Abstract
Background: Duchenne Muscular Dystrophy (DMD) is a fatal muscular dystrophy of pediatric population coupled with other secondary comorbidities including mental retardation and neuropsychological impairments. Mutation location in the dystrophin gene, have been associated with neuropsychological functioning in DMD.
Purpose: We investigated temporal changes in the neuropsychological functioning of DMD subjects, hitherto understudied.
Methods: Subjects with suspected DMD were enrolled according to the ethical guidelines. Genetic confirmation by Multiplex Ligation Dependent Probe Amplification was carried out to identify pathogenic deletion or duplication in dystrophin gene. Intellectual and neuropsychological functioning was assessed by using standardized batteries. Investigated neuropsychological domains included visual, verbal and working memory, selective and sustained attention, executive functioning, verbal fluency, and visuo-constructive and visuo-spatial abilities. The assessments were carried out at baseline and followed for one time point in 30 cases.
Result: The follow-up assessment revealed that neuropsychological functioning did not worsen with time. Improvements were seen in block designing task (p = 0.050), serial positioning primacy effect (p = 0.002), Stroop incongruent task (p = 0.006), visual long-term memory (p = 0.003) and attention (p = 0.001). DMD cases with mutation location affecting short dystrophin isoform (Dp140) also showed improvement in these domains.
Conclusion: No temporal alterations were found in DMD subjects, though improvements in few domains were observed. Neuropsychological rehabilitation may be useful in improving the quality of life in DMD subjects.
Introduction
Duchenne muscular dystrophy (DMD) is a fatal X-linked genetic neuromuscular disorder, characterized clinically by rapidly progressive and disabling muscle weakness, present from birth and exclusively occurring in males. DMD is caused by an X-linked recessive frameshift mutation in the dystrophin gene that ensues absent or non-functional muscle dystrophin protein and resultant muscle fibre degeneration, leading to chronic peripheral inflammation. 1 Dystrophin functions as a direct signalling molecule and connects the extracellular matrix to the cytoskeleton. It is a part of the dystrophin-associated glycoprotein complex.2, 3 It is the most common childhood muscular dystrophy with an estimated incidence of 200 per million male live births. 4 By the age of 3, patients with DMD exhibit motor inabilities in such as walking, running, climbing, jumping, waddling gait, difficulty in standing, followed by upper limb weakness and pseudohypertrophy by the age of 5. This is followed by progressive worsening of the symptoms and with death due to respiratory failure or cardiac arrhythmia before the third decade of life. 5
In addition to skeletal muscle pathology and loss of physical strength, a subset of children with DMD is characterized by global cognitive impairment. Previous works suggest that in DMD patients, intelligence quotient (IQ) distribution is downshifted one standard deviation with a lower verbal IQ than performance IQ. It is reported that DMD patients might also have specific neuropsychological deficits including poor performance in working memory, executive function, attention deficits, and impaired reading and language acquisition skills.1, 6 Previous studies have led to hypothesis that these specific neuropsychological deficits resonate with cerebellar lesions due to similarity in cognitive impairments. 7 Even though dystrophin is often characterized in muscles, it is also found in various other tissues including the brain. Multiple studies have shown the association between the loss of dystrophin and cognitive impairments. Multiple studies from both clinical and animal models attribute the lack of dystrophin expression in the brain to the development of the cognitive and behavioural alterations in DMD.8–10 Some patients with DMD also have a higher incidence of neurobehavioral disorders including attention-deficit/hyperactivity disorder (ADHD), anxiety disorder, autism spectrum disorders (ASD), epilepsy and obsessive-compulsive disorder. Experimental studies have found that dystrophin is expressed in neurons within specific brain regions including the cortex, cerebellar Purkinje cells, Cornu Ammonis (CA) region of the hippocampus, retina and the peripheral nerve. These might be responsible for some of the neuropsychological deficits. 11
It is important to note that myelination is critical in the central nervous system (CNS) for complex brain processing and therefore the disorders affecting the neuronal myelination, by a process regulated by oligodendrocytes in the CNS, may produce neurological deficits. 12 In a recent study, researchers have found that for proper maturation of oligodendrocytes and effective myelination during postnatal brain development, normal expression of dystrophin isoforms is required. Oligodendrocytes express three different forms of dystrophin, Dp427, Dp140 and Dp71, and loss of oligodendroglial dystrophin, particularly Dp427, was found to be contributory to neurodevelopmental deficits in their experimental mdx mouse model of DMD. In this study, in mice without functional Dp427 dystrophin protein had late development of myelination with significantly affecting the cerebral cortex. 13 A past review identified lack of Dp427 to be associated with progressive muscle weakness in all DMD patients, likely responsible for both muscle degeneration and brain dysfunction. 14
Despite involvement of common gene isoforms, Wingeier et al. in their study found no correlation of declining cognitive function with the progression of muscular deterioration. 7 Another study reported that cognitive impairment in DMD is non-progressive and unrelated to the severity of muscle disease. Additionally, varying phenotypic expressions of specific neuropsychological impairments is also notable in DMD patients. 15 The reason for this divergence is inconclusive, but this might be associated with the timing and localization of human dystrophin isoforms expression. 1 In contrast, previous studies reported that intellectual functioning in DMD patients deteriorates as the disease progresses with progressive reduction in all IQ scores. 15 As previously noted, varying neuropsychological deficits affect overall cognitive performance of the boys with DMD. For example, boys with DMD often have problems in short-term verbal working memory and increased risk of learning disability resulting from poor phonological awareness/processing. They often encounter problems with reading as discussed in a study, whereby 40% of boys with DMD have been shown to have reading problems. It is also found that they have lower academic achievement scores than expected of their level of cognitive functioning. 16 In addition to academic performance, they also face poor health-related global quality of life potentially posing them at risk of depression, anxiety and stress.17, 18 A successful care of DMD patients thus requires comprehensive, multidisciplinary plan including psychosocial care, in addition to a pharmacological approach.
In order to plan clinical trials to establish efficacy of interventions targeting different neuropsychological impairments, longitudinal studies in DMD patients are required. This will help to explore how, over the course of time, neuropsychological function changes with progression of DMD. Additionally, this can help with risk stratification and screening and offering specific neuropsychological rehabilitation. Future studies could include acquisition of longitudinal data in order to examine which cognitive and neuropsychological functions in DMD are non-progressive or progressive. This is important in counselling and future planning. Previous studies suggested that more research is needed about characterizing the features of neuropsychological profile in determining the use and effectiveness of cognitive rehabilitation and retraining for children with DMD. 5 In-depth review of the literature has revealed that there are no longitudinal studies that have investigated whether the cognitive and neuropsychological impairment in DMD is progressive. To the best of our knowledge, this is the global first longitudinal study which has described the neuropsychological function in DMD patients. The aim of this longitudinal study was to use a battery of intelligence, learning and memory tests to characterize the neuropsychological profile in boys with DMD by following them up for long-term changes in various domains.
Methods
Statistical Analysis
We used SPSS version 21 to analyse the neuropsychological data. Normal distribution was analysed by Kolmogorov–Smirnoff statistics. Normally distributed data was further analysed by paired t test. Level of significance was analysed at p < 0.05.
Results
Details of Participants
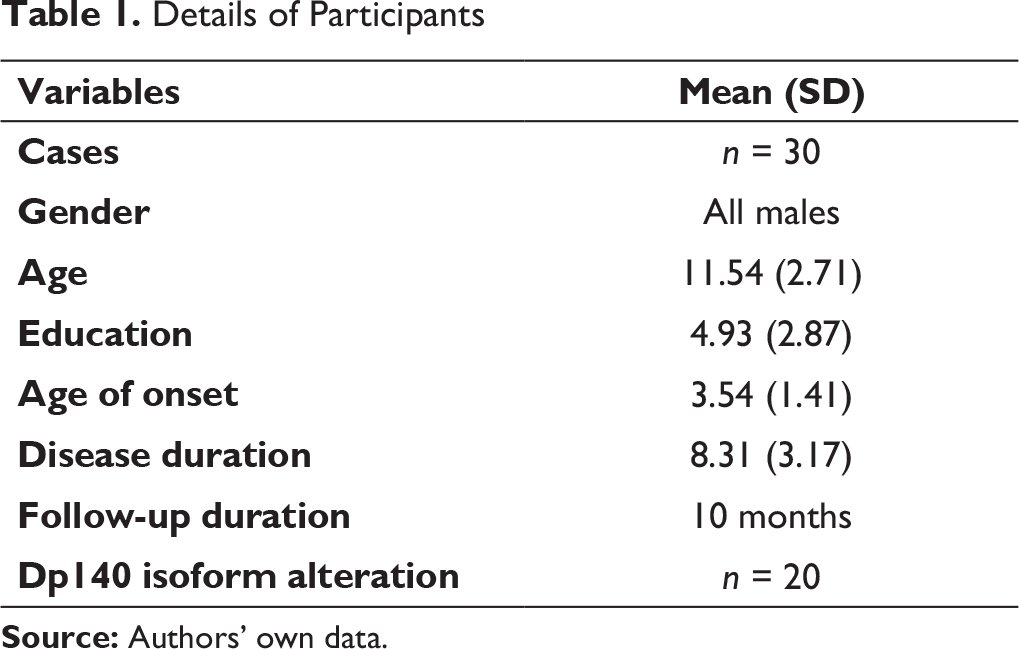
Longitudinal Analysis of Cognitive and Neuropsychological Profile in DMD Subjects
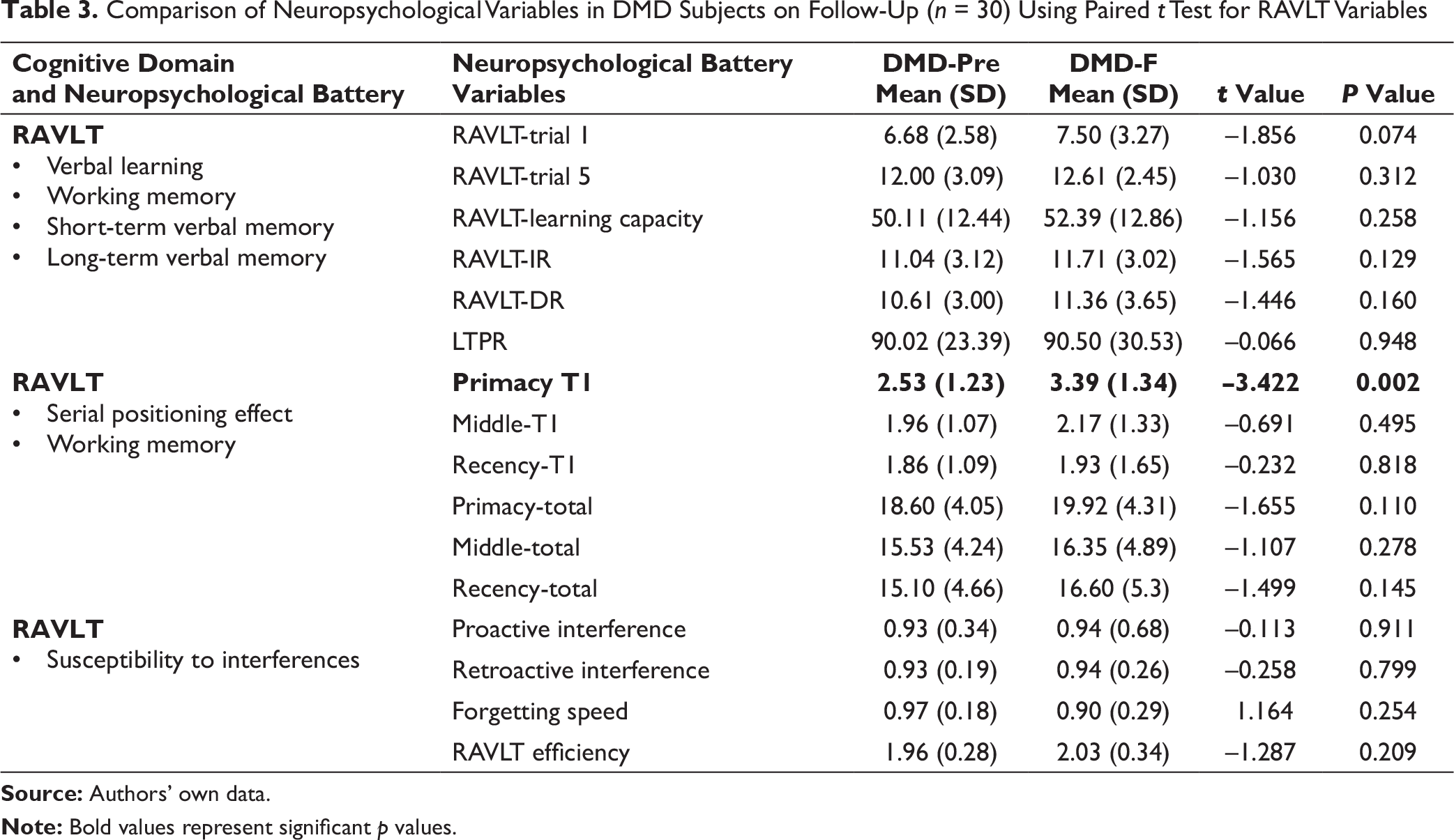
Comparison of General Intelligence on Pre and Post Follow-Up in DMD Subjects (n = 30) Using Paired t Test
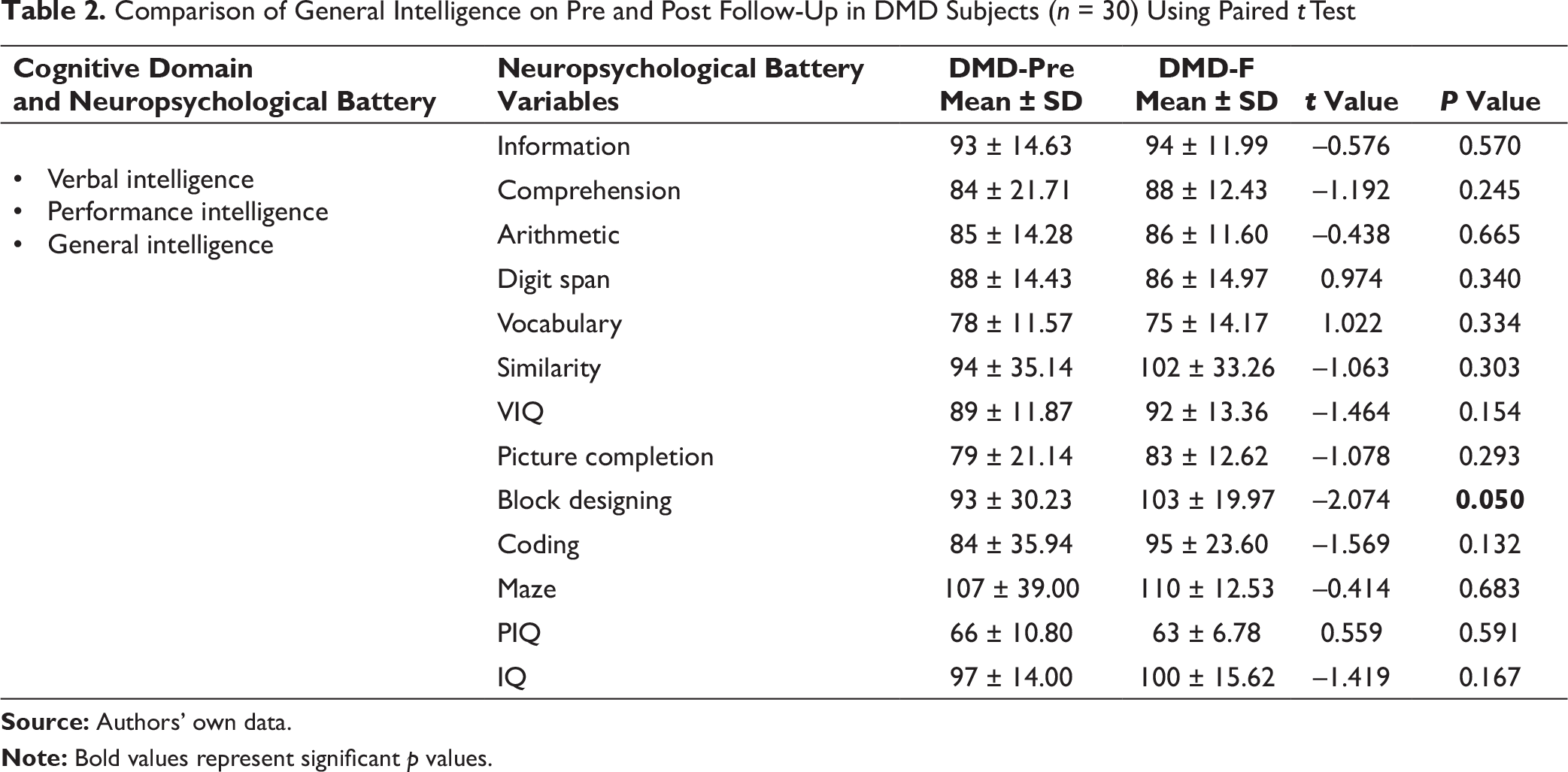
Comparison of Neuropsychological Variables in DMD Subjects on Follow-Up (n = 30) Using Paired t Test for RAVLT Variables
Comparison of Neuropsychological Variables in DMD Subjects on Follow-Up (n = 30) Using Paired t Test
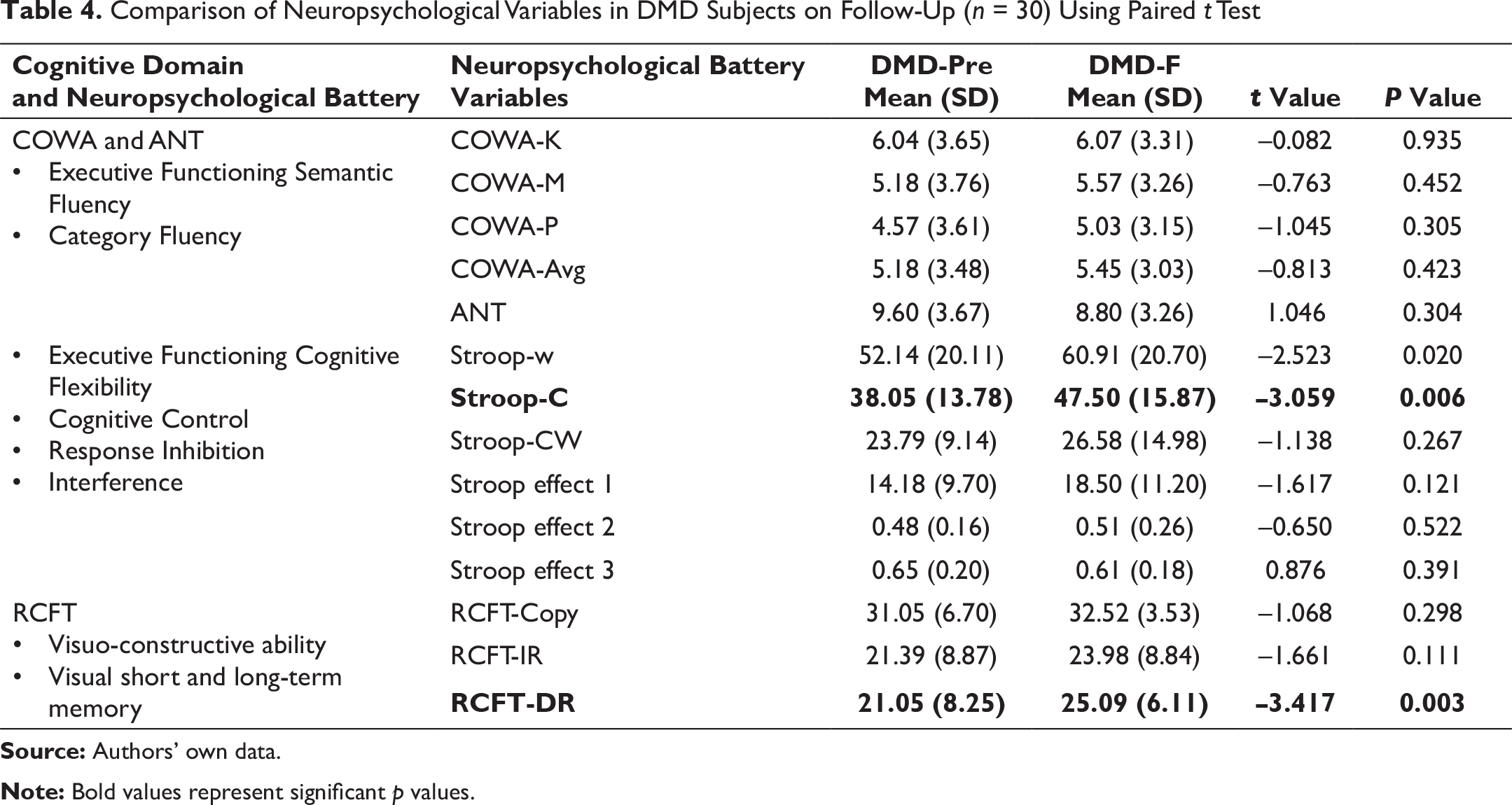
Comparison of Neuropsychological Variables in DMD Subjects on Follow-Up (n = 30) Using Paired t Test
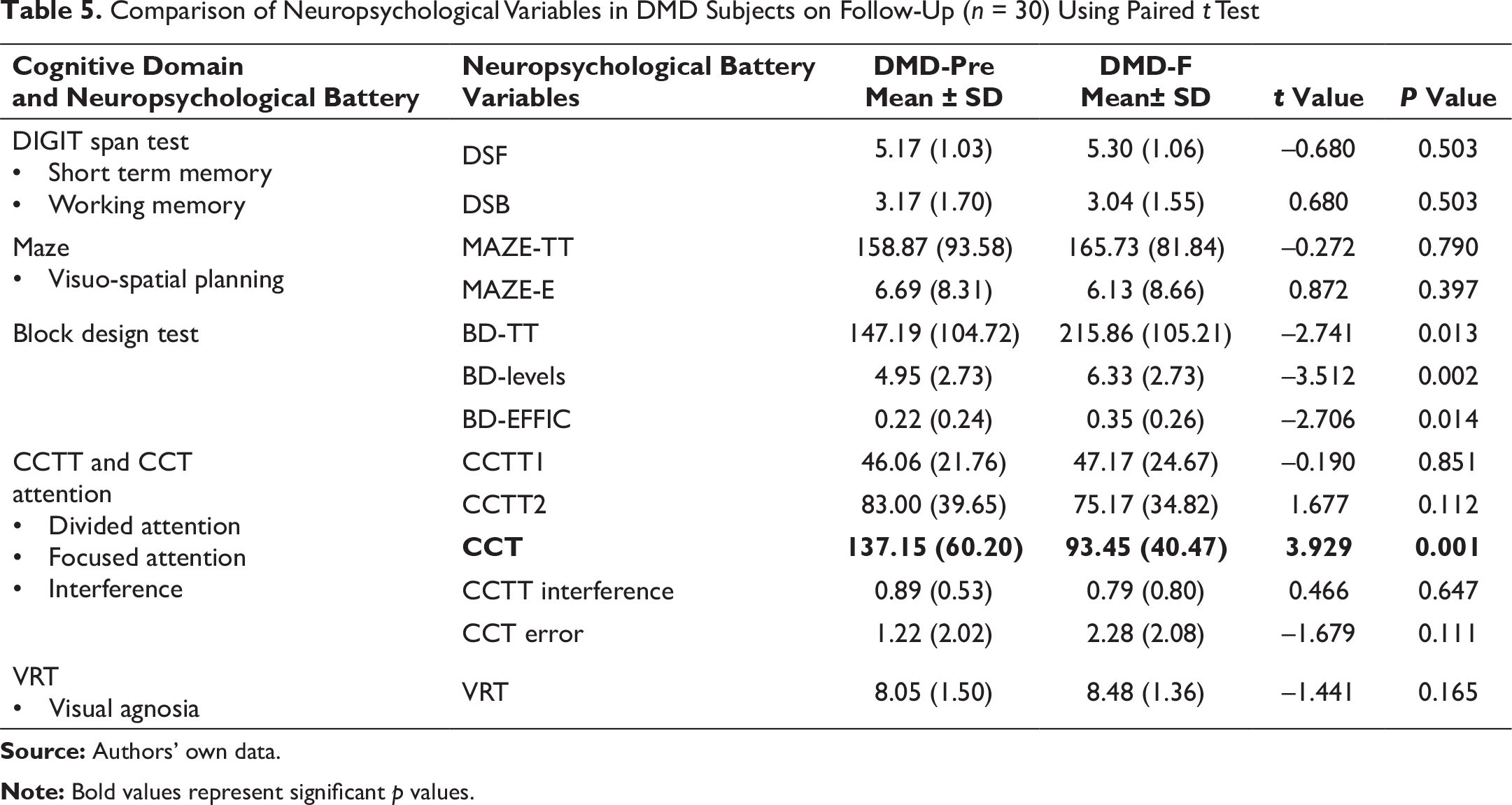
Representing Temporal Changes in Neuropychological Functioning Due to DMD Gene Mutation Affecting Dp140 Isoform
Effect of Mutation Location on Temporal Change in Neuropsychological Functioning
We analysed the trends of neuropsychological functioning in cases with distal mutation location affecting Dp140 isoform. Among 30 DMD subjects, 20 had mutations in the DMD gene affecting Dp140 isoform, that is, exon 44 or upstream. No changes in the cognitive and neuropsychological functioning were observed over time in majority of parameters except primacy, Stroop colour and word task-colour component, and RCFT-delayed recall, which showed improvement from baseline assessment as shown in Table 6.
Discussion
We provide a comprehensive longitudinal analysis of cognitive and neuropsychological profile in DMD subjects. The detailed analysis of neuropsychological domains and their progressive nature in boys with DMD provide better understanding of the use and effectiveness of specific rehabilitation regime required for retraining these patients. Additionally, this will enable future interventional studies targeting specifically impaired neuropsychological function.
When investigating cognitive process, analysing different aspects of the function is critical. In the present study, 30 boys with DMD were assessed for the progression of impairment in the general and specific cognitive domains over a mean follow-up duration of 10 months. The findings of this study showed that after a mean follow-up of 10 months, boys with DMD had no change in their general, verbal and performance intelligence. Data regarding non-progressive nature of intelligence was consistent with previous findings. DMDs have lower verbal IQ score than performance IQ score, and all IQ scores progressively reduce as the disease progresses.15, 21 The risk of cognitive deficit is determined by the location of mutation in the DMD gene that ensues specific functional dystrophin isoforms as described earlier. For example, patients who get lower IQ score were found to have a mutation in the distal region of the gene, whereas those with full-length mutation had highest scores. 22 However, our study confirmed superior cognitive performance on block design task, designed to assess visuospatial ability, with significant improvement in the designing efficiency.
The study also undertook the neuropsychological assessment of boys with DMD for the RAVLT. We found a significant improvement in serial positioning effect of primacy component. In this effect, the person is assessed for the tendency to better recall the first items in a list than those in the middle or last. The finding that DMD patients had improvement in primacy component reflects their ability to improve the long-term memory after repeated exposures. However, there is a paucity of evidence that showed this effect in DMD patients. A previous study investigating serial positioning memory of boys with DMD found their inability to sustain attention to the task; however, temporal changes were not investigated. 23
Furthermore, executive function and information processing speed were assessed with Stroop Colour Test (SCT), Stroop Colour and Word Test (SCWT), COWA test. Stroop test is used to measure cognitive flexibility and selective attention. 24 Examination was performed at baseline and during follow-up rounds. Our study found significant improvement in the SCT during follow-up, suggesting improvement in the executive function of this population. The improved performance on tests assessing executive functions such as cognitive flexibility is in contrast to a past study which showed poor performance on tests for executive function among DMD patients. 25 Chamova et al. reported poor performance on all neuropsychological tests (general cognitive abilities, verbal memory, attention and executive functions) in patients with non-functional Dp140 isoforms. 9 Remmelink et al. examined the effect of an absent full-length dystrophins (Dp427) on behavioural consequences in DMD patients and found a deficit in cognitive flexibility. 26
In our study, all other neuropsychological functions remained unchanged over the period. However, improvement in colour cancellation task, block design task, visual long-term memory and primacy effect indicate possibilities of improvement in cognitive domains. The domains that remained unchanged can be further analysed in future studies, by profiling the expression of dystrophin isoforms in post-mortem brain samples of the DMD patients. This will help elucidate underlying genetic basis for the observed variable phenotypic changes in the specific neuropsychological function. Additionally, interventional studies can enhance characterization of clinical and genetic variability and develop newer interventions specific to neuropsychological deficits. This may also serve to explore genotype-phenotype relationship in subsets of DMD patients with other coexisting neurodevelopmental disorders such as ADHD and ASD.
The significant improvement of executive functions in our study suggests that genetic prediction models can be developed to facilitate risk assessment, early detection and targeted treatment in such patient populations. Bailey et al. have recently developed a bioinformatics tool, called DMD Open access Variant Explorer (DOVE), to facilitate effective analysis of pathologic DMD gene variants, resulting in scope of precision medicine treatment for DMD. 27
The functional improvement observed during the follow-up period shows that boys with DMD may be more amenable to neurocognitive rehabilitation. The substantial economic burden of physical and neuro-developmental disability makes DMD patients vulnerable. Several studies have shown such economic burden of DMD on patients and their family.28, 29 Since the advent and progress in multidisciplinary management for DMD, the functional outcome, quality of life and longevity of the patients have significantly been improved.
Conclusion
The neuropsychological profiling of DMD patients provides a well-recognized pattern of cognitive strengths and weaknesses among DMD patients. This opens new vistas to explore other comorbid neurodevelopmental and neuropsychiatric disorders. The variation in phenotypic manifestation of neuropsychological deficits was found to vary with location of the DMD gene and effect of the mutation on CNS-expressed isoforms. Further research with larger sample size and multi time point analysis will be required to understand the involvement of various domains. The neuropsychological domains that remained unchanged need to be explored in future interventional studies with increased sample size in order to explore the changes on such domains and develop newer targeted neurocognitive interventions. Additionally, improved executive function in our study population reflects their receptibility to neurocognitive interventions. Future longitudinal studies with increased sample size and long-term follow-up are imperative.
Footnotes
Acknowledgements
We acknowledge Dr Mitali Mukerjee and their team at Institute of Genomics and Integrated Biology for providing resources and assistance. We thank Ms Sanjana Goyal, President, IAMD, for providing patient resources.
Author Contributions
Akshay Anand: Conceptualization, management of the study, editing and final approval the manuscript.
Rahul Tyagi: Co-conceptualization under supervision, genetic and neuropsychological data acquisition, experiments and analysis, statistical analysis, drafting and editing the manuscript.
Vivek Podder: Drafting the manuscript.
Harshia Arvind: Neuropsychological data acquisition.
Manju Mohanty: Supervision in neuropsychological assessment, analysis and validation of data.
Ethical Statement
The study was approved by Institute Ethics Committee of PGIMER, Chandigarh vide no. INT/IEC/2015/732 dated 19 November 2015.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding support was provided by Department of Atomic Energy, Mumbai, Government of India [Sanction No: 37(1)/14/53/2014-BRNS]. Fellowship support was provided by Indian Council of Medical Research (ICMR). We thank American Society for Human Genetics for travel award to first author.
Supplemental Material
Supplemental material for this article is available online.
