Abstract
Background
Stress can affect the morphology and synaptic organization of the telencephalon. These structural changes at the cellular level can lead to the development of various psychopathologies.
Purpose
Given that the telencephalon plays a major role in stress responses, the current study aimed to investigate the role of Spirulina platensis as a neuroprotectant supplement in the early life of zebrafish in averting the alteration of synapse morphology in the telencephalon caused by chronic unpredictable stress (CUS) in the later stage.
Methods
5dpf larvae were divided into two groups: one group was fed with a commercial fish diet and a second group with a 1% Spirulina-supplemented diet for 90 days. After 90 days, the adult zebrafish were exposed to CUS with different chronic stressors for 15 days. The synaptic plasticity was evaluated by morphometric analysis of synapse in telencephalon of zebrafish by transmission electron microscopy.
Results
The ultrastructural study demonstrated the protective role of Spirulina in the CUS model as no significant alterations in the length of the active zone, postsynaptic density, and synaptic cleft were observed as compared to the control group in the CUS model.
Conclusion
Thus, suggesting that the Spirulina supplementation can avert the remodeling effect of stress on synapse ultrastructure.
Introduction
Neuroplasticity is a main characteristic of the brain which includes synaptogenesis and the functional changes it causes. This property enables the brain to respond adaptably to environmental challenges. 1 For instance in an animal model, the restraint stress or chronic unpredictable mild stress (CUMS) might cause a transformation of neuronal structure.2, 3 The forebrain is a key functional part of the brain that controls behavior by receiving and processing sensory data. The anterior region of the forebrain, that is, telencephalon is mainly associated with the control of emotion, memory, and social behavior.4, 5 According to Gray’s theory, two types of the synapse are involved in various neuronal cells of the forebrain, that is, type I (asymmetric) and type II synapses (symmetric). Type I synapses are the most common subtype and have an excitatory function. Whereas type II synapses are less common and inhibitory in function. The function and plasticity of synapses are related to the proteins that mediate the communication of neurons in their postsynaptic density (PSD).6, 7 Li et al. showed that chronic unpredictable stress (CUS) in rat animal model induced a considerable remodeling in Type I synapse involving a change in length of the active zone, synaptic cleft, and PSD. 8 Therefore, maintaining brain health is crucial to ensuring that it functions properly.
To prevent or lessen the effects of brain illnesses, one must maintain a healthy lifestyle and consume a nutrients rich diet. A healthy diet can influence and help prevent the formation of a range of neurological and mental conditions, especially when key chemicals required for brain function are not present. 9 Spirulina platensis (Arthrospira) is a planktonic, filamentous cyanobacteria with a plethora of high nutritional profile.10, 11 Among its several properties, it has neuroprotective advantages as reported in several in vivo studies on a variety of animal models of depression, neuroinflammation, neurodegeneration, and neurotoxicity.12–14 Despite having several neuroprotective properties, the role of S. platensis as a protectant of synaptic plasticity in zebrafish telencephalon against CUS has not been investigated so far. In order to better understand the synaptic plasticity in the telencephalon of zebrafish, we have looked at how stress affected the morphology of asymmetric synapses in the telencephalon of zebrafish that were fed normally and those that were given S. platensis supplements.
Methods
Animals and Feed Supplementation
Adult wild-type zebrafish (Danio rerio) were purchased from the local fish farm, raised, and bred in laboratory conditions for further experiments. 15 The 5dpf zebrafish larvae (n = 10/group) were divided into two groups: the first group was fed with commercial fish feed “Tetra Bits Complete” (Tetra GmbH, Germany) and the other one was fed a 1% Spirulina-supplemented diet (in commercial feed) ad libitum for 90 days. The fish were housed at 28 ± 2°C with a 14:10 h light/dark cycle, regular filtration, and aeration. All animals were kept in accordance with the “Guide for the Care and Use of Laboratory Animals” and the study was approved by Institutional Animal Ethics Committee, Panjab University (PU/45/99/CPCSCA/2017/32).
Exposure to Chronic Stress
After feeding for 90 days, adult zebrafish were exposed to different chronic stressors following the method of Piato et al. 16 Zebrafish were exposed to two distinct stressors daily for 15 days. For heat and cold stress, the zebrafish were transferred to new tanks having temperatures 33°C and 23°C, respectively, for 30 min. Restraint stress was given for 90 min by restraining each zebrafish in a 2 mL Eppendorf micro-centrifuge tube perforated at both ends to allow free flow of water. Chasing stress was given by chasing zebrafish by a net with constant slow and sometimes frequent movements for 8 min. For social isolation, each zebrafish was placed solely in separate beakers without any visual contact with its counterpart for 60 min. Overcrowding stress was given for 60 min by placing 10−15 fish in a 250 mL beaker containing 150 mL water. For alarm pheromone stress, epidermal cells washings of sliced skin of 10 donor zebrafish in 100 mL distilled water were added in a concentration of 0.5 mL to 5-L housing tank containing 12−15 fish for a period of 30 min. 17 Tank change stress was given by transferring the fish from one tank to another six times in succession. For dorsal body exposure stress, zebrafish were placed for 2 min in tanks having low water levels enough to expose the dorsal surface of zebrafish. Images of Convict cichlid (Amatitlania nigrofasciata) in close proximity for 60 min were exposed to zebrafish for predator stress. Although the fish were subjected to stressful situations, neither extreme suffering nor an unusual number of deaths was observed.
High-resolution Electron Microscopy
Brain samples were prepared for ultrastructural studies in accordance with the protocol proposed by Bozzola and Russell.
18
The zebrafish were euthanized in tricaine (500 mg/L), brain samples were excised, fixed in 4% glutaraldehyde (pH 7.2−7.4) for 2 h, microdissected and kept in 2% glutaraldehyde (overnight at 4°C). The fixed tissues were rinsed with Sorenson’s buffer (0.1 M, pH 7.4) and post-fixed in 1% osmium tetraoxide for 1 h at 4°C. The tissues were then washed and dehydrated in ascending series of acetone. Following infiltration and embedding in Beem capsules with spurr low viscosity media, tissues were polymerized at 60°C.
19
Using an ultra-microtome (Leica-Ultra cut EM UC 7), ultrathin sections (50−60 nm) were cut and stained using uranyl acetate and lead citrate. The sections were observed under High-Resolution Transmission Electron Microscope (HR-TEM, JEOL JM 2100 PLUS) at SAIF, CIL, Panjab University, Chandigarh, India. The asymmetric synapses were captured and the length of active zone (LAZ), width of synaptic cleft (SC), the thickness of postsynaptic density (TPSD), and the synapse curvature were analyzed using the open-source imaging software ImageJ (
Statistical Analysis
The data was statistically analyzed at 0.05 statistical significance level using the Mann-Whitney U test to compare the control and Spirulina-supplemented group before and after CUS, and the Wilcoxon signed-rank test was used to compare ultrastructural changes within groups before and after CUS. The results were expressed as mean ± SEM.
Results
In the present study, the effect of 15 days of exposure to CUS on neuronal plasticity in the zebrafish brain of the commercial diet group and Spirulina-supplemented group was investigated. In the ultrastructural study of zebrafish brain before CUS exposure, the control group (Figure 1a) and Spirulina-supplemented group (Figure 1c) showed similar morphometry of asymmetric synapse in the telencephalon (Figure 1a and c) when compared with Mann-Whitney U test, no significant change in the LAZ (U = 146, p = 0.149) (Figure 2c), the width of SC (U = 162, p = 0.314) (Figure 2d), the TPSD (U = 147, p = 0.157) (Figure 2e), and synaptic curvature was observed (U = 155, p = 0.231) (Figure 2f). After 15 days of CUS exposure, control group (Figure 1b) showed a significant increase in the LAZ (p = 0.006) (Figure 2c) and TPSD (p < 0.001) (Figure 2e). However, a significant reduction in the width of SC (p < 0.001) (Figure 2d) but no change in synaptic curvature (p = 0.709) (Fig. 2f) was observed. As compared to the commercial diet group, Spirulina-supplemented group showed no change in the morphometry of synapse (Figure 1d) with no significant change in the LAZ (p = 0.794) (Figure 2c), the TPSD (p = 0.156) (Fig. 2e), the width of SC (p = 0.736) (Figure 2d), and synaptic curvature (p = 0.881) (Figure 2f). After 15 days of CUS exposure, when both groups, that is, commercial diet group and Spirulina-supplemented group were compared with Mann-Whitney U test, the control group showed a significant increase in the LAZ (p = 0.026) (Figure 2c) and TPSD (p < 0.001) (Figure 2e). It showed a significant narrowing of SC (p < 0.001) (Figure 2d) with no change in synaptic curvature (p = 0.301) (Figure 2f) compared to the Spirulina-supplemented group. As 15 days of CUS exposure had no effect on Spirulina-supplemented group, thus Spirulina supplementation might offer substantial neuroprotection against stress. Thus it can be concluded that Spirulina supplementation prevents any structural changes in the synapse of the telencephalon which were caused due to 15 days of CUS exposure in zebrafish.

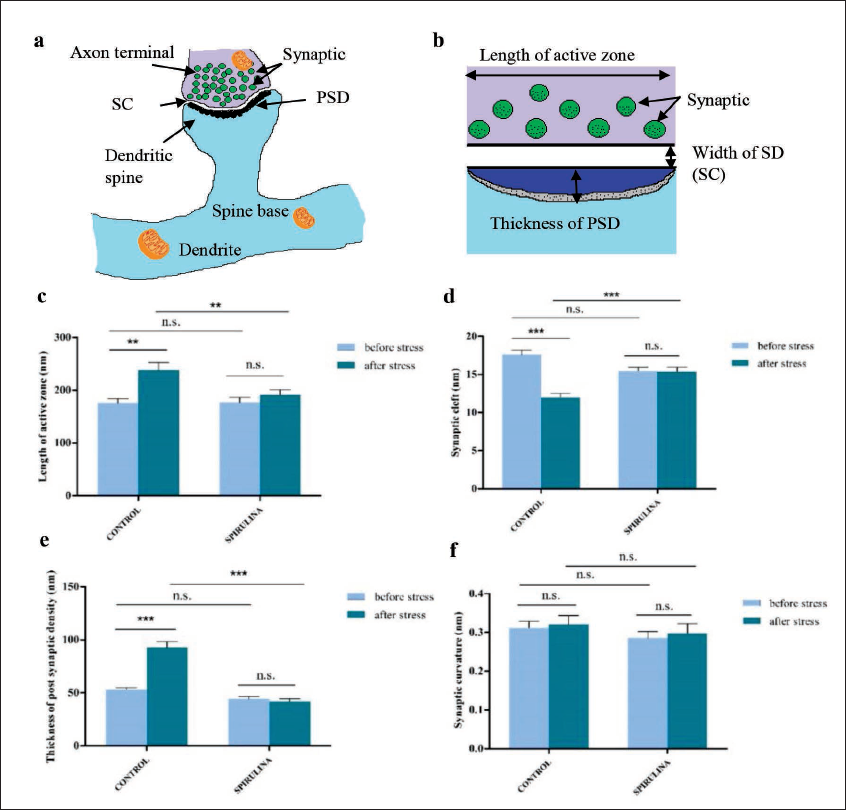
(a) Schematic representation of asymmetrical synapse; (b) schematic illustration of the measures of synaptic structure such as the LAZ, width of SC, and the TPSD; (c) CUS resulted increased LAZ (in nm) in control group (p = 0.006) and without any effect on S. platensis supplemented group; (d) CUS caused the narrowing of SC (in nm) in control group (p < 0.001), whereas, S. platensis supplemented group remained unaffected; (e) CUS increased the TPSD (in nm) (p < 0.001) in control group without any alteration in S. platensis supplemented group; and (f) synaptic curvature remained unaffected in both control and S. platensis supplemented group after 15 days of CUS exposure. Data represented as mean ± SEM, n = 16 (total number of synapses studied), GraphPad Prism v5 software. n.s., not significant; CUS, chronic unpredictable stress; SC, synaptic cleft; LAZ, length of active zone; PTSD, thickness of postsynaptic density. *p < 0.05; **p < 0.01; ***p < 0.001.
Discussion
The current study investigated the shielding role of S. platensis in averting the effect of stress on synaptic plasticity. The goal of the current study was to determine whether the S. platensis supplementation to zebrafish from early juvenile stages to the adult stages has a neuroprotective effect on the morphology of asymmetrical synapse against 15 days of CUS exposed at a later stage. This was assessed by studying the morphometry of asymmetric synapses in the telencephalon of zebrafish at the ultrastructural level.
In this study, 15 days of CUS exposure to a commercial diet-fed control group resulted in altered synaptic morphology, explained by the increased LAZ and TPSD, and decreased width of SC. No change in synaptic curvature was observed. The presynaptic and postsynaptic component of a synapse is curved rather than flat which is known as synaptic curvature. It ensures released neurotransmitters reach their intended target rather than diffuse to other areas. 21 This observation was similar to Lalert et al. in which a long-term treatment with paracetamol (15 and 30 days) to rats induce structural changes in the active zone and synaptic cleft with no changes in synaptic curvature of the synapse. 22 Thus, from the present reported observation 15 days of CUS may not be enough to alter the synaptic curvature. In contrast, the supplementation of S. platensis prevented all these structural abnormalities in the asymmetric synapse of zebrafish induced with CUS. The 15 days of chronic stress, failed to alter the morphology of asymmetric synapses in S. platensis-supplemented group. It showed no significant variations in the LAZ and TPSD, the width of SC and synaptic curvature. These results were consistent with the findings of Li et al. that showed chronic stress and escitalopram can alter the synapse structure in the brain and reconstruct synaptic ultrastructure in the CUMS model of rats. 8 However, these structural remodeling of the synapse may vary with animal species, sex, brain part, and the type of stress.2, 3 Therefore, more studies are needed to understand the relationship between synaptic plasticity and the role of S. platensis in the development of resilience against stress.
Conclusion
In conclusion, CUS caused changes in the morphometry of synapses in the telencephalon region of the zebrafish. However, early life supplementation of S. platensis acted as a shield against stress and prevented these changes from occurring. Therefore, these findings suggest that early supplementation of S. platensis can be used as a therapeutic agent to reduce the negative consequences of stress in a later stage.
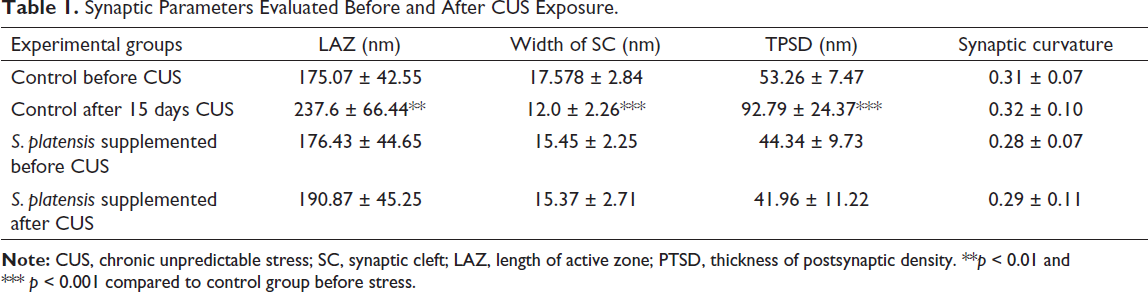
Synaptic Parameters Evaluated Before and After CUS Exposure.
Footnotes
Acknowledgements
We are grateful to the Department of Zoology, Panjab University, Chandigarh, for providing research facilities, the Sophisticated Analysis Instrumentation Facility (SAIF), CIL, Panjab University, Chandigarh, for ultrastructural studies, and the National Centre for Conservation and Utilization of Blue Green Algae (NCCUBGA) and the Indian Agricultural Research Institute (IARI), New Delhi, for providing Spirulina platensis culture.
Authors’ Contribution
HK: conceptualization; data curation; formal analysis; methodology; resources; software; roles/writing − original draft; writing − review & editing. VG: methodology; writing − review & editing. KK: conceptualization; supervision; validation; visualization; resources; writing − review & editing. RK: conceptualization; supervision; validation; visualization; methodology; resources; writing − review & editing.
Statement of Ethics
The study was approved by Institutional Animal Ethics Committee, Panjab University (PU/45/99/CPCSCA/2017/32).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed the receipt of following financial support for the research, authorship, and/or publication of this article: This research is supported by the Indian Council of Medical Research (ICMR), New Delhi, India, for financial support as an ICMR-JRF-2016 fellowship (No. 3/1/3/JRF-2016/HRD, Dated: 16-09-2016) and Panjab University for partially funding the publication.
ICMJE Statement
I/we had full access to all of the data in this study and take the responsibility for the integrity and accuracy of the data.
