrogen isoflavone in a cerebral ischemic-reperfusion-induced rat model. Pretreatment with daidzein significantly prevents the free radical generation and decline in the antioxidant status of MCAO injured rats. It significantly decreased the levels of acetylcholinesterase and NO levels and increased the brain ATP suggesting its neuroprotective role. It also inhibited the levels of NF-κB thereby preventing neuroinflammation and neuronal apoptosis. Our analysis of daidzein role in MCAO injured rats confirms daidzein is a potent anti-ischemic drug which significantly prevents post-ischemic complications in MCAO injured rats. Additional studies are still recommended in the future to clearly understand the therapeutic roles of daidzein on ischemic stroke.
Footnotes
Abbreviations
MCAO: Middle cerebral artery occlusion; ANOVA: One-way analysis of variance; SOD: Superoxide dismutase; NO: Nitric oxide; NF-κB: Nuclear factor-kappa B.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Laizhou City people’s Hospital, No.1718, Wuli street, Laizhou City, Shandong Province, 261400, China.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Summary
Daidzein treatment significantly prevented brain edema and cerebral infarction thereby decreasing neurological deficit in cerebral I/R injured rats.
Pretreatment with daidzein significantly prevents the free radical generation and decline in antioxidants status of MCAO injured rats.

