Abstract
Background
Ketamine (KT) is known to have analgesic and sedative effects. Intranasal (IN)/inhalational KT has been used in different trials involving paediatric patients for analgesic and anxiolytic function. The present meta-analysis was conducted to establish the role of IN/inhalational KT compared to that of inhalational opioids (OPs).
Summary
A systematic literature search was performed through the Cochrane Library, Pub Med and
Key Message
KT had efficacy outcomes comparable to those of OP, but KT had very high adverse effects. OP seems to have better tolerability than KT. However, as the number of patients was less than the RIS, it was not possible to draw any conclusions.
Introduction
Effective pain management is crucial in the paediatric population, as uncontrolled pain can lead to adverse physiological consequences, impaired healing and long-term psychological effects.1, 2 However, achieving adequate analgesia, particularly in acute settings, remains a significant challenge for both adults and children.3, 4 In current practice, opioid analgesics (OPs) are commonly used as the standard of care (SoC) for severe acute pain in children. Despite their efficacy, OPs are associated with adverse effects such as respiratory depression, nausea, vomiting and the risk of addiction.5, 6 Furthermore, obtaining intravenous (IV) access to administer parenteral OPs can be challenging, time-consuming and distressing for children.7, 8 Rescue analgesics are often required to manage breakthrough pain or insufficient response to initial analgesic regimens, further complicating pain management strategies.
Ketamine (KT), a dissociative anaesthetic and N-methyl-D-aspartate (NMDA) receptor antagonist has emerged as a promising alternative to OPs for acute pain management in children. At sub-dissociative doses (0.1–0.4 mg/kg IV), KT provides potent analgesia with minimal respiratory depression compared to OPs9, 10; it also exhibits anti-inflammatory and anti-hyperalgesic effects, helping to prevent central sensitisation and OP-induced hyperalgesia. 11
While IV KT is effective, its administration requires IV access, which can be challenging in children. Alternative routes such as intranasal (IN), nebulised or inhalational delivery offer non-invasive options for KT administration, potentially simplifying the process and improving the patient experience.7, 12 Several studies have evaluated the efficacy and safety of IN KT versus IN OPs, for example, fentanyl, for acute pain management.13–16 However, the role of IN, nebulised or inhaled KT as a first-line analgesic versus an adjunct to OPs remains unclear.
Accurate assessment of pain in children is crucial for effective management. A variety of self-reported pain scales, including the faces pain scale-revised (FPS-R), visual analogue scale (VAS) and numeric rating scale (NRS), have been validated for use in the paediatric population.17, 18 The selection of an appropriate scale depends on factors such as the child’s age, cognitive development and clinical context. For instance, the FPS-R and other facial scales are commonly used in younger children, while the NRS or VAS may be more suitable for older children and adolescents.19, 20 Behavioural observation scales, such as the FLACC (face, legs, activity, cry, consolability) scale, can also be useful, particularly in nonverbal or cognitively impaired children. Additionally, sedation levels must be monitored to ensure safety during analgesic administration. The University of Michigan Sedation Scale (UMSS) is commonly employed to assess sedation depth, enabling timely detection of oversedation and facilitating appropriate management. It is essential to choose a developmentally appropriate and validated pain assessment tool to accurately quantify pain intensity and guide treatment decisions.18, 21
The aim of this systematic review and meta-analysis was to comprehensively evaluate and integrate findings from randomised controlled trials (RCTs) concerning the efficacy, safety and tolerability of IN, nebulised or inhalational KT administration, relative to equivalent routes of OP delivery, in achieving analgesia and sedation in paediatric population across diverse clinical contexts.
Methods
Search Strategy and Study Selection
Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2020 guidelines were used to conduct the present systematic review and meta-analysis.
22
The MEDLINE, Clinical Trial Registry (
Data Extraction and Risk of Bias Assessment
The review authors extracted all the information about the studies relevant for meta-analysis, that is, demographic information, interventions (dose and route), indications and country of trial, from the published articles. Additional sources of information, such as protocols, statistical analysis plans and other reviews, were also accessed for required information. The authors applied both tools to assess the risk of bias (RoB).23, 24 The older RoB tool was used to prepare the summary of findings (SoF), whereas the RoB2 tool available since 2020 is currently used to assess the RoB in the included studies. 24 There are six domains in the older tool, that is, selection bias, performance bias, detection bias, attrition bias and reporting bias, whereas the RoB2 tool has five domains, including bias due to the randomisation process, bias due to deviations from the intended interventions, bias due to missing outcome data, bias in the measurement of the outcome, and bias in the selection of the reported result. Funnel plots and Egger’s test were used to assess publication bias. A one-tailed P < .05 indicated publication bias. The SoF table was prepared by using the Grades of Recommendation Assessment, Development and Evaluation (GRADE) approach, which includes the overall RoB, inconsistency, indirectness, imprecision and publication bias. Subsequently, the quality of the evidence was rated as ‘high’, ‘moderate’, ‘low’ or ‘very low’. 25
Outcomes
The efficacy and safety outcomes are as follows:
Proportion of patients who achieved a clinically significant pain reduction (defined as a >20 mm decrease in the VAS). Proportion of patients requiring rescue analgesics. Proportion of patients who achieved mild to moderate sedation (defined according to the UMSS) with scores of 1 and 2. Mean change in pain score after 30 minutes of administration. Proportion of patients with any adverse events (AEs). Proportion of patients with nausea and/or emesis. Proportion of patients with dizziness. Proportion of patients with an unpleasant taste.
Data Analysis
The present meta-analysis was performed using both fixed-effect and random-effects models; however, the data were analysed with only a random-effects model. Continuous and dichotomous data are presented as standardised mean differences (SMDs) and risk ratios (RRs), respectively, with 95% confidence intervals (CIs). The heterogeneity was assessed by Cochrane’s Q and I 2 statistics. A P value of .10 was considered significant for heterogeneity. 26 The meta-analysis was carried out with Review Manager 5.4 for Windows. For all the statistical tests, P < .05 was considered significant. Furthermore, the authors also performed trial sequential analysis (TSA) using the TSA 0.9.5.10 beta version. 27 The tool provides the required information size (RIS), which indicates the threshold for adequate sample size for a statistically significant treatment effect considering heterogeneity among included studies. The sample size was used as a measure for the RIS. For all endpoints, the type I error and power were 5% and 80%, respectively. The expected RR reduction was set at 20%, and the mean difference was calculated based on low bias. The variance and incidence in the control arm were calculated based on the data included in the studies, and the heterogeneity correction was model variance-based. Z-curves crossing sequential boundaries with the achievement of an RIS were considered a significant intervention effect. 28 In a few studies, continuous data are not presented as the mean ± SD; hence, the medians with ranges/interquartile ranges or CIs were converted into standard deviations by using different formulae.29, 30
Results
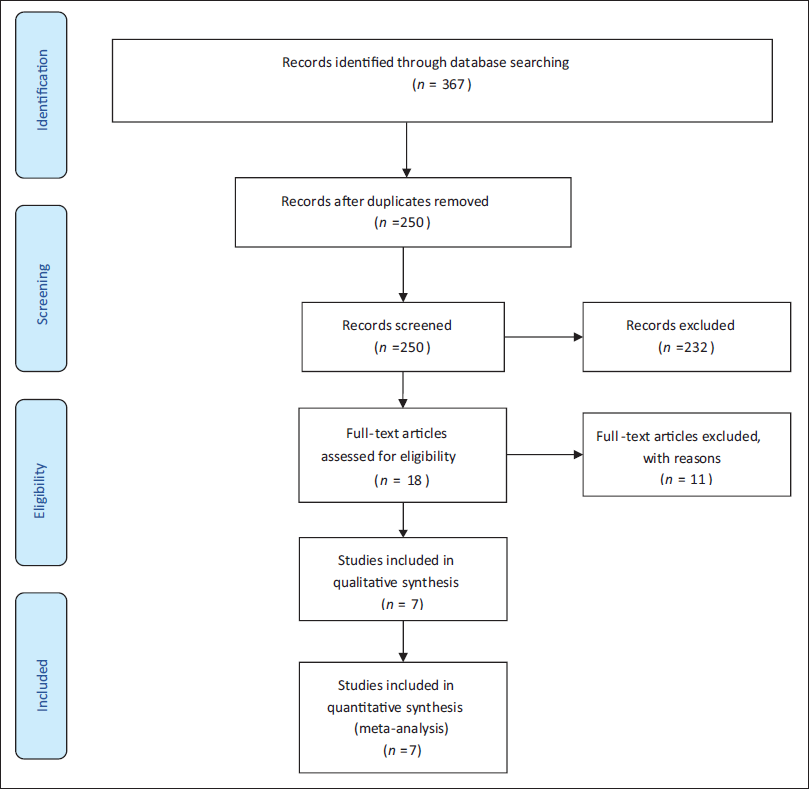
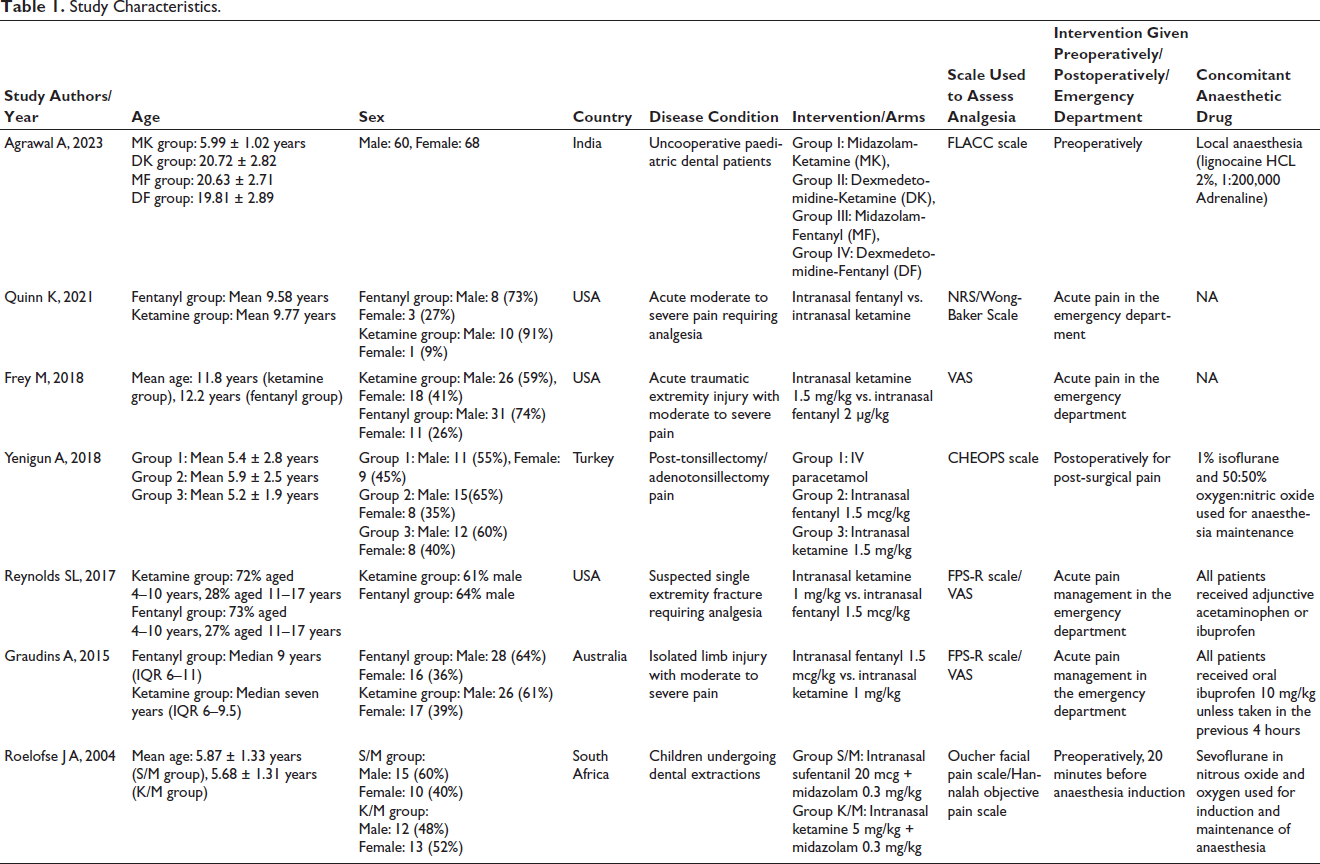
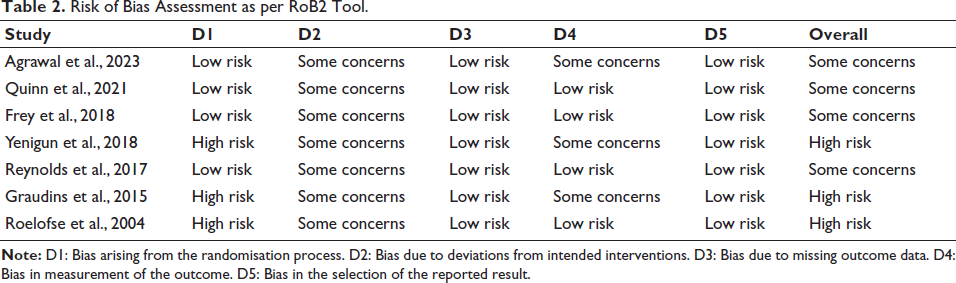
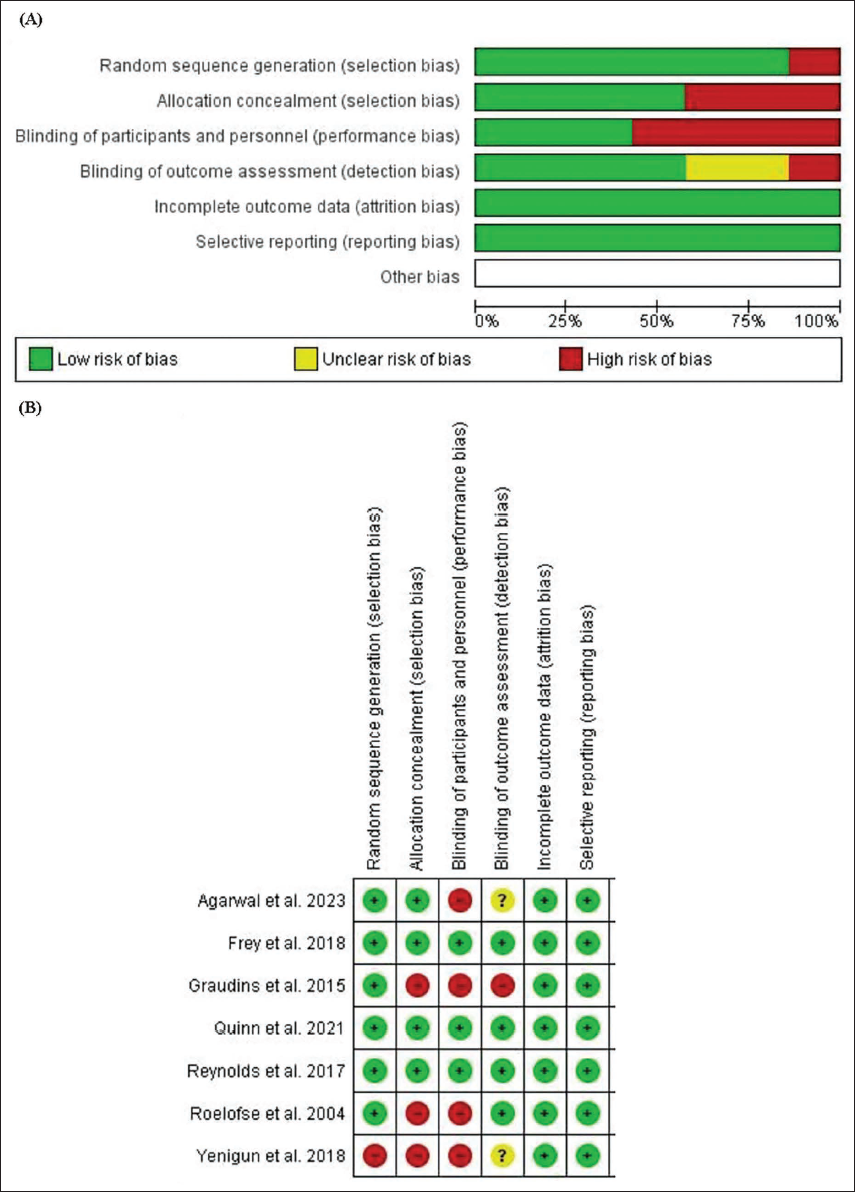
Upon systematic search, initially, a total of 367 abstracts from various databases were found. Out of 367 articles, 117 were excluded because they were duplicates; subsequently, a total of 250 articles were assessed for eligibility. Of the 250 articles, the full texts of 18 articles were assessed, and 11 were excluded. The reasons for exclusion were RCTs involving the adult population (01), a different SoC/no OP in the comparator arm/different route of administration of KT (09), and the use of esketamine (01). Eventually, 07 RCTs were selected for carrying out a meta-analysis (Figure 1).31–37 The current meta-analysis included 07 RCTs published from inception to February 2024 including 489 patients aged <18 years who required analgesia and who received IN KT or OP (fentanyl/sufentanil). The patients also received other drugs as a part of their SoC. Among the seven studies, four compared KT to OPs for analgesia in the emergency department.32, 33, 35, 36 Most of the patients were male (62%). In the included RCTs, IN KT and OPs were administered in addition to SoC,31, 34–37 and in the other two studies, no other concomitant drug was administered.32, 33 The SoC was not uniform and varied according to the indications mentioned in Table 1. KT and OPs were administered intranasally according to patient weight. The doses of KT and fentanyl were 1–4 mg/kg and 2 µg/kg, respectively. Only one study used sufentanil. 37 According to the RoB2 tool, three RCTs were at high risk34, 36, 37 (Table 2; Figure 2).
Flow Diagram of Study Selection Process.
Study Characteristics.
Risk of Bias Assessment as per RoB2 Tool.
(A) Risk of Bias Graph for Included Studies. (B) Summary of Risk of Bias for the Included Studies.
Efficacy Endpoints
The RCTs had two groups, that is, whether the group received KT or OP. For each outcome, publication bias was assessed by funnel plots and Egger’s test. All funnel plots are presented in the supplementary files (S2). For the efficacy endpoint, that is, the proportion of patients achieving clinically significant pain reduction, the funnel plot obtained was symmetrical, suggesting no publication bias, while Egger’s test could not be performed because the number of studies was <3. A forest plot indicated mild heterogeneity (I 2 = 26%, P = .25) (Figure 3). A slightly greater percentage of patients achieved the endpoint in the OP group than in the KT group, but the difference was not statistically significant (82.6% vs. 77.3%; RR = 0.94; 95% CI = 0.78–1.13; P = .52). For another endpoint, the proportion of patients requiring rescue analgesics, the funnel plot indicated no publication bias (P = .16). There was slight heterogeneity observed (I 2 = 26%, P = .26) (Figure 4). The need for rescue analgesics was lower in the KT group than in the OP group (17.2% vs. 21.6%), but this difference was not statistically significant (RR = 0.80; 95% CI = 0.44–1.43; P = .45).
Forest Plot for Pain Responders.
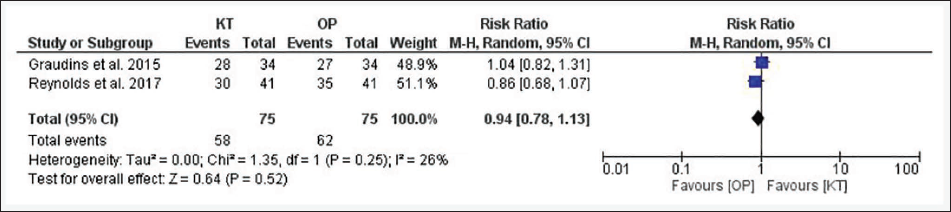
Forest Plot for Patients Requiring Rescue Analgesia.
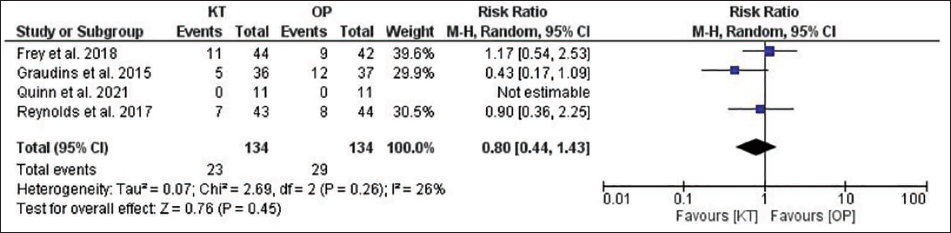
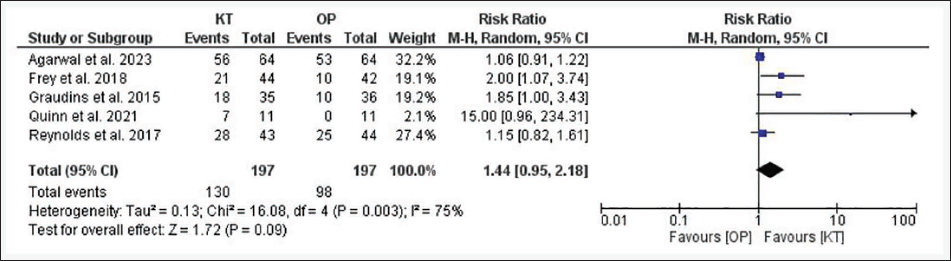
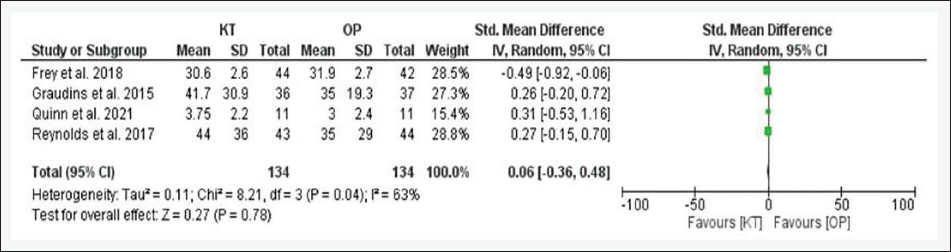
For another endpoint, that is, the proportion of patients who achieved mild to moderate sedation, publication bias was observed (P = .004), and significant heterogeneity was noted, that is, I 2 = 75%, P = .003. A greater number of patients in the KT group achieved mild to moderate sedation than did those in the OP group (66% vs. 49.7%), but this difference was not statistically significant (RR = 1.44; 95% CI = 0.95–2.18; P = .09) (Figure 5). Similarly, for the endpoint mean change in pain score after 30 minutes of administration, no publication bias was detected, and significant heterogeneity was noted (I 2 = 63%, P = .04). The KT group had slightly greater pain reduction than the OP group, but the difference was not statistically significant (SMD = 0.06; 95% CI = −0.36 to 0.48; P = .78) (Figure 6). The estimates of different endpoints did not change even after applying a fixed-effects model, except for the proportion of patients who achieved mild to moderate sedation, which became statistically significant (RR = 1.33; 95% CI = 1.13–1.56; P = .0004).
Forest Plot for Sedation.
Forest Plot for Change in Pain Score.
Safety Endpoints
A greater number of patients experienced any AEs in the KT group than in the OP group (58.2% vs. 28.2%), and this difference was statistically significant; RR = 1.99; 95% CI = 1.47–2.69; P < .00001 (Figure 7). The funnel plot was asymmetrical and suggested publication bias (P = .04), and mild heterogeneity was observed (I 2 = 36%, P = .18). The proportion of patients with nausea and/or emesis was greater in the KT group than in the OP group (5.6% vs. 2.2%), but the difference was not statistically significant; RR = 2.19; 95% CI = 0.79–6.09; P = .13 (Figure 8). The funnel plot was asymmetrical and was indicative of publication bias (P = .02), and the review authors did not observe any heterogeneity (I 2 = 0%, P = .57). There were significantly more patients who experienced dizziness in the KT group than in the OP group (50% vs. 8.4%; RR = 5.47; 95% CI = 3.12–9.58; P < .00001) (Figure 9). The funnel plot was asymmetrical, and publication was suggested (P = .03), with no heterogeneity (I 2 = 0%, P = .83). More patients felt an unpleasant taste in the KT group than in the OP group (52.1% vs. 17.5%), and the effect was statistically significant (RR = 2.91; 95% CI = 1.51–5.61; P = .001) (Figure 10). The funnel plot was symmetrical, and no publication bias was observed (P = .44). This endpoint had moderate heterogeneity (I 2 = 52%, P = .13). The estimates of safety were similar after applying the fixed-effects model, and no significant change was observed.
Forest Plot for Any Adverse Event.
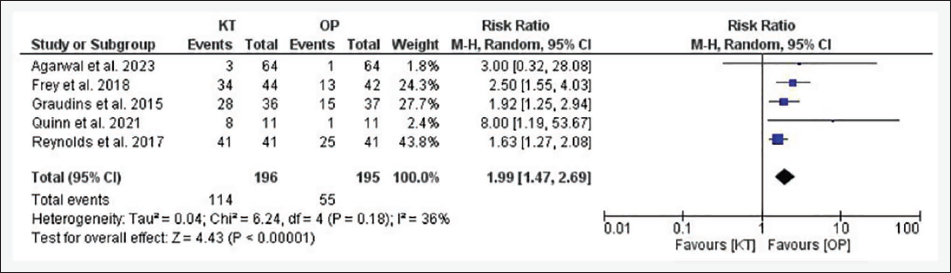
Forest Plot for Nausea.
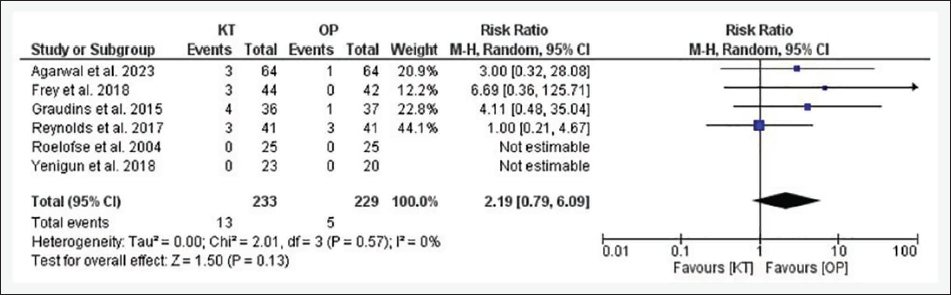
Forest Plot for Dizziness.
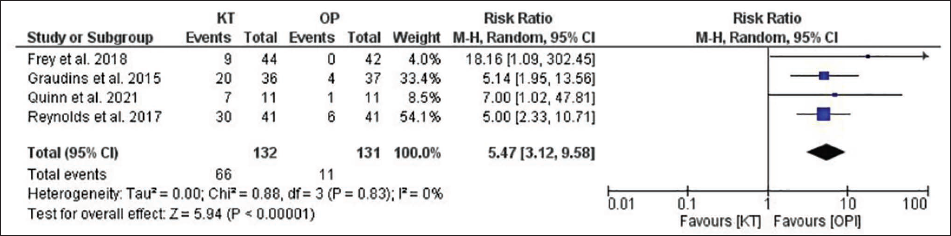
Forest Plot for Unpleasant Taste.
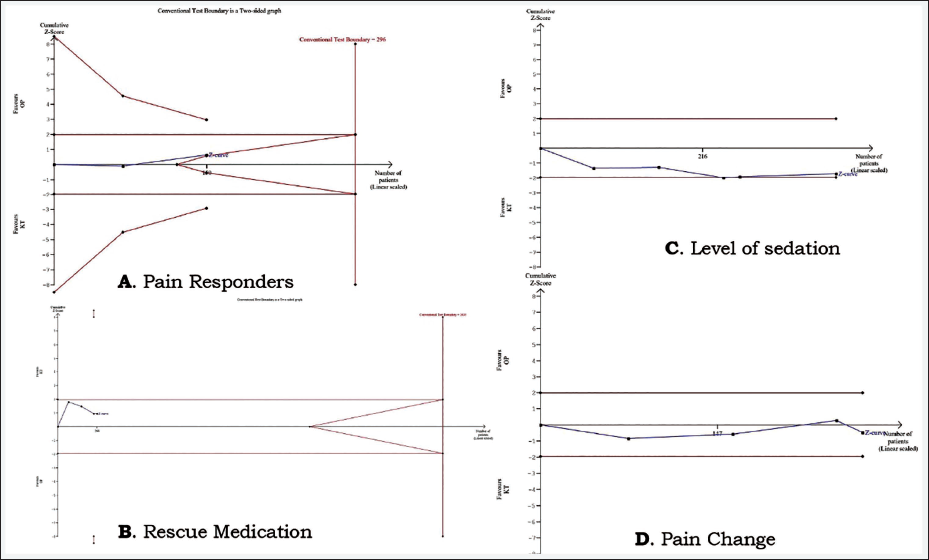
The information size obtained for numerous endpoints was compared with the RIS obtained by TSA. In the present review, no efficacy endpoint could be reached for RIS (Figure 11). The Z-curves obtained did not cross the trial sequential boundary for any outcome. According to the GRADE approach, the overall certainty of the evidence was very low for all endpoints except for the use of rescue analgesia, and for any AEs, the certainty of the evidence was low. The detailed estimates are presented in Table 3.
(A) Pain Responders. (B) Rescue Medication. (C) Level of Sedation. (D) Pain Change.
Summary of Findings.
aThe study Graudins et al. is at significantly high risk of bias.
bBoth boundaries of confidence interval do not lie on one side of the threshold, optimal information size criterion is not met.
cThree out of four studies were at low risk of bias, hence a majority of estimates came from low risk of bias.
dFour out of five studies are at low risk of bias.
eSignificant heterogeneity observed, that is, I 2 = 75%; P = .003
fThe funnel plot as well as Egger’s test (P = .004) strongly suggest publication bias.
gSignificant heterogeneity observed, that is, I 2 = 73%; P = .01.
hOptimal information size criteria not met.
iThe funnel plot as well as Egger’s test (P = .04) strongly suggest publication bias.
jThe funnel plot as well as Egger’s test (P = .02) suggest publication bias.
kVery wide confidence interval, OIS not met.
lThe funnel plot as well as Egger’s test (0.03) suggest publication bias.
mThe I 2 is 52% with a wide variation of CI in included studies.
Discussion
In the present meta-analysis, review authors compared IN KT to IN OP in paediatric patients undergoing painful procedures under different settings. OPs are part of the SoC in paediatric patients for such procedures and have few potential serious ADRs. KT provides a nonopioid alternative for analgesia. In this study, a total of seven RCTs with 489 patients were included, and four studies were included in the previous meta-analysis. 38 The majority of the patients were male, which is similar to the findings of a previous meta-analysis. 38 Among seven studies, three studies were judged to be at high risk of bias34, 36, 37 according to the RoB2 tool, whereas others had some concerns.31–33, 35 The major reason for the high risk of bias was the failure to implement randomisation and/or allocation concealment. Among the included studies, no study provided information about protocol deviation. These were the major shortcomings of the included studies.
In all the efficacy endpoints, KT and OP were comparable, that is, both groups achieved similar analgesia and sedation; however, KT performed better than OP. The use of rescue analgesics was also comparable. Our findings match those of a previous study that reported comparable pain relief. 38 An important meta-analysis that compared KT to the control group also revealed a decrease in pain intensity and a reduction in OP requirements but no opioid-sparing effect. 39 In another meta-analysis, KT did not decrease pain intensity and had no opioid-sparing effect. 40 Moreover, previous meta-analyses showed mixed results for KT’s analgesic effect, with some studies indicating limited efficacy in specific contexts. 41 Considering the findings of previous meta-analyses, the findings of the present analysis should be interpreted cautiously, as the overall effect generated had significant biases, inconsistency, imprecision and publication bias (Table 3). The major reasons for heterogeneity were the wide age range of patients, different geographical locations of clinical trials, different SoCs, variable disease conditions, different timings of administration and different methods of evaluating analgesia. The major reasons for imprecision in the overall estimate were failure to achieve an RIS and inappropriate CIs. These factors led to a reduction in the overall strength of evidence (low/very low). Low certainty was also observed in other studies.40, 41 As the total number of patients was much less than that of the RIS, it is difficult to draw a valid conclusion regarding the efficacy of IN KT. A similar limitation was also observed in a previous study. 40
The patients in the KT group had significantly greater adverse effects (any AE, dizziness and unpleasant taste) than those in the OP group. Nausea/vomiting was also more common in the KT group. The major limitations in these findings are a very wide CI, a smaller number of patients compared to the RIS, and significant publication bias. A very wide CI in the included studies made the results less accurate. Despite these limitations, significantly more AEs, especially nausea/vomiting, and unpleasant tastes can drastically reduce the acceptance of KT. These AEs significantly reduce tolerability in paediatric patients. Similar findings regarding safety have also been observed previously. 42 In a previous meta-analysis that evaluated IN KT to SoC in the adult population, the profile of AEs was similar to that in our analysis. 43
Despite these limitations, KT can be used as an alternative to OPs in paediatric patients in certain situations where the patient is known to have adverse effects from OPs, or the patient has developed tolerance. 33 It can also be preferred in patients who are at risk of hypotension or bronchospasm. KT should be avoided in patients prone to myocardial ischaemia. 44 The major limitation of this meta-analysis is that the total number of patients was far less than the number of patients included in the RIS. It is not possible to draw a sound conclusion.
Conclusions
With these limited data, KT and OP administered intranasally seem to be comparable in attaining analgesia in paediatric patients. The most important limitation of KT use is AEs, which are much more common than those associated with OP. As pain was measured by different scales, a uniform scale in clinical trials will help in generating good quality data. Moreover, to achieve an RIS and certainty of evidence, more well-designed, adequately powered clinical trials are indispensable.
Footnotes
Abbreviations
CENTRAL, Cochrane Central Register of Controlled Trials; CIs, Confidence intervals; FPS-R, Faces Pain Scale-Revised; FLACC, Face, Legs, Activity, Cry, Consolability; GRADE, Grades of Recommendation Assessment, Development and Evaluation; IN, Intranasal; IV, Intravenous; KT, Ketamine; NMDA, N-methyl-D-aspartate; NRS, Numeric Rating Scale; OPs, Opioids; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-analyses; RIS, Required Information Size; RoB, Risk of Bias; RR, Risk ratio; SMDs, Standardised mean differences; SoF, Summary of Findings; TSA, Trial Sequential Analysis; UMSS, University of Michigan Sedation Scale; VAS, Visual Analogue Scale.
Authors’ Contribution
All authors framed the concept. Search and meta-analysis were performed by MPS and AS. AS and MPS drafted the manuscript. AS and EK critically evaluated the manuscript and all authors approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Ethics
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
