Abstract
Detection of spermatozoa from seminal stain is a vital piece of evidence in the examination of sexual assault cases, both in the case of a victim as well as a suspect. The conventional method of detection of spermatozoa from seminal stain uses acid, which many times leads to damage to the spermatozoa, resulting in poor detection. To overcome the issue, a new technique is proposed where there is no use of acid in the extraction of the sample from the seminal stain and subsequent preparation of the slides for microscopic examination. In the present study, both conventional and proposed techniques are compared in terms of the detection rate of spermatozoa and also microscopy scoring. It was found that the rate of detection of spermatozoa was far better in the proposed technique. In 80.4% slides, intact spermatozoa were detected with the proposed technique, whereas with the conventional technique, no intact spermatozoa were detected in any of the slides. The microscopy scoring was also found to be high in proposed technique compared to conventional technique.
Introduction
Sexual assault is one of the most tragic and unfortunate incidents in any society and India is not an exception.1, 2 Detection of spermatozoa is one of the most important pieces of evidence in the investigation into a case of sexual assault; many a time, the seminal stain detected may serve as vital evidence in such cases. There are various methods for the detection of semen stains in practice, which include alternate light source examinations, the acid phosphatase test and cytological methods. 3 However, detection of spermatozoa in the suspected stain is still the gold standard for the confirmation of semen. The different cytological stains used in forensic routine are hematoxylin and eosin stain, ‘Christmas tree’ stain using nuclear fast red and picro-indigocarmine and alkaline fuchsin.3, 4 However, hematoxylin and eosin is the cytological stain most commonly described in the scientific literature. 5 Semen stains suspected to be from a sexual assault can be found on clothing and the crime scene, resulting in a large array of potential materials for stains to be present. 6 Thus, these items provide an important source of legal evidence in forensic medicine. 7 There are various methods for preparing a seminal smear from these items in forensic fields.
The conventional method for semen extraction is by the rubbing method with the use of acid for extraction; although there are various other methods for improving the extraction rate of spermatozoa, these methods are either time-consuming or costly. 8 However, there is only a small number of published studies investigating the detection and identification of semen or spermatozoa in these items.7, 9 The proposed method of preparation of the slide is less time-consuming and cheaper and at the same time, the detection rate is also higher.
The main objective of the research was to compare the extraction of the sperm by the rubbing method without using acid with the conventional method using 1% HCL or 3% acetic acid.
Material and Methods
This was an analytical laboratory-based study done in the Department of Forensic Medicine at a tertiary care medical college. Institutional Ethical Committee approval was obtained.
The extraction was done by two designated technical staff at the Department of Forensic Medicine, one using the conventional methods and the other using the proposed method separately. The microscope slides prepared by the proposed method and the conventional method are examined by three designated faculty members individually for the detection rate of spermatozoa.
Sample Preparation
Samples of seminal fluid were collected from volunteers in sterile containers and were first studied for the motility, count and morphology of the sperm. Only those samples showing a normal study were considered for the study. The seminal fluid samples were then used to prepare seminal stains on Whatman filter papers; a total of 200 seminal stains/spots were prepared on Whatman filter papers using a micro pipette. For each stain/spot, 150 µl of seminal fluid was poured on the Whatman filter paper. Each stain/spot was air-dried and kept under normal conditions for use at various time intervals in this study.
Smear Preparation
The stained filter papers are then taken out and the stain is cut into two equal halves. These two halves are then used separately to make the smears on two separate glass slides using the proposed technique and the conventional technique.
The first slides were examined at the end of the first week after the sample preparation. The process is repeated with the remaining sample preparation at an interval of 1-week for the first 6 months, then every fifth day in the next 6 months. A total of 168 smears were prepared using both methods, separately and were used for the study.
Proposed Technique of Smear Preparation
One-half of the dried stain/spot on the Whatman filter paper is taken and a smear is prepared by gently rubbing the stain/spot on a clean, grease-free glass slide for 3 minutes. The slides are allowed to stand at room temperature for 10 minutes and then fixed in absolute alcohol for 10 minutes.
Conventional Technique of Smear Preparation
The other half of the dried stain/spot on the Whatman filter paper is moistened with a few drops of 1% HCl or 3% acetic acid in a glass Petri dish and kept for 1 hour. A smear is prepared by rubbing the moistened portion of the filter paper on a clean, grease-free slide. The slides are air-dried at room temperature for 10 minutes and then fixed in absolute alcohol for 10 minutes. 5
Staining
Slides prepared by both the conventional and proposed techniques of smear preparation are stained with hematoxylin and eosin stain. 10
Examination of Slides
Stained slides prepared by both conventional and proposed techniques of smear preparation are examined by three designated faculty members individually for the detection of spermatozoa under the 40× light microscope and the results are noted in a specially designed proforma for the study separately, which are then compiled and the data is analysed using SPSS version 26 for Windows.
Scoring Pattern: Scoring of the microscopy was done as described below.
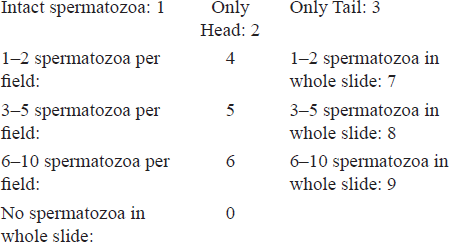
Results
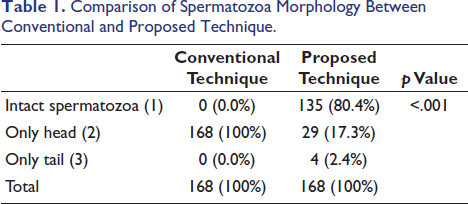
The present study shows that there are significant differences between the proposed technique and the conventional technique in the detection of intact spermatozoa from semen-stained filter paper. Out of the total 168 slides examined, starting from end of first week till end of the study, not a single slide showed intact spermatozoa in the slides prepared using the conventional method, however with proposed technique in 135 slide we could detect intact spermatozoa that is in conventional method detection rate of intact sperm was 0.0% whereas in the proposed technique it was 80.4%. In the conventional technique, in all 168 (100%) slides, only the head of the spermatozoa was detected. In the proposed technique, only the head was detected in 29 (17.3%) slides and the tail was detected in 4 (2.4%) slides out of the total (Table 1).
Comparison of Spermatozoa Morphology Between Conventional and Proposed Technique.
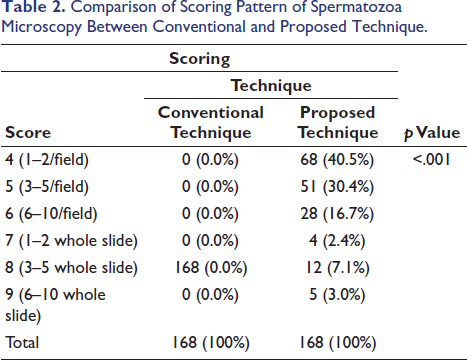
The study also shows that the scoring of microscopy was better with the proposed technique. While in the conventional technique, none of the slide spermatozoa were detected in every field of the slide (score >6), in all slides, the spermatozoa detected were 3–10/whole slide (score 8), which was 100% of the total slides examined. However, in the proposed technique, in 147 slides out of 168, spermatozoa were detected per field examined (score 4–6), which is 87.5% of the total. Out of these 147 slides, 68 (40.5%) scored 4, 51 (30.4%) scored 5 and 28 (16.7%) scored 6. In the proposed technique, there were 21 (12.5%) slides where spermatozoa were not detected in every field examined, which is much less compared to the conventional technique (Table 2).
Comparison of Scoring Pattern of Spermatozoa Microscopy Between Conventional and Proposed Technique.
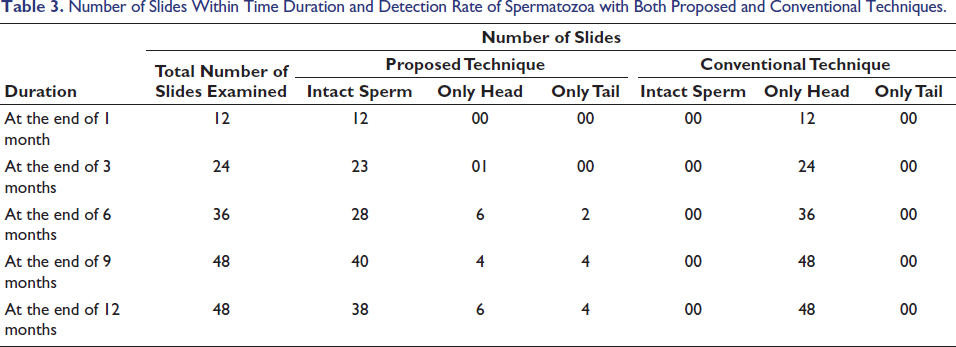
The study shows that in the proposed technique chances of getting intact spermatozoa from a sample which is up to 1 year is considerably higher compared to the conventional method. In our study, intact spermatozoa were detected when samples were smeared, stained and examined with the proposed technique even after 1 year, which is the maximum duration of our study. Out of 48 slides examined after 1 year, in 38 slides we could find intact spermatozoa (79%). However, in the conventional method, in none of the slides were any intact spermatozoa detected even after 1-week (Table 3).
Number of Slides Within Time Duration and Detection Rate of Spermatozoa with Both Proposed and Conventional Techniques.
Smear After 1 Month.
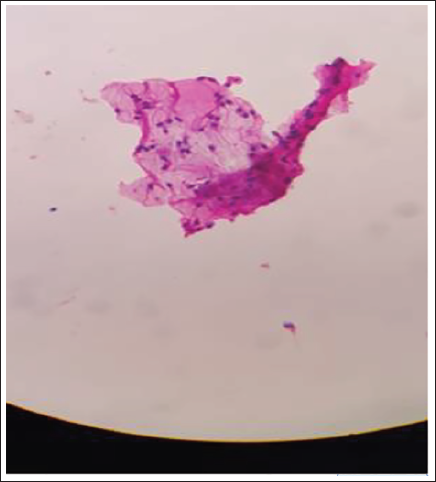
Smear After 3 Months.
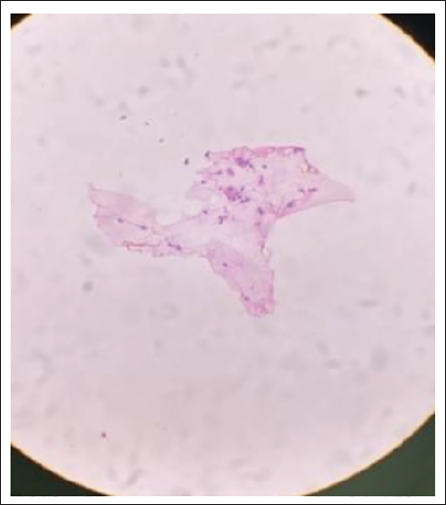
Smear After 6 Months.
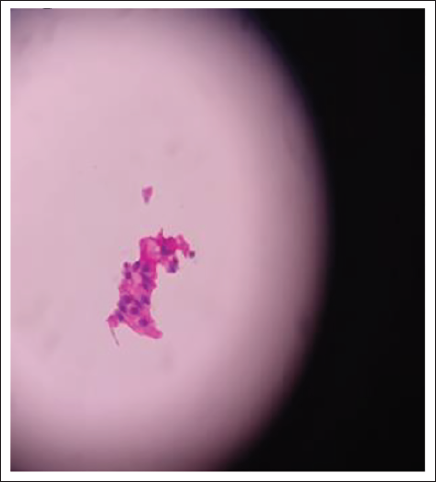
Smear After 9 Months.
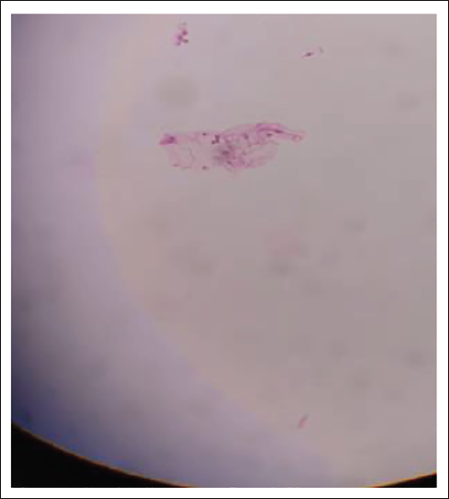
Smear After 1 Year.
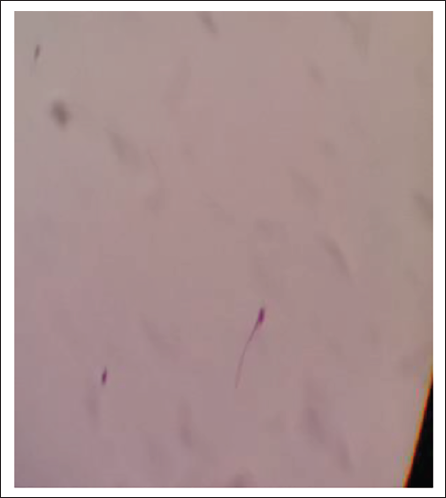

Discussion
It is always a challenge for forensic medicine specialists while examining the samples of suspected seminal stains. Many a time, the detection of sperm is not possible because of the time elapsed and also the method used for extraction. The morphology, including the dimensions of the sperm, depends on careful preparation of the smear, its fixation and staining, as it can affect the sperm dimensions significantly. 11 In this present study, an attempt was made to apply a novel technique for the detection of spermatozoa from seminal stain and the result was compared with the conventional method. It was found that the rate of detection of spermatozoa (both intact and fragmented) is better than the conventional method, particularly when the stain is older than 72 hours. With the conventional method detection of intact spermatozoa when examined after 72 hours is very difficult. 12 The acid in the conventional technique may damage the already fragile old spermatozoa. However, in the proposed novel technique, acid was not used. Hence detection rate of intact spermatozoa was better.
The study shows that in the proposed technique chances of getting intact spermatozoa from a sample even after passes of a long time are considerably higher compared to the conventional technique. In this study, samples were examined up to 1 year and with the proposed technique, intact spermatozoa were detected till the end of 1 year; however, the authors believe that had the sample been examined even longer than 1 year, intact spermatozoa could have been detected longer than 1 year.
Conclusions
The detection rate of intact spermatozoa and microscopy scoring of spermatozoa is better with our proposed technique compared to the conventional technique; it can be a better alternative.
Limitation and Need for Further Research
Authors feel that in this study, seminal stains were taken from Whatman filter paper, from where, by rubbing the sample could be collected, which may not be compatible when the stain is in garments; So, there is a necessity for more study in this regard, especially to see the compatibility of this technique in stains present in garments and other materials.
Footnotes
Acknowledgement
Prof. (Dr) Star Pala, Professor, Department of Community Medicine, for the support in analysing the data.
Dr S. Kishanth, currently pursuing DM in Forensic Radiology at AIIMS, Rishikesh, for his support when he was doing a PG course at the Department of Forensic Medicine, NEIGRIHMS, Shillong.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
Institutional Ethical Committee approval was obtained for the study and informed consent was also taken.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
