Abstract
Introduction: High consumption of smokeless tobacco in adult Indian population increases the risk of developing oral cancers leading to high morbidity and mortality. Though the influence of abstinence from smoking on cognitive performance has been widely studied, the effect of smokeless tobacco on cognitive performance and its association with withdrawal symptoms is less understood. This study comparatively investigates the effect of short-term conscious abstinence and distraction during abstinence from smokeless tobacco consumption on the craving, withdrawal symptoms, sympathetic response, and cognitive performance in tobacco addicts.
Methods: Age, sex, education and socioeconomic status matched control (N = 15) and smokeless tobacco addicts (N = 60) were recruited from residential areas in Bhubaneswar for the study. Following randomization of the addicts, conscious abstinence (N = 30) was induced by informed abstinence from tobacco consumption for 8 hours, while distracted cessation (N = 30) was induced by involving the participants in a cognitively engaging task for 8 hours during uninformed tobacco abstinence.
Results: The results of the study show higher withdrawal symptoms and reduced cognitive performance in volunteers with conscious abstinence which was positively correlated. The decreased cognitive performance in conscious cessation was independent of tobacco-induced increase in the LF:HF ratio and cotinine concentration in saliva.
Conclusion: While conscious abstinence results in higher withdrawal symptoms, distraction during abstinence lowers these symptoms. Inclusion of distraction sessions during cessation can, therefore, be a new element in tobacco control strategies.
Introduction
Tobacco consumption is a leading and avertable cause of morbidity and mortality worldwide. India stands third in tobacco production and second in consumption of tobacco in the world, resulting in more than 1.3 million deaths.1, 2 Incidence of oral cancer is one of the highest in the world accounting for almost 50% of all cancers in men and 25% in women. 3 According to Global Adult Tobacco Survey 2016–2017 (GATS-2), 28.6% of adults aged greater than 15 years in India use tobacco in some form or the other. Recent findings indicate growing popularity of smokeless tobacco especially among adults. 4 Unlike western countries, India has higher number of smokeless tobacco consumers who mostly consume tobacco in the form of gutka, pan, khaini and snuff in comparison to tobacco smokers. Gutka, a tobacco–lime–areca nut mixture, is a widely used smokeless tobacco product in the urban areas of the Indian subcontinent (GATS-2 survey, 2017). Gutka contains and comparatively delivers higher nicotine than smoking cigarettes. 5
Nicotine, the addictive substance in tobacco, activates nicotinic receptors and modulates acetylcholine-mediated neurotransmission. Though the carcinogenicity and adverse effects of tobacco consumption on human health has been widely reported, its influence on brain and cognitive performance still remains debatable. Repeated use of nicotine is reported to induce physiological neuroadaptations. 6 There has been scientific evidence suggesting that tobacco dependence may partly be an outcome of its cognitive-promoting actions.7, 8 Piper et al. have shown benefits of smoking on attention and memory. 9 Nicotine abstinence in smokers is reported to be associated with decreased working memory performance and impairment of sustained attention and concentration. 10 Studies also suggest that tobacco enhances attention, working memory and performance of complex tasks in satiated smokers and nonsmokers. 11 Mitchell demonstrated an attention deficit when smokers were deprived of nicotine. 12 Smokeless tobacco (gutka) users report of addictive symptoms that include positive expectancies about the effects and experiencing subjective pleasure on tobacco consumption. 13 Contradictory to these findings, several studies show no effect of nicotine on cognitive performance of human volunteers. 14 While most of these studies on neuropsychological effects of nicotine have been conducted on smokers, information on the effect of smokeless tobacco is sparse despite increasing prevalence of smokeless tobacco consumption in young population. A study by Lindgren et al., however, reports increase of arousal along with shifting of activity within the alpha band towards higher frequencies in male users of oral snuff. 15 The physiological effects of nicotine also include activation of the splanchnic nerves and stimulation of the adrenal medulla to release adrenaline which influences the cardiovascular functions. Acetylcholine released from the preganglionic sympathetic fibres of splanchnic nerves acts on nicotinic acetylcholine receptors (nAChRs) which in turn causes the release of epinephrine (and norepinephrine).16, 17 The predominant metabolite of nicotine is cotinine that has been widely used as a biomarker for tobacco consumption. With similar mechanism of action as nicotine, cotinine also activates the SNS. Studies on short-term effects of chewing gutka on heart rate variability (HRV) in young adults have concluded that the LF/HF ratio was elevated for 5 min on chewing of tobacco. 18 The association of these autonomic changes resulting due to physiological effects of smokeless tobacco consumption with cognitive performance of addicts, however, remains to be established. A recent study on geriatric population in Odisha shows association of chewing tobacco with depression and anxiety. 19 While several studies describe the cause–effect relationship of tobacco consumption, withdrawal or cessation on various cognitive domains, scientific evidence on the effect of conscious awareness of abstinence on cognitive performance is sparse. While conscious cessation involves focused awareness of abstinence of tobacco consumption, distracted abstinence refers to cognitive activity resulting in distraction and abstinence from tobacco consumption without being explicitly aware of it. A recent study by Joshi et al. suggests that the act of chewing gutka is almost always preceded by conditioned reflex indicating role of conscious abstinence in craving. 20 The relative contribution of craving and withdrawal symptoms due to focused awareness during conscious abstinence and its effect on cognitive performance, however, remains to be studied.
Previous studies by Keenan et al. also show significant effect of short-term smokeless tobacco deprivation on cognitive performance in a study population of 47 individuals. 21 Studies by Meil et al. have demonstrated significant alterations in executive functioning in a study population of 17 individuals undergoing short-term tobacco cessation therapy. 22 A study conducted by Sarkar et al. had a population size of 24 participants for assessment of the craving in nicotine-dependent subjects using smokeless tobacco. 23 In this study, we investigated the effect of short-term conscious abstinence and distracted abstinence from smokeless tobacco consumption on craving, withdrawal symptoms and cognitive performance. The association of sympathetic physiological responses induced by smokeless tobacco consumption with cognitive performance was also studied.
Methods
Study Setting and Study Population
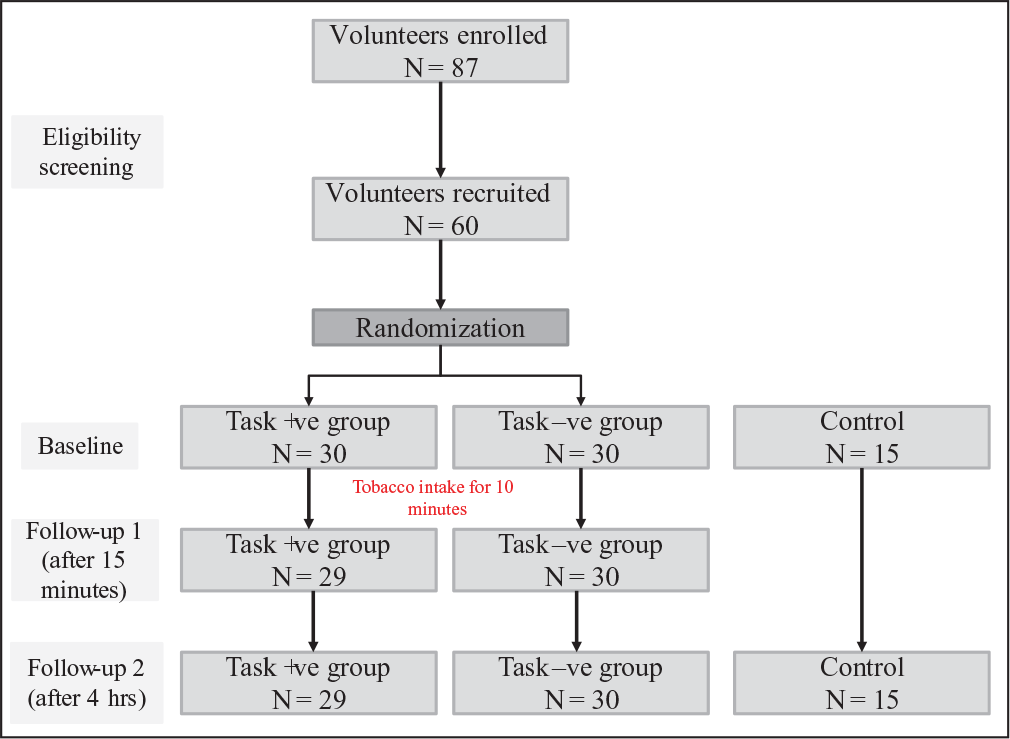
Inclusion Criteria (Eligibility Criteria 1 and 2) for Enrolling Volunteers
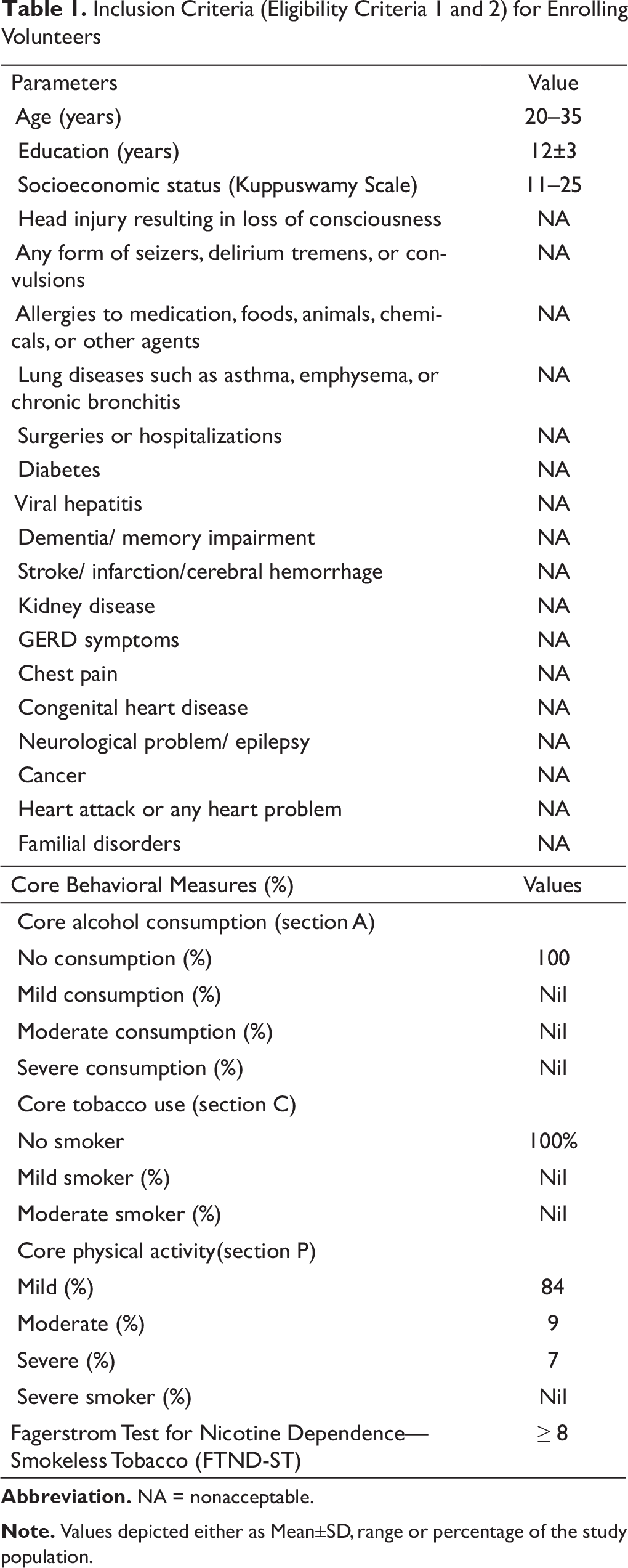
Randomization
60 tobacco chewers who had FTND-ST scores ≥8 and qualified the inclusion criteria were randomly assigned to task +ve (N = 30) or task –ve groups (N = 30) using computer-generated 1:1 randomization using the web programme
Study Design
The study design involved conscious and distracted abstinence for a period of 8 hours. At baseline neuropsychological tests, namely, mini-mental state examination (MMSE), 26 multi-domain cognitive screening test (MDCST), 27 color Stroop test (CST) 28 and Frontal Systems Behavioral Scale (FrSBe) 29 were administered. The volunteers were then permitted to chew their usual tobacco brand and report back within a gap of 10 minutes; the volunteers were not instructed to take any precise quantity of tobacco but rather were asked to take their regular dose. Fifteen minutes after having chewed tobacco, the volunteers were again evaluated (follow-up 1) with MMSE, MDCST and CST. An additional scale of Minnesota Nicotine Withdrawal Scale (MNWS) was administered to examine the withdrawal symptoms. The randomized groups were assessed separately, and all the volunteers were briefed about the study protocol. Conscious tobacco abstinence was introduced by making the task –ve volunteers wait in the waiting room and explicitly informing them that they were not permitted to consume tobacco for the next 8 hours. The task –ve volunteers had access to food, water and newspapers in the waiting room and were free to interact with each other.
Distracted abstinence was induced by engaging the volunteers in a task without informing them that they could not chew tobacco for the next 8 hours. The volunteers in task +ve group were engaged into 8-hour sessions, comprising six activities using standardized teaching materials for both trainers and trainees. First activity was to introduce about themselves to other participants. Second activity included 1 hour of paper craft task where subjects were asked to follow the instructor to make small paper crafts. Third activity was to surf the internet for 1 hour to gather maximum information regarding their city and their state. Fourth activity for 1 hour was to pen down their thoughts on a topic of social relevance in the language of their preference and then to exchange this information with fellow volunteers, to engage socially. Fifth activity was to watch a movie of choice from a playlist. Sixth activity for 1 hour was to play a board game where participants had to arrange the pieces in logical manner to complete a picture. The volunteers had free access to food and water during the activity sessions. The MMSE, MDCST, CST and FrSBe were administered in continuation to the sixth activity to adhere to the unconscious cessation paradigm (follow-up 2). The withdrawal symptoms were further followed up using the MNWS scale. The overall motive for the 8-hour task +ve intervention was to create a paradigm where randomized subjects involved in tobacco withdrawal task were kept continuously engaged in some activity requiring their conscious effort, so that they have minimal time to consciously think about the withdrawal. The studied parameters at follow-up 2 remained same for both task +ve group and task –ve group at the end 8 hours. The study design has been depicted in Figure 1.
Study Design
Tobacco Withdrawal Symptoms
MNWS was used to estimate the withdrawal symptoms and severity. MNWS is a five-point scale (none, slight, mild, moderate, severe), which evaluated withdrawal severity in the following eight domains, namely, angry, irritable, frustrated; restless, impatient; depressed mood, sad; increased appetite, hungry, weight gain; difficulty in concentrating; insomnia, sleep problems, awakening at night; anxious, nervous and desire/craving. Each of the eight withdrawal symptoms in the past week were rated for their severity on a scale from 0 (not present) to 4 (severe).
Saliva Cotinine Measurement
The volunteers were instructed to drool the saliva into collection tube through a small funnel until the tube was half full. The tube was sealed after discarding the funnel. The samples were stored at –80° C until further analysis. Cotinine levels were assessed by NicAlertTM (Nymox Pharmaceutical Corporation, St. Laurent, QC, Canada) enzyme-linked immunosorbent assay (ELISA) kit, as per manufacturer’s instruction. The results were recorded as units 0–6, where level 0 corresponds to 0–10 ng/ml cotinine concentration, level 1 for 10–30 ng/ml, level 2 for 30–100 ng/ml, level 3 for 100–200 ng/ml, level 4 for 200–500 ng/ml, level 5 for 500–1000 ng/ml and level 6 for >1000 ng/ml of cotinine concentration, respectively.
Heart Rate Variability Measurements
Sympathetic activity was calculated from heart rate (HR) and HRV that was acquired using Zephyr physiology monitoring system with MP150 hardware (BIOPAC Systems, CA, USA) and AcqKnowledge 3.9 software. Short-term HRV was acquired in compliance to the guidance of Task Force on HRV using previously standardized protocol. 30 Briefly, the volunteers were habituated to test environment in a relaxed sitting position for 30 minutes to minimize inter-variability among the subjects due to tobacco intake prior to data acquisition. Data was acquired for beat-to-beat intervals, HR and breathing rate in block durations of 1 minute at a sampling rate of 25 Hz in sitting position while maintaining a breathing rate of 15–20 breaths per minute for 3 minutes. Subsequently, HR and HRV was acquired for 5 minutes under normal breathing for analysis purposes. A consistent 5-minute RR interval series was selected after removal of artefacts and ectopic from the recorded tachogram, for analysis of HRV parameters. Nonparametric Fast Fourier Transform method was used to determine powers of low-frequency (LF) domain, high-frequency (HF) domain and the LF:HF ratio. While frequency in the range of 0.04–0.15 Hz was considered as LF, frequency between 0.15 and 0.4 Hz was considered as HF.
Cognitive Assessment
Assessment of cognitive performance was performed using a battery of neuropsychological tests described previously.27, 31 Assessment of mild cognitive impairment was performed using MMSE wherein scores ≥25 were considered to be cognitively normal. 26 A previously validated 45-point scale MDCST was also administered to screen performance of nine different cognitive domains. 27 Subjects scoring ≥36 in MDCST were considered as cognitively normal. Information-processing rate, parallel processing of attended and unattended stimuli and attention was assessed by CST. 28 The time needed to complete each Stroop subtask served as dependent measures (Stroop I, Stroop II and Stroop III, respectively). While the participants were allowed to spontaneously correct the errors they noticed, the number of errors that were not self-corrected was also recorded for each Stroop subtask (Error I, Error II and Error III, respectively). Higher inference score in CST indicated poor performance. FrSBe 29 was administered to evaluate behaviour allied with executive functions. FrSBe includes 46-item measures where volunteers were to rate themselves on how frequently they performed certain behaviours on a 0–5-point scale. The FrSBe has three subscales, namely, apathy (poor initiation, reduced drive and interest), disinhibition (restlessness risk taking, socially inappropriate behaviour) and executive dysfunction (difficulty with learning, mental flexibility and working memory). The mean score of the three subscales was considered for statistical evaluation. All scores were converted to T-scores corrected for age, education and gender as per the ‘FrSBe Administration Manual.’ 32 High FrSBe score was an indicator of poor executive function.
Statistical Analysis
For the purposes of data analysis, all statistics were calculated using SPSS 22.0. Chi square and one-way analysis of variance (ANOVA) with Duncan’s post hoc test were used to compare the two-randomized group and control group on age, socioeconomic status, neuropsychological performance and physiological parameter. Analysis of covariance (ANCOVA) was used for comparison of differences in neuropsychological tests (MMSE, MDCST, CST and FrSBe at the follow-up session between randomized volunteers using pretest scores as a covariate. All variables were tested for normality, multicollinearity and homogeneity and for the presence of outliers. The effectiveness of task +ve intervention or subconscious tobacco cessation on executive functioning was evaluated using paired-sample t-test of neurocognitive score prior to and after intervention. The correlation between neuropsychological performance on tobacco cessation with physiological, biochemical and psychological parameters was assessed by calculating Pearson product-moment correlation coefficients. Statistical significance in all cases was defined as P value less than 0.05.
Results
Population Characteristics
Population Characteristics for Randomized Tobacco Users and Control Subjects Enrolled for the Study
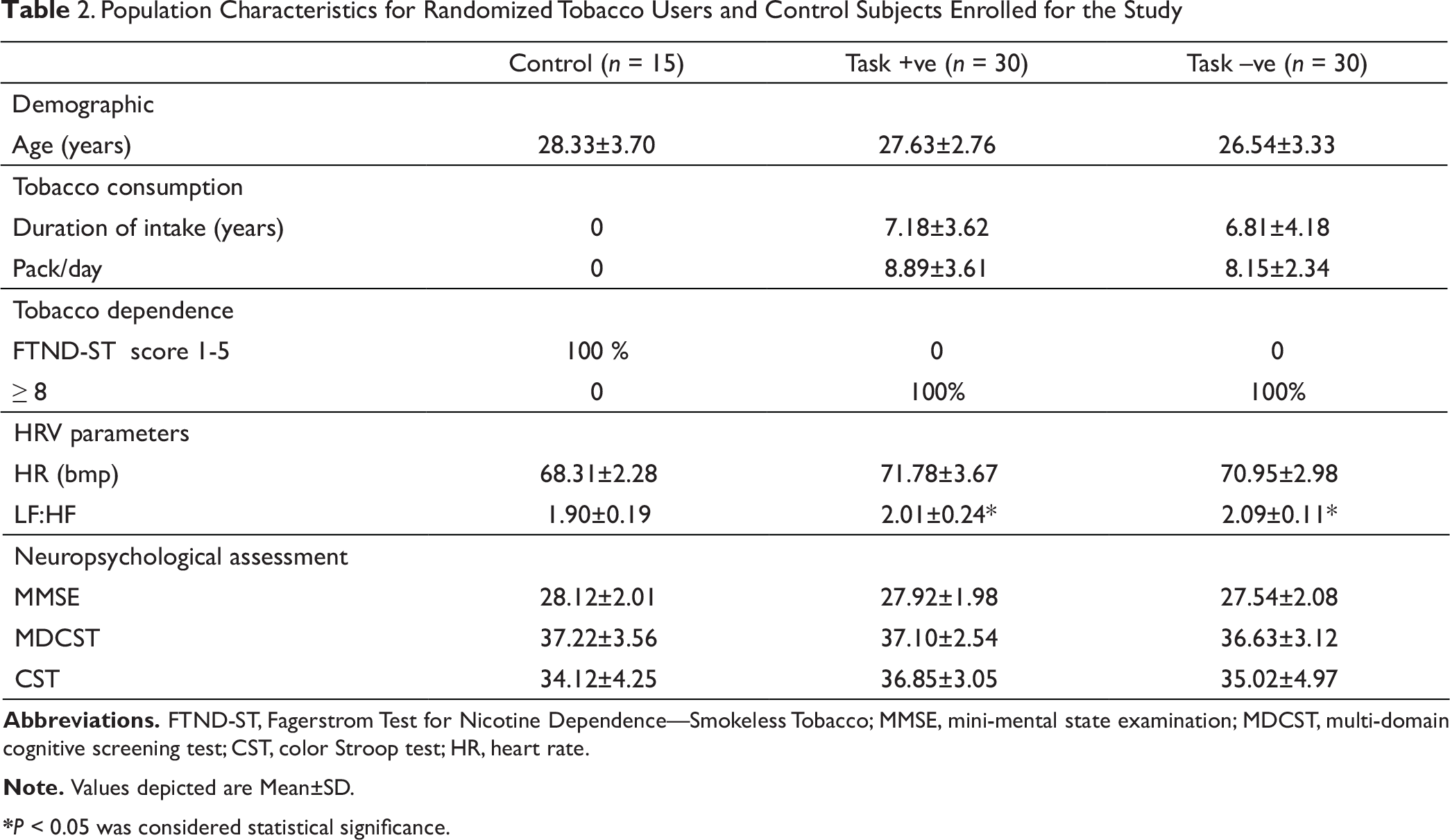
*P < 0.05 was considered statistical significance.
Physiological Measures of Task +ve and Task –ve Chewer Groups
Frequency domain analysis of HRV after 15 minutes of chewing gutka showed an increase in sympathetic nervous system (SNS) activity compared to baseline as indicated by an increase in LF and HF ratio (P < 0.01). No significant difference was observed between task +ve and task –ve randomized group at baseline (Table 3). After 15 minutes of tobacco intake, both the groups showed significant increase in sympathetic activity (P < 0.05). Similarly, after 8 hours of cessation (follow-up 2), both groups showed slight decrease in SNS activity, though the mean LF:HF values remain significantly higher than baseline values taken prior to gutka intake (P < 0.05; Table 3).
Saliva Cotinine in Task +ve and Task –ve Groups
Mean saliva cotinine concentration of task +ve and task –ve groups was significantly higher at follow-up 1 (P < 0.001) and follow-up 2 (P < 0.01) when compared to baseline. The inter-group difference at any particular follow-up showed no statistical difference in the cotinine level (Table 3).
Tobacco Withdrawal Symptoms
Tobacco withdrawal symptoms were measured at follow-up 1 and follow-up 2 using MNWS. At follow-up 1, the withdrawal symptoms were recorded slightly higher in the task –ve group (4.19 ± 2.20) when compared to the task +ve group (3.25 ± 1.44; P = 0.041). However, at follow-up 2, the difference between the two groups was significantly larger. The task +ve volunteers had the mean MNWS score of 6.05 ± 4.25, while task –ve volunteers had an MNWS score of 17.25 ± 3.01 (P < 0.001; Table 3).
Neuropsychological Measures of Task +ve and Task –ve Group
Physiological, Biochemical and Cognitive Characteristics of Study Population at Baseline and Follow-Up
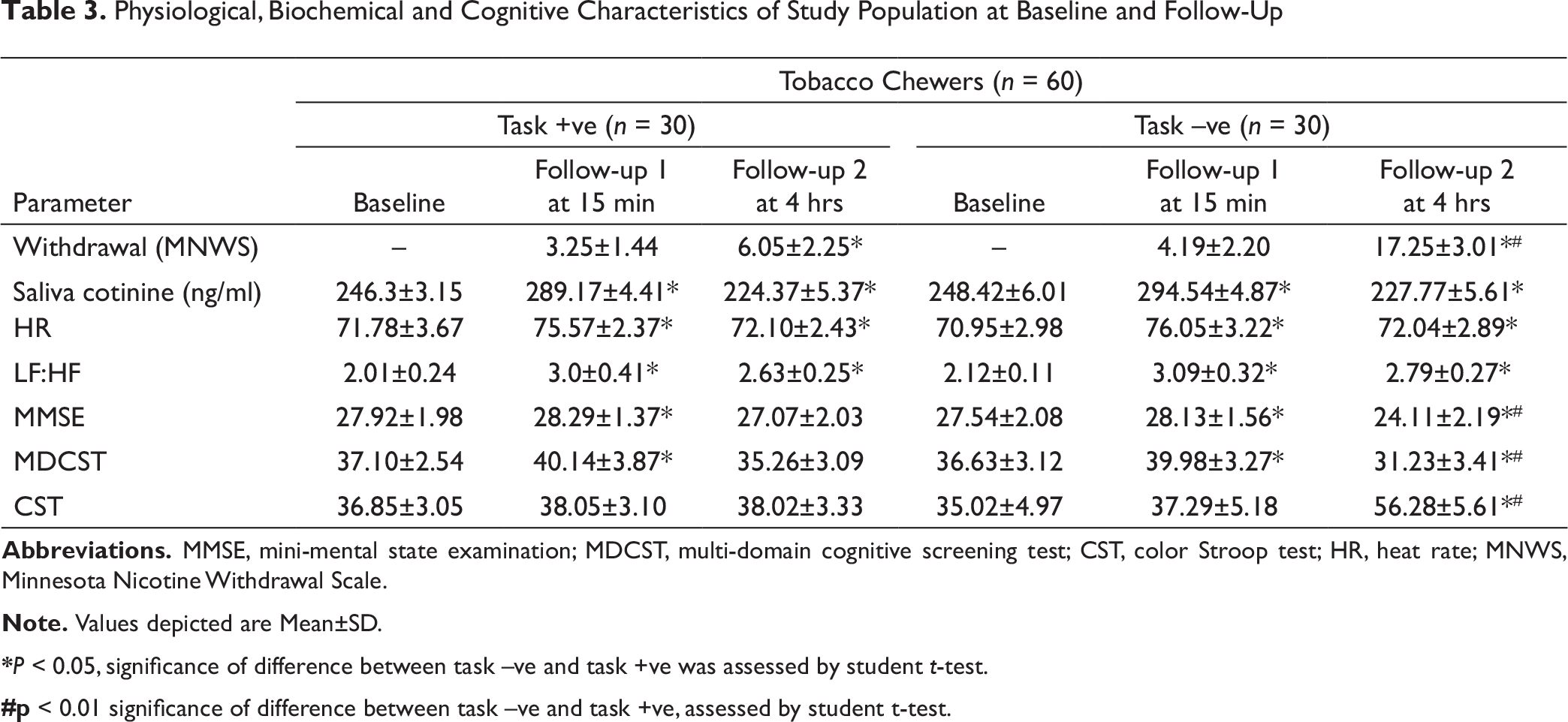
*P < 0.05, significance of difference between task –ve and task +ve was assessed by student t-test.
#p < 0.01 significance of difference between task –ve and task +ve, assessed by student t-test.
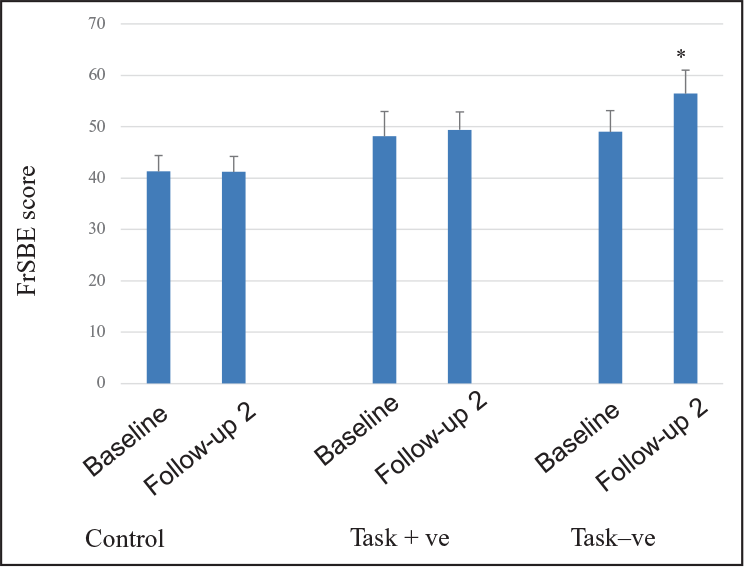
FrSBe Scores for Executive Function
ANCOVA was performed to evaluate differences in FrSBe scores after 8 hours of cessation (follow-up 2) between two-randomized group after controlling for baseline FrSBe scores. The linear relationship between baseline and follow-up scores between groups and homogeneity of variances was ascertained using scatterplot, visual inspection and Levene’s test of homogeneity of variance (P = 0.51), respectively. Since standardized residuals > ±3 SD was not found, no outliners in the data were assessed.
The baseline FrSBe scores showed statistically significant difference when compared to follow-up 2 FrSBe scores (F = 1.27, P = 0.013) among the task –ve group. Paired sample t-test indicated that volunteers in the task +ve tobacco cessation group showed no change in mean FrSBe scores at baseline (48.12 ± 5.12) and follow-up 2 (49.41 ± 3.48). There was significant increase in FrSBe scores of the task –ve group when compared to baseline (49.01 ± 4.10) and follow-up (56.47 ± 4.51). The FrSBe scores of control subjects were significantly lower than both randomized groups at baseline and follow-up 2 (P < 0.001; Figure 2).
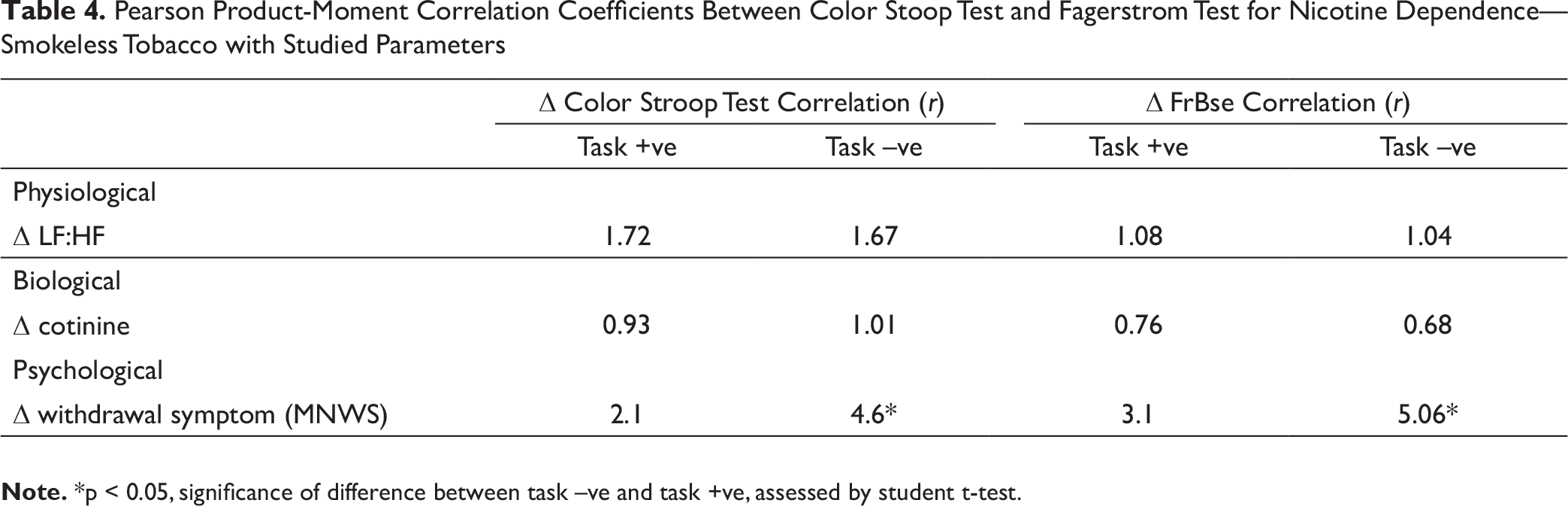
Correlation of FrBSE Score and CST Score with Physiological, Biochemical and Tobacco Withdrawal Symptoms
At the end of second follow-up, the changes in the FrBSE and CST scores from baseline were correlated with changes in LF:HF ratio, saliva cotinine and MNWS scores, in order to evaluate their respective contribution towards the observed change in neuropsychological performance (Table 4). Among task +ve subjects, the correlation between change in CST with LF:HF ratio was r = 1.72 (P = 1.02), saliva cotinine r = 0.93 (P = 2.3) and MNWS r = 2.1 (P = 0.94). Among task –ve subjects, the correlation between change in CST with LF:HF ratio was r = 1.67 (P = 1.15), saliva cotinine r = 1.03 (P = 1.5) and MNWS r = 4.6 (P < 0.01). Among task +ve subjects, the correlation between change in FrBSE with LF:HF ratio was r = 1.08 (P = 0.95), saliva cotinine r = 0.76 (P = 1.9) and MNWS r = 3.1 (P = 0.61). Among task –ve subjects, the correlation between change in FrBSE with the LF:HF ratio was r = 1.04 (P = 0.99), saliva cotinine r = 0.68 (P = 1.23) and MNWS r = 5.06 (P < 0.01).
Discussion
Pearson Product-Moment Correlation Coefficients Between Color Stoop Test and Fagerstrom Test for Nicotine Dependence—Smokeless Tobacco with Studied Parameters
Smokeless tobacco products, namely, gutka, tambaku, zarda pan are popularly consumed by Indians particularly in eastern regions including Odisha. 33 Previous literatures indicate potential health risks including oral cancers, degenerative dental conditions, cardiovascular diseases, hypertension and asthma, among others. The World Health Organization (WHO) estimated nearly 60 lakh deaths worldwide every year due to tobacco consumption, with majority of these deaths being reported from low- and middle-income countries like India. Despite mandatory statuary warnings on these smokeless tobacco products, a large number of individuals consume these products regularly. 34 The consumption of tobacco despite awareness of its adverse effects can at least partially be explained by the ‘Cognitive Dissonance Theory.’ 35 This theory provides a theoretical explanation to the fact that despite known ill effects of tobacco, the regular users fail to refrain themselves from using these products. It refers to a situation involving conflicting attitudes, beliefs and opinions.35, 36 A popular belief that tobacco users have is that tobacco consumption keeps them alert and cognitively more active and withdrawal of the product will hamper their performances. A recent survey shows greater satisfaction and psychologically rewarding experience on regular snus use when compared to occasional use. In the study, snus intake produced an early and transient cognitive improvement on Iowa Gambling Task (IGT) in abstinent snus users. 37 It is this disengagement that poises a great challenge for tobacco abstinence, thereby requiring further research for development of tangible strategies to promote de-addiction of tobacco.
Tobacco abstinence is the biggest challenge amongst users and has been attributed to poor concentration, impatience, fatigue, insomnia and restlessness. 38 Decline in cognitive performance during nicotine withdrawal has been reported in animal models as well.39, 40 Several studies conducted on human volunteers also report the effect of tobacco cessation on cognitive performance. 41 However, the duration of withdrawal that triggers the effect on cognitive performance has been widely debated. Smoking cessation for 17 hours is reported to increase reaction time on continuous performance task. 42 Conversely, no changes in cognitive performance were reported following 24 hours of cessation 43 or overnight cessation. 44 Hendrick et al., however, observed slower reaction time among subjects refrained from tobacco use as early as 30 minutes post cessation. 45 In this study, we report significant decrease in cognitive and executive performance in task –ve group volunteers after 8 hours of conscious tobacco withdrawal (Table 3). However, we also observed a positive effect of tobacco consumption on cognition when subjects were followed up 15 minutes after the tobacco intake (Table 3). This positive effect of chewable tobacco could be an outcome of two possible reasons. Firstly, the nicotine present in tobacco products is an agonist of nAChRs in brain and enhances the brain activity 46 and secondly, since the neuropsychological tests were administered at very short intervals between baseline and follow-up 1, there is a possibility of practice-related effect. 47
Earlier reports demonstrate short-term effects of tobacco cessation on autonomic response, thereby affecting the cognitive performance. Studies conducted by Keenan et al. showed higher craving score, increased reaction time and self-rated withdrawal symptoms along with decreased HR on smokeless tobacco cessation. 21 Similar decrease in HR was reported among volunteers after 24 hours of tobacco cessation. 48 During this study, we also observed the initial phase of higher HR followed by decrease in HR on tobacco cessation in both task +ve and task –ve group of tobacco chewers (Table 3). However, these physiological changes were not found to be associated with changes in cognitive performance. We observed that despite interventional variance in both randomized task +ve and task –ve groups of tobacco chewers, the individuals in both the groups had similar physiological response to the tobacco cessation. Both the groups of tobacco chewers, however, varied on the basis of withdrawal symptoms and neuropsychological assessment scores. The task –ve group individuals had significantly higher withdrawal symptoms and lower cognitive performance than task +ve group (Tables 3 and 4).
Though western countries have reported overwhelming research studies on tobacco prevention and cessation, but majority of these studies have been done on smoke tobacco products and lesser emphasis has been given to smokeless tobacco use. 49 Hence, most of the reports claiming downfall in mortality due to tobacco consumption over last decade contradict with demographics data collected in South Asian and Indian population, where smokeless tobacco is widely used.33, 50
Novel approaches are warranted to inhibit the disengagement beliefs among tobacco users and to encourage individuals to quit tobacco successfully. A recent study on patients recruited from ENT and dental OPD in Bhavnagar shows that 20% of the patients were highly dependent on smokeless tobacco and 61% had low willingness to quit tobacco. 51 Previous studies report that self-administered, online support and internet-based nonpharmacological interventions have lower effectiveness when used alone for successful cessation of tobacco consumption and fail on cost-effectiveness measures.52, 53 Contrarily, our findings suggest that use of interventions that could result in distraction from tobacco during abstinence for long durations, delivered through day-to-day life-related tasks can result in lower withdrawal symptoms and has a potential to provide effective support against disengagement beliefs of tobacco chewers.
Ranabhat et al., during a recent explorative systematic policy review investigation using available scientific database, have emphasized the need for youth-focused creative policies for effective tobacco control. 54 In 2009, the Government of India made a 30-second audio-visual advertisement showing an oral cancer surgeon explaining the serious illnesses in his young patient caused by smokeless tobacco use. Response of smokeless tobacco users regarding the campaign showed that it was well recalled and provided new information. 55 However, the primary outcome of such methods of tobacco cessation campaign to promote successful tobacco quittance is yet to be evaluated. We suggest that such novel approaches cannot provide the expected outcome if employed in isolation. Based on findings of our study, we propose the inclusion of distraction through cognitively involving tasks during abstinence as a new element in tobacco control intervention strategy, which can be employed in combination with other novel and conventional tobacco cessation therapies to prevent withdrawal relapse.
Conclusion
This study provides evidence for increased withdrawal symptoms and impaired cognitive abilities in volunteers on conscious abstinence from chewing tobacco despite being aware of its adverse health effects. On the contrary, distraction due to cognitively involving tasks resulted in lower withdrawal symptoms and better cognitive performance in tobacco addicts when compared to the conscious abstinence group. The withdrawal symptoms and cognitive performance were independent of cotinine levels and physiological influence of nicotine on the autonomic functions. Based on our findings, we propose inclusion of cognitively engaging tasks as a part of de-addiction programs and strategies for tobacco cessation.
Footnotes
Acknowledgments
The authors acknowledge the support of the volunteers participating in this ICMR nonfunded project. We convey our gratitude to Dr Gitanjali Batmanabane, Director, All India Institute of Medical Sciences, Bhubaneswar, and Dr Om Prakash Chaurasia, Director, Defence Institute of High Altitude Research, for providing access to equipment required to conduct the study.
Author Contributions
KKB: Overall planning and execution of the project, study design, acquisition of cognitive parameters related data, preparation of manuscript.
SB: Recruitment of volunteers, acquisition of physiological data and analysis.
VV: Analysis of physiological data.
SCM: Data interpretation.
Ethical Statement
Informed consent was obtained from all the volunteers who participated in the study. The project proposal was cleared by institutional ethical committee of All India Institute of Medical Sciences, Bhubaneswar, with ethical approval number IEC/AIIMS BBSR/STS_UG/2017-18/21.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
No funding was received from any funding agency for undertaking the project. The project was carried out under Indian Council of Medical Research (ICMR) STS scheme vide reference number ICMR-STS-2017-03819.
