Abstract
Background
Gamma-aminobutyric acid (GABA) is the main inhibitory neurotransmitter in the mammalian brain and is a non-proteinogenic amino acid. Doxorubcin (DOX) or adriamycin is one of the most potent chemotherapy drugs for breast cancer.
Purpose
This study focused on diminishing the brain injury and neurotoxicity of doxorubicin (DOX) by GABA administration.
Methods
Rats were randomly divided into four groups (8 rats each), which were the control group, DOX group (3 mg/kg for 4 weeks, then 2 mg/kg for 2 weeks), GABA group (2 mg/kg for 21 days), and DOX + GABA group (treated as the second and third groups). Neurotoxicity and brain injury were assessed by determining CSF biomarkers, serum inflammatory markers, and histopathological evaluation of the cerebral cortex.
Results
DOX treatment significantly increased the levels of all CSF biomarkers (S100B, IL-1β, ubiquitin carboxy-terminal hydrolase L1 (UCH-L1), glial fibrillary acidic protein (GFAP), spectrin breakdown products (SBDP145), and C-C motif chemokine ligand 2 (CCL2) and all inflammatory markers (IL-6, TNF-α, and IFN-γ), causing extensive neutrophilic infiltration and great alteration in the cerebral cortex architecture as evidence of neurotoxicity. The oral administration of GABA significantly reduced the levels of all CSF biomarkers and inflammatory markers and restored the normal architecture of the cerebral cortex, with observed ameliorations in neutrophilic infiltration.
Conclusion
GABA administration can ameliorate neurotoxicity and protect the brain against the negative effects of DOX treatment.
Introduction
Gamma-aminobutyric acid GABA (γ-aminobutyric acid) is the main inhibitory neurotransmitter in the mammalian brain and has been involved in the control of excitability, information processing, plasticity, and the synchronization of neural activity. 1 Low GABA levels in the blood indicate a brain disorder; 2 thus, GABA plays a vital role in humans, principally in the central nervous system.
Outside the CNS, GABA has a physiological role in different systems, including the regulation of circulatory functions, 3 the suppression of the metastasis of cancer cells, 4 the balancing of pituitary functions and growth hormone release in rats, 5 the fertilization of mammals, 6 and the excretory modulation function. 7 In addition, it has been well established that GABA or GABA-mimic compounds, such as the amino acid L-theanine, play a major role in stabilizing mood disorders. 8
Antitumor drugs such as doxorubicin (DOX) have strong effects on various malignance cells. 9 DOX is a prototype active ingredient that produces reactive oxygen species and, in the presence of molecular oxygen, delivers the reactive superoxide radical anion (O•2) through redox cycles of its quinone component. 10 Oxidative damage increases the formation of tumor necrosis factor-alpha (TNF-α) in peripheral tissues. This proinflammatory cytokine penetrates the blood brain barrier (BBB) and leads to CNS toxicity, including further TNF-α formation in the brain, oxidative and nitrosative damage to the most important biomolecules (proteins, lipids, DNA, and RNA), dysfunction of the mitochondria, and neuronal death. 10 Neurotoxicity is related to the early onset of high levels of serum cytokines such as IL-6, IFN-γ, and TNF-α that can pass through the BBB. 11
Methods
Experimental Animals
At the Breeding Animal House of the Faculty of Medicine, Zagazig University, Egypt, 32 healthy adult male Wistar albino rats were collected and kept. They were between the ages of 12 and 14 weeks and weighed 180–200 g. Animals were housed for 1 week of acclimation in plastic cages with stainless steel wire-bar covers at a regulated humidity level of 55 ± 5% and temperature of 23 ± 1°C with a 12:12 h light:dark cycle in a completely chemical-free room. They were fed, given unrestricted access to it, and allowed to freely consume water. All rats were treated with the approval of the chair of the zoology department under the principles of humane care, in accordance with the regulations of Zagazig University’s Ethical Committee (ZU-IACUC committee) and the NIH Guidelines for the Care and Usage of Laboratory Animals (Science Faculty, Zagazig University).
Experimental Design
The rats were categorized into Groups I–IV as follows:
At the end of the experiment, all rats were anesthetized with intraperitoneal injections of 25 mg/kg sodium thiopental. 14 CSF was extracted. 15 Rats were killed to recover their brains, and blood samples were also taken. 16
Experimental Procedures
CSF Collection Technique
The animals were placed on a sterile surface and anesthetized with intraperitoneal injections of 25 mg/kg sodium thiopental. Once the rat was anesthetized, its neck was shaved and flexed by placing the head at an angle of 90°, enabling visualization of a diamond-shaped surface between the occipital bone and the cervical column. A 24-G needle was inserted at a perpendicular position (deep 0.5 and 1 mm) with the oblique edge upward. Two researchers must perform this step, one fixing the animal in the right position and the other introducing the needle. CSF was collected with a syringe. The CSF was transferred to an Eppendorf tube and kept frozen at –80°C. After collecting all the required samples, the animals were sacrificed. 15
Blood Collection Technique
Under general anesthesia, blood samples were taken from the retro-orbital venous plexus using a tiny, heparinized capillary tube inserted into the rat’s medial epicanthus. 16 Blood samples were drawn into a clean, graduated centrifuge tube, allowed to clot for 10 min at room temperature, and then centrifuged for 20 min at 5000 rpm using a Remi cold centrifuge. For the purpose of determining the biochemical parameters, the serum supernatant was collected in a sterile tube.
S100 β Protein Determination in the CSF
Enzyme-linked immunosorbent assay kit for S100 was obtained from CUSABIO Inc. 17
IL-1β Determination in the CSF
The level of IL-1β in the CSF was measured with a commercially available sandwich ELISA. 18
UCH-L1, GFAP, SBDP145, and CCL2 Determination in the CSF
Enzyme-linked immunosorbent assay analysis of CSF samples was performed using CUSABIO Inc., ELISA kits.
IL-6 Determination in the Serum
Interleukin-6 was determined by ELISA according to Allen et al. 19 using the rat interleukin-6 kits.
TNF-α Determination in the Serum
TNF-α was determined by ELISA according to Barbuio et al. 20 using the rat interleukin-6 kits.
IFN-γ Determination in the Serum
IFN-γ was determined using the ELISA kit for IFN-γ from CUSABIO Inc.
Histopathological Study
All procedures were carried out at Zagazig University’s Faculty of Medicine’s Histology and Cell Biology Department. All animal brain samples were embedded in paraffin wax, dehydrated, and fixed in 10% neutral formal saline before being processed into 5-m-thick slices. Hematoxylin and eosin were used for the staining. In an image analysis unit, light microscopy was used to examine stained slides. 21
Statistical Analysis
The mean and standard deviation were used to express the study’s data. The Statistical Package for the Social Sciences software, version 13.00, was used to conduct statistical analysis (Chicago, Illinois, USA). For discrepancies between the means of the groups under study, one-way ANOVA followed by multiple group comparisons using the post hoc Tukey test to determine statistical significance. Statistics were expressed as probability values
Results
S100B Concentrations in the CSF
The S100B values of rats in the DOX group were significantly greater than those of healthy control rats (
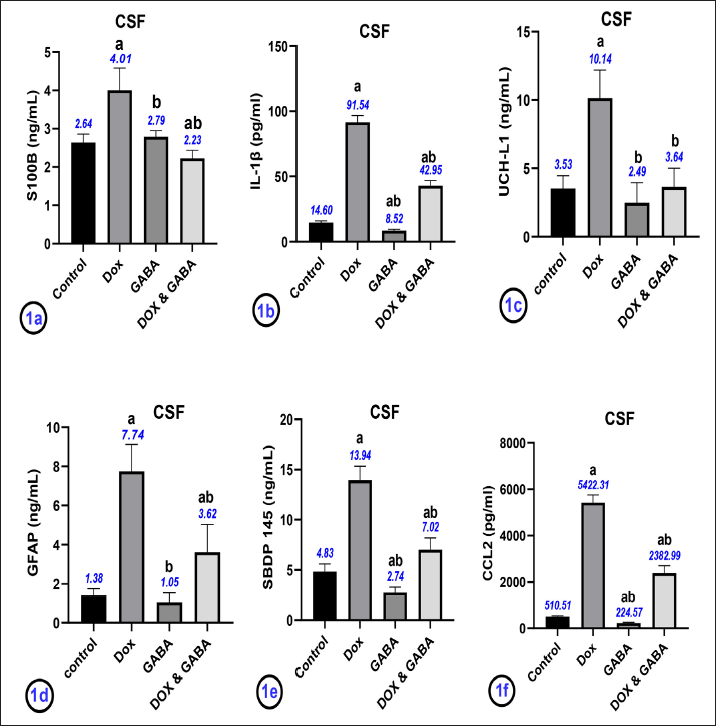
IL-1β, UCH-L1, GFAP, SBDP145, and CCL2 Concentrations in the CSF
As shown in Figure 1b–1f, rats in the DOX group had significantly increased levels of IL-1β, ubiquitin carboxy-terminal hydrolase L1 (UCH-L1), glial fibrillary acidic protein (GFAP), spectrin breakdown products (SBDP145), and C-C motif chemokine ligand 2 (CCL2) in the CSF, as detected by ELISA (
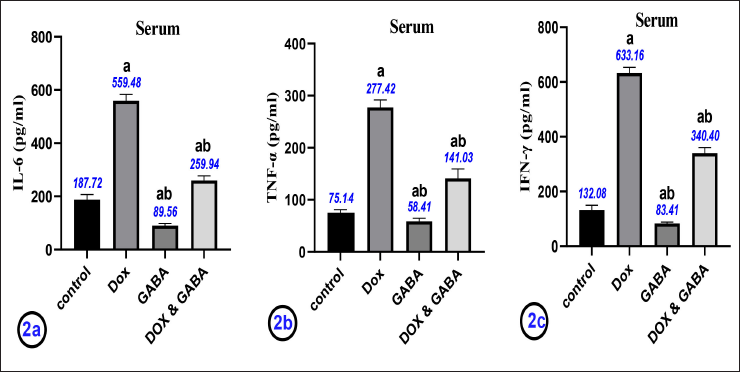
IL-6, TNF-α, and IFN-γ Concentrations in the Serum
The levels of serum IL-6, TNF-α, and IFN-γ in the DOX group were significantly higher than in the control group (
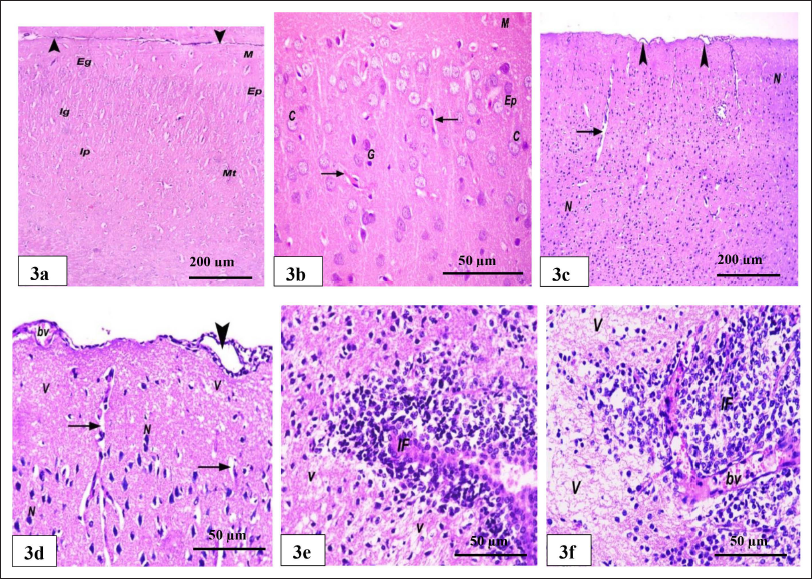
Histopathological Results
Light microscopic examination of the control group showed no detectable differences from the normal histology of the cerebral cortex of healthy rats. H&E-stained sections of the cerebral cortex of the rats in the control group revealed normal cranial architectures where molecular, external granular, external pyramidal, internal granular, internal pyramidal, and multiform, which is the deepest layer, are the six levels that are definitely identified. Multiform is the sixth layer. The dura mater seems to be healthy. It was possible to see cortical neurons with spherical, pale nuclei that were encircled by basophilic cytoplasm. Different varieties of neuroglia and blood arteries with condensed perivascular gaps were visible in intercellular neuropils (Figure 3a and 3b). The rats in the DOX group showed disruptions of the layers of the cerebral cortex, with the layers not clearly defined. When compared with the rats in the control group, blood vessels with a slightly wide perivascular space were observed. A higher magnification of the previous section showed the outer portion of the cerebral cortex. The dura mater, with separations and dilated blood vessels, is visible. Most of the neurons had darkly stained nuclei bounded by white spaces. Wide perivascular spaces surround vacuolated neutrophils and blood vessels compared with control (Figure 3c and 3d). Also, in the DOX group (Figure 3e and 3f), the cerebral cortex showed neutrophils with extensive vacuolations. An area of extensive mononuclear inflammatory cellular infiltration around congested, dilated blood vessels is well visible.
The GABA group showed the six layers of the cerebral cortex, as in the control group (Figure 4a and 4b). The DOX + GABA group showed some improvements in the layers of the cerebral cortex compared with the DOX group; however, the layers were still not easily identified. Its dura mater appears normal. Few blood vessels and little white matter are noticed (Figure 4c). Also, in the DOX + GABA group, H&E-stained sections confirmed the restoration of the normal architecture of the six layers, defined as the molecular, external granular, external pyramidal, internal granular, internal pyramidal, and multiform layers. The blood vessels were in longitudinal sections with slightly wide perivascular spaces. Its dura mater appears with little separation. Normal cortical neurons with rounded, pale nuclei and basophilic cytoplasm were present. Distinct types of blood arteries and neuroglia were visible in the intercellular neuropils, which also had slightly wider perivascular gaps (Figure 4d and 4e).
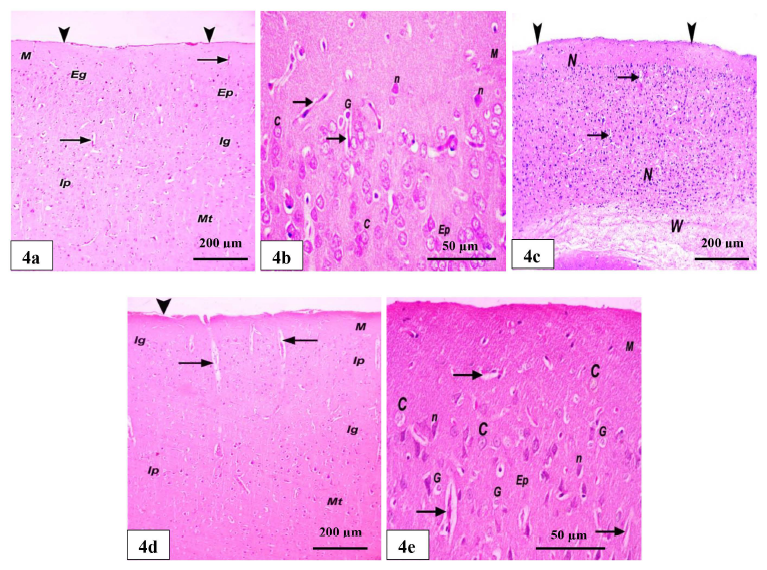
Discussion
Protein biomarkers are biological molecules that are present in brain cells and that enter the CSF or serum after the brain cells have been damaged. 22 Examples of protein biomarkers are the glia proteins S-100 beta (S100), the fibrillar glia protein (GFAP), and the carboxy-terminal hydrolase of ubiquitin (UCH-L1). S100 is the major low-affinity Ca-binding protein in astrocytes and is reflected in the activation, injury, or death of astrocytes. 23 For the past decade, molecular biomarkers derived from acute neuronal, axonal, glial, and endothelial injuries have been measured in bio-fluids such as CSF and blood. They have received a lot of attention as promising candidates for the diagnosis and prognosis of traumatic brain injuries. Biomarkers for structural damage such as NF-L, MAP2, UCHL1, GFAP, and S100B were identified and examined.24, 25
In the current study, DOX treatment caused a significant increase in the CSF S100B, SBDP145, CCL2, GFAP, and UCH-L1 concentrations; however, GABA administration caused a significant decrease in the levels of these parameters to be approximately close to the standard levels of the control group. These findings are in line with those of Babets et al., 26 administering DOX increases the concentration of S-100B in the hippocampus, thalamus, and cerebral cortex by 20%–26% compared to control rats. Babets et al. proposed the hypothesis that the increase in S100B and GFAP could occur due to the progression of the ischemic state under the influence of DOX treatment.
The principal calpain degradation product SBDP145’s spatiotemporal distribution characteristics in rat brain tissue are consistent with results from studies utilizing comparable neurotoxicity models. 27 Rodent models of the toxicant-induced, selective loss of hypothalamic dopamine neurons seen in Parkinson’s disease have shown overexpression of UCH-L1. 28
According to the following study, GABA signals affected S100B secretion in astrocyte cultures and hippocampus slices, explaining how it affects the aforementioned parameters.
29
In astrocytes, glutamate and GABA transporters can raise intracellular Na+ levels, which in turn raise Ca2+
In the present study, DOX treatment caused significant elevations in serum inflammatory markers such as IL-6, TNF-α, and IFN-γ. In contrast, GABA administration caused a significant downregulation in the levels of these parameters to be approximately near the normal levels of the control group. These results are in line with those of Liao et al., 35 who stated that DOX motivated the formation of TNF-α, subsequently causing the stimulation of NF-B and iNOS and increasing the gene expression of IL-1 and IL-6 as well as the protein expression of p65, which shows severe inflammation in the hippocampus. These inflammatory conditions can lead to nerve death and behavior changes. TNF-α, which crosses the BBB, causes increased TNF manufacture by activating glial cells, which leads to mitochondrial damage. 36 The continued stimulation of glial cells and the further increase in TNF-α play a role in chronic inflammation, which can lead to neuronal death or neurodegenerative diseases. Therefore, an increase in TNF levels could be a possible linkage between DOX-induced oxidative stress and CNS destruction. 37 Clinical doses of DOX can increase IL-1 and IL-6 expression and levels. 38 In addition, sarcoma, melanoma, and leukemia tumor cells secrete IFN-α in response to DOX treatment. 39
In agreement with the results of this study, administration of GABA reduced levels of inflammatory cytokines such as TNF-α, IFN and IL-12 in colitis mice. GABA has opposite effects on immune and inflammatory damage in colitis vs. autoimmune syndromes, although it elicits the same inhibitory effect on proinflammatory cytokines. GABA suppresses pro-inflammatory cytokines in tissues and increases the expression of anti-inflammatory cytokines. 40 In addition, GABAA activation has anti-inflammatory effects in some pathological situations. 41 Chronic inhibition of the GABAA receptor in rats induced mild peripheral inflammation that increased IL-6 and decreased IL-10. 42
The histopathological investigation also confirmed that DOX produced severe structural abnormalities in the brain tissue. The cerebral cortex showed neutrophils with extensive vacuoles, an area of extensive mononuclear inflammatory cellular infiltration around clogged dilated blood vessels, results that are in line with the findings of Ramalingayya et al. 43 Also, Zickri et al. 44 explained that large vacuoles were seen in the superficial layers of the cerebral cortex after treatment with adriamycin (DOX), which also produced a partial separation of the surface from the deeper layers. In the deeper layers, many vacuoles filled with cell debris were found. Several of the undersized neurons exhibited vacuoles surrounding their dark nuclei.
Treatment with adriamycin (DOX) makes the brain mitochondria more vulnerable to oxidative stress, which puts the brain cells at risk of senescence and death. 45 When delivered alone or in combination with other active ingredients, DOX can harm healthy, non-cancerous cells, which can result in cognitive problems brought on by chemotherapy. 46 Histological results of the DOX + GABA group confirmed the restoration of the normal architecture of the six layers of the cerebral cortex after GABA treatment. There is not much separation in the dura mater. Cortical neurons with spherical, light-colored nuclei were also seen. Different forms of neuroglia and blood vessels with slightly wider perivascular gaps can be seen in intercellular neuropils. This is consistent with the findings of Brailowsky et al. 47 who observed that, when GABA was injected into the somatomotor cortex of both young and old rats, a progressive restoration of motor functions occurred. The latter phenomenon’s precise mechanism is unknown. Tolerance to the effects of similarly administered chronic GABA infusions has been reported at the cellular level, suggesting that desensitized or downregulated GABA receptors may have such an effect. 48
Conclusion
GABA demonstrated protective and recreational effects against DOX toxicity on CSF biomarkers and the cerebral cortex of the brain. This study also showed that GABA has anti-inflammatory properties, which mitigated the DOX-induced inflammation. As a result, it is advised that GABA be supplied while receiving DOX, but carefully under supervision for the other systems, such as the GIT, CVS, and hepatic system, as further research is needed to fully understand how GABA affects these systems.
Footnotes
Abbreviations
Acknowledgements
I am appreciative of those who taught me the importance of education and the benefits and chances it may provide. In particular, I am grateful to my father, who gave me his passion, support, and innovative insight. His insightful comments on the paper helped me improve this thesis’s conceptualization, and I sincerely appreciate his keen interest in the subject and unwavering support.
Authors’ Contribution
HA conducted the physiological, biochemical, and anatomical studies; participated in the sequence alignment; and drafted the manuscript.
Availability of Data Materials
On Mendeley Data (
Statement of Ethics
The Zagazig University Ethical Committee and the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals were followed, and all rats received humane treatment according to those guidelines.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
