Abstract
Fixation is the first and most critical step in histopathological diagnosis, preventing autolysis and putrefaction while preserving tissue morphology. Conventional formalin fixation provides good cellular preservation but requires prolonged fixation time and poses health hazards. Microwave-assisted isopropyl alcohol fixation offers a rapid, formalin-free alternative that may reduce turnaround time without compromising diagnostic quality, particularly relevant in forensic autopsy tissues prone to rapid autolytic changes. To compare the efficacy of microwave-assisted isopropyl alcohol fixation with conventional formalin fixation in oral autopsy tissues. This study was carried out for a duration of one month. Five paired gingival tissue samples obtained during routine autopsy were fixed using either formalin or microwave-assisted isopropyl alcohol fixation. Slides were evaluated by three trained oral pathologists using predefined histological criteria. Formalin-fixed tissues showed better sectioning quality, whereas microwave-fixed tissues demonstrated superior staining characteristics. Other parameters showed no appreciable differences. Microwave isopropyl alcohol fixation demonstrated comparable efficacy to formalin with superior staining characteristics and reduced turnaround time, highlighting its potential role in forensic histopathology.
Introduction
Fixation is the first and most crucial step in histopathology, preventing autolysis and putrefaction while preserving tissue morphology. 1 With modernization, faster and more efficient techniques such as microwave-assisted fixation have emerged. 2 Rapid tissue stabilization is especially important in forensic pathology, where autopsy tissues are highly susceptible to postmortem autolysis and decomposition.3, 4 Microwave-assisted fixation accelerates fixative penetration, improves staining quality, and reduces exposure to toxic chemicals such as formalin and xylene, making it a promising alternative in forensic settings.5, 6
Despite these advantages, literature on microwave-assisted fixation in oral autopsy tissues remains sparse. Hence, the present study was undertaken to compare microwave-assisted isopropyl alcohol fixation with conventional formalin fixation in oral autopsy specimens.
Materials and Methods
This preliminary study was conducted at KLE Hospital, Belagavi, after obtaining approval from the Institutional Ethics Committee. Five routine autopsy cases were included, representing a limited sample size. Paired gingival tissue samples (2 mm thickness) were obtained during postmortem examination. One specimen from each pair was fixed in 10% neutral buffered formalin, while the corresponding specimen was fixed using microwave-assisted isopropyl alcohol fixation at 67°C for 10 minutes.
Sections were processed, stained with hematoxylin and eosin, and independently evaluated by three trained oral pathologists (experienced in forensic oral histopathology) for cell morphology, cellular details, nuclear details, staining characteristics, and sectioning quality. Each parameter was scored as poor (0), fair (1), or good (2). In case of disagreement, a consensus score was assigned. Statistical analysis was performed using appropriate non-parametric tests, with p < .05 considered statistically significant.
Results
All five paired samples were diagnostically adequate. On comparison between the two fixation methods, formalin-fixed tissues showed significantly better sectioning quality (p < .001), whereas microwave isopropyl alcohol fixed tissues demonstrated superior staining characteristics (p < .001). No statistically significant differences were observed for cellular morphology and nuclear details between the two fixation methods (p > .05).
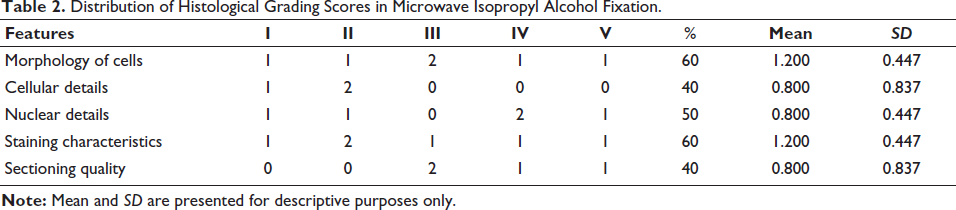
Detailed assessment of histological parameters in conventionally formalin-fixed tissues is summarized in Table 1, while the corresponding findings for microwave-assisted isopropyl alcohol fixation are presented in Table 2.
Distribution of Histological Grading Scores in Routine Formalin Fixation.

Distribution of Histological Grading Scores in Microwave Isopropyl Alcohol Fixation.
Discussion
There are numerous procedural innovations that have been developed in histopathology laboratory processes. Formalin fixation remains the standard method in routine surgical and forensic pathology; however, it is associated with several disadvantages, including delayed diagnosis, adverse chemical reactions, and degradation of nucleic acids. In addition, formalin is classified as a carcinogen, prompting the exploration of safer alternative fixatives.
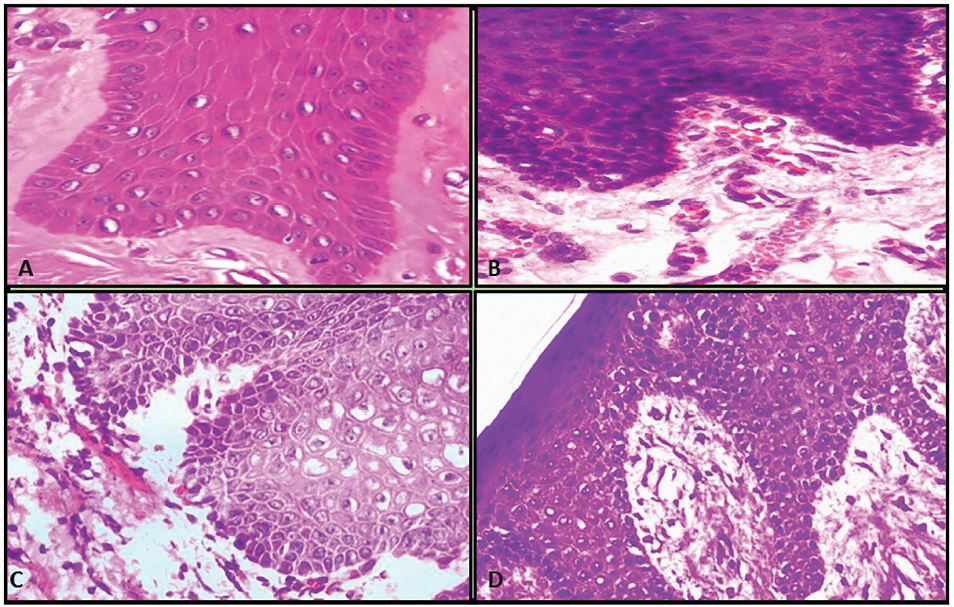
In the present study, isopropyl alcohol fixative demonstrated good preservation of tissue morphology, histochemical characteristics, and antigenic properties, comparable to formalin fixation (Figure 1C and 1D).
A & B: H&E Section Showing the Architectural Details of Gingiva (Autopsy Tissue) Using Routine Fixation. C & D: H&E Section Showing the Architectural Details of Gingiva (Autopsy Tissue) Using Microwave Fixation.
Previous studies have also highlighted the role of alternative fixatives and evolving histotechnology techniques in improving safety and efficiency in forensic histopathology.7, 8
These findings suggest that isopropyl alcohol can serve as an effective alternative fixative, particularly when rapid processing is required. In forensic pathology, microwave irradiation is employed to achieve rapid tissue stabilization, thereby preventing autolysis and putrefaction that may otherwise compromise tissue morphology (Figure 1). 5
Modifications in tissue processing techniques have enabled the diagnosis of small biopsy specimens within a shortened time frame; however, such approaches often require expensive equipment and highly skilled manpower. Although cryostat sectioning offers rapid results, it remains cost-intensive and is not routinely feasible in many forensic settings. In contrast, procedures such as fixation and impregnation are more convenient and time-efficient when performed using microwave-assisted fixation compared to conventional processing methods.
However, widespread adoption of microwave fixation may be limited by equipment cost and the need for trained personnel in forensic laboratories.
The innovative technique utilized in the present study accelerates fixation and tissue processing by eliminating multiple sequential steps of conventional processing, such as passage through graded alcohols followed by paraffin impregnation, and instead employs a simplified single-step approach. This approach minimizes intermediate processing steps, thereby reducing the need for additional clearing procedures (Figure 1). 9 Furthermore, the exclusion of hazardous chemicals such as xylene and formalin significantly enhances laboratory safety.
This reduction in processing time facilitates faster diagnostic decision-making, duplication of sections, evaluation of special stains, and minimizes the need for repeat biopsies. Our findings demonstrated no significant compromise in sample quality or diagnostic accuracy when microwave-assisted fixation was compared with conventional formalin fixation (Figure 1). Similar observations have been reported by other authors.10–13
On comparative evaluation of both fixation methods, most assessed parameters received scores of fair (1) to good (2), indicating that microwave-assisted isopropyl alcohol fixation is comparable to traditional formalin fixation in preserving morphological and staining characteristics of oral autopsy tissues (Figure 1). Thus, the present study demonstrates that microwave-assisted fixation produces good-quality tissue sections and holds promise for application in forensic histopathology.
One limitation of the present study is the relatively small sample size; therefore, further studies with larger sample sizes are warranted before recommending routine implementation of this technique in forensic practice.
Conclusion
Microwave-assisted isopropyl alcohol fixation provides rapid and reliable tissue preservation in oral autopsy samples. With further validation through larger studies, it may complement or replace formalin fixation in forensic histopathology, particularly in settings requiring rapid turnaround time. This technique shows promise as a time-efficient alternative to conventional fixation in medico-legal practice.
Footnotes
Acknowledgement
I thank all the teaching and non-teaching staff of the Department of Oral Pathology & Microbiology for helping and guiding me in this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Prior approval was obtained from the Institutional Ethics Committee (EC/NEW/INST/2021/2435).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
As the study involved postmortem tissues obtained during routine medico-legal autopsies and did not involve living human participants or identifiable personal information, informed consent from relatives was not required. The study was conducted following approval from the Institutional Ethics Committee.
