Abstract
Background
Sequential changes in brainstem and spinal cord neurons after traumatic injury to peripheral nerves are related to neuropathic pain symptoms.
Purpose
This study was conducted to elucidate the influence of nerve insult on stimulus-induced c-Fos expression and ERK phosphorylation by brainstem neurons.
Methods
The brainstem trigeminal sensory nuclear complex (BTSNC) was examined for neuronal profiles immunolabeled with c-Fos and phosphorylated ERK (p-ERK) antibodies elicited by stimulation of the tongue with capsaicin after lingual or inferior alveolar nerve (IAN) injury.
Results
Abundant neuronal profiles immunolabeled for c-Fos and p-ERK elicited by capsaicin were distributed in the spinal trigeminal nucleus caudalis (Vc) without nerve injury. The spinal trigeminal nucleus oralis (Vo) contained limited numbers of these neuronal profiles after stimulation of the tongue. A significant reduction of these neuronal profiles in the ipsilateral Vc was detected after lingual nerve injury. After IAN injury, an increased number of neuronal profiles immunolabeled for c-Fos elicited by capsaicin was noted, while that of p-ERK was left unchanged in the ipsilateral Vc. On the both sides of the Vo, an increased number of capsaicin-induced neuronal profiles immunolabeled for c-Fos and p-ERK was detected after lingual or IAN injury.
Conclusion
Differential effects of lingual or IAN injury on stimulus-induced c-Fos expression and ERK phosphorylation by Vo and Vc neurons may be involved in the complex nature of symptoms of trigeminal neuralgia.
Keywords
Introduction
c-Fos is known to be one of the activation markers of neurons.1, 2 Noxious stimuli-induced c-Fos expression by medurally and spinal neurons is enhanced after nerve injury when a stimulus is applied to the sites surrounding the denervated area.3–5 We previously reported anomalous nociceptive inputs from intact nerves after nerve injury, which appeared to be involved in hyperalgesia of the peripheral tissue innervated by the intact nerve, but close to the injured nerve.6, 7 Therefore, the induction of c-Fos has been utilized to indicate abnormal sensory processing as well as normal nociceptive responses.2, 8, 9
Extracellular signal-regulated kinase (ERK) is a specific subtype of mitogen-activated protein kinases (MAPKs). 10 ERK is activated by MEK phosphorylation and activated ERK plays a pivotal role in neuronal plasticity, which is responsible for several functions in the nervous system.11, 12 Phosphorylated ERK (p-ERK) has also been demonstrated to be induced in brainstem and spinal cord neurons following noxious stimuli.13, 14 However, temporal changes in induction by noxious stimuli markedly differ from c-Fos. Stimulus-induced c-Fos is detectable after 30 min and reaches the peak after approximately 2 h. By contrast, the induction of p-ERK is detectable within a few minutes and reaches the peak after approximately 5 min.13, 15 A previous study indicated that p-ERK regulated the expression of IEGs. 16 The expression of c-Fos as well as ERK phosphorylation elicited by mechanical pressure were diminished by pretreatment with intrathecal MEK 1/2 inhibitor. 17 Hence, ERK phosphorylation appears to contribute to c-Fos expression. Nevertheless, intracellular signal transduction pathways involved in c-Fos expression following peripheral nerve injury have not yet been elucidated in detail.
The brainstem trigeminal sensory nuclear complex (BTSNC) transmits somatosensory information from the orofacial region. The BTSNC consists of the trigeminal principal sensory nucleus (PrV) and the trigeminal spinal tract nucleus. Three subdivisions, the spinal trigeminal nucleus oralis (Vo), interpolaris (Vi), and caudalis (Vc) make up the trigeminal spinal tract nucleus. The Vc has an exclusive function in orofacial nociceptive transmission.18, 19 It has been reported that subdivisions of the BTSNC other than the Vc are also involved in orofacial nociceptive responses.20–26 The enhanced excitability of Vc neurons induced by nerve injury has been identified as one of the factors involved in the development of trigeminal neuralgia.4, 27, 28 On the other hand, changes in rostral subdivisions of the BTSNC after nerve injury have not yet been examined in detail. Therefore, we herein investigated neurons in the BTSNC for stimulus-induced c-Fos expression and ERK phosphorylation 14 days after injury to the lingual nerve (LIN) or inferior alveolar nerve (IAN), when the excitability of spinal neurons has been reported to be enhanced after sciatic nerve injury.29–31
Methods
Surgery
Male Sprague-Dawley rats (200–250 g) were anesthetized with 4% isoflurane/O2 inhalation and maintained on 2% isoflurane/O2 for surgery. In the right LIN transection (LIN-TS), a skin incision in the submandibular region and retraction of the muscles of the oral floor were made to expose the right LIN. The exposed nerve was firmly ligated using 7-0 silk and transected by fine scissors just distal to the ligation (n = 10). In the right IAN transection (IAN-TS), a facial skin incision and retraction of the masseter muscle were made to reveal the lateral aspect of the mandible. The lateral surface of mandible was drilled off to expose the IAN. The exposed nerve was firmly ligated using 7-0 silk and transected by fine scissors just distal to the ligation (n = 10). As sham-operated controls, the right LIN or IAN was exposed without further procedures (n = 10 for each sham-operated group).
The Expression of c-Fos and ERK Phosphorylation Elicited by Capsaicin
Naïve rats (n = 6) and nerve-injured or sham-operated rats 14 days after surgery were anesthetized with an i.p. injection of pentobarbital sodium (40–50 mg/kg). Using a micropipette, a 10-µl of capsaicin (Wako Co., Japan), absorbed at 1.0% in a vehicle (10% Tween 80, 10% ethanol, and 80% saline), was dripped onto the surface of the tongue. The application of capsaicin was carefully performed to avoid contact with the micropipette tip to oral tissues except for the tongue. 32 Two rats in each surgery group applied the same volume of the vehicle used as sham-stimulated controls.
Rats were perfused intracardially with saline 5 min (n = 5 after each surgery and 3 naïve rats; for p-ERK) or 2 h (n = 5 after each surgery and 3 naïve rats; for c-Fos) after the application of capsaicin, followed by 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4. The brainstem was dissected out and further fixed in a fresh volume of fixative at 4°C for 24 h, and then replaced with 20% sucrose at 4°C. Frozen sections (40 µm) of the lower brainstem including the BTSNC were cut on a transvers plane, and alternate series of sections were serially collected in phosphate-buffered saline (PBS), pH 7.4. The peroxidase anti-peroxidase (PAP) method was used for immunohistochemical staining. 3 Sections were blocked with 3% goat serum in 0.3% Triton X-100 at room temperature (RT) for 1 h and incubated with primary antibodies at 4°C for 72 h. Commercially available rabbit polyclonal antibodies for p-ERK (1:8,000; Cell Signaling, Beverly, MA, USA) and c-Fos (1:8,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA) were used as primary antibodies. After a series of PBS rinses, sections were incubated for 1 h at RT with peroxidase-conjugated goat anti-rabbit IgG (1:600; Rockland, Gilbertsville, PA, USA), followed by several PBS rinses, and then incubated with the PAP reagent (1:3,000; Rockland) at RT for 1 h. Sections were reacted for 10 min with 0.002% 3,3′-diaminobenzidine-tetrahydrochroride (DAB) in the presence of 0.006% hydrogen peroxide in 0.05 M Tris buffer, pH 7.4, intensified by 0.08% nickel ammonium sulfate.
The number of immunolabeled neuronal profiles was counted. Horizontal bar charts were used to summarize the rostrocaudal distribution by plotting the number of immunolabeled neuronal profiles sorted rostrocaudally. For statistical analyses, five consecutive sections containing the largest number of immunolabeled neuronal profiles were picked for each subdivision of the BTSNC.
Statistical Analysis
Summarized data in each surgery group are represented as means ± SEM. The Student’s t-test was used to compare the surgery groups. Differences between the ipsilateral and contralateral sides were compared using a paired t-test. (P < .05 was set as the level of statistical significance).
Results
Induction of c-Fos and p-ERK in Naïve Rats
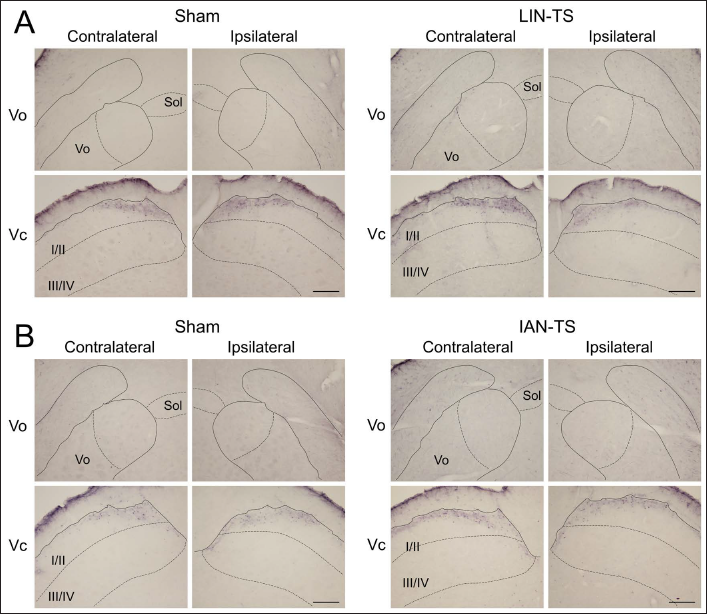
Stimulation with capsaicin induced many immunolabeled neuronal profiles in the BTSNC on both sides in naïve animals (Figure 1). Immunohistochemical labeling with c-Fos antibody was detected exclusively within the neuronal nucleus, whereas that with p-ERK antibody extended in the neuronal perikaryon and diffused as fibrous labeling. The neuronal profiles immunolabeled for p-ERK were confined to neuronal perikaryal labeling since the fibrous one was considered to be ascribed to dendrites or axons.
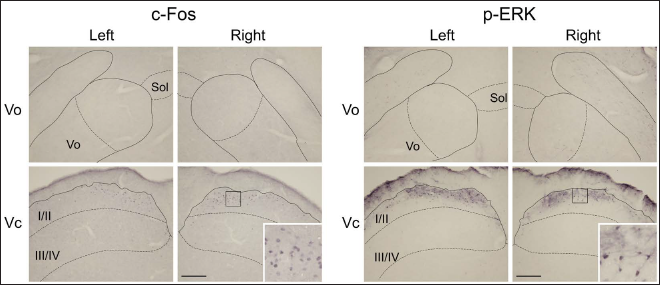
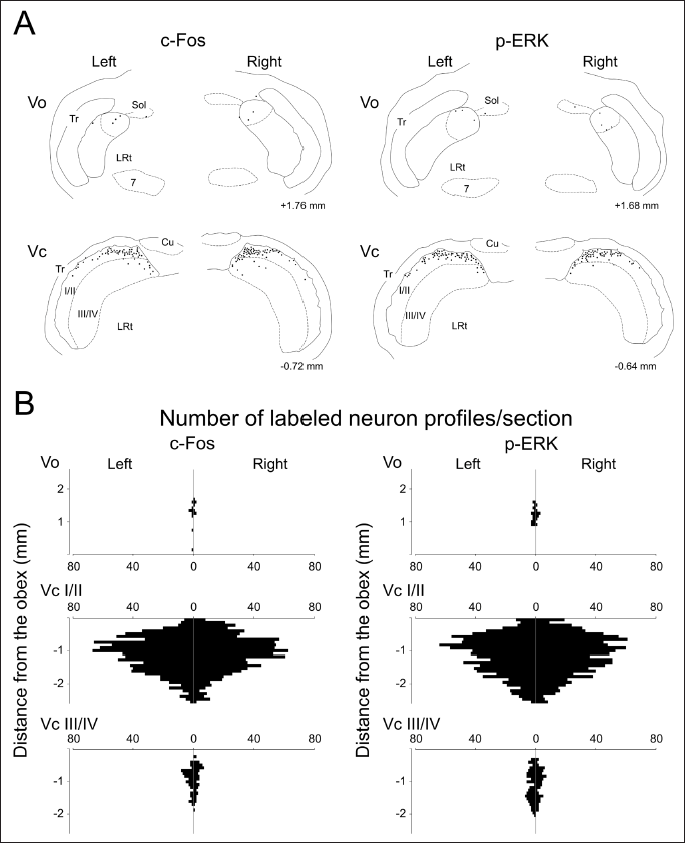
In the Vo, limited numbers of neuronal profiles immunolabeled for c-Fos and p-ERK were located at the level between 1.0 and 2.0 mm rostral to the obex after noxious tongue stimulation with capsaicin. These neuronal profiles confined largely to the dorsomedial portion of the Vo on both sides (Figures 1 and 2). These neuronal profiles were almost undetectable in the ventral part of the Vo. The most prominent induction was noted in the Vc. Superficial laminae of the Vc contained abundant immunolabeled neuronal profiles on both sides. Deeper laminae of the Vc comprised fewer of these neuronal profiles (Figures 1 and 2). They localized within the dorsomedial portion of the Vc crossing through laminae (Figure 2A). In the rostrocaudal distribution, they were mainly located in rostral sections (Figure 2B). These neuronal profiles were almost undetectable in the PrV and Vi. In contrast to capsaicin, each subdivision of the BTSNC was almost devoid of these neuronal profiles after the application of vehicle to the tongue. Therefore, these neuronal profiles found in the Vo and Vc were assumed to be elicited by noxious tongue stimulation with capsaicin. Similar distributions and numbers of immunolabeled neuronal profiles were observed for c-Fos and p-ERK immunolabeled neuronal profiles.
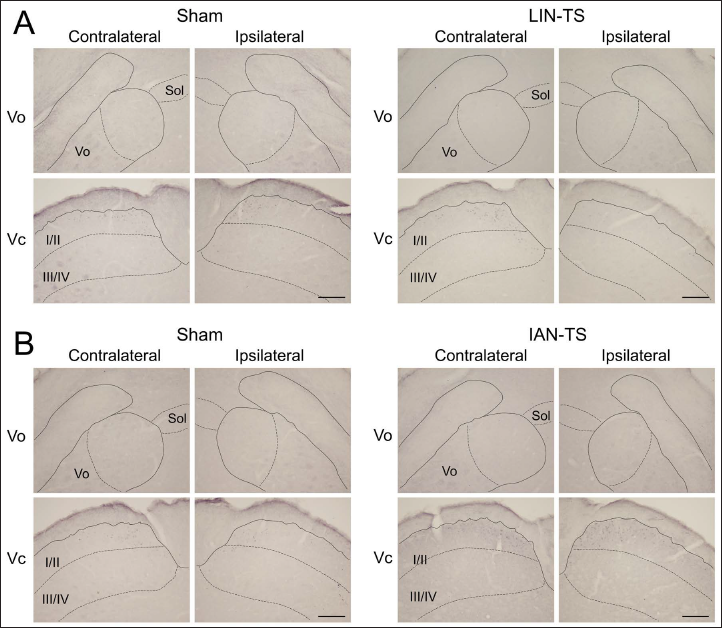
Capsaicin-Induced c-Fos Immunolabeled Neuronal Profiles in Rats with Injured LIN or IAN
Neuronal profiles immunolabeled for c-Fos were barely detected in each subdivision of the BTSNC after the application of vehicle to the tongue 14 days after any surgeries, including LIN- or IAN-TS or the respective sham surgeries (data not shown). The application of capsaicin to the tongue of rats that underwent LIN- or IAN-TS or the respective sham surgeries induced a lot of immunolabeled neuronal profiles. The Vo and Vc contained many immunolabeled neuronal profiles in their dorsomedial portions, which were similar to those in naïve rats (Figures 3 and 4). Despite the LIN or IAN injury, no marked differences were noted in the territorial distribution pattern of these neuronal profiles. Alternatively, the effects of nerve injuries became distinct in comparisons of the number of them elicited by noxious tongue stimulation with capsaicin (Figures 4 and 5). In the respective sham-operated groups, stimulation of the tongue with capsaicin induced a few c-Fos immunolabeled neuronal profiles in the Vo on both sides (Figure 4). However, the same treatment induced significantly larger number of these neuronal profiles in the Vo bilaterally 14 days after LIN-TS or IAN-TS (Figures 4 and 5).
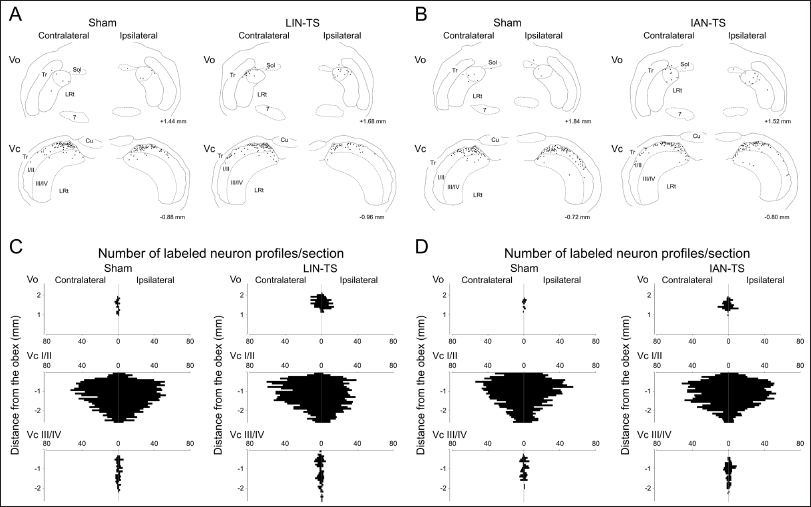
Drawings for the Location of Immunolabeled Neuronal Profiles in the Vo and Vc with Capsaicin 14 Days After LIN-TS and the Corresponding Sham Surgery (A) or IAN-TS and the Corresponding Sham Surgery (B). Numbers on the Lower Right Side in Each Drawing Indicate Distances from the Obex. Horizontal Bar Charts Show the Rostrocaudal Distribution of Immunolabeled Neuronal Profiles in the Vo, and Superficial Laminae of the Vc (Vc I/II) and Deeper Laminae of the Vc (Vc III/IV) 14 Days After LIN-TS and the Corresponding Sham Surgery (C) or IAN-TS and the Corresponding Sham Surgery (D).
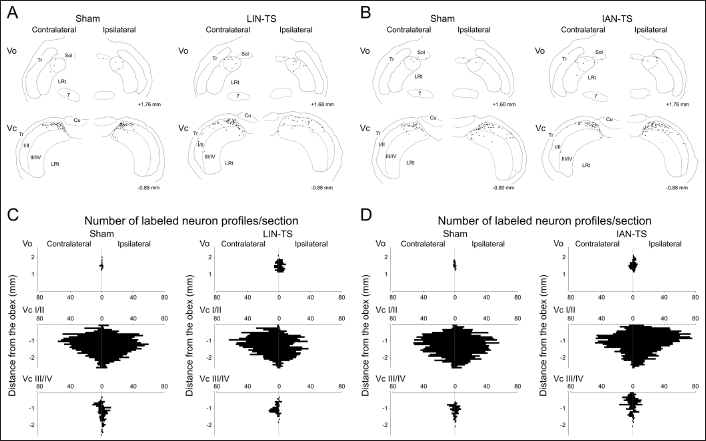
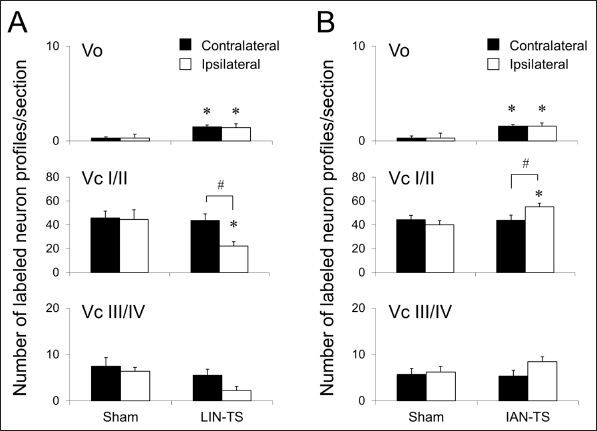
In the Vc, capsaicin-induced c-Fos immunolabeled neuronal profiles varied in number depending on the types of surgery and the side. Numerous immunolabeled neuronal profiles were induced in superficial laminae on both sides after noxious tongue stimulation with capsaicin in respective sham-operated rats (Figure 3). Deeper laminae of the Vc contained fewer immunolabeled neuronal profiles on both sides. Similar numbers and distributions of these neuronal profiles were observed on both sides (Figures 4 and 5). Fourteen days after LIN-TS, a significant reduction of capsaicin-induced neuronal profiles was noted in superficial laminae of the ipsilateral Vc (Figure 5). A similar reduction was also observed in deeper laminae of the Vc, although the difference was not statistically significant. The number of immunolabeled neuronal profiles on the contralateral side was left unchanged after the same surgery (Figures 4 and 5). On the contrary, the number of labeled neuronal profiles in superficial laminae of the ipsilateral Vc in IAN-TS group was significantly larger than that in sham-operated group (Figure 5). Although this increase was also found in deeper laminae, the difference was not statistically significant. The number of these immunolabeled neuronal profiles on the contralateral side was left unchanged after IAN-TS (Figures 4 and 5).
Capsaicin-Induced p-ERK Immunolabeled Neuronal Profiles in Rats with Injured LIN or IAN
Neuronal profiles immunolabeled for p-ERK were barely detected in each subdivision of the BTSNC after the application of vehicle to the tongue 14 days after any surgeries, including LIN- or IAN-TS or the respective sham surgeries (data not shown). In contrast, the application of capsaicin to the tongue induced many p-ERK immunolabeled neuronal profiles in the BTSNC. The Vo and Vc contained many p-ERK immunolabeled neuronal profiles in their dorsomedial portions (Figures 6 and 7). Regardless of the types of surgery, similar territorial distribution patterns of these neuronal profiles were observed. In the respective sham-operated groups, a few capsaicin-induced these neuronal profiles were found in the Vo bilaterally (Figure 6). However, the same treatment induced significantly larger number of these neuronal profiles in the Vo bilaterally after LIN-TS or IAN-TS (Figures 7 and 8).
Drawings for the Location of Immunolabeled Neuronal Profiles in the Vo and Vc 14 Days After LIN-TS and the Corresponding Sham Surgery (A) or IAN-TS and the Corresponding Sham Surgery (B). Numbers on the Lower Right Side in Each Drawing Indicate Distances from the Obex. Horizontal Bar Charts Show the Rostrocaudal Distribution of Immunolabeled Neuronal Profiles in the Vo, and Superficial Laminae of the Vc (Vc I/II) and Deeper Laminae of the Vc (Vc III/IV) 14 Days After LIN-TS and the Corresponding Sham Surgery (C) or IAN-TS and the Corresponding Sham Surgery (D).
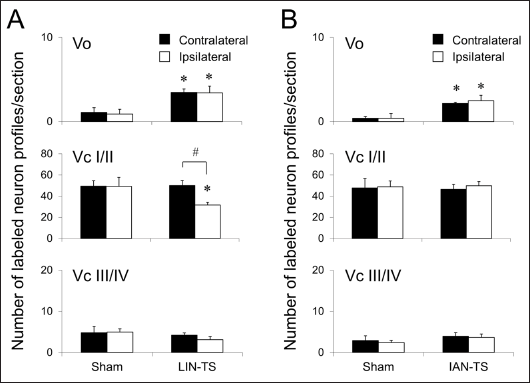
Numerous p-ERK immunolabeled neuronal profiles were induced in superficial laminae on both sides after noxious tongue stimulation with capsaicin in respective sham-operated rats (Figure 6). Deeper laminae of the Vc contained a fewer immunolabeled neuronal profiles on both sides. Similar numbers and distributions of these neuronal profiles were observed on both sides (Figures 7 and 8). Fourteen days after LIN-TS, a significant reduction of capsaicin-induced p-ERK immunolabeled neuronal profiles was noted in superficial laminae of the ipsilateral Vc (Figure 8). A similar reduction was also observed in laminae of the Vc, although the difference was not statistically significant. By contrast, no remarkable differences in the number of these neuronal profiles for the ipsilateral Vc were detected between IAN-TS and sham surgery groups (Figure 8). Neither LIN-TS nor IAN-TS affected the induction of these neuronal profiles by Vc neurons on the contralateral side (Figures 7 and 8).
Discussion
We demonstrated the following findings: (1) capsaicin-induced neuronal profiles immunolabeled for c-Fos and p-ERK markedly reduced in the ipsilateral Vc after injury to the LIN; (2) after injury to the IAN, the number of capsaicin-induced neuronal profiles immunolabeled for c-Fos was increased in the ipsilateral Vc, while that for p-ERK was left unchanged; and (3) injury to the LIN or IAN caused an increased number of capsaicin-induced neuronal profiles immunolabeled for c-Fos and p-ERK in the Vo bilaterally.
The expression of c-Fos is induced by Vc neurons after intense stimulation of the facial skin. This induction occurred in a somatotopically-organized manner, with stronger stimuli inducing a larger number of c-Fos immunolabeled neuronal profiles.33, 34 An intraoral noxious stimulation was also shown to induce a certain number of these neuronal profiles in the Vo in addition to the Vc.24, 25 Regardless of the surgery type, most of immunolabeled neuronal profiles observed in this study were localized within the dorsomedial portions of the Vo and Vc, where somatosensory afferents in the LIN terminate in.35, 36 Therefore, these neuronal profiles appear to have been elicited by noxious stimulation with capsaicin to the tongue, which is mainly innervated by the LIN.
After injury to the LIN, a decreased number of capsaicin-induced c-Fos immunolabeled neuronal profiles was noted in the ipsilateral Vc, but that on the contralateral side remained unchanged. This decrease was attributed to dysfunctional nerve fibers conveying nociceptive information because the injured nerve was tightly ligated on the proximal nerve stump to prevent regeneration. However, the enhanced induction of c-Fos was noted after IAN-TS in the ipsilateral Vc, suggesting that IAN-TS enhanced the transmission of nociceptive signals conveyed by the uninjured LIN. Consistent with these results, it has been reported that c-Fos expression by Vc neurons induced by intense skin stimulation was enhanced after IAN-TS. 4 The expression of c-Fos by spinal neurons after subcutaneous formalin injection into the hind paw was exaggerated by tibial nerve injury. 3 These changes have been implicated in hyperalgesia of the peripheral area close to the denervated area after nerve injury. Primary afferents of the LIN and IAN terminate in the dorsomedial part of the rostral Vc and synaptic inputs interact with each other, which may cause the overexpression of c-Fos after IAN injury when the stimulation is applied to the peripheral tissue innervated by the LIN. We previously indicated that Vc neurons receive nociceptive inputs from primary afferents derived from different nerves simultaneously, which enhanced the excitability of second-order nociceptors.7, 37, 38 In addition, intact nerve fibers may invade to denervated peripheral areas after nerve damage.
Similar to c-Fos, the induction of p-ERK by noxious stimuli applied to peripheral tissues has been demonstrated.14, 39–42 In this study, the location of these neuronal profiles was equivalent. Corresponding to changes in c-Fos, a decreased number of capsaicin-induced p-ERK immunolabeled neuronal profiles was noted in the ipsilateral Vc 14 days after LIN-TS. ERK phosphorylation is responsible for the activation of cAMP-response element binding protein (CREB) neurons, 40 which regulates many genes, including c-fos. 43 The attenuation of ERK phosphorylation by pretreatment with intrathecal MEK 1/2 inhibitor decreased c-Fos expression. 17 These findings suggest that ERK phosphorylation regulates the c-Fos expression for normal nociceptive responses. Neuronal profiles immunolabeled for p-ERK in the ipsilateral Vc did not increase in number 14 days after IAN-TS, which was inconsistent with c-Fos. These differential results suggest that c-Fos expression is responsible for both the anomalous and normal nociceptive responses, while ERK phosphorylation is engaged mainly in normal nociceptive transmission. Other signal pathways that are not accompanied by ERK activation may contribute to the overexpression of c-Fos. Previous studies demonstrated that the protein kinase A or Ca2+/calmodulin kinase pathways may be involved in CREB phosphorylation, which enhances c-Fos expression.44, 45
The number of capsaicin-induced c-Fos and p-ERK immunolabeled neuronal profiles in the Vo was significantly increased on both sides after LIN-TS or IAN-TS. This increase may be attributed to changes in neuronal excitability. Unilateral LIN-TS significantly down-regulate nicotinamide adenine dinucleotide phosphate-diaphorase (NADPH-d) expression in the Vo bilaterally. 46 NADPH-d colocalizes with GABA and glycine in the spinal dorsal horn 47 and cuneate nucleus. 48 Therefore, nerve insult may cause dysfunctions in inhibitory neurons in the Vo, thereby increasing excitability. Neurons in the dorsomedial Vo connect to the trigeminal motor nucleus 49 and parabrachial nucleus, 50 but not to the thalamus 51 and are involved in reflexes rather than sensory processing of the orofacial region. The enhanced c-Fos expression and ERK phosphorylation by Vo neurons may be involved in pathological neural circuits related to orofacial reflexes after peripheral nerve injury.
Conclusion
Injury to the LIN or IAN resulted in changes in stimulus-induced c-Fos expression and ERK phosphorylation by Vo and Vc neurons. These differential effects may be involved in the complex nature of symptoms of trigeminal neuralgia.
Footnotes
Authors’ Contribution
RT was responsible for the conceptualization of the research and carried out the investigations. Immunohistochemistry and analysis of data were done by KT, HF, and DNPM. The manuscript was prepared by RT and reviewed by AN and KU. The overall supervision was provided by RT.
Statement of Ethics
All experiments and protocols were approved by The Animal Care and Use Committee, Hiroshima University (Approval No. A20–164).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors wish to thank Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (19K10042) for financial assistance.
ICMJE Statement
This manuscript has been edited in accordance to the guidelines of the International Committee of the Medical Journal Editors.
