Abstract
Background
Progressive supranuclear palsy (PSP) and multiple system atrophy (MSA) are less responsive to usual antiparkinsonian drugs.
Purpose
To assess the treatment outcome of the combination of low-dose amantadine and escitalopram in the study population.
Methods
A retrospective study of PSP and MSA patients from June 2021 to December 2022 treated with the dose of amantadine 100 mg and escitalopram 5 mg per day with a follow-up of 2 months at a tertiary care centre in India. The primary outcome measure was a change in the PSP and MSA severity rating scale scores, while cognition, neuropsychiatric symptoms and functionality scale scores were secondary outcomes on follow-up visits. Repeated measures ANOVA and Friedman rank test were used to analyse the data.
Results
A total of 21 patients (8 PSP and 13 MSA), including 19 males, with a mean age of 72.84 years, were included. The mean total PSP rating scale score had a significant decline from baseline to week 2 and 8 (p < .001), along with the scores for limb symptoms (p < .001), ocular (p = .001), history (p = .003), gait (p = .007), mentation (p = .014) and bulbar (p = .018) symptoms. Similarly, the mean score of the MSA rating scale significantly decreased on follow-up visits (p < .001). The cognition, functional disability and behavioural symptoms scores also had significant improvement with p < .01.
Conclusion
The PSP and MSA patients had clinically significant improvement with the combination treatment in both motor and non-motor domains. Further prospective trials for longer durations are needed to establish the effect size and stability of response.
Introduction
Parkinsonism is a hypokinetic syndrome that is characterised by the presence of resting tremors, rigidity, bradykinesia and postural instability. The most common cause of parkinsonism is idiopathic Parkinson’s disease (PD). However, a small proportion of patients with parkinsonism have atypical features, which include cognitive impairment, frequent falls, ocular motility disturbances, ataxia and autonomic dysfunctions; these are called atypical parkinsonian disorders (APD). 1 The common differentiating features of APD from PD are rapid progression, poor response to levodopa and distinctive pathological characteristics. 2 APD is a rare entity with a prevalence of around 5%–7% of cases of all types of parkinsonism.3, 4 Progressive supranuclear palsy (PSP) and multiple system atrophy (MSA) are the most common APDs with predominant motor manifestation, and both have a prevalence of approximately 10 per 100,000 persons. 5 Notably, the onset of illness is usually after 55 years, with a median survival duration of 5–10 years. 5
PSP is characterised by gaze palsy, backward falls and axial rigidity, while MSA is characterised by autonomic dysfunction and cerebellar motor symptoms. Pathologically, PSP is grouped under the tauopathies, while MSA is a synucleinopathy. PSP involves the basal ganglia and the brainstem, and MSA involves the cerebellum, pons and basal ganglia. The involvement of a myriad of neurotransmitters in different brain pathways, both pre-synaptic and post-synaptic, in atypical parkinsonism is posing a challenge in devising a pharmacotherapy for its cure.
Since the recognition of PSP as a pathological entity in 1964, followed by MSA in 1969, no intervention has been demonstrated to modify the disease progression, despite relentless efforts. However, mild to moderate effectiveness of supportive treatments based on neurotransmitter modulation for individual symptoms in atypical parkinsonism has been reported in studies.
6
Amantadine, an N-methyl-
Methods
Study Design
The present study is a retrospective case note review of the use of low-dose amantadine and escitalopram in patients with a diagnosis of PSP and MSA. The study was conducted among the patients attending a tertiary care centre, Department of Geriatric Mental Health, King George’s Medical University, Lucknow. The combination of the two medications was initially used for the purpose of treatment based on existing evidence for similar symptoms and not for research. Considering the clinical improvement noticed in the patients, a retrospective case note review was planned to substantiate the evidence.
Case Selection
Participants were patients aged 60 years and older with the diagnosis of ‘possible or probable progressive supranuclear palsy’ according to the National Institute of Neurological Disorders and Stroke and the Society for Progressive Supranuclear Palsy (NINDS-SPSP) criteria and ‘possible or probable multiple system atrophy’ according to current consensus criteria by Gilman et al. (2008).12, 13 Participants who received amantadine 100 mg/day in divided doses and escitalopram 5 mg/day for a minimum period of 8 weeks were included in the study. Cases in which adequate information pertinent to this study was identified by reviewing the medical records from 1 June 2021 to 31 December 2022.
Data Extraction
Patients’ case notes during admission and follow-up notes in the outpatient department were compiled. A semi-structured proforma was used to collect socio-demographic and clinical variables, such as age, sex, age of onset of illness/duration of illness, progression of symptoms and duration of hospital stay. The severity of symptomatology was assessed using progressive supranuclear palsy rating scale (PSPRS), 14 multiple system atrophy rating scale (MSARS) 15 or whichever was applicable; cognition was assessed using Hindi Mental State Examination (HMSE), 16 neuropsychiatric symptoms were assessed using neuropsychiatric inventory (NPI) 17 and functioning was assessed using Hoehn and Yahr scale, 18 during admission and follow-up visits were extracted from the records. The last follow-up record of the patients was extracted using a non-fixed end-point methodology for assessing the stability of response to the treatment.
Outcomes
The change in PSPRS and MSARS scores from baseline to week 2 and 8 after receiving the combination treatment of amantadine 100 mg/day in the divided dose and escitalopram 5 mg/day in the subjects was considered the primary outcome measure. The change from baseline in the HMSE, NPI scale score and Hoehn and Yahr scale at 2nd and 8th week in the same subjects were the key secondary outcome measures. Any reported adverse events and stability of the effect of treatment received till the last follow-up were the other secondary measures.
Analysis
The baseline socio-demographic and clinical profile data analysis was done using descriptive statistics like percentages and frequencies, and comparison was done using the unpaired t-test, Fisher exact test and chi-square test, wherever applicable. The rating scale scores of PSPRS, MSARS, HMSE and NPI at baseline and at the end of the 2nd and 8th week were compared using a repeated measures ANOVA test. The Friedman rank test was used to compare the MSARS global disability subscale and Hoehn and Yahr stages at baseline, as well as 2nd and 8th week. The stability of the effect of treatment was assessed by comparing the scores of PSPRS and MSARS at 8 weeks and the last recorded follow-up visit using a paired t-test. A p value of less than .05 was considered to be significant.
Results
Out of the total of 28 cases who met the diagnosis of MSA and PSP within the study duration, 21 subjects fulfilled the inclusion criteria for the study. All the subjects were treated as inpatients and were followed up in outpatient clinic after discharge.
Socio-demographic and Clinical Characteristics
A total of 21 participants (19 males) were included in the study, 13 with a diagnosis of MSA and 8 with a diagnosis of PSP (Table 1). The average age of the subjects was 72.84 ± 7.88 years, and patients with MSA were older in comparison to PSP (76.30 ± 4.66 vs. 64.75 ± 3.61). Of note, the mean age of onset of illness was later in MSA in comparison to PSP (72.07 ± 5.82 vs. 60.87 ± 3.31). Notably, autonomic symptoms in MSA (53.35%) and ocular symptoms in PSP (50%) were the most common presenting symptoms, and they were statistically significant.
Baseline Demographic and Clinical Characteristics of the Subjects with Atypical Parkinsonian Disorder.
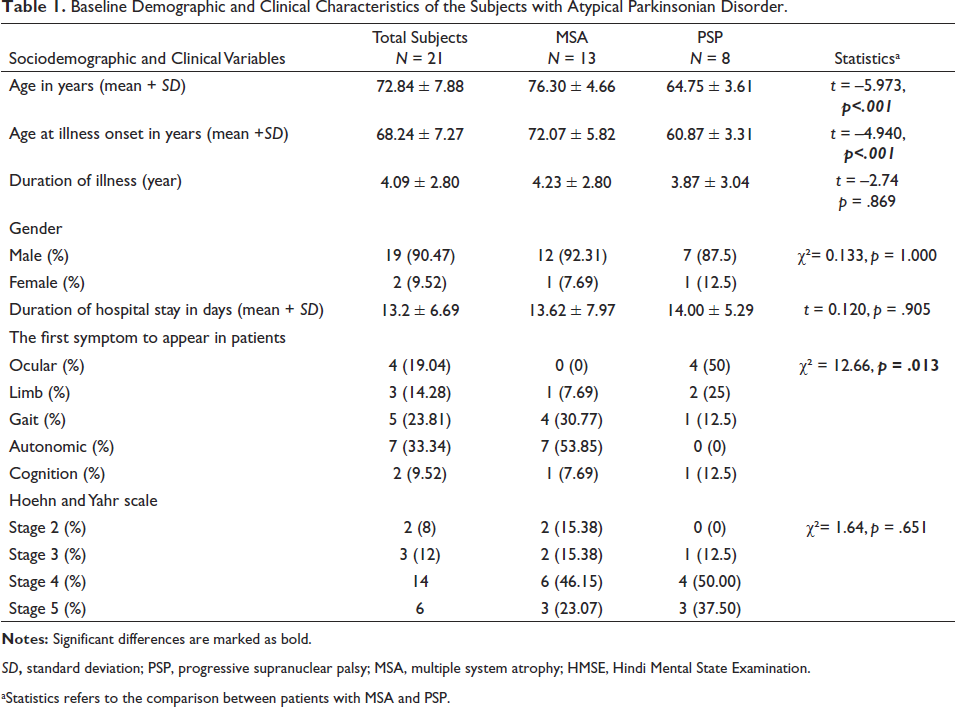
SD, standard deviation; PSP, progressive supranuclear palsy; MSA, multiple system atrophy; HMSE, Hindi Mental State Examination.
aStatistics refers to the comparison between patients with MSA and PSP.
Response on a Combination Treatment of Amantadine and Escitalopram in Severity, Neuropsychiatric Symptoms, Cognition and Functional Disability
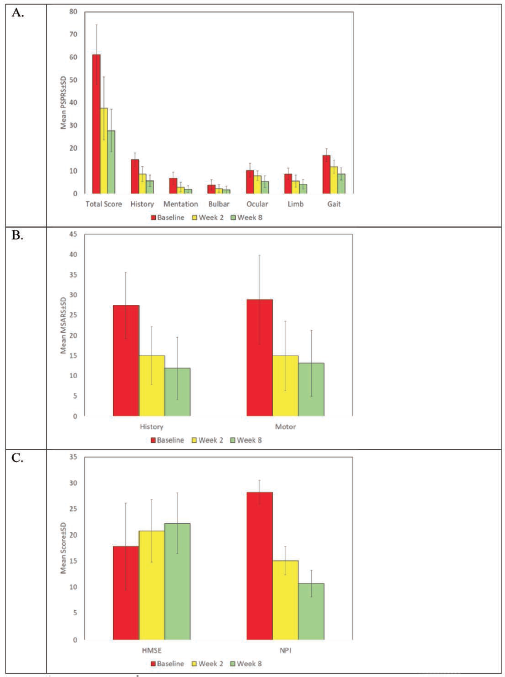
Table 2 shows a comparison of PSPRS of eight patients, showing a statistically significant decline in mean total score from baseline to week 2 and 8 (p < .001). The significance was highest for limb symptoms (p < .001), followed by ocular (p = .001), history (p = .003), gait (p = .007), mentation (p =.014) and bulbar (p = .018) symptoms. Similarly, the history and motor subscale mean score of MSARS in 13 patients significantly decreased on follow-up visits (p < .001). The autonomic symptoms were present in 12 out of 13 patients at baseline, which decreased to 3 in week 2 and 0 in subsequent visits. The baseline mean score of NPI and HMSE in 21 patients also had a significant decline in weeks 2 and 8 with p < .001 and p = .001, respectively (Figure 1). Table 2 represents the statistically significant change (p < .001) in the functionality of the patients with PSP and MSA on combination treatment over the follow-up visits based on the MSARS global disability subscale and Hoehn and Yahr scale.
Scores of PSP Rating Scale, MSA Rating Scale, HMSE, NPI Scale, Stages of MSARS Global Disability Subscale and Hoehn and Yahr Scale at Baseline and Post-treatment at Different Time Points.
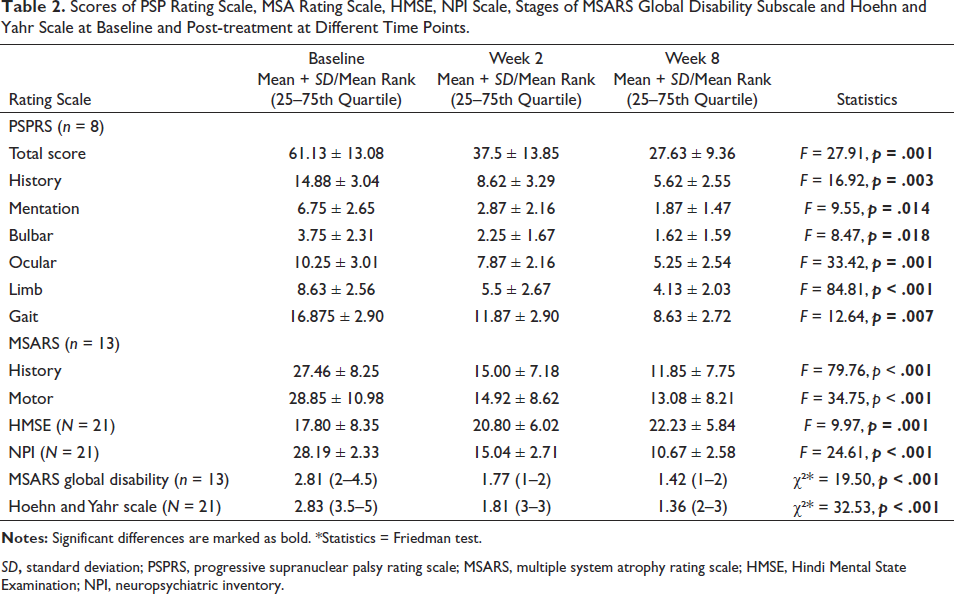
SD, standard deviation; PSPRS, progressive supranuclear palsy rating scale; MSARS, multiple system atrophy rating scale; HMSE, Hindi Mental State Examination; NPI, neuropsychiatric inventory.
Comparison of Severity Scale Scores of Patients at Baseline, Second- and Eighth-week Follow-up Intervals. (A) PSPRS Total and Domain Scores. (B) MSARS History and Motor Domain Scores. (C) HMSE and NPI Scores.
Scores of PSP Rating Scale, MSA Rating Scale at Week 8 and Last Documented Visit.
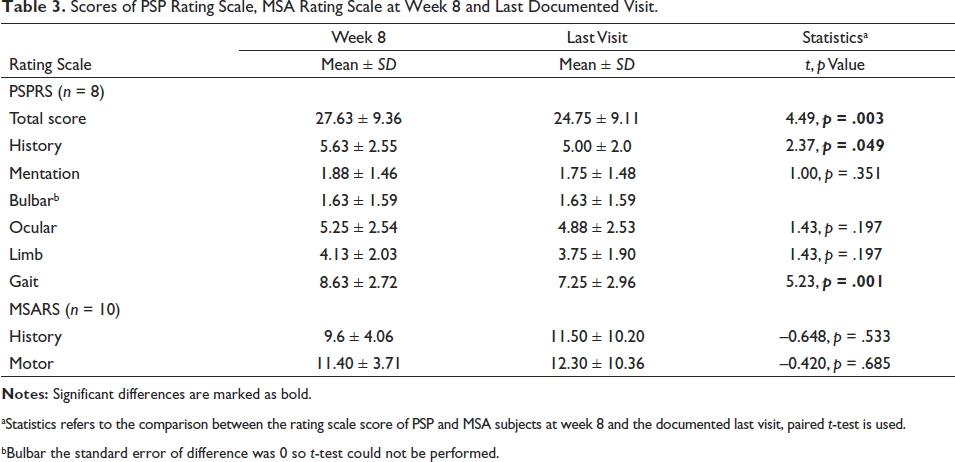
aStatistics refers to the comparison between the rating scale score of PSP and MSA subjects at week 8 and the documented last visit, paired t-test is used.
bBulbar the standard error of difference was 0 so t-test could not be performed.
Side Effects and Stability of Treatment Response
A sensation of heaviness of the body, nightmares sleep disturbances and nausea were reported by 19.05%, 9.52% and 4.75% participants, respectively, but the symptoms were short-lasting and did not require any treatment. The stability of treatment response was assessed by comparing the PSPRS and MSARS scores at week 8 and the non-fixed last documented visit (Table 3). In Table 3, the total mean score of PSPRS was significantly higher on week 8 in comparison to last visit (p = .003). However, besides the mean scores of history and gait, the mean score of other subscales of PSPRS and MSARS were comparable.
Discussion
The study was conducted to assess the effectiveness of combination treatment of sub-therapeutic low-dose amantadine and escitalopram in patients with PSP and MSA. Notably, this is the first study to report the use of combination treatment of an antidepressant with an antiparkinsonian medication in APD. Of the total 21 participants (8 PSP and 13 MSA), the average mean age of onset of PSP in our study was 55.77 years, which is comparable to other studies, but the onset of MSA was in the 70s, which is more than a decade later in comparison to patients in other studies.3, 9, 19, 20 This may be due to either failure in prompt recognition of non-motor symptoms or misdiagnosis as idiopathic PD. Interestingly, the study population comprised only one female each with the diagnosis of PSP and MSA, which is not in concordance with other studies as gender differences have not been reported in PSP and MSA.5, 9, 19, 20 However, a study has reported male preponderance (70%) in PSP, but MSA did not show any gender predilection. 3 The male preponderance in the current study can be attributed to differences in service utilisation among the Indian population rather than a gender predilection.
Of the total 21 participants, 10 were previously diagnosed with idiopathic PD, 6 with major depressive disorder and 5 with Alzheimer’s dementia, and this is in accordance with previous studies reporting a prior diagnosis of Idiopathic PD before patients were diagnosed with APD. 19 Misdiagnosis at the onset of PSP and MSA could be explained on the basis of overlapping symptomatology in APD, which can present with cognitive, neuropsychiatric or ophthalmological symptoms prior to the onset of frank motor symptoms. Notably, four participants in the PSP group reported ocular (blurring, difficulty in seeing downward and fixed gaze) symptoms as the first symptom. However, concerns with limb movement were found in two participants, and one participant reported gait abnormality and cognitive symptoms as the first symptom, which disapproves findings in previous studies where nuchal rigidity and bradykinesia were reported as the presenting symptoms along with dizziness, fall, postural instability, speech difficulty, memory problems and axial rigidity.7, 21 Interestingly, most of the participants with ocular symptoms at the onset of disease visited to ophthalmologist frequently with minimal or no benefit. In contrast, 7 out of 13 MSA patients presented with autonomic symptoms, followed by gait abnormality, limb involvement, ocular symptoms and cognitive impairment, which is not in accordance with other studies where motor symptoms were the most common initial manifestation, followed by autonomic failure, cerebellar ataxia and REM sleep behaviour disorder in MSA.7, 22 Contrasting findings in this study compared to previous studies highlight the need to reflect upon the non-motor symptoms for early diagnosis of PSP and MSA.
Various neurotransmitter abnormalities have been postulated in APD as PSP, including reduction of dopamine along with D2 receptor density in the nigrostriatal pathway, cholinergic deficiency due to involvement of the nucleus basalis of Meynert and pedunculopontine nucleus along with acetyl-cholinesterase deficiency, a moderate decrease in Gamma-aminobutyric acid (GABA) in the basal ganglia system, an increase in glutamate toxicity, upregulation of serotonin receptors and a decrease in adrenergic neurotransmitter in the basal ganglia.23 –28 However, loss of both pre- and post-synaptic dopaminergic transmission in preferential striatonigral areas, loss of GABAergic neurons and loss of brainstem neural control of the cardiovascular and respiratory centres lead to a constellation of clinical presentations in MSA. 29 Moreover, loss of striatonigral neurons and a decrease in both D1 and D2 receptor density in APD have been advocated as possible explanations for unresponsiveness to levodopa treatment, but the exact reason is yet to be elucidated.19, 30 Further, serotonin has a role in the modulation of basal ganglia, limbic system, brainstem structure and cerebellum, which might contribute to the pathophysiology of movement disorders. 31 Additionally, serotonergic neurotransmission plays an important role in autonomic dysfunction, mood, cognition and movement, which are the core symptoms of APD.32, 33 Considering the involvement of various neurotransmitters in APD, targeting more than one neurotransmitter can help alleviate several symptoms in patients with APD.
Escitalopram is a relatively pure SSRI that not only increases serotonin concentration at all doses but also has no or very less affinity for other receptors. 34 The elderly age group itself poses some specific challenges in terms of drug tolerability, side effects and efficacy. The dose of escitalopram used was 5 mg/day, which is below the minimum effective dose for depression with the purpose of increasing serotonergic transmission and to curtail the possible side effects. On the other hand, amantadine, a weak NMDA receptor antagonist, not only increases dopamine synthesis, but also blocks dopamine reuptake and protects the neurons through suppressing microglial activation as well as inducing glial cell-derived neurotrophic factor (GNDF) expression through astroglia, and potentiates the antidepressant actions of the antidepressants in combination.34, 35 Considering the pharmacokinetic property of amantadine and its use in the elderly, resulting in decreased dopamine receptors in the striatum along with dopamine neurotransmitters, the dosage was kept at 100 mg/day in two divided doses to avoid side effects. Moreover, amantadine has a risk-benefit ratio of 0.80 in PSP patients after levodopa, rendering it a safe molecule. 10
The PSP patients had significant improvement not only in the total PSPRS score but also in individual domains (history, mentation, bulbar, ocular, limb and gait) from baseline to 2nd and 8th week of the combination treatment, which is in concordance with previous studies reporting symptomatic improvement with amantadine in PSP patients.19, 20. Moreover, 43% of autopsy-confirmed cases of PSP reported improvement with amantadine at a dose of 200 mg/day, which is double the dose used in the current study. 7 Notably, the severity of MSARS also significantly improved at the 2nd and 8th week follow-up of the combination treatment, and this has been supported by a previous study reporting improvement in 61.5% of cases but on relatively high doses of amantadine. 7 Furthermore, use of parenteral amantadine on MSA patients with predominant ataxia and parkinsonism in a study revealed significant improvement in symptoms, as evidenced by the change in the MSA severity rating scale, but the treatment duration was less than a week.20, 36 In contrast, a placebo-controlled trial of 400–600 mg of amantadine per day in eight patients with levodopa non-responsive MSA reported no improvement on follow-up. 37 There have been a few anecdotal studies in the form of case reports, retrospective studies and clinical trials in the literature to elucidate the role of the antidepressant in parkinsonism, and some of the case reports have reported improvement in motor symptoms with low-dose bupropion and amitriptyline in elderly patients with PSP.6, 11, 38 Further, imipramine, amitriptyline, bupropion and fluoxetine had moderate to marked benefits in a retrospective study on PSP patients, but the dosage used was not mentioned in the study. 10 Similarly, a double-blind placebo-controlled trial of paroxetine 30 mg bid as an add-on to levodopa in patients with MSA had significant improvement in motor symptoms and speech but had a relatively short duration of follow-up of 2 weeks. 39 In contrast, no effect was seen on stridor and survival in MSA patients with SSRI in the study, but rather higher rates of falls and parkinsonism with SSRI, which is not in accordance with this study where the combination of escitalopram and amantadine not only helped to improve motoric symptoms but also had significant and sustained improvement in non-motoric symptoms of PSP and MSA. 9 The symptomatic improvement in our patients in both groups after initiation of combination treatment was very prompt, as the change was appreciated within 2 weeks and was sustained till the last visit, which is not in concordance with other studies. 7 The serotonergic regulation of neurotransmitters in the subcortical circuits and cerebellum, along with dopaminergic and NMDA antagonist properties of amantadine, can be a possible explanation for early and steady response in PSP and MSA patients. The longest duration of follow-up recorded was at the 21st month after the initiation of the treatment, with an average duration of approximately 8 months. The severity scores were comparable in the eighth week, and at the last assessment, and notably the gait, history and total scores in PSPRS showed considerable improvement even in the last visit. Similarly, amantadine therapy for a period of long follow-up of 40 months in patients with olivopontocerebellar atrophy had sustained improvement in motor function. 40
The neuropsychiatric symptoms, cognition and functional ability also showed significant improvement on follow-up visits at the 2nd and 8th week of the combination treatment. The effect on behavioural symptoms and cognition can be primarily attributed to SSRI, but amantadine, through its dopaminergic property, can also help alleviate behaviour, mood and subsequently cognitive symptoms. The improvement in functional ability itself depicts improvement in the broader domain of motor, cognitive and behavioural symptoms in PSP and MSA patients. There is a lack of literature in this context, as motor symptoms were given precedence over neuropsychiatric and cognitive symptoms, and the results are inconclusive.11, 38, 41
Strengths and Limitations of the Study
The retrospective design of the study is the major limitation of the study, but considering the low prevalence of APD in population and the considerable sample size in the study duration, the design of the study proved to be advantageous. The diagnosis of APD was based on clinical criteria, but there is a probability of misdiagnosis, and an autopsy is not confirmed for a definite diagnosis. The duration of the last visit of the patients was not uniform, so the stability of the response to combination treatment for a definite duration cannot be generalised. Data related to comorbidities and medication use by patients other than escitalopram and amantadine were not collected, and they could be potential confounders in the study. Uniform dosage and response to amantadine and escitalopram in patients with a diagnosis of PSP and MSA provide a definite treatment regime for these patients with limited treatment options.
Conclusion
In conclusion, the combination of low-dose amantadine and escitalopram is an effective treatment modality for difficult-to-treat APD patients. A long-term prospective study on this combination is further warranted to establish the treatment regime in APD patients.
Abbreviations
MSA, multisystem atrophy; PSP, progressive supranuclear palsy; PD, Parkinson’s disease; APD, atypical Parkinsonian disorders; NMDA, N-methyl-
Footnotes
Acknowledgements
The authors would like to thank the patients involved in the study, without whom this research would not be possible. The authors would also like to extend their sincere gratitude to faculties and residents of the Department of Geriatric Mental Health, as without their contribution to the patient’s assessment and record maintenance, this study was not possible.
Authors’ Contribution
S.M.T. designed the study, supervised the data collection, analyzed the data and critically revised the article. P.C. collected the data and wrote the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Ethics
Approval for audit and a ‘consent waiver’ were obtained from the Institutional Ethics Committee, King George’s Medical University, Lucknow (Ref. code: 122nd ECM IIA/P1), and the study was conducted in accordance with relevant regulations and guidelines.
ICMJE Statement
The authors declare that the manuscript is in compliance with ICMJE guidelines.
