Abstract

On 23 June 2020 a criminal complaint was filed in a court located in Bihar state of India, against yoga 1 guru Swami Ramdev and Acharya Balkrishna, Managing Director, Patanjali Ayurved. The complainant alleged that the duo were misleading and putting at risk the lives of millions by claiming to treat the disease (COVID-19) caused by novel coronavirus, with a new Ayurvedic 2 medicine ‘Coronil’. Indian law barred companies from advertising any cure without proper government approval. During the televised press conference to launch Coronil (manufactured and marketed by Patanjali Ayurved), Ramdev had claimed that the product had shown 100% favourable results on mild and asymptomatic COVID-19 infected patients during clinical trials. Ayush Ministry of Government of India (GOI), the appropriate authority for granting permission to Ayurvedic drugs, had, however, notified that the details of any such scientific study were not known to it (The Hindu, 2020, June 24).
Ramdev had immediately retorted to all such allegations in national television by claiming ‘It seems that the work of yoga and Ayurved inside India is a crime. FIRs 3 have been lodged at hundreds of places in the same way cases are registered for a traitor and a terrorist’ (Prashasti, 2020). Taking a strong exception to those questioning Ayurved-based Coronil’s effectiveness to cure COVID-19, he pointed out at the multinational corporations that were making profits of ₹10 billion 4 were trying to run down the Indian cure for COVID-19 due to their vested interests. He assured that Patanjali had followed all the existing guidelines in developing Coronil and reaffirmed that the use of Coronil and Swasari 5 (together called Corona Kit) had shown 100% results in curing COVID-19 within 7 days. Balkrishna, in his interviews to newspapers, also stated that this controversy was a well-planned conspiracy to discredit Patanjali and Coronil. The ingredients, the claims and the license for Corona Kit were all clear (Amar Ujala, 2020).
On 30 June 2020, Ayush Ministry notified that the Coronil formulation was only an immunity booster and not a medicinal cure for COVID-19. On the same day, Ramdev used this notification to address many interviews in nationally broadcast television news channels to assure people who wanted to try these medicines. He claimed that as per the directions of the Ayush Ministry, the company would now be using the term COVID-19 management in place of COVID-19 treatment. He stuck to his claim that this formulation cured all mild-to-moderately ill patients. He also claimed that the Corona Kit would be now available across the country (The Hindu, 2020, July 1).
These events fuelled intense debate on broad topic of ‘information disorder’ on social media platforms, television, etc., questioning what could be construed as medical misinformation during a global pandemic (Seth & Waghre 2020)? There were also broader discussions about what should be the standards of ethics and legality in marketing of pharmaceutical products? Patanjali Ayurved launching the Corona Kit prematurely, and announcing Coronil as a cure for COVID-19 was intensely debated. Many were critical about the controversial promotional tactics, while another group considered it to be a brilliant marketing strategy. Debate also raged on the role to be played by different regulatory bodies of GOI like the Ayush Ministry, especially considering the fact that any effective vaccine or medicine against COVID-19 was still not in sight. Whether to use Corona Kit or not, was left to consumer’s judgement. This choice was made even more complicated by the news that the World Health Organization (WHO) was now acknowledging emergence of significant evidences of the airborne spread of the novel coronavirus (The Economic Times, 2020, June 9).
Coronavirus Pandemic: Origin, Growth, Impact and Its Treatment
The COVID-19 pandemic was a global health crisis and possibly the greatest challenge faced by the world since World War II. The virus emerged in Asia in late 2019 and, within a few months, spread to all continents except Antarctica. This pandemic, apart from being a health crisis, was also an unprecedented socio-economic crisis. Every day, for many months now, people were losing lives and livelihood, with no signs of return to normality. The International Labour Organization estimated that 195 million jobs could be lost. The World Bank projected a US$110 billion decline in remittances this year, which could result in 800 million people not being able to meet their basic needs. Overall, the pandemic had created devastating social, economic and political effects, which needed the entire world to join forces and fight the disease. The United Nations Development Programme (UNDP) was leading the socio-economic recovery, alongside WHO, which coordinated the health response, and the Global Humanitarian Response Plan were working under the leadership of the UN Resident Coordinators (United Nations Development Programme, 2020).
COVID-19 was a very contagious disease with symptoms like fever, cough, cold and vomiting. The risks were higher for people exposed to COVID-19 patients or had travelled to areas with ongoing community spread of the COVID-19 virus. Depending on the location, age and presence of co-morbidities of the suspected COVID-19 patient, the need for testing was decided. Generally, a long swab was used, to take a sample from the nose or throat, for conducting the test. At present, no medication was recommended to treat COVID-19, and no cure was available. Antibiotics did not work effectively against viral infections like COVID-19. Researchers across the world were testing a variety of possible treatments, vaccinations, etc. Some countries had approved or recommended some treatments like use of Remdesivir and corticosteroid dexamethasone to treat severe and emergency patients who required supplemental oxygen or mechanical ventilation (Mayo Clinic, 2019).
The COVID-19 disease was unlikely to go away soon and was expected to afflict a large proportion of the global population in the coming months. The situation was grim as recent studies concluded that there was a high chance of reinfection among patients if they were exposed to the virus again (Myupchar, 2020). The number of patients infected with COVID-19 and the death tolls as per the WHO reports were alarming (see Table 1).
COVID-19 Cases and Deaths in the World
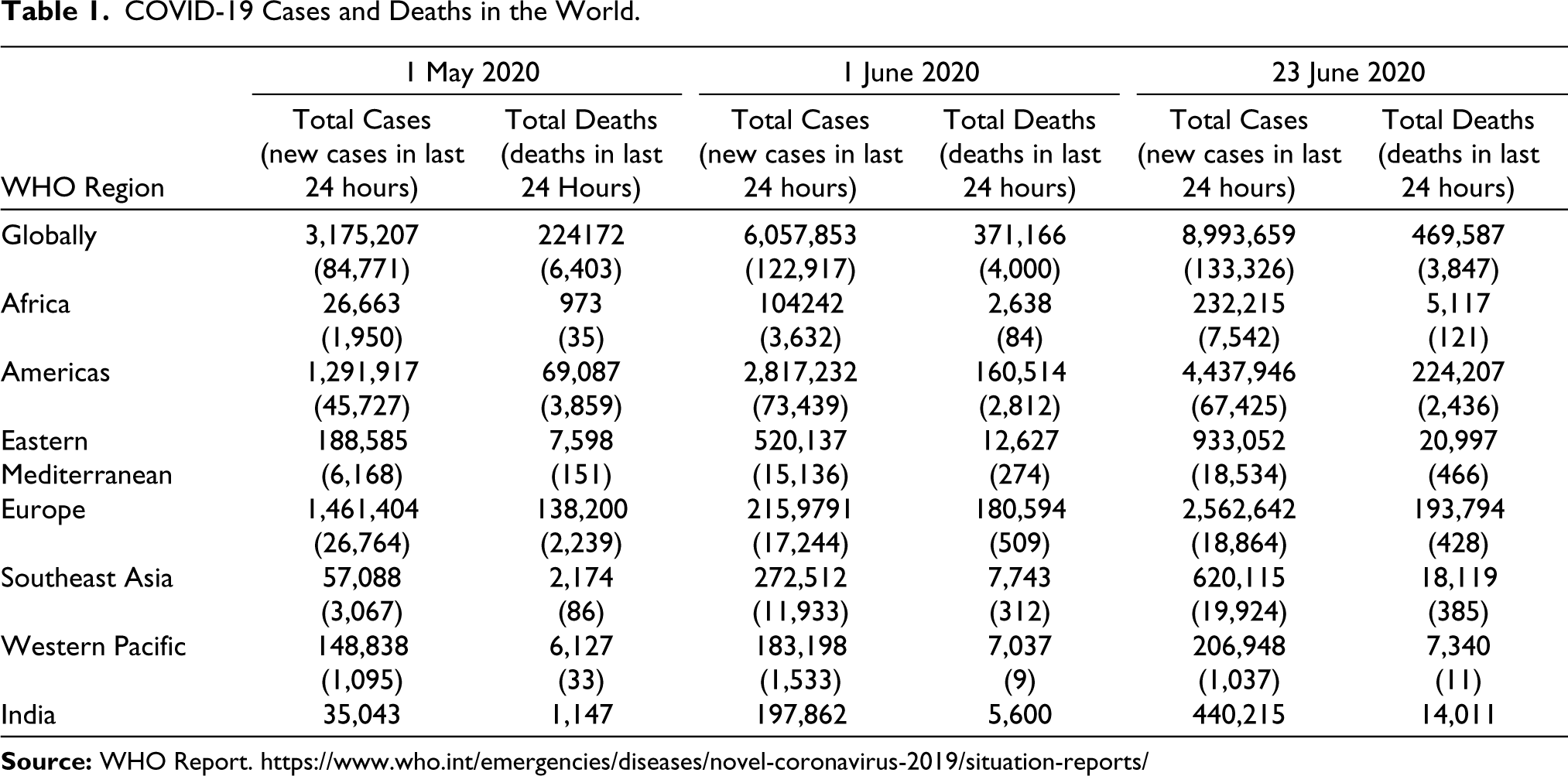
Research on COVID-19 in different parts of the world found that patents with comorbidities such as chronic pulmonary obstructive disease (COPD), diabetes, hypertension and malignancy did not respond as well to treatment; showed higher rate of complications; and higher rate of fatalities as compared to patients without such comorbidities (Guan et al. 2020).
Approval for Coronil
The Ministry of Ayush was formed by the GOI on 9 November 2014. It aimed at providing focused attention for the development of education and research in traditional Indian practices of Ayurveda, Yoga and Naturopathy, Unani, Siddha and Homoeopathy. Its specific target areas were to upgrade the educational standards of traditional systems of Indian medicines, as well as develop homoeopathy colleges in the country, developing research institutions to deliver time-bound research on diseases for which these systems were known to have effective treatment. It also designed and implemented schemes for promotion, cultivation and regeneration of medicinal plants used in these systems. Finally, it also focused on evolving pharmacopoeia and standards for Indian systems of medicine and homoeopathic drugs.
Ayurveda had very vivid analytical description of the stages and events that took place, since the causative factors started operating in the human body, till the final manifestation of disease. Hence, it was possible to know the possible onset of disease much before the latent symptoms became apparent. Thus, Ayurveda focused on preventive action against disease, by taking proper and effective steps in advance, to arrest growth and curb the disease in its earliest stage of onset. The principal objectives of Ayurveda were maintenance and promotion of health, prevention of disease and cure of sickness. Ayurvedic treatment focused on avoiding causative factors by the use of Panchkarma procedures, medicines, appropriate diet, activities and regimen for strengthening the body mechanisms to prevent or minimize re-occurrence of the disease (Ministry of Ayush, 2020). The Ayush Ministry prescribed a standardized process for development of Ayurveda-based drugs (see Figure 1).
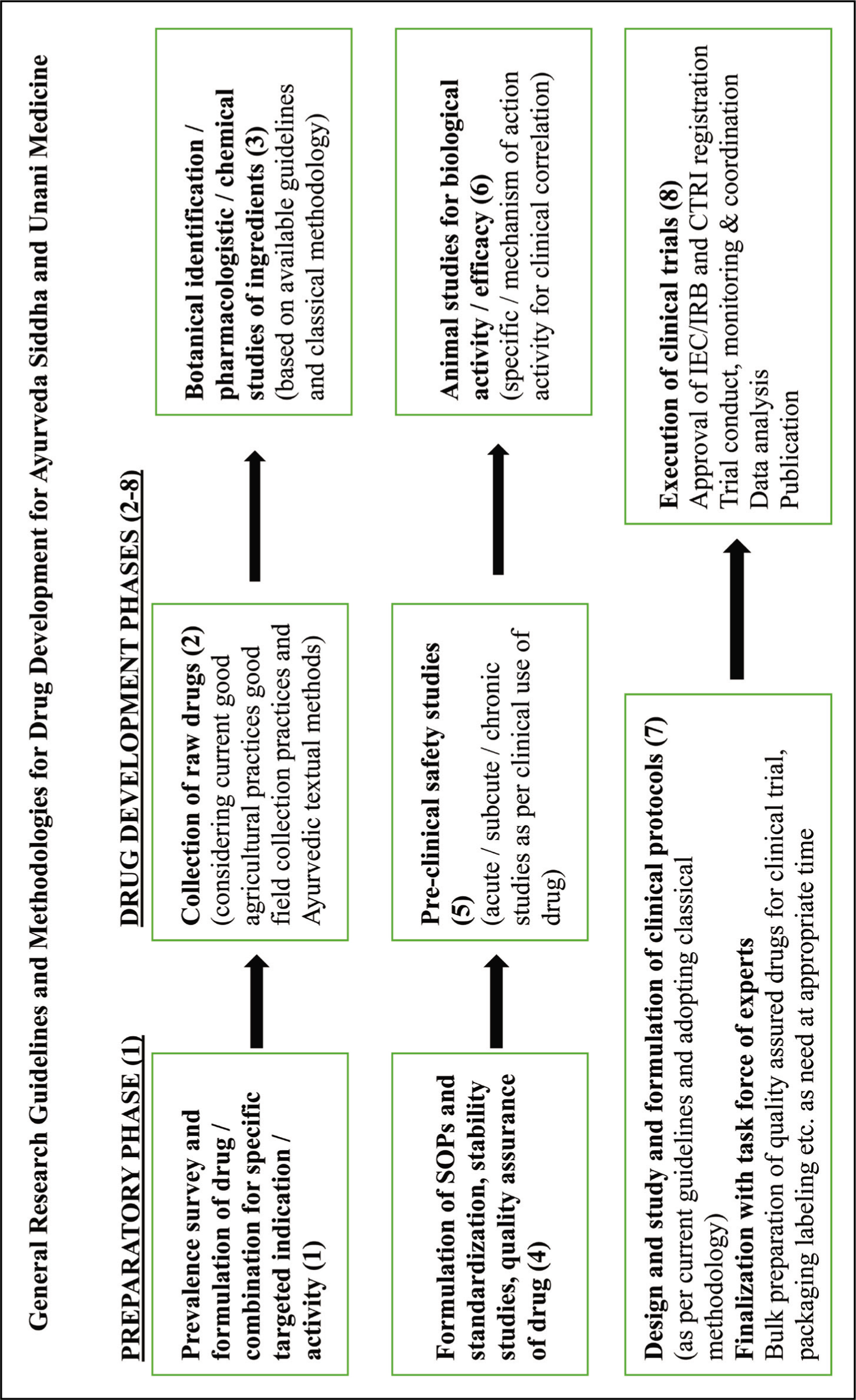
The Drugs and Cosmetics Act, 1940, exempted Ayurvedic formulations that were listed in ancient texts from clinical tests on efficacy. If the medicines were manufactured and sold for the ailments mentioned in those ancient texts, the licensing authorities only checked their composition. Despite this leeway, most Ayurvedic medicine manufacturers in India preferred to play safe. The packaging and marketing of such compositions typically described the contents as ‘beneficial in’, ‘helps fight’ or ‘boosts immunity’ against specific diseases. Patanjali, too, had often used similar descriptions for many of its products (Khandekar, 2020).
Ayurveda Department of Uttarakhand state was the designated office, under the Ayush Ministry of GOI, for granting license to Patanjali on Coronil. Immediately after the controversy broke out on 24 June 2020, the office confirmed receiving an application from Patanjali on 10 June 2020, which was approved by an examination panel on 12 June 2020. However, this permission was for manufacturing only 2–3 drugs as immunity boosters against cough and fever, and not a drug for COVID-19. The office was going to issue a notice to Patanjali demanding explanation about the launch of ‘Corona Kit’. The Ayush Ministry also issued a statement that Patanjali Ayurved Ltd. had been asked to clarify at the earliest the relevant details about the medicines being claimed will treat COVID-19, including the details of site(s)/hospital(s) where the research study was conducted, the protocol followed, sample size details, Institutional Ethics Committee clearance, copy of the registration in Clinical Trials Registry—India and the results of the studies conducted. Also, Patanjali was asked to immediately stop all advertising and claims about Coronil, till the issue was thoroughly examined and approval granted (Express Web Desk, 2020).
The decision of the Ayush Ministry of GOI—just to halt advertising and sale, but not taking any immediate legal action against Patanjali Ayurved—was criticized by many. There was a strong possibility that people were desperate for a COVID-19 cure, and if a well-known and popular personality assured that this product may help, it would definitely sell. This would help the company, but it could adversely impact the health of people, who would pay for that (The Wire, 2020)?
Patanjali Ayurveda and Coronil
Patanjali Ayurved Limited, one of the fastest growing consumer products company in India, was established in 2006. It was headquartered in the industrial areas of Haridwar, in the Uttarakhand state of India. The company manufactured and marketed around 450 products in the personal care and foods segments, and over 300 Ayurvedic medicines for the treatment of a range of body ailments (The Economic Times, 2019). Ramdev had announced, in 2018, that Patanjali Ayurved would be turned into a non-profit charitable trust, and all its revenues and profits would be used for charity, also its reins could be passed on only to a sanyasi. 6 ‘Turnover and profits have never been our goal and this has been the recipe of our success’, said Balkrishna (Shashidhar, 2018).
All Patanjali products were made from natural components and Ayurveda. Dental cream (Dant Kanti), hair oil (Kesh Kanti), herbal bath soap and honey were among its best-selling products (Indian Brand Equity Foundation). The company focused on the Indian middle-class consumers, promoting natural and pure Ayurvedic products, selling at significantly lesser prices than their competitors. In financial year (FY 7 ) 2018–2019, Patanjali Ayurved had reported a revenue of ₹0.833 billion, while the entire group’s turnover was much higher than this figure. The company had achieved a revenue of ₹0.35 billion during the first half of FY 2019–2020, and it expected to cross a revenue of ₹1 billion in FY 2019–2020 (The Economic Times, 2019). Patanjali Ayurved Limited was awarded the prestigious Certificate for Significant Achievement in Food Safety and Certificate for Strong Commitment to Food Safety, at the 14th Confederation of Indian Industry (CII) Food Safety, Quality and Regulatory Summit, on 11 December 2019, held at New Delhi. 8
The planned launch of Corona Kit was propagated in the media interviews given by Balkrishna as well as Ramdev since early June 2020. The company claimed that it had appointed a team of scientists, immediately after the COVID-19 outbreak, for finding a cure. First, a simulation study was carried out and compounds were identified, which could fight the virus and stop its spread in the body. Then, a clinical case study was conducted on hundreds of COVID-19-positive patients, and it showed 100% favourable results. ‘After taking our medicine, Covid patients recovered in 3–14 days and then tested negative. So, we can say the cure for Covid-19 is possible through Ayurveda. We are performing controlled clinical trials only. In the next 4–5 days, evidence and data will be released by us’, Balkrishna had claimed (Kumar, 2020a). Ramdev further announced that apart from Coronil, performing yogasans 9 and pranayam 10 daily was also very helpful in keeping the COVID-19 infection away (India TV Lifestyle Desk, 2020).
Some minerals with therapeutic effects and herbs Giloy, Ashwagandha and Tulsi were the main ingredients of Coronil. All the ingredients used were known for medicinal benefits from ancient times in India (Kumari, 2020). The recommended dosages were 2:2 tablets of Coronil and Swsari, to be consumed with hot water half an hour after meals for persons between 15 years and 80 years of age, and 1:1 tablet for those between the age of 6 years and 14 years (Nayak, 2020).
The Patanjali team claimed the following during the press conference held on 23 June 2020 to launch Corona Kit, which was widely attended by the mainstream national media in India (Kumar, 2020a):
We’ve prepared the first Ayurvedic-clinically controlled, research, evidence and trial based medicine for Covid-19. A total of 100 coronavirus positive patients were recruited for the controlled clinical trial. Of which 95 were studied, 1 patient was lost on the follow up details and 4 patients withdrew consent between the study. These were RT-PCR
11
confirmed cases, between ages 15–60 years. The study was conducted on patients with mild to moderate symptoms of Covid-19. Clinical case study & clinical controlled trial found 69% patients recovered in 3 days and 100% patients recovered in 7 days. These trials were jointly conducted by Patanjali Research Institute, Haridwar and National Institute of Medical Sciences, Jaipur. We are testing the medications on mice and rats, and shall even conduct tests on people on ventilators. We have done every research and will answer all questions. This is not only an immunity booster but also a Covid-19 cure. We have used minerals with herbs to make this medicine even more effective. There are no side-effects of the corona medicines which is being launching. We will also launch an e-commerce app for the delivery of the medicines within 2 hours for the order placement. This medicine kit is not available anywhere for now, will be made available at Patanjali stores in a week. The price would be just ₹545 for a corona kit, which will have medicines for 30 days. More technical details about the clinical trials and its results were made available to the media (see Table 2).
Details of the Clinical Trials of Corona Kit by Patanjali

#Interleukin-6—Previous retrospective studies indicated that an elevated level of interleukin-6 (IL-6) was associated with a high case fatality of the COVID-19 infection.
$COVID-19 infection is accompanied by an aggressive inflammatory response with the release of a large amount of pro-inflammatory cytokines in an event known as ‘cytokine storm’.
After the Ayush Ministry granted the status of only immunity booster for Coronil, the company announced that Patanjali’s Coronil medicine will be home delivered free of cost to poor people, who cannot afford to spend money to buy the medicine (India TV News Desk, 2020).
Controversies About Patanjali
In the past, Patanjali had been reprimanded many times for flouting rules. The first was for its atta 12 instant noodles, launched in 2016, without the necessary approval granted by the Food Safety and Standards Authority of India (FSSAI). The second was the sale of its Amla 13 juice, which was found to be ‘unfit for consumption’, by a state laboratory test on the product, and hence it was barred from Ministry of Defence’s canteen stores department.
In 2019, Business Standard 14 revealed that the company had acquired over 400 acres of forested, hilly common land in Kot in the Aravalli region by bending norms. It also highlighted that in June 2019, the Government of Maharashtra state had handed over another 400-acre plot of land in Latur district to Patanjali, which was earlier reserved for a prominent public sector undertaking in India. The Print, 15 in 2020, reported that the Government of Uttar Pradesh offered Patanjali a substantial land subsidy to set up a proposed food park in Greater Noida. This was carried out despite reports of the company’s unplanned expansion, poor supply chain, inconsistent product quality and business practices.
Patanjali had strong ambition to grow and drive indigenous products in India. It had made a range of products easily accessible to the masses. For instance, its aloe vera 16 gel helped a lot of people overcome acne, skin allergies and other dermatological problems, gaining it very encouraging reviews in Amazon, a popular e-commerce website in India. However, many of the over 2,500 products of Patanjali did not have sufficient empirical evidence to stand on, which were being offered at unbelievably affordable prices, available for countrywide delivery. A case in point being its Amla Churna, priced at ₹24 per 100 g, had ‘Amla’ as the listed ingredient. The benefits being claimed were ‘Boosts digestion and reduces constipation, cures eye problem, reduces hair fall, cures cold, has antioxidant properties’. However, only two of the six claims made and displayed were verified in the past research. Thousands of Patanjali’s formulations did not have verified tests available in the public domain, which cast doubts about the many of the claims made by the company about the benefits of its products and exposed the consumers to take on significant degree of risk. While systems around alternative medicine were far from developed, Patanjali products had encouraged a large base of consumers to consider Ayurveda as an accessible option for the first time. However, the ethics were questionable, and when it came to medicine, no matter what the system was, no ethics meant no medicine. More tangibly, the science of alternative medicine had very limited research. This was the common situation in many countries worldwide, not just India (Narayanan, 2020).
Patanjali Ayurved claimed in its website as well as sold ‘cures’ for many diseases such as dengue, chikungunya, swine flu, cataract and high blood pressure apart from many others. However, these claims neither had proper approval nor had adequate clinical evidences in the public domain. Many experts claimed that these actions mostly violated the provisions of the Drugs and Magic Remedies (Objectionable Advertisements) Act, 1954, of GOI, which strictly forbade the promotion of medicines for certain categories as ‘cures’ (Khandekar, 2020).
Ramdev was personally involved in and supported the Bharatiya Janata Party (BJP’s) 17 national election campaign in 2014. Within a year, he was not only named brand ambassador for the BJP-ruled state of Haryana, he was also granted a cabinet minister rank. Consequently, Ramdev was provided with all the facilities of a minister, which included a car with a red beacon, security personnel and an escort vehicle whenever he visited the state (Sanyal, 2020). Patanjali’s business also boomed immediately after BJP came to power in India—the world’s largest democracy, in 2014. Revenues of the company soared tremendously, and it also received more than an estimated ₹3.2 billion in discounts for land acquisitions in states controlled by the BJP (according to a Reuters review of state government documents, its interviews with relevant officials as well as real estate estimates). Also, the company gained access to many other lands free of charge. Patanjali, seemed to also enjoy an official approval from central government, as well as tacit support from many BJP leaders. On its part, advertisements for Patanjali’s products stress that they are Ayurvedic, meaning they are rooted in ancient Indian tradition. These advertisements echoed the talking points of the Indian Prime Minister Modi’s support base by appealing to consumers’ patriotism, urging them to avoid giving cash to foreign firms (Hindustan Times, 2017).
Patanjali had mostly been on the side of the Indian government, supporting the government’s initiatives from time to time. On 12 May 2020, when Prime Minister Modi called upon Indians to be ‘vocal for local’, Patanjali Ayurved Managing Director and Chief Executive Balkrishna announced the launch of ‘OrderMe’ application, supplying and endorsing only swadeshi 18 products. The application was designed to support the swadeshi movement by connecting all the local retailers and small shop owners, so that those selling swadeshi products could benefit from this platform. All the suppliers were allowed to join this platform and the plan was to deliver their products free of cost. Micro, small and medium enterprises (MSMEs) distributing domestic items would be encouraged to join this e-commerce site and benefit from it (ET Now Digital, 2020).
COVID-19 Prevention and Treatment in India
As the knowledge about, and the experience with, the COVID-19 increased, the medical treatment protocol also evolved. From the beginning of the outbreak, a series of measures were taken by the central and state governments in India to break the chain of transmission. One among these was to isolate all suspected and confirmed cases of COVID-19. Any suspected patient was advised home quarantine, along with undergoing a test to confirm the illness. In case the patient tested positive for the virus, healthcare professionals would assess if the residential setting was appropriate for home care. It meant that the patient was stable enough to receive care at home, and appropriate caregivers were available at home (who did not have high risk of contracting the virus themselves) (Centre for Disease Control & Prevention, 2020).
Patients suffering mild symptoms normally recovered on home treatment, and available data in India suggested that nearly 70% of all confirmed COVID-19 cases were such. However, rest of the 30%, who developed more severe forms of the disease, typically developed within 5–8 days, required hospitalization. Most patients required hospitalization of 10–14 days before being discharged. Some hospitals were classified as Covid Care Centres, who addressed cases that were clinically assigned as mild or very mild or as suspect cases. Dedicated Covid Health Centres were hospitals that handled patients clinically assigned as moderate. These were either a full hospital or a separate block in a hospital with preferably separate entry and exit zones. The Dedicated Covid Hospitals offered comprehensive care for patients clinically assigned as severe. Such patients were administered antiviral medicines, oxygen supply, and, in case of the intensive care unit (ICU) admissions, put on ventilators (The Economic Times, 2020, April 7),
On 31 Mar 2020, the GOI recommended use of the combination of Hydroxychloroquine (HCQ) and azithromycin for patients with severe infection, and those requiring intensive care. In May 2020, the main treatment changed to human immunodeficiency virus (HIV) drugs like the combination of lopinavir and ritonavir. On 2 June, the government approved the use of ‘remdesivir’ in case of emergency to treat the COVID-19 patients. Even for doctors and caregivers who were likely to have higher chances of contact with infected patients, the government recommended the prophylactic use of HCQ (
The cost of treatment for COVID-19 was nominal in government hospitals, its availability was limited and the service level was not satisfactory in many such facilities. In contrast, the private hospitals were charging exorbitant amounts, forcing the state governments to put a cap on such charges. For COVID-19 treatment, private hospitals were now allowed to charge between ₹13,000 and ₹15,000 per day for a bed without ventilators, between ₹15,000 and ₹18,000 per day for a bed with ventilators, while charges for a bed in isolation ward of a hospital had to be between ₹8,000 and ₹10,000 (Pati, 2020).
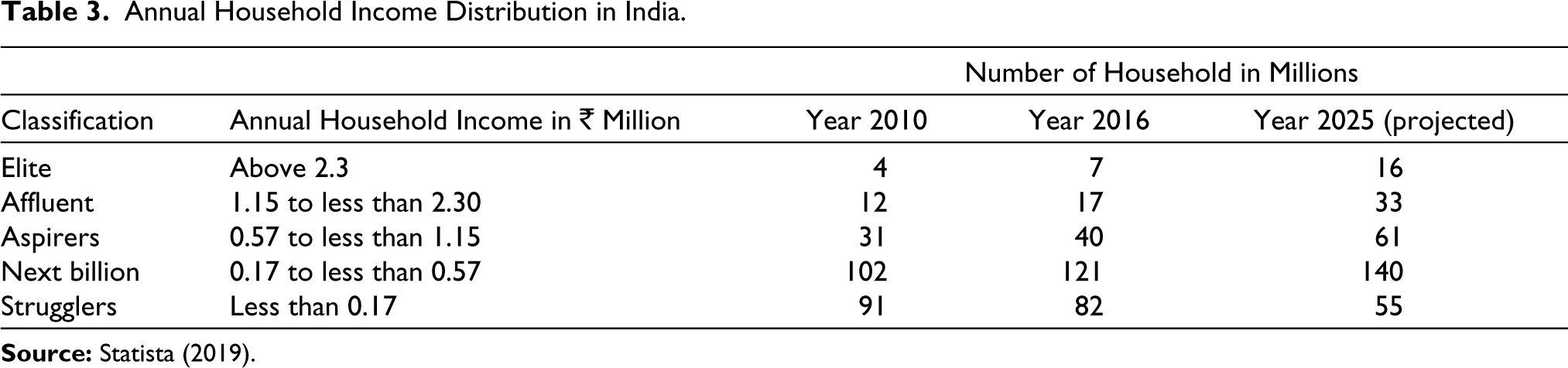
Most Indians did not have appropriate health insurance; hence, the hospitalization costs were mostly out of pocket for the ongoing COVID-19 pandemic. Even COVID-19-positive patients, who were covered by insurance, ended up spending as much as half of their hospital bills on personal protective equipment as such consumables were not covered by insurance (The Indian Express, 2020, July 7). Thus, the cost of hospital treatment for COVID-19 was very high for a large part of the Indian population (see Table 3 for annual household income). Thus, the GOI’s efforts to deal with the COVID-19 pandemic was on preventive approach, in consonance with the WHO guidelines (The Print, 2020). The GOI also extended free testing and treatment for COVID-19 in the public facilities at empanelled hospitals, benefitting more than 500 million citizens (Financial Express Online, 2020). To reduce the spread of the disease, the government imposed a lockdown in India from 23 March 2020 to 31 May 2020—the world’s largest lockdown, covering 1.3 billion people (BBC News, 2020). This controlled the spread of COVID-19 and provided valuable time to gear up the medical infrastructure for the fight against the virus. However, within a month of easing the lockdown restrictions, the COVID-19 growth rate became the highest among the world’s worst-affected countries. It was estimated that the COVID-19 cases in India were growing at a rate which was twice as fast as the rest of the world between 1 June and 28 June 2020. India recorded a growth rate of 3.85% in June, followed by Brazil at 3.5%, Russia at 1.6%, the USA at 1.3% and the UK at 0.5% (Rampal, 2020).
Annual Household Income Distribution in India
Six Indian companies were developing vaccines against COVID-19. In the first week of July 2020, the Indian government had given two firms the permission for the most promising vaccines, named COVAXIN and ZyCov-D, to start phase I and phase II human clinical trials (Kumar, 2020b).
Responding to the public interest case, the Ministry of Health and Family Welfare of GOI filed an affidavit in the Supreme Court, stating
The number of cases of Covid-19 are constantly increasing and at some point of time in near future, apart from existing hospitals, large number of temporary make-shift hospitals will have to be created in order to accommodate Covid-19 patients requiring admission, medical care and treatment. (The Indian Express, 2020, June 5)
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
