Abstract
Background
The hypothalamus, a small yet crucial neuroanatomical structure, integrates external (e.g., environmental) and internal (e.g., physiological/hormonal) stimuli. This integration governs various physiological processes and influences cognitive, emotional, and behavioral outcomes. It serves as a functional bridge between the nervous and endocrine systems, maintaining homeostasis and coordinating bodily functions.
Summary
Recent advancements in the neurobiology of the hypothalamus have elucidated its functional map, establishing a causal relationship between its responses—such as respiration, sleep, and stress—and various physiological processes. The hypothalamus facilitates and coordinates these complex processes by processing diverse stimuli, enabling the body to maintain internal balance and respond effectively to external demands. This review delves into the hypothalamus’s intricate connections with cognition, emotion, and physiology, exploring how these interactions promote overall well-being and adaptability.
Key Message
Targeted external stimuli can modulate hypothalamic neuronal activities, impacting the physiological, cognitive, and emotional landscape. The review highlights non-invasive techniques, such as controlled breathing exercises, optimized sleep architecture, and stress management, as potential methods to enhance hypothalamic function. Ultimately, this comprehensive review underscores the multifaceted role of the hypothalamus in integrating signals, maintaining homeostasis, and influencing cognition, emotion, and physiology.
Introduction
Hypothalamus is a small, pea-sized neuroanatomical structure located deep within the brain, specifically in the lower central part of the diencephalon. It is an essential constituent of the complex brain and operates as a silent sentinel, governing our primal impulses and intricate physiological functions. In essence, the hypothalamus serves as a vital intermediary connecting the nervous and endocrine systems and is pivotal in preserving homeostasis and overseeing diverse physiological processes. In recent decades, technological advancements in functional neurobiology, such as perturbation-based functional assays, electrophysiology, optogenetics, neuronal tracings, behavioural readouts and others, have facilitated the gaining of profound depths in the functional space of this unique anatomical structure. It is now established that the hypothalamus serves as a critical hub for maintaining the body’s internal balance, coordinating various physiological processes, integrating signals from both the external environment and internal body systems, shaping our experiences, decision-making and the delicate equilibrium between our primal instincts and multifaceted behaviours (Figure 1). From a functional perspective, the hypothalamus is recognised as the primary regulator of the pituitary gland, exerting direct control over numerous systems within our body, such as the nervous, endocrine and respiratory systems (Figure 2). Being an ultracomplex structure, the hypothalamus can be influenced by various endogenous and exogenous factors, including genetic, environmental and lifestyle factors. Breathing techniques, such as deep diaphragmatic breathing, can help regulate the autonomic nervous system, which indirectly influences hypothalamic activity and helps modulate stress responses. Similarly, sleep cycles also indirectly influence hypothalamic activities by modulating various physiological processes and neurochemical signalling. Consequently, by mastering breathing dynamics, fostering healthy sleep patterns and adeptly managing stress responses, it is feasible to establish a harmonious resonance within the intricately orchestrated hypothalamic symphony. The existing evidence for hypothalamic functions opens up possibilities for more innovative interventions and therapies to promote optimal well-being and enhance cognitive abilities. Unfortunately, only little supporting evidence is available in the biomedical literature that comprehensively explains the direct causal relationship between these vital physiological processes and the hypothalamus. It, therefore, demands an urgent need for extensive sample-size prospective studies for further strengthening the link between these vital physiological processes and hypothalamic functions. While the pharmacological interventions directly targeting the hypothalamus offer an alternative approach to modulate its activity and hence its associated functions, the existing solutions harbour significant side effects, suggesting the need for non-invasive techniques or methods.
Functional Map of the Hypothalamus, Depicting Distinct Subparts (Nuclei) of the Hypothalamus and Their Associated Known Functions.
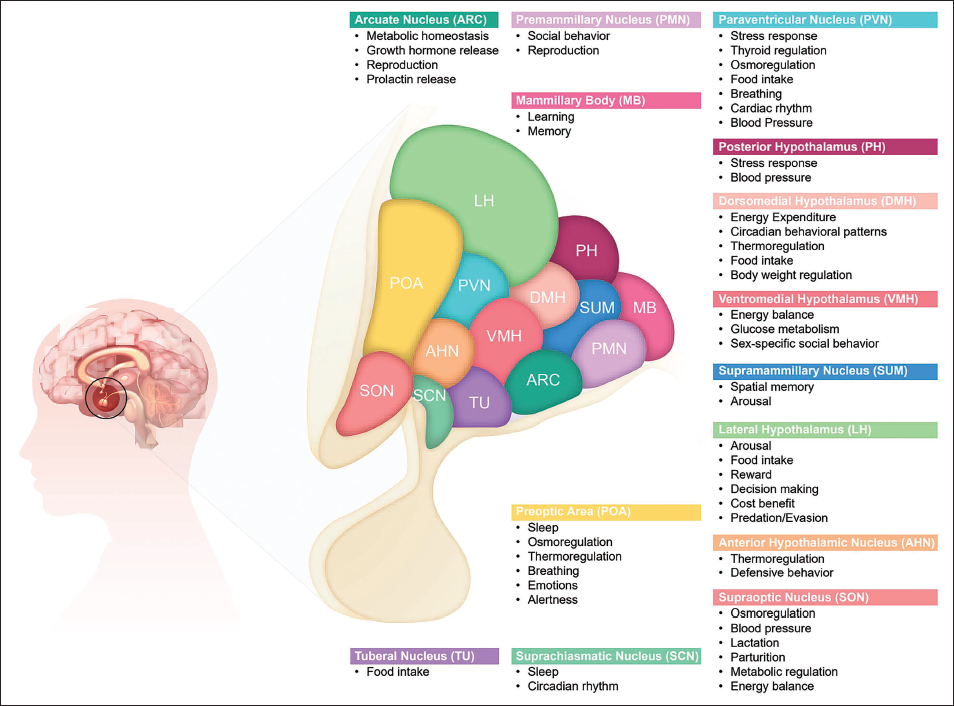
Hypothalamus: The Master Regulator of the Key Physiological Processes.
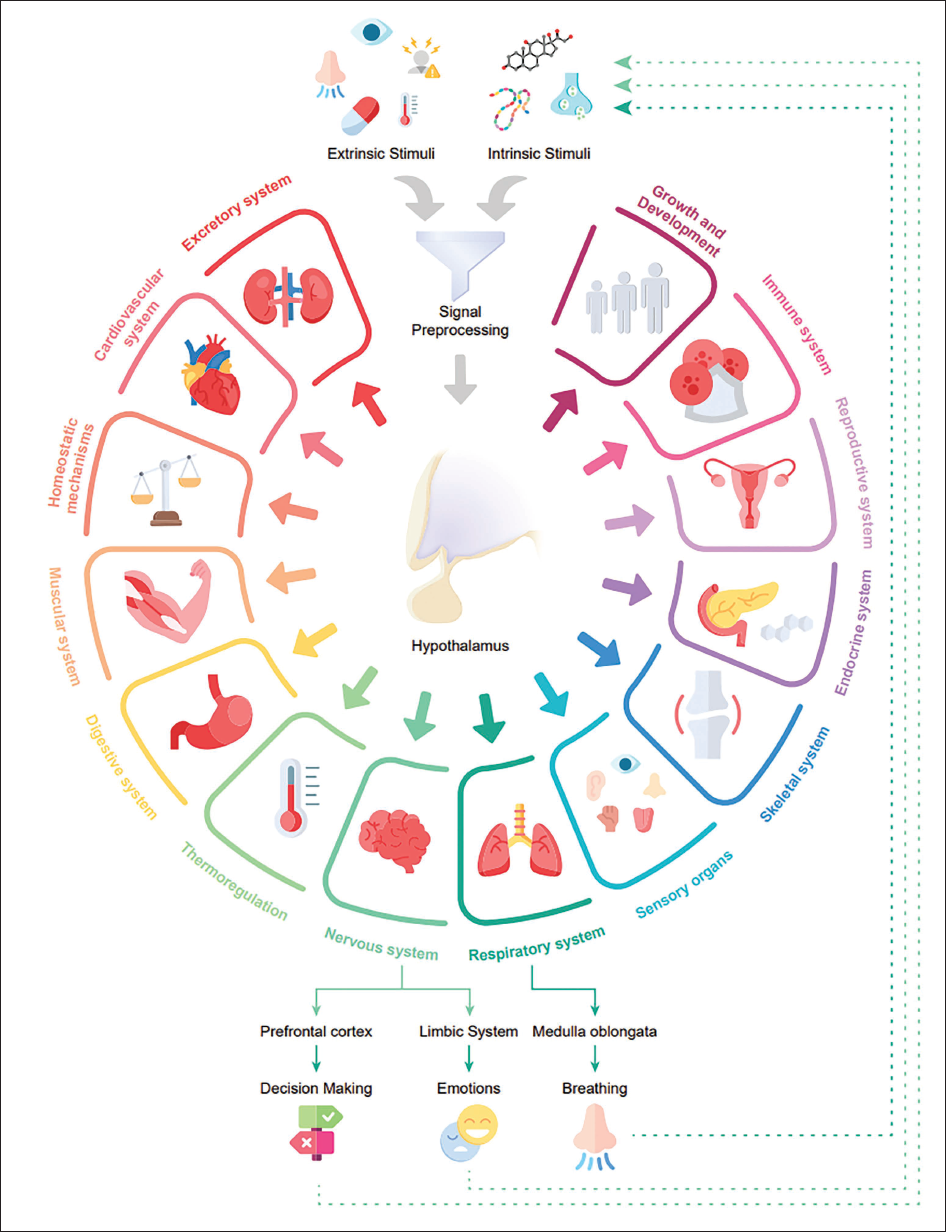
In this review article, we comprehensively discuss and highlight the hypothalamic functions and how they regulate cognition, emotion and body physiology. In particular, we discuss the varied intrinsic and extrinsic stimuli that trigger hypothalamic responses and their interplay. Moreover, we also discuss the existing literature about the functional regulation of various physiological processes by the hypothalamus and how it can be manipulated via a feedback mechanism using controlled breathing exercises, fostering optimal sleep architecture and adeptly navigating stress, a necessity for maintaining overall health and well-being.
Intrinsic Factors in Hypothalamic Regulation and Neurochemical Modulation
Holistic comprehension of the intricate operational mechanisms of the hypothalamus, an enigmatic neuroanatomical structure, demands multifaceted experimentally validated shreds of evidence, including both the loss and gain of function assays. Decades of research has identified a myriad of extrinsic and intrinsic stimuli that singly or synergistically contribute to the orchestrations of hypothalamic functionality. It is, therefore, indispensable to delve deeper into the complicated dynamics governing the interrelationship between extrinsic and intrinsic stimuli to gain mechanistic insights into the mode of action of hypothalamic neurons. Intrinsic factors encompass neurotransmitters, hormones, circadian rhythms, metabolic state, genetic predisposition, feedback mechanisms, neuropeptides and neuronal circuits. Neurotransmitters such as GABA, serotonin and dopamine are well characterised and have been proven to play a crucial role in regulating hypothalamic functions. For example, Saper and colleagues unveiled that the GABA release from inhibitory neurons in the ventrolateral preoptic nucleus inhibits the arousal system and promotes sleep. 1 Further, Li and colleagues in 2018 identified that upon confrontation with predatory assaults, a cohort of GABAergic neurons within the hypothalamus regions instigates a cascade of aggressive behavioural responses. Conversely, in the context of evasive behavioural responses, glutamatergic neurons seize control, precipitating a repertoire of actions aimed at facilitating escape and ensuring survival. 2 Notably, the delicate balance of dopamine and serotonin systems is known to influence glutamate release from Vglut2 neurons in the hypothalamus, contributing to the induction of aversive response. 3 Integration of the hypothalamus with various brain regions is essential for its multifaceted functions. Karimi et al. conducted a series of behavioural experiments utilising the T-maze task, revealing the interconnectedness between the perifornical area of the lateral hypothalamus (PeF-LH) and the ventral tegmental area (VTA), a region recognised for its involvement in reward processing. 4 The interactions between the hypothalamus, specifically Orx neurons, and the VTA (ventral tegmental area) play a role in decision-making and motivated behaviours. 5 Significantly, the presence of direct neural pathways linking the paraventricular nucleus of the hypothalamus (PVN) with phrenic motor neurons suggests the participation of PVN neurons in the coordination of cardiovascular and respiratory functions.
Hormonal regulation is a critical factor affecting the hypothalamic activity. The hypothalamus acts as a central hub for hormone regulation, secreting, releasing and inhibiting factors that control hormone release from the pituitary gland. The intricacies of elaborate hormonal networks orchestrate key physiological processes encompassing reproduction, stress response, growth and metabolism. Significant disruptions in the regulation of sleep, particularly concerning the hypothalamic–pituitary–adrenal (HPA) axis, have been associated with abnormally elevated levels of adrenocorticotropic hormone (ACTH) and cortisol. These changes have been correlated with the development of chronic insomnia and increased arousal within the central nervous system. 6 Moreover, the lateral hypothalamus, particularly the activity of corticotropin-releasing factor (CRF) neurotransmission, plays a crucial part in regulating the tachycardic response (elevated heart rate) observed in rats during instances of acute emotional stress. By blocking the CRF1 receptors in the lateral hypothalamus, researchers have observed a reduction in stress-induced rapid heart rate, highlighting the involvement of the hypothalamus in regulating cardiovascular responses to stress. 7 The pivotal role of the hypothalamus in stress response involves the secretion of the corticotropin-releasing hormone (CRH), which triggers the activation of the HPA axis. Upon activation, this process leads to the secretion of stress hormones like glucocorticoids, which effectively regulate diverse physiological systems and orchestrate adaptive reactions to stress. 8 Inhibitory neurons in the ventrolateral preoptic nucleus, through the release of GABA, promote sleep and inhibit the arousal system. 1 Studies on the developmental patterns of the sleep–wake cycle in infant rats have emphasised the significance of the hypothalamus. Experimental interventions, such as lesions and surgical manipulations targeted at specific hypothalamic regions, have been found to impact sleep–wake states. 9 Circadian rhythms, internal biological processes following a 24-hour cycle, also significantly influence the hypothalamic function. The lateral hypothalamus interacts with the circadian rhythm to regulate food intake, as demonstrated by Delgado and Anand in their study on electrical stimulation in cats. 10
Neuropeptides assume a pivotal role as intrinsic determinants influencing the intricate activities of the hypothalamus. The hypothalamus exerts its influence on the modulation of alcohol consumption via the actions of neuropeptides, which are specialised neurochemicals instrumental in shaping brain function. Specifically, orexigenic neuropeptides, namely galanin, enkephalin and orexin/hypocretin, exert their effect by augmenting the processes of reward and reinforcement, thereby promoting alcohol-drinking behaviour. Conversely, anorexigenic neuropeptides such as dynorphin, CRF and melanocortins act as inhibitors of alcohol intake, countering the effects elicited by orexigenic neuropeptides and impeding their proclivity for enhancing alcohol consumption. Hence, the dysregulation of neuropeptide signalling can contribute to excessive alcohol consumption. 11 Moreover, orexin neuropeptides, produced by hypothalamic neurons, play a crucial role in regulating breathing. 12
Extrinsic Factors Modulate Hypothalamic Functions
Hypothalamus activity is also known to be influenced by various extrinsic factors. Some well-characterised examples include stress, light, temperature, nutritional status, drugs and medications, and other environmental factors. When faced with stress, the HPA axis becomes activated within the hypothalamus–pituitary–adrenal system, prompting the synthesis of CRH, 8 which further sets off a series of biochemical responses that ultimately culminate in the release of cortisol. As discussed in the previous section, CRF neurotransmission in the lateral hypothalamus specifically plays a crucial role in modulating the tachycardic response, characterised by an increased heart rate during acute emotional stress, and the blockage of CRF1 receptors results in a significant reduction in the stress-induced rapid heart rate. 7 Drugs and medications are also known to have direct or indirect effects on the hypothalamus and therefore constitute extrinsic stimuli. In a study involving 120 hens, administration of N-acetyl-L-cysteine (NAC) alongside a regular diet reduced heat stress–induced oxidative stress and inflammation in the hypothalamus. The protective effects of NAC on the hypothalamus were attributed to the suppression of the NF-kB pathway activity, leading to the mitigation of inflammation and oxidative stress. 13 In rats, diazepam has been found to enhance the function of the HPA axis by inhibiting phosphodiesterase 4 (PDE 4). This particular effect is mediated via a cyclic AMP-dependent pathway, which involves the activation of cyclic AMP-dependent protein kinase (PKA). 14
It has been established that the hypothalamus regulates hunger and satiety via hormones such as leptin and ghrelin. 15 Leptin receptor expression on POMC neurons of obese and diabetic mice shows positive outcomes in weight loss and blood sugar normalisation. 16 Hypothalamic activity is also known to be modulated by external temperature and to trigger responses such as shivering and sweating to regulate body temperature. For example, research utilising functional magnetic resonance imaging (fMRI) in rats has demonstrated that alterations in body temperature can influence the activity of the hypothalamus. When the body temperature was elevated from 38°C to 39°C, a notable increase in activity was observed in the hypothalamic preoptic area (PO), indicating the role of this region in temperature regulation. 17 Furthermore, investigations into the control of body temperature have led to the identification of underlying hypothalamic circuits. This pathway comprises specific groups of temperature-sensitive neurons located in the ventral portion of the lateral preoptic nucleus (vLPO) and the dorsal portion of the dorsomedial hypothalamus (DMD). Within the vLPO, GABAergic neurons were observed to suppress the activity of thermogenic neurons in the DMD, leading to a reduction in body temperature and physical activity. Conversely, activation of DMD neurons resulted in an elevation of body temperature and increased energy expenditure, whereas their inhibition induced hypothermia. 18
Environmental factors, such as noise and odours, can influence the hypothalamic function. In a study involving rats exposed to varying intensities of loud noise, it was observed that higher noise intensities triggered an elevation in hormone levels and the activation of specific brain regions. These findings further suggest that the hypothalamus, along with other brain areas, initiates the body’s stress response to perceived threats, highlighting its involvement in regulating stress-related processes. 19 Furthermore, olfactory stimuli too can modulate the hypothalamic activity. Research on monkeys investigated the response of glucose-sensitive (GS) and glucose-insensitive (GIS) neurons in the lateral hypothalamus to odours. GS neurons exhibited a broader sensitivity to various odours, with their responses influenced by the hedonic value of the odour and inhibited by dopamine. Conversely, GIS neurons demonstrated responses to specific cues and were facilitated by dopamine, implying their role in regulating feeding behaviour. 20 Taken together, the hypothalamus is intricately influenced by various intrinsic and extrinsic factors. Investigating the complex interactions between these factors provides profound insights into the functioning of the hypothalamus. Understanding and harnessing the collective power of these factors can support optimal hypothalamic functioning, leading to a deeper understanding of various physiological processes and potentially enabling novel therapeutic interventions in the future.
Stimuli mix is a phenomenon where many inputs, both from the external environment and from the body itself, converge in the hypothalamus. By assimilating this diverse range of information, the hypothalamus gains a comprehensive understanding of the body’s internal and external states. With this comprehensive picture, the hypothalamus processes the data and orchestrates appropriate responses. For instance, on a hot summer day outdoors, the hypothalamus receives a ‘Stimuli mix’ from sources like thermoreceptors detecting temperature changes, 21 hormones like cortisol influencing stress responses 22 and sensory input from skin receptors detecting environmental cues. 23 It integrates these inputs to regulate temperature by activating cooling mechanisms, increasing skin blood flow and promoting thirst. 18 The significance of this integration lies in its ability to safeguard the body’s internal equilibrium while enabling effective responses to the external world. This harmonious coordination of inputs ensures that the body remains in a state of balance, facilitating its ability to navigate through life’s varying challenges with precision and efficiency. Through intentional adjustments of physiological inputs, one can effectively modulate the stimuli mix, influencing the body’s adaptive capabilities and promoting a harmonious balance in its functioning. For instance, regular exercise changes the mix of physiological stimuli in the hypothalamus. It increases heart rate, triggers hormone release, elevates metabolic rate, regulates temperature and influences stress and recovery signals. As a result, individuals may experience reduced appetite, improved sleep, better stress regulation and enhanced overall well-being. By influencing the mix of physiological stimuli in the hypothalamus, exercise not only impacts physical health but also contributes to emotional well-being 24 and cognitive performance. 25 This holistic approach to well-being highlights the interconnectedness of the body and mind and underscores the importance of intentional adjustments in physiological inputs for achieving a more balanced and harmonious state of being. Understanding and harnessing this power opens doors to enhancing adaptability and achieving a more balanced and harmonious state of being.
Stimuli Preprocessing: Integrating External and Internal Stimuli for Hypothalamic Function
Sensory preprocessing precedes hypothalamic processing through complex neural cascades involving sensory transduction, refinement and modulation. Sensory information undergoes sequential processing along specialised pathways, culminating in the thalamic relay and specific hypothalamic nucleus engagement. Integrating sensory signals within distinct hypothalamic nuclei results in a comprehensive representation of the organism’s milieu. Feedback from diverse brain regions and endocrine glands contributes to homeostatic regulation. Ultimately, processed and integrated hypothalamic stimuli drive regulatory responses crucial for organismal survival and well-being. For instance, the PVN has been linked to regulating respiratory output. Experimental investigations conducted on rats have shown that activation of the PVN enhances the activity and frequency of contractions in the diaphragm muscle, leading to changes in the timing of respiration. 26 Furthermore, damage to the suprachiasmatic nucleus (SCN) has been associated with the development of insulin resistance, underscoring its role in metabolic regulation. 27 In addition to processing sensory inputs, the hypothalamus receives crucial feedback signals from various brain regions and endocrine glands. These feedback signals provide contextual information and contribute to the regulation of homeostasis. By incorporating feedback signals, the hypothalamus can adjust its responses and orchestrate appropriate physiological adaptations. Activation of the PVN impacts cardiorespiratory responses through the oxytocinergic innervation of neurons located in the pre-Bötzinger complex (pre-BötC). The stimulation of PVN neurons containing oxytocin (PVN-OT) results in an augmentation of respiratory drive to the diaphragm and genioglossus muscle, along with an increase in arterial blood pressure and heart rate. 28 Furthermore, during fasting, tanycytic VEGF-A protein increases, enhancing the plasticity of the blood–hypothalamus barrier and allowing metabolic signals to reach the arcuate nucleus in the hypothalamus, influencing energy and metabolism regulation. 29 The processed and integrated stimuli within the hypothalamus ultimately initiate regulatory responses. These responses are vital for maintaining homeostasis and coordinating essential physiological functions, including temperature regulation, thirst and satiety, circadian rhythms, reproduction and stress responses. Acting as a central hub, the hypothalamus integrates and interprets sensory information to ensure the survival and well-being of the organism (Figure 1).
All-powerful Hypothalamus: The Master Regulator of the Body’s Functions
The hypothalamus plays a crucial role as a central connection point between the nervous system and the endocrine system. Despite its relatively modest size, the hypothalamus significantly influences the human brain and organismal well-being. Structurally, the hypothalamus comprises several nuclei, clusters of specialised neurons. The human hypothalamus is a diminutive yet intricately organised brain structure that regulates diverse physiological functions encompassing vegetative, emotional and reproductive processes. 30 Anatomically, it is segregated into three primary regions: the anterior, tuberal and posterior hypothalamus. 31 The anterior region contains the PO, which regulates the body temperature, sleep–wake cycles and reproductive behaviours. The hypothalamus, being a structurally complex and functionally diverse brain region, consists of distinct nuclei and neuronal populations that possess specific functional roles. Studies have shown that exposure to low oxygen levels, also known as a hypoxic condition, affects the functioning of the anteroventral preoptic region (AVPO) in the hypothalamus, which plays a role in regulating breathing and thermoregulation. 32 The supraoptic nucleus (SON) is a conglomerate of magnocellular neurosecretory cells situated in close proximity to the optic chiasma, comprising approximately 3,000 neurons. It was found that oxytocin neurons in the SON of mice are crucial for successful breastfeeding. Mice with a conditional knockout of the oxytocin gene in both the paraventricular and supraoptic hypothalamic nuclei displayed normal parturition and maternal behaviours but severe impairments in breastfeeding, leading to the death of their pups. 33
Additionally, neurons in the lateral hypothalamus (LHA) have been found to distinguish between rewarding and aversive stimuli, enabling animals to learn and discriminate cues associated with these stimuli. In contrast, neurons in the lateral preoptic-anterior hypothalamic area (IPOA-AHA) respond similarly to cue tones regardless of their predictive value, suggesting their involvement in arousal or attention. 34 The tuberal region of the hypothalamus houses important nuclei, including the arcuate nucleus, involved in regulating appetite and metabolism, and the paraventricular nucleus, responsible for controlling stress responses, blood pressure and hormone release. Ghrelin, a hormone involved in food intake regulation, passes through specialised brain capillaries in the median eminence and rapidly binds to specific neurons in the ventromedial arcuate nucleus, such as NPY and POMC neurons, which plays a crucial role in regulating food intake. Ghrelin binding to hypothalamic neurons varies depending on the feeding status. 15 During fasting, the tanycytic VEGF-A protein increases, enhancing the plasticity of the blood–hypothalamus barrier. This allows metabolic signals to reach the arcuate nucleus in the hypothalamus, influencing energy and metabolism regulation. 29 The cardiorespiratory responses, such as increased respiratory drive, elevated arterial blood pressure and heart rate, are influenced by the stimulation of the PVN. This effect is achieved through the oxytocinergic innervation of neurons in the pre-BötC, as the activation of PVN-OT-containing neurons leads to these physiological changes. 28 Furthermore, the PVN plays a role in modulating respiratory output, as its stimulation increases the activity and frequency of diaphragm muscle contractions, resulting in changes in respiratory timing. Direct connections between the PVN and phrenic motor neurons indicate the involvement of PVN neurons in coordinating cardiovascular and respiratory functions. 26 The PVN also plays a crucial role in controlling penile erection in male rats. Oxytocinergic neurons originating in this nucleus and projecting to other brain areas are activated by various substances, including dopamine, oxytocin and excitatory amino acids, leading to penile erection. Inhibition of these neurons by substances such as GABA and opioid peptides suppresses the sexual response. 35 During lactation, the activity of neuropeptide Y (NPY) neurons in the arcuate nucleus (ARH) and dorsomedial nucleus (DMH) is heightened. These NPY neurons project to the PVN, indicating their involvement in regulating physiological changes during lactation and potentially impacting food intake through the modulation of PVN neuronal activity. 36
The posterior region of the hypothalamus includes the SCN, responsible for regulating circadian rhythms, and the mammillary bodies, involved in memory processes. Through a network of activating and inhibitory neurons, the hypothalamus regulates sleep and circadian rhythms. Inhibition of the arousal system by inhibitory neurons in the ventrolateral preoptic nucleus promotes sleep by releasing the neurotransmitter GABA. The flip-flop switch circuit controls the balance between wakefulness and sleep. Sleep regulation involves both homeostatic factors, such as the accumulation of adenosine, and circadian factors controlled by the SCN. 1 Damage to the SCN, the body’s internal clock, disrupts energy balance, resulting in weight gain and fat accumulation. Additionally, damage to the SCN can lead to the development of insulin resistance, highlighting its role in metabolic regulation. 27 Glutamate-releasing neurons in the supramammillary region (SuMvglut2) play a crucial role in the arousal system, promoting wakefulness and regulating sleep stages, including rapid eye movement (REM) sleep. Activation of SuMvglut2 neurons leads to sustained behavioural and EEG arousal, while their inhibition decreases wakefulness and suppresses theta and gamma frequency EEG activity. 37
In summary, the hypothalamus is a master regulator of various physiological processes. It maintains the body temperature through thermoregulation, regulates feeding behaviour and energy balance, controls sleep and wakefulness, orchestrates the stress response and emotional behaviours, governs the release of hormones from the pituitary gland and influences reproductive behaviours and functions. Understanding the intricacies of the hypothalamic function is crucial for diagnosing and treating disorders associated with hypothalamic dysfunction and developing innovative therapeutic interventions (Figure 3).
Hypothalamus Connections to the Other Brain Regions.
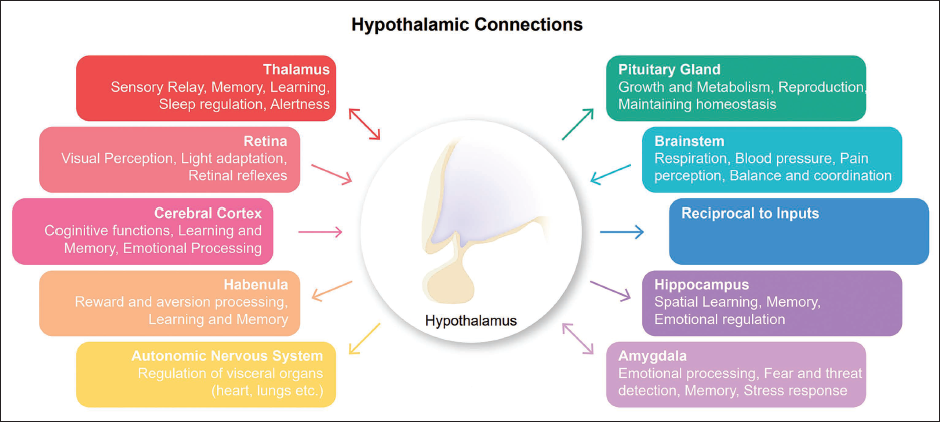
Integrative Interactions: Hypothalamic Cross-talk with Other Brain Regions
One of the remarkable aspects of the hypothalamus is its intricate cross-talk with other brain regions. This phenomenon allows the hypothalamus to assimilate information originating from distinct brain areas and subsequently orchestrate well-suited responses. The hypothalamus integrates neural input from the brainstem, limbic system, cerebral cortex and retina and non-neural input such as blood temperature and osmolarity. It subsequently generates outputs to the pituitary gland (both neural and endocrine pathways), the autonomic nervous system (neural pathway), the habenula and the thalamus (neural pathway). The hypothalamus, a vital structure in the brain, is closely connected with the brainstem, specifically the midbrain, pons and medulla oblongata. 38 The brainstem performs vital functions such as regulating vital bodily functions, including breathing, 39 heart rate 40 and blood pressure; 41 controlling cranial nerves; modulating sleep; 42 regulating pain perception; 43 maintaining balance and coordination; regulating digestion 44 and integrating autonomic functions such as temperature regulation. 45 For instance, hypothalamic arcuate nucleus neurons exhibit responsiveness to fluctuations in circulating leptin levels, playing a pivotal role in the modulation of eating behaviour and energy balance. Gastrointestinal signals are conveyed to the brainstem through the vagus nerve and other pathways, engaging with hypothalamic–brainstem circuits that govern meal timing, meal size and overall food consumption. 46 The hypothalamus exhibits bidirectional neural connectivity with the thalamus, encompassing both afferent and efferent fibre pathways. 47 Afferent fibres originating from the hypothalamus facilitate the transmission of sensory information from diverse bodily regions to the thalamus, thereby enabling the hypothalamus to receive pertinent input regarding both internal and external environmental conditions. Conversely, efferent fibres stemming from the hypothalamus serve as conduits for relaying signals from the hypothalamus to the thalamus, thereby exerting regulatory influence over an array of functions, including autonomic nervous system activity, hormone secretion and behavioural responses. The thalamus is a vital structure located in the brain that plays a crucial role in sensory processing, 48 motor control, 49 emotion processing, 50 pain perception, 51 memory and spatial navigation 52 and the regulation of consciousness and sleep. 53
The hypothalamus and the limbic system are closely connected and interact extensively. 47 The limbic structures, particularly the amygdala, exhibit bidirectional neural connectivity to the hypothalamus, which is responsible for integrating emotional information and influencing a range of physiological and behavioural responses. The limbic system plays a role in processing basic emotions such as happiness, 54 sadness, 55 anger 56 and fear 57 as well as complex emotions such as love, 58 jealousy, 59 shame 60 and guilt. 61 It is also involved in memory formation and retrieval, 62 learning, 63 decision-making, 64 and the regulation of essential physiological processes such as sleep. 65 One of the primary sources of input to the hypothalamus is the cerebral cortex. The cerebral cortex, a wondrous web of neurons, weaves thoughts, emotions and perceptions into the tapestry of our conscious experience. Impaired hypothalamic-subgenual cortex communication in psychotic major depression is associated with reduced functional connectivity, increased cortisol secretion and symptom severity, suggesting its role in the pathophysiology of depressive symptoms in individuals with psychosis. 66 The cerebral cortex performs a multitude of functions, including sensory processing, 67 motor control, 68 memory, 69 executive functions such as decision-making, 70 problem-solving, 71 attention regulation 72 and emotional processing. 73
The hypothalamus sends efferent fibres to the habenula, specifically to the lateral habenula (LHb) and the medial habenula (MHb). The hypothalamus, through its connections with the lateral habenula, a part of the brain stem, integrates internal and external information related to an organism’s physiological state and influences decision-making processes by encoding risk preferences. 74 These connections are part of a complex neural circuit involved in various functions, including the regulation of mood, 75 motivation 76 and reward processing. 77 The hypothalamus sends efferent fibres to the hippocampus, although the specific connections and their functions are still a topic of ongoing research. The major pathway through which the hypothalamus communicates with the hippocampus is known as the hypothalamo-hippocampal pathway. 78 The hippocampus is a crucial brain structure involved in various cognitive processes, including learning, 79 memory 80 and spatial navigation. 81 The hypothalamus sends efferent (outgoing) fibres to various components of the autonomic nervous system, including the parasympathetic and sympathetic divisions. These efferent fibres transmit signals from the hypothalamus to influence the activities of organs and tissues throughout the body, including organs such as the liver and pancreas. Research has identified a distinct circuit between the hypothalamus and pancreatic β cells, which are responsible for insulin secretion. A distinct hypothalamus-to-pancreatic β cell circuit involving PVNOXT neurons modulates insulin secretion, suppressing it upon stimulation and increasing it when silenced, impacting glucose levels. 82 The hypothalamus communicates with the liver through neural connections involving the ventromedial hypothalamic nucleus (sympathetic signals), the lateral hypothalamic area (parasympathetic signals) and the periventricular hypothalamic nucleus (integrates autonomic signals). Additionally, the hypothalamus influences liver function through neuroendocrine connections, including the hypothalamus–pancreas, hypothalamus–adrenal and hypothalamus–pituitary axis. 83
In conclusion, the hypothalamus is a highly interconnected structure that engages in intricate cross-talk with various brain regions. These integrative interactions enable the hypothalamus to orchestrate appropriate responses, regulating essential bodily functions such as hormone secretion, autonomic nervous system activity, eating behaviour, sleep–wake cycles and emotional processing. Furthermore, the hypothalamus demonstrates bidirectional neural connectivity with key structures such as the thalamus and limbic system, emphasising its role in sensory processing, motor control, memory formation and decision-making. Overall, the complexity and extensive connections of the hypothalamus highlight its pivotal position in orchestrating and regulating numerous physiological and behavioural responses throughout the body.
Hypothalamic–Pituitary Axis: A Dynamic Partnership
The interplay between the hypothalamus and the pituitary gland is a highly intricate and essential process in neuroendocrine regulation within the body. The hypothalamus, situated at the base of the brain, serves as a central control centre that orchestrates the secretion of hormones from the pituitary gland. The pituitary gland comprises two main lobes: the anterior lobe, which releases hormones directly into the bloodstream, and the posterior lobe, which releases neurohormones through its neurovascular connection with the hypothalamus. Neurohormones produced within the hypothalamus are transported through the pituitary stalk to reach the posterior lobe, from where they are released into the bloodstream via coiled capillaries and portal vessels. 84 The hypothalamus–hypophysiotropic area (HTA) is a specialised region within the hypothalamus that plays a crucial role in regulating the secretion of hormones from the anterior pituitary. It comprises specialised hypophysiotropic or neurosecretory cells that produce releasing and inhibiting hormones directly controlling the release of pituitary hormones. The HTA is a crucial link between the hypothalamus and the anterior pituitary, allowing precise regulation of hormone secretion that influences various physiological processes in the body. The HTA plays a critical role in maintaining the normal structure and function of the anterior pituitary gland when transplanted, while grafts outside the HTA do not exhibit the same histological maintenance. 85 The HTA serves as a critical interface between the hypothalamus and the anterior pituitary, autonomously regulating pituitary function through a direct supply of tropic and releasing substances. The involvement of external neural structures further enhances the two-tiered control system, allowing for intricate modulation of the anterior pituitary hormone secretion. 86 The hypothalamus–pituitary system not only encompasses the neuroendocrine system but also includes a neurosecretory pathway. This pathway originates in the anterior hypothalamus and traverses through the ventral hypothalamus, ultimately terminating in the neurohypophysis or posterior lobe (PL) of the pituitary gland. Along this pathway, the hypothalamus synthesises hormones such as oxytocin and vasopressin, which are subsequently transported through nerve fibres to the neurohypophysis. Upon reaching the neurohypophysis, these hormones are released into fenestrated blood vessels, which possess high permeability to facilitate the passage of hormones into the systemic circulation. Once in circulation, these hormones exert their effects on target tissues throughout the body, thereby influencing various processes, including water balance, uterine contraction and lactation. Furthermore, the hypothalamus exerts neural control over the pars intermedia (PI), a distinct region within the pituitary gland. Nerve fibres originating from the hypothalamus predominantly exhibit aminergic characteristics and project to the PI. The hypothalamus produces and releases hypothalamic-releasing or hypothalamic-inhibiting hormones, which are then transported to the anterior pituitary gland through the hypophyseal portal system—a specialised network of blood vessels. This system enables direct communication between the hypothalamus and the anterior pituitary, facilitating precise regulation of hormone secretion. The hypothalamic-releasing hormones have specific effects on the anterior pituitary, either stimulating or inhibiting the release of tropic hormones. For example, gonadotropin-releasing hormone (GnRH) stimulates the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which are vital for reproductive processes. Thyrotropin-releasing hormone (TRH) stimulates the release of thyroid-stimulating hormone (TSH) and prolactin, while growth hormone-releasing hormone (GHRH) stimulates the release of growth hormone (GH), which is involved in growth, metabolism and tissue repair. Conversely, somatostatin acts as an inhibitory hormone, suppressing the release of GH and TSH from the anterior pituitary. Pituitary hormones, particularly GH and prolactin (PRL), also serve as neurotrophic signals in the hypothalamus. In hypopituitary dwarf mice lacking GH and PRL, the expression of inhibitory neurohormones (dopamine and somatostatin) is reduced, while the expression of stimulatory GHRH is increased. This indicates the crucial roles of GH and PRL in the survival, phenotypic differentiation and axonal connectivity of hypophysiotropic neurons in the hypothalamus. 87 In addition to controlling the anterior pituitary, the hypothalamus also exerts direct control over the posterior pituitary gland, an extension of the hypothalamus. Apart from controlling the anterior pituitary, the hypothalamus also exerts direct control over the posterior pituitary, which serves as an extension of the hypothalamus. The posterior pituitary stores and releases two hormones the hypothalamus synthesises: antidiuretic hormone (ADH) or vasopressin and oxytocin. ADH regulates water balance by controlling water reabsorption in the kidneys, thereby influencing urine concentration. Oxytocin plays a critical role in childbirth by stimulating uterine contractions during labour and promoting milk ejection during breastfeeding. Hypothalamo-pituitary disconnection (HPD) refers to the disruption of communication between the hypothalamus and the pituitary gland, leading to hormonal imbalances and dysfunction in various physiological processes. The surgical procedure in sheep known as HPD disconnects the pituitary gland from hypothalamic control but maintains its blood supply, resulting in decreased LH and FSH levels and increased prolactin levels, indicating altered pituitary hormone secretion. 88
The HPA axis is a complex neuroendocrine system that is crucial in regulating the body’s stress response. It involves intricate interactions between the hypothalamus, pituitary gland and adrenal glands. When the body experiences stress or other stimuli, the HPA axis is activated, starting with the release of CRH by the hypothalamus. This hormone stimulates the anterior pituitary gland to release adrenocorticotropic hormone (ACTH) into the bloodstream. ACTH then travels to the adrenal glands, specifically the adrenal cortex, and triggers the release of glucocorticoid hormones, with cortisol being the primary glucocorticoid in humans. Cortisol, as the main glucocorticoid, plays a pivotal role in regulating various physiological processes, including metabolism, immune function and the stress response. It affects target tissues throughout the body, influencing glucose metabolism, suppressing inflammation and modulating the sleep–wake cycle. A negative feedback mechanism governs the functioning of the HPA axis. When cortisol levels are elevated, they inhibit the release of CRH and ACTH, leading to a reduction in cortisol production. This feedback loop helps maintain cortisol levels within a normal range. In a study investigating the effects of cortisol infusion on the pituitary–adrenal axis in HPD foetal sheep, it was found that the infusion of cortisol increased foetal plasma cortisol levels without affecting plasma ACTH concentrations. Cortisol had a direct tropic effect on the maturation of corticotrophs in the foetal pituitary, but an intact hypothalamo–pituitary axis was necessary for complete maturation during late gestation. 89 Disruptions in HPA axis regulation can have significant health implications. Dysfunctions in this axis are associated with an increased risk of developing anxiety disorders in adulthood, including generalised anxiety disorder, panic disorder and phobias. 90 The HPA axis, which regulates stress response, is implicated in developing depression and schizophrenia. In major depressive disorder, there is an overactivation of the HPA axis, leading to cognitive impairment and low mood. 91 On the other hand, insufficient HPA axis activity or cortisol deficiency can manifest as Addison’s disease, characterised by low cortisol levels. Hyperactivity of the HPA axis is observed in certain patients with anxiety and mood disorders. Benzodiazepines, tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs) have been studied for their effects on various components of the HPA axis and their potential in treating anxiety disorders. 92
Taken together, both the hypothalamus and the pituitary gland exhibit intricate interactions to regulate hormone release and uphold homeostasis in the body. The hypothalamus serves as a central control centre, generating and releasing hypothalamic-releasing and -inhibiting hormones that modulate the function of the anterior pituitary gland. Consequently, the anterior pituitary gland secretes various tropic hormones that act on target organs and tissues throughout the body, ensuring the appropriate functioning of physiological processes. The posterior pituitary, an extension of the hypothalamus, stores and releases hormones synthesised by the hypothalamus, further contributing to hormonal regulation and overall homeostasis.
Empowering Hypothalamus Through Breathing Techniques, Optimised Sleep Architecture and Stress Management
To truly achieve well-being and cognitive enhancement, it is essential to take a holistic approach acknowledging the intricate interplay between various factors influencing our mental and physical health. This article explores and discusses a comprehensive strategy that focuses on empowering the hypothalamus—the master regulator of the brain—to optimise well-being and cognitive function. In this section, we explicitly discuss whether and how breathing techniques, optimal sleep cycles and related physiological processes could modulate hypothalamic activity and thus promote wellness and cognitive enhancement. Empowering the hypothalamus is essential for achieving overall well-being and cognitive enhancement. Breathing techniques, such as deep breathing, diaphragmatic breathing and alternate nostril breathing, have been used for centuries to induce relaxation, reduce stress and improve focus. These techniques have a direct impact on the hypothalamus by activating the parasympathetic nervous system, which promotes a state of calm and balance. Slow breathing techniques lead to increased activity in the hypothalamus, along with other cortical and subcortical structures. It is also associated with psychological and behavioural outputs such as increased comfort and relaxation and reduced symptoms of arousal, anxiety, depression, anger and confusion. These changes in psychological well-being are likely mediated by the modulation of the hypothalamus and its interactions with other brain regions. The precise mechanisms through which slow breathing influences the hypothalamus are not yet fully understood. However, two hypotheses have been proposed to explain the psychophysiological changes induced by slow breathing. One hypothesis suggests that the voluntary regulation of slow breathing may involve the voluntary regulation of internal bodily states (enteroception), which in turn influence the hypothalamic activity. The other hypothesis proposes that mechanoreceptors within the nasal vault, stimulated by slow breathing, could modulate olfactory bulb activity, leading to cascading effects on the entire cortical mantle, including the hypothalamus. 93 The hypothalamus also plays a crucial role in decision-making by influencing motivation, reward-seeking behaviour and emotional regulation. Computational methods and electrophysiological investigations provided a preliminary understanding of decision-making within the ventromedial nucleus (VMN). Network models elucidated emergent properties such as rhythm generation and bistability, crucial for effective decision-making in the VMN. 94 Investigations centred on the perifornical region of the PeF-LH have provided compelling evidence of its vital contribution to cost-benefit decision-making in rats. Through a T-maze task, researchers have uncovered the connection between PeF-LH and the ventral tegmental area, which is involved in reward processing. 4 One crucial question that stems from these research findings is whether the reverse of this event also holds. Studies have shown that by adopting effective decision-making strategies, such as setting clear goals, weighing pros and cons and cultivating mindfulness, we can optimise the functioning of the hypothalamus, improving cognitive abilities and overall well-being.
Quality sleep is also vital for cognitive function, emotional well-being and physical health. The hypothalamus regulates the sleep–wake cycle by producing melatonin and other hormones. Implementing sleep hygiene practices, such as maintaining a consistent sleep schedule, creating a conducive sleep environment and practicing relaxation techniques, can optimise hypothalamic function and enhance the restorative power of sleep. Sleep deprivation in rats led to increased somatostatin mRNA levels in the arcuate nucleus (ARC) and increased GHRH mRNA levels in the PVN. In contrast, the periventricular nucleus (PE) showed no significant effect. These findings suggest that sleep loss modulates SRIF and GHRH cells in specific hypothalamic nuclei, supporting their role in sleep regulation. 95 REM sleep deprivation in rats led to a decrease in the expression of growth GHRH in PVN and arcuate nucleus of the hypothalamus. The number of cells expressing somatostatin (SRIF) increased in the arcuate nucleus after 24 hours of REM sleep deprivation but not after 72 hours. Serum GH levels were decreased in rats subjected to both short and prolonged REM sleep deprivation. 96 Chronic stress disrupts hypothalamic functioning, leading to a cascade of adverse effects on mental and physical health. By adopting stress management techniques, such as mindfulness meditation, physical exercise and engaging in hobbies, one can modulate the hypothalamic response to stress and promote overall well-being and cognitive enhancement. Stress, a prevalent factor in modern lifestyles, negatively affects the HPA and GBA axes, disrupting equilibrium and increasing susceptibility to inflammatory diseases and infections. 97 This study emphasises the impact of chronic stress on the cellular function and structure of the hippocampus and hypothalamus in rats. Extended periods of stress disrupt the regulation of the HPA axis, heighten glutamate transmission, impede neurogenesis and promote apoptosis, inhibit synaptic plasticity and diminish responses to serotonin. These results offer promising avenues for developing novel therapeutic approaches in addressing brain disorders associated with stress. 98
The hypothalamus, a vital brain region, possesses complex connectivity with emotional processing centres, thus playing a role in the regulation and manifestation of emotions. Situated below the thalamus, the hypothalamus forms neural connections with regions such as the amygdala, hippocampus, limbic system and prefrontal cortex, working together to facilitate emotional modulation. These interconnections enable the hypothalamus to receive and integrate sensory inputs and information from these regions, thereby engaging in the generation and regulation of emotional responses. The medial temporal limbic system (TLS) is an integral neuroanatomical network within the brain that plays a pivotal role in the processing and regulation of emotions. 99 Thyrotropin-releasing hormone (TRH), localised within the limbic system, exerts regulatory influence over mood and eating behaviours. TRH exerts direct effects via its receptors and indirect effects by modulating neurotransmitter systems in the brain, including glutamate, gamma-aminobutyric acid, acetylcholine and dopamine. 100 Major depression involves separate circuits for anhedonia (lack of pleasure) and dysphoria (lack of happiness), with the limbic system regulating misery-fleeing behaviour and the extrapyramidal system regulating reward-seeking behaviour. 54 The ventrolateral hypothalamic area, specifically its tuberal portion, has long been suspected of triggering positive emotional responses, as electrical stimulation of this region has been found to provoke bursts of laughter in various species. 101 Specific neurons in the ventromedial hypothalamus can independently initiate defensive behaviours and are necessary for such behaviours in different situations, demonstrating that the hypothalamus plays an active role in generating emotional states rather than just being a downstream effector of other emotion centres like the amygdala. 102 When electrical stimulation was applied near the ventromedial area of the hypothalamus in a human patient, it evoked intense emotions of shame, sadness and fear, while rage or anger was not observed. In contrast, stimulating other cortical structures did not produce the same intricate negative emotional responses witnessed with hypothalamic stimulation. This finding provides compelling evidence for the involvement of the hypothalamus in human emotional experiences and enhances our understanding of the interconnectedness between the hypothalamus and other brain regions engaged in regulating emotions. 103 Considering the acknowledged role of the hypothalamus in emotional regulation, an inquiry emerges: Is it possible to harness our emotions to exert influence and modulation over the activity of the hypothalamus?
In conclusion, by adopting a holistic approach that empowers the hypothalamus through breath control, sleep hygiene and stress management, it seems possible to tune hypothalamic activities and pave the way for well-being. These earlier scientific proofs support the notion that by nurturing the hypothalamus, it is possible to unlock the potential for a fulfilling and successful life, promoting resilience, cognitive function and overall wellness.
Conclusion
In conclusion, this comprehensive review underscores the central and indispensable role of the hypothalamus in integrating internal and external signals to regulate a diverse array of physiological processes, cognition, emotion and behaviour. Serving as a vital intermediary between the nervous and endocrine systems, the hypothalamus maintains homeostasis and coordinates various bodily functions. The review also investigates the modulation of hypothalamic activity by intrinsic and extrinsic factors, highlighting the potential of non-invasive techniques such as controlled breathing exercises, optimised sleep architecture and stress management in enhancing hypothalamic function and promoting overall well-being and cognitive function. Specific techniques such as meditation, breathing exercises and cold exposure are reported to trigger epinephrine release, reducing inflammation and promoting anti-inflammatory responses, and offering the potential for treating conditions involving chronic inflammation. 104
Based on these, there are many apparent questions that need to be systematically addressed. Some of the prevalent ones include the following: Can we leverage our emotions to influence and regulate the activity of the hypothalamus? Can certain techniques, such as controlled breathing, sleep cycle management, decision-making strategies, stress reduction, and exercise, be utilised to influence the hypothalamus and potentially improve cognitive abilities? What are the potential risks or drawbacks associated with attempting to manipulate the hypothalamus through emotional means, and how can these be addressed or mitigated? What role does decision-making play in hypothalamic regulation or vice versa? Are there specific decision-making approaches that can positively influence hypothalamic function? Most importantly, it is important to devise strategies to decode the relationship between the types of stimuli mix perceived by the hypothalamus and its downstream effects. However, it is essential to recognise that the current scientific evidence in this area is limited, necessitating further research to uncover fascinating discoveries. While further research is required to establish direct causal relationships between hypothalamic functions and specific physiological processes, this review provides a comprehensive understanding of the multifaceted role of the hypothalamus. By acknowledging the influence of breathing techniques, sleep patterns and stress management on hypothalamic activity, interventions can be devised to optimise health and cognitive abilities. It is crucial to adopt a holistic approach that encompasses the intricate interplay between the hypothalamus and various factors impacting mental and physical well-being. Empowering the hypothalamus is a promising avenue for individuals seeking to enhance overall well-being and cognitive performance. Further investigations and prospective studies are warranted to deepen our knowledge and facilitate the development of innovative interventions targeting hypothalamic function, with the ultimate goal of improving health outcomes.
Authors’ Contribution
MG, VJ, GA and AJ wrote the original draft; SM, AJ and KK conceptualised the review content; GA and AJ designed the framework. AM performed the graphical illustrations, AB provided critical comments and suggestions while reviewing and editing the manuscript. All authors have read the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable for this review article.
Statement of Ethics
Not applicable for this review article.
