Abstract
Background:
Exposure to lead has been linked to biochemical changes similar to those patients suffering from Alzheimer’s disease. Trévo is a phytonutrient-rich product with antiaging and antioxidant properties.
Purpose:
To investigate the neuroprotective activity of trévo against lead-induced biochemical changes in male Wistar rats.
Methods:
The study involves 35 animals that were randomly divided into five groups of seven rats each. Group I (Control): Orally administered distilled water; Group II (Induced): Administered 15 mg/kg of lead acetate (PbA) intraperitoneally; Group III (Treatment group): Orally administered 2 mL/kg of trévo for two days before co-administration with PbA for 12 consecutive days; Group IV (Treatment group): Orally administered 5 mL/kg of trévo for two days prior to coadministration with PbA for 12 consecutive days; Group V: Orally administered 5 mL/kg of trévo for 14 consecutive days. Animals were anesthetized with diether and the brain excised and processed for the following biochemical assays: Malonedialdehyde (MDA), glutathione (GSH), catalase (CAT), superoxide dismutase (SOD), glutathione-S-transferase (GT), acetylcholinesterase (AChE), beta-amyloid, glutamate, Na+/K+ ATPase, and glutamate dehydrogenase (GD).
Results:
PbA caused significant oxidative stress (increased MDA concentration, decreased GSH concentration, suppressed the activity of CAT, SOD), decreased GT activity, increased activity of AChE, increased the concentration of beta-amyloid, and caused glutamate excitotoxicity (increased concentration of glutamate, decreased activity of Na+/K+ ATPase, and GD) in rat brains. Treatment with trévo at the two different doses significantly prevented oxidative damage, beta-amyloid aggregation, glutamate excitotoxicity, and acetylcholine breakdown induced by lead acetate.
Conclusion:
Our findings added to the reported pharmacological activity of trévo and supported the antiaging potential of trévo.
Introduction
Lead (Pb) exposure is one of the global challenges that humans face daily.1, 2 This is because of its high industrial usage and presence in most household utilities. 3 The presence of Pb has been discovered in contaminated water, batteries of various types, petroleum products, fumes from generators, children’s toys, and some contaminated candies. 4
While developing countries might not be using Pb, most of the developed and industrialized countries such as the USA and China transport industrial waste and expired products to the underdeveloped countries in addition to them being the end-users of their products.5–7 This has made underdeveloped countries such as Nigeria one of the countries affected by Pb poisoning. 8 The permeability and mobility of Pb across various tissues in the body make it a heavy metal of concern. 9 Exposure to a high concentration of Pb can lead to loss of consciousness, convulsion, and possible death. 10 Pb exposure has been linked to different types of neurodegenerative diseases, such as Parkinson’s disease and Alzheimer’s disease (AD).11, 12 AD is the number one cause of dementia among the aged. It is characterized by memory loss, cognitive impairment, learning disability, and character defect. It is estimated that more than 150 million people will suffer from dementia by 2050, 13 making it a global challenge of health concern. Biochemical indicators of AD are the accumulation of 39 to 43 aa peptides, generally referred to as beta-amyloid in the brain. 14 While the root cause of AD is unknown, it is generally agreed that the development of AD is linked to the environmental factors and genetic alteration that occurs during the development of the brain. 14 Apart from the amyloid plaque formation, the cholinergic system is also implicated in the etiology of AD. 13 It was observed that there is a low concentration of acetylcholine in the brain of patients suffering from AD. Thus to manage AD, the use of acetylcholinesterase (AChE) inhibitors is prescribed. 15 AChE is an enzyme that catalyzes the breakdown of acetylcholine (ACh) to acetate and choline. Thus, its inhibition can prolong the half-life of acetylcholine, thereby improving the patient's memory. Some anticholinesterase drugs include rivastigmine, galantamine, and donepezil. Apart from ACh, the role of glutamate excitotoxicity in AD has led to the development of N-methyl-D-aspartate (NMDA) receptor antagonists in the symptomatic treatment of AD. 15 Glutamate is an excitatory amino acid that can act as a neurotransmitter in the brain. However, a high concentration of glutamate because of brain impairment can cause excitotoxicity in the brain.16–18 There is a strong connection between oxidative stress and the above-listed pathologies (amyloidogenesis, cholinergic, and glutaminergic). Oxidative stress can be a cause or outcome of these pathologies. These have also led researchers to prescribe antioxidant drugs that can prevent or delay the progression of AD.19–22 While the approved drugs for the treatment of AD are expensive and often developed side effects in patients. 23 This has led to the search and development of better and effective drugs with multiple targets. Trévo is a rich nutritional supplement that contains different types of phytochemicals that have been confirmed to boost the immune system, slow down aging, and improve the health well-being of humans. The numerous health benefits in reported scientific publications include hepatoprotective,24,25 neuroprotective,26 and cardioprotective27. However, there is no scientific report on the neuroprotective effect of trévo against lead acetate poison; we decided to investigate the potential of trévo to counter the neurotoxic effect of lead acetate in this study.
Materials and Methods
Chemicals and Reagents
Lead acetate was purchased from Sigma-Aldrich, USA, while trèvo, a product of Trèvo limited liability company (LLC), Oklahoma City, USA, was purchased from a pharmacist. The kits used for the biochemical assays were supplied by RayBiotech Inc. (Norcross, GA, USA), and other chemicals were of analytical grade.
Experimental Design
Thirty-five male Wistar rats weighing 170 ± 10 g were purchased from the Central Animal House, University of Benin, Edo State, Nigeria, and used for this experiment. The animals were housed in well-ventilated cages and provided water and food ad libitum. Animals were divided into five groups of seven rats each as described below and pretreated with 2 and 5 mL/kg of trévo for two days prior to coadministration with 15 mg/kg of lead acetate (PbA) for 12 consecutive days.
Processing of the Brain
Around 24 h after last administration, animals were sacrificed via cervical dislocation and the brain excised, rinsed, and homogenized in a phosphate buffer saline (0.1 M, pH 7.4) to obtain a 10% w/v homogenate. The homogenate was centrifuged at 15,000 rpm for 10 min with the temperature set at 4°C to obtain a clear supernatant that was used for biochemical assays.
Biochemical Assay
Beta-Amyloid
The level of β-amyloid 1–42 in the brain was determined, according to the ELISA kit instruction manual supplied by RayBiotech Inc. (Norcross, GA, USA).
Acetylcholinesterase, Na+/K+ ATPase, Glutamate Dehydrogenase, and Glutamate
The activities of acetylcholinesterase, Na+/K+ ATPase were evaluated as described by Ilesanmi and Co. 28 and Ilesanmi and Co., 29 respectively, while glutamate dehydrogenase and glutamate concentration in the brain was estimated according to the instruction from the kit manual.
Estimation of Oxidants
The level of oxidative stress was determined by measuring the amount of malondialdehyde (MDA) formed from lipid peroxidation in the brain tissue according to the method of Varshney and Kale. 30
Estimation of Antioxidant and Antioxidant Enzymes in Brain Tissues
The concentration of glutathione (GSH) was measured according to study of Jollow and Co. 31 Catalase (CAT) activity was determined as described by Aebi. 32 The activity of superoxide dismutase (SOD) was measured as described by Misra and Fridovich. 33
Glutathione-S-Transferase Assay
The activity of Glutathione-S-Transferase (GST) was assessed as described by Habig et al. 34
Statistical Analysis
The software used for stsatistical analysis was Graphpad prism 6.0 developed by Graphpad software. Differences among groups were evaluated by one-way analysis of variance followed by Duncan’s multiple range tests. All values were expressed as the mean ± standard deviation of seven animals per group.
Results
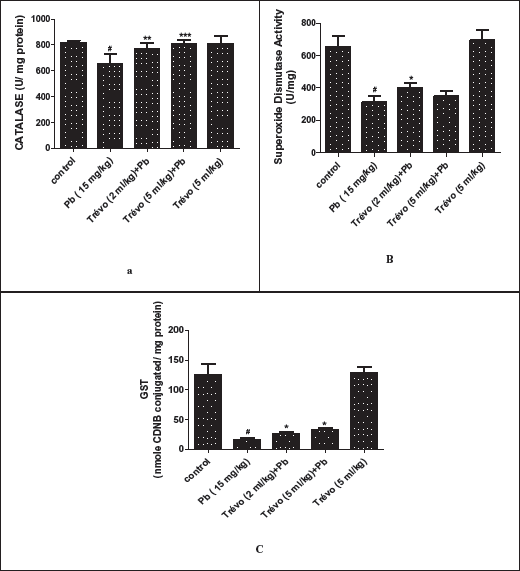
(a ) Acetylcholinesterase (AChE) Activity in the Brain of Male Wistar Rats After two day- Pretreatment With Trévo and 12-Day Co-Administration With Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control vs. PbA; *P < .05 = PbA vs. Treatment Groups. (b ) Ameliorative Activity of Trévo on Accumulation of β Amyloid in the Brain of Male Wistar Rats After Two Day-Pretreatment With Trévo and 12-day Co-Administration With Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; **P < .01 = PbA vs. Treatment Groups
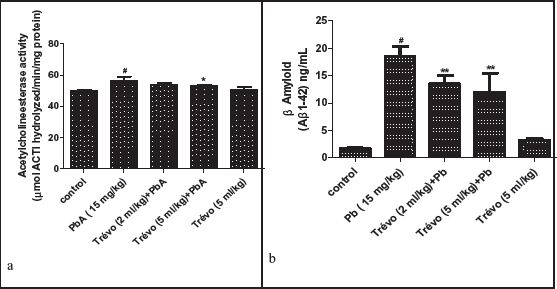
(a) Effect of Trévo on Na+/K+ ATPase Activity in the Brain of Male Wistar Rats Following Lead Exposure. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; **P < .01 = PbA vs. Trévo (5 mL/kg) + PbA. (b ) Effect of Trévo on Glutamate Dehydrogenase Activity in the Brain of Male Wistar Rats Following Lead Exposure. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; *P < .05 = PbA vs. Trévo (5 mL/kg) + PbA. (c ) Effect of Trévo on Glutamate Content in the Brain of Male Wistar Rats Following Lead Exposure. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; ***P < .001 = PbA vs. Treatment Groups.
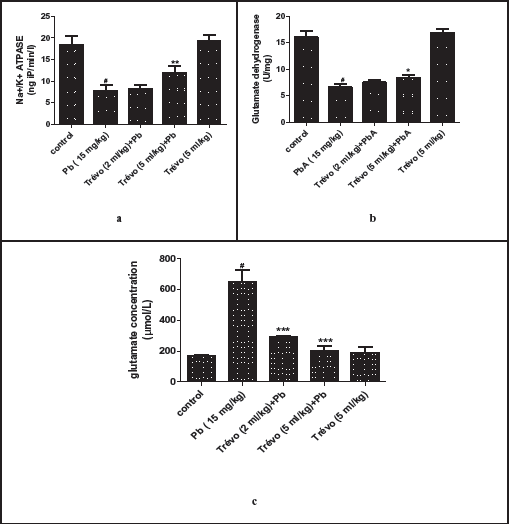
(a) The Concentration of Lipid Peroxides Product—Malondialdehyde (MDA), in the Brain Tissue of Male Rats After Two Day-Pretreatment With Trévo and 12-Day Co-Administration With Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; ***P < .001 = PbA vs. Treatment. (b ) The Concentration of Nonenzymatic Antioxidant-Reduced Glutathione (GSH) in the Brain Tissue of Male Rats After Two-Day-Pretreatment With Trévo, Followed by 12-Day Concomitant Exposure to Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; *P < .05 = PbA vs. Treatment Groups.
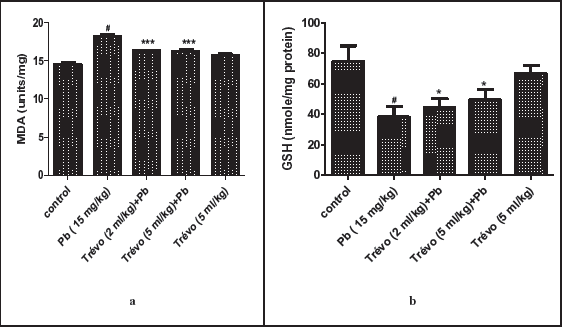
(a) The Catalase Activity in the Brain Tissue of Male Rats After Two Day-Pretreatment With Trévo and 12-Day Co-Administration With Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; ** P < .01 = PbA vs. Trévo (2 mL/kg) + PbA; ***P < .001 = PbA vs. Trévo (5 mL/kg) + PbA. (b ) The Superoxide Dismutase Activity in the Brain Tissue of Male Wistar Rats After Two Day- Pretreatment With Trévo and 12-Day Co-Administration With Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; *P < .05 = PbA vs. Trévo (2 mL/kg) + PbA. (c ) The Glutathione-S-Transferase (GST) Activity in the Brain Tissue of Male Rats After 2 Day-Pretreatment With Trévo and 12-Day Co-Administration With Lead Acetate (PbA; 15 mg/kg) via Intraperitoneal Administration. Data Are Shown as Mean ± Standard Deviation (SD) for Seven Animals. Statistically Significant Differences: #P < .05 = Control Group vs. PbA; *P < .05 = PbA vs. Treatment.
Effect of Trévo on Acetylcholinesterase (AChE) Activity and β-Amyloid Following PbA-Toxicity.
Figures 1a and 1b show the effect of PbA (15 mg/kg) and trévo (2 and 5 mL/kg) on AChE activity and β-amyloid level.
Figure 1a shows that the treatment of animals with 15 mg/kg of lead acetate caused a significant increase in AChE activity as compared to the control (P < .05). It also shows that pretreatment with 2 and 5 mL/kg of trévo caused a significant reduction in the activity of AChE as compared to the untreated group (P < .05). The treatment of animals with trévo only had no significant effect on AChE activity as compared to the control.
Figure 1b shows that the administration of lead acetate caused a significant increase in the brain level of β-amyloid as compared to the control (P > .05). However, pretreatment with trévo at both 2 mL/kg and 5 mL/kg caused a significant decrease in the level of β-amyloid when compared with the untreated group (P < .01). The administration of trévo had no significant effect on the β-amyloid level as compared to the control group (P > .05).
Effect of Trévo on the Activity of Na+/K+ ATPase, Glutamate Dehydrogenase (GD), and Concentration of Glutamate Following Lead Acetate (PbA) Exposure.
Figures 2a and 2b show the modulatory effect of pretreatment of trévo on PbA-induced alteration on Na+/K+ ATPase and GD activity, while Figure 5 shows the effect of trévo on PbA-induced increase in glutamate concentration. The exposure of animals to PbA causes a significant decrease in the activity of Na+/K+ ATPase as compared to the control (P < .05). The pretreatment of animals with 2 mL/kg of trévo caused a mild increase in the activity of Na+/K+ ATPase when compared with the untreated group (P > .05). However, pretreatment with 5 mL/kg of trévo caused a significant increase in the activity of Na+/K+ ATPase as compared to the untreated group (P < .01). Treatment of the rats with 5 mL/kg of trévo only had no significant effect on Na+/K+ ATPase when compared to the control (P > .05).
As shown in Figure 2b, exposure of the animals to PbA caused a significant decrease in the activity of GD as compared to control (P < .05). Pretreatment with trévo at the dose of 5 mL/kg caused a significant increase in the activity of GD as compared to the untreated group (P < .05).
Figure 2c shows the effect of pretreatment with trévo on the concentration of glutamate in the brain of rats exposed to PbA. PbA caused a significant increase in glutamate concentration as compared to the control (P < .05). Pretreatment with 2 and 5 mL/kg of trévo caused a significant decrease in the concentration of glutamate when compared to the untreated group (P > .001).
Antioxidant Activity of Trévo Against Lead Acetate (PbA) Induced Oxidative Stress in the Brain of Male Wistar Rats
Figures 3a, 3b, 4a, and 4b show the effect of the pretreatment of animals with trévo and exposure to PbA on the level of MDA, reduced GSH, and activity of CAT and SOD, respectively.
As observed in Figure 3a, PbA caused a significant increase in the concentration of MDA as compared to the control (P < .05). Pretreatment with 2 and 5 mL/kg of trévo caused a significant decrease in the concentration of MDA as compared to the untreated group (P < .001). Treatment with 5 mL/kg of trévo only had no significant effect on the concentration of MDA as compared to the control (P > .05).
Figure 3b showed that PbA caused a significant depletion in the concentration of GSH as compared to the untreated group (P < .05). Pretreatment with trévo at 2 and 5 mL/kg caused a significant increase in the concentration of GSH as compared to the untreated group (P < .05). Treatment of the rats with 5 mL/kg of trévo without exposure to PbA had no significant effect on the GSH level when compared to the control (P > .05).
Figure 4a shows that PbA significantly suppressed the expression of CAT activity when compared to the control (P < .05). Pretreatment with 2 and 5 mL/kg of trévo was able to prevent 0the inhibitory effect of PbA on CAT activity as observed in the high activity of CAT in the pretreatment group as compared to the untreated group (P < .01 and P < .001, respectively). Treatment of the rats with 5 mL/kg of trévo had no significant effect on CAT activity when compared to the control group (P > .05).
The effect of pretreatment of rats with trévo before exposure to PbA on SOD activity is summarized in Figure 4b. It showed that PbA caused a significant decrease in SOD activity as compared to the control (P > .05). Pretreatment with 2 mL/kg of trévo prevented the inhibitory activity of PbA on SOD activity as observed in the significant increase in SOD activity as compared to the untreated group (P < .05). It was also observed that a low dose of trévo was more effective than a high dose. Administration of 5 mL/kg of trévo had no significant effect on SOD activity as compared to the control.
Effect of PbA and Trévo on Glutathione-S-Transferase Activity
Figure 4c shows the activity of GST because of pretreatment with trévo and PbA exposure. PbA caused a significant decrease in the activity of GST as compared to control (P < .05). Pretreatment with 2- and 5 mL/kg of trévo caused a significant increase in GST activity as compared to the untreated group (P < .05). Treatment of rats with 5 mL/kg of trévo only had no significant effect on the activity of GST when compared to the control (P > .05).
Discussion
Plant-rich phytochemicals have high health benefits to humans. Their consumption has been reported to delay aging, prevent cancer, and reduce the risk of neurodegenerative and cardiovascular diseases. 35 In addition, the result also suggests that some of the properties of these natural compounds such as anticholinesterase, antioxidant and amyloidogenic, and antiapoptotic properties enable them to prevent this disorder. The prophylactic effect of trévo was investigated against Pb-induced neurotoxicity on male Wistar rats. The biochemical assays investigated include AChE, β-amyloid accumulation, glutamate dehydrogenase, and Na+/K+ ATPase. In addition to the above, we also determine the activity of GST, SOD, CAT, MDA, and GSH in the brain of Wistar rats. Lead acetate at the administered doses caused a significant increase in the accumulation of β-amyloid and glutamate concentration, decreased the activity of ATPase and GD, increased glutamate and MDA concentration, decreased GSH concentration in the activity of CAT, SOD, and GST.
The inhibition of acetylcholinesterase is one of the key determinants of drugs to treat AD prolonging the neuronal effect of ACh by inhibiting the activity of AChE, which has a positive effect on memory loss and motor balance. 36 Our result shows that trévo delayed the degradant acetylcholine by suppressing the activity of AChE. This anticholinesterase activity of trévo might be linked to some of the functional plants present in trévo such as Phyllanthus emblica (Amalaki fruit) and Garcinia mangosteen (Mangosteen), with reported cholinesterase activity against different models of neurodegenerative diseases.37, 38
The antiamyloidogenic effect of trévo was also investigated and our results show that trévo was able to prevent the aggregation of β-amyloid. AD is mostly characterized by the deposition of β-amyloid. Most reported natural products against AD often work by preventing the aggregation of β-amyloid. 39 Our results show that there is a low level of β-amyloid in the brain of animals pretreated with trévo. This antiamyloidogenic effect of trévo might be linked to some of the various fruits present in trévo that has been reported to prevent the aggregation of β-amyloid in the brain. Some of the plants include Schisandra (fruit), mango (stem), and pomegranate.37, 38, 40
Glutamate dehydrogenase and NA+/K+ ATPase are two important enzymes involved in regulating the availability of glutamate in the brain. Thus, their activity has been implicated in glutamate excitotoxicity, a mechanism that has been reported to contribute to the pathogens of AD. 41 In our experiment, the activities of Na+/K+ ATPase and glutamate dehydrogenase were lower, while the concentration of glutamate was higher in the brain of rats exposed to lead. This is similar to the work of Li et al. 42 and Neal et al., 43 who reported the alteration of glutamate transmission following lead exposure, as well as Thangarajan et al. 44 The antiglutaminergic effect of trévo was reflected in the increased activity of NA+/K+ ATPase and glutamate dehydrogenase with the concomitant decrease in glutamate concentration in rats’ brains. One of the key components of the trévo is pomegranate, a plant that has been reported to increase the activity of NA+/K+ ATPase. 45 The pomegranate might contribute to the antiglutaminergic effect of trévo observed in our study.
Both animal experiments and human surveys on the effect of Pb exposure on MDA and GSH revealed an increase in MDA and a decrease in the GSH in blood and brain.46–48 A similar result was observed on the effect of lead on GSH and MDA levels in our experiment. In addition, the activity of CAT, SOD, and GST was also reduced in the brain of rats following lead exposure as observed in the report of other researchers.49–52 The antioxidant effect of trévo in effecting neuroprotective effect has been reported in our previous work. 26 This was also confirmed in the present experiment. The antioxidant activity of trévo might be linked to the antioxidant-rich plant present such as manqué berry, a Malaki fruit, pomegranate, mangosteen, and schizandra fruits.
Conclusion
Our results show that administration of 15 mg/kg of lead acetate for 12 consecutive days to male Wistar rats causes biochemical changes observed in the brain of patients suffering from Alzheimer’s diseases; these include increased oxidative stress, low antioxidant activity, glutaminergic, low acetylcholine, and formation of β amyloid. Pretreatment with trévo was able to counter the neurotoxic effect of lead acetate. The neuroprotective properties of trévo can be linked to the abundance of natural compounds with proven antioxidant, antiaging, and neuroprotective properties.
Footnotes
Acknowledgments
The authors would like to appreciate the undergraduate students that participated in the conduct of the experiment and Taif University supporting project TURSP 2020/235.
Authors’ Contribution
OBI designed and supervised the experiment. MJ, IC, BC, and CA assisted in the experiment, TTO assisted in data analysis, OJA, FOA, OO, were involved in the draft, EIA, NKAA, GM, OO, GEB were involved in manuscript editing and revising.
Statement of Ethics
All the rats used in this study were healthy and treated according to the guidelines of the Helsinki Declaration of 1975 for the care and use of laboratory animals. The experimental design was approved by the ethics committee on animal research and treatment (ART) of the Federal University Otuoke, Nigeria (approval code: ART2021005). In specific terms, the experiments were conducted in the animal house of the Department of Biochemistry, Faculty of Science, Federal University Otuoke between February and June 2021.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
