Abstract
The quality of death certificates (DCs) in many countries, including Ethiopia, is often inadequate, leading to significant implications for medical, legal, and administrative processes. This study evaluated the quality of DCs issued at St. Paul’s Hospital Millennium Medical College and Ras Desta Damtew Memorial Hospital in Addis Ababa. A retrospective review of 595 randomly selected DCs from September 11, 2020 to September 10, 2021, was conducted using a standardized evaluation tool developed by the University of Melbourne. All reviewed DCs contained errors, with the most prevalent being the omission of the time interval between symptom onset and death (100%) and ill-defined underlying causes of death (72.8%). Both hospitals in this study used paper-based DCs, but neither had implemented the internationally recommended format for DCs. The high frequency of errors underscores the need for targeted training for certifiers, implementation of quality assurance mechanisms, and adoption of standardized formats to enhance the consistency and accuracy of DCs.
Keywords
Introduction
Medical Certification of Cause of Death (MCCD) is a document of utmost importance in the medical, legal, and social realms. 1 It serves as a means of documenting and registering the facts and causes surrounding an individual’s death.1, 2 The MCCD holds significant value for various purposes, including legal, administrative, and health-related matters. Death certificates (DCs) provide important information for government officials, public health authorities, health studies, and families.1–3 However, concerns regarding the quality of DCs and their application in vital statistics often arise.1–5
Death certification is essential to furnish legal evidence regarding the cause of death (COD), to establish whether criminal activity was involved, and to resolve matters related to inheritance, insurance claims, and other legal affairs. Death certification is helpful not only to provide health-related data regarding COD and preceding illnesses but also to identify the leading COD, trends and patterns of mortality, risk factors, and determinants of health outcomes. They also guide health policy decisions and assist in setting priorities.1–5
The quality of DC and its applicability to mortality statistics depend on several factors, including data availability, completeness, accuracy, consistency, and timeliness. These factors are influenced by the format and process of death certification, as well as the knowledge and skills of the certifier. The accuracy of DCs relies on various factors, such as the certifiers’ proficiency in accurately identifying the sequence of events that led to death and their understanding of the concept and significance of the underlying cause of death (UCOD). 3 To ensure a standardized format for death certification, the World Health Organization (WHO) has recommended the use of MCCD. This certificate consists of two parts: (a) the UCOD and (b) other significant conditions contributing to the death. The information from the MCCD is then coded using the latest International Classification of Diseases (ICD) codes, which classify and aggregate COD for statistical purposes.6, 7
Several studies have reported common problems and issues with the quality of MCCD and its use for mortality statistics. Some of these problems include vague descriptions of conditions, such as modes of dying (e.g., cardiac arrest or respiratory failure) or mechanisms, rather than specifying the UCOD, like listing hemorrhage without detailing the specific injury. Other concerns involve clinically implausible sequences of COD, listing multiple CODs on a single line, incomplete or omitted information (e.g., failing to include relevant comorbidities or the duration of illness), and the use of abbreviations and illegible handwriting.1–5 These problems can affect the validity and comparability of mortality data and statistics, thereby compromising their usefulness for mortality studies.
In Ethiopia, the official and functioning outline of vital event registration was established by adopting a widespread rule. This rule was made obligatory by National Proclamation No. 760/2012. Vital events registration was implemented nationwide starting on August 6, 2016 and was reinforced by Proclamation No. 1049/2017.8, 9 However, the internationally recommended DC format prescribed by the WHO has never been used in Ethiopia. 10
In Ethiopia, there is limited information regarding the quality of MCCD and its common problems. Identifying common errors and concerns associated with death certification will be beneficial for improving hospital-based data and national statistics. It will also guide how to enhance the quality of certification and records in healthcare institutions. Therefore, we assessed the quality of DC in selected public health facilities in Addis Ababa, Ethiopia.
Materials and Methods
Study Setting, Design and Period
This study was conducted in Addis Ababa, the capital of Ethiopia, with a population of 3,774,000 as of the July 2021 Ethiopian Statistical Service projection report. Among the 12 public hospitals in Addis Ababa, St. Paul’s Hospital Millennium Medical College (SPHMMC) and Ras Desta Damtew Memorial Hospital (RDDMH) were randomly selected for this hospital-based cross-sectional study. DCs issued over a year from September 11, 2020 to September 10, 2021, were reviewed retrospectively. The data were collected from May to June 2022.
Population
Records of all deaths that occurred at SPHMMC and RDDMH during the study period were the source population. The sampled records of deaths in both hospitals were the study population. Cases referred for autopsy were excluded from the study.
Sample Size and Sampling Procedure
The sample size was calculated using a single population proportion formula considering a 95% confidence level (Zα/2 = 1.96), a 5% margin of error, and a 48.5% estimated prevalence of a major error on DCs, based on a previous study conducted at Tikur Anbessa Specialized Hospital. 10 After adjusting for 10% of non-response for the unreadable or lost records and a design effect of 1.5, the final estimated sample size was 634.
The lists of medical record numbers of deaths registered at the facilities were separately obtained from Health Management Information System logbooks and used as a sampling frame. Over the study period, SPHMMC and RDDMH recorded 1,892 and 769 deaths, respectively. To ensure representativeness, the samples were allocated proportionally to each hospital based on the total number of DCs during the study period. Participant records were selected using a simple random sampling technique using computer-generated numbers.
Data Collection
The quality of the DCs was assessed by a structured data abstraction template designed using a rapid assessment tool adopted from the University of Melbourne. 5 The data collection tool comprised patients’ information on the socio-demographic data of the deceased, duration of stay, time and place of death, qualification of the certifier and treating physicians, manner of death, DC format, and a rapid assessment checklist. The data collection form was pretested on 50 cases, and the necessary amendments were made. Data were collected by well-trained medical residents and supervised by the principal investigators. All collected data were examined for completeness and consistency. Incomplete or incorrect entries were corrected daily by reviewing the original record using the assigned code.
Assessment Tool
The data collection tool served as a checklist to identify common errors in MCCDs. The assessment criteria classified errors as either major or minor, based on their impact on determining the UCOD. Major errors were defined as errors that directly affect the determination of UCOD, such as listing multiple causes per line, incorrect sequencing of events leading to death, entering ill-defined conditions as the UCOD, and illegible handwriting. Minor errors were defined as errors that did not directly affect the determination of UCOD, such as the presence of blank spaces within the sequence of events, the absence of disease time intervals, and the use of abbreviations. 5
Data Analysis
Data were analyzed using IBM SPSS Statistics for Windows (version 26). Descriptive statistics were utilized to summarize categorical variables with proportions and frequency tables. Continuous variables were summarized using the mean and standard deviation.
Results
After excluding the lost records, we reviewed 620 sampled DCs and corresponding medical records; 441 were obtained from SPHMMC, and 179 were obtained from RDDMH. Among these, (25 DCs, 4.03%) had illegible handwriting and were omitted from further review. The study included 595 DCs with a 94% retrieval rate: 71.6% (n = 426) from SPHMMC and 28.4% (n = 169) from RDDMH. The mean age at death was 46 ± 17.06 years, with males representing 58.2% (n = 346) of the cases. Most deaths (65.7%) occurred in the 25–65 age group, followed by individuals older than 65 years, accounting for 21.7% of the deaths (Table 1).
Demographic and Health Facility-related Characteristics of the Deceased for Whom Death Certificates Were Issued at the SPHMMC and RDDH From September 11, 2020 to September 10, 2021.
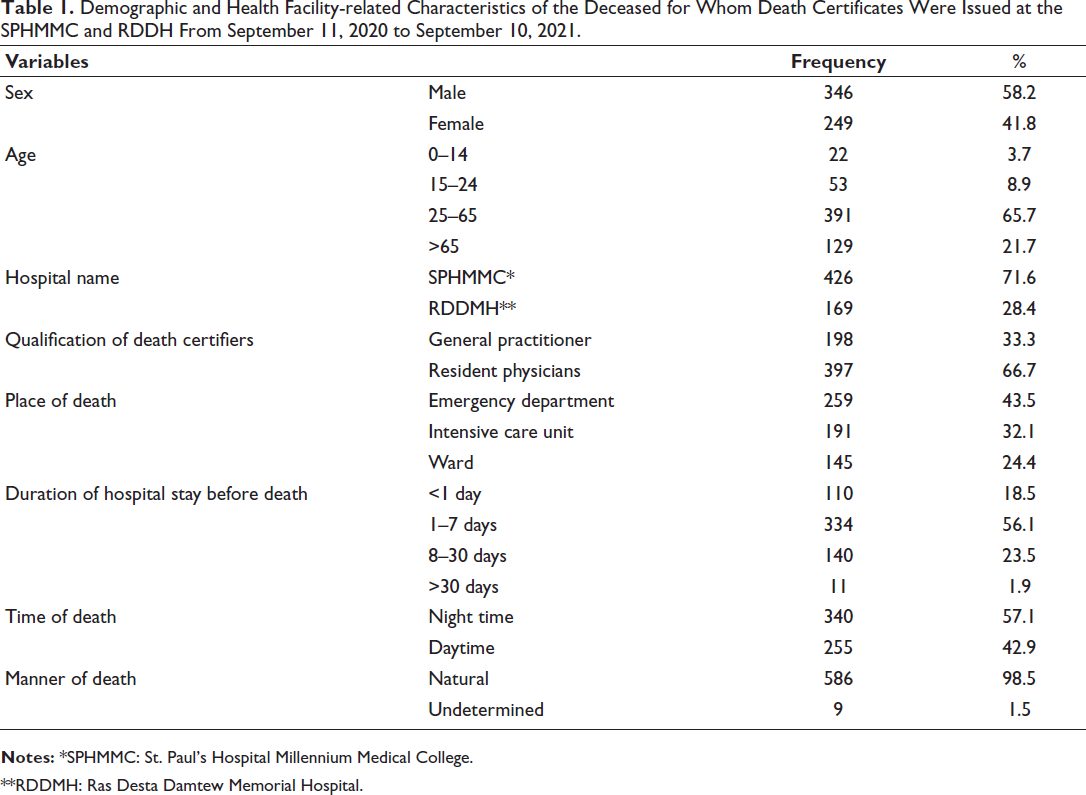
**RDDMH: Ras Desta Damtew Memorial Hospital.
Medical residents issued about two-thirds (66.7%) of DC, while general practitioners issued the remaining third (33.3%). A substantial number of deaths (56.1%) occurred after hospital stays lasting between 1 and 7 days. Most DCs (98.5%) indicated a natural manner of death, with only 1.5% classified as undetermined. The majority of deaths occurred in the emergency department, accounting for 43.5% of the cases (Table 1).
Errors in Death Certificates
Both hospitals in this study used paper-based DCs, but neither adhered to the standardized format recommended by the WHO. The DC form used in both hospitals did not include the standard part one and part two formats, making it impossible to evaluate errors such as listing multiple CODs on a single line or leaving blank spaces.
All the issued DCs (100%) contained errors. The most common error was the omission of the time interval between the onset of symptoms and death (100%), followed by an ill-defined UCOD. Major errors included ill-defined UCOD (72.8%; 95% CI: 69.1%, 76.2%), clinically improbable sequences of COD (46.8%; 95% CI: 42.8%, 50.8%), and unspecified neoplasms (6.4%; 95% CI: 4.7%, 8.6%). Among these ill-defined conditions, the mode of death was the most frequently listed at 60.9%, followed by the mechanism of death at 33.9%. Furthermore, ill-defined conditions such as laboratory values, signs, and symptoms were mentioned in 4.7% and 0.5% of the certificates, respectively. Table 2 summarizes the frequency of major and minor errors identified on DCs.
Types of Errors in Death Certificates Issued at SPHMMC and RDDMH From September 11, 2020 to September 10, 2021.
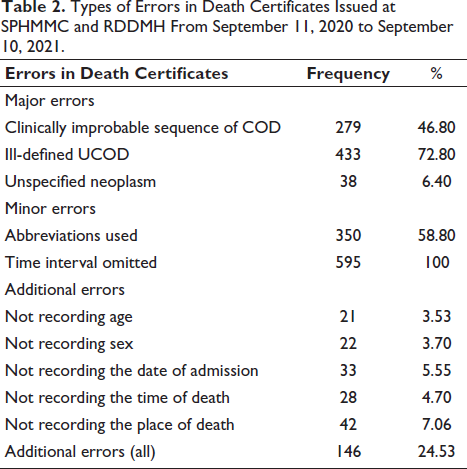
Overall, all the DCs reviewed contained errors. Among these, approximately 3.7% of the DCs had one, 25.9% had two, 40% had three, and 30.4% had four or more errors. When considering the severity of these errors, 11.9% of the DCs had no major errors, while 50.9% had one major error. In addition, 36.3% of the DCs contained two major errors, and only 0.8% had three major errors. Regarding minor errors, 34.1% of the DCs had a single minor error, 57.5% had two minor errors, and 8.4% had three minor errors.
Discussion
This study evaluated the quality of and challenges related to the certification of deaths in selected public health institutions in Addis Ababa, Ethiopia. It was observed that healthcare facilities in Ethiopia have not yet adopted the internationally recommended DC format provided by the WHO. A systematic review conducted to evaluate common errors in DCs revealed that the use of non-standard DC forms was a key factor leading to inaccuracies in the completion of DCs. 2 Adhering to these guidelines and forms is crucial for guaranteeing the precision and excellence of medical death certification, as well as for producing mortality data that can be compared across countries, national and subnational populations, specific population groups, and different periods. Consequently, we urge immediate adoption of the standard DC format recommended by the WHO in Ethiopia.
The most significant finding of this study is that all the DCs examined had errors. This finding is consistent with previous research conducted in Addis Ababa and other countries, indicating that errors in death certification are a universal problem. A review of studies from various countries revealed that the percentage of DC errors ranged from 20% to 100%, with most studies reporting error rates above 90%. 1 Additionally, several studies from Egypt, Saudi Arabia, India, and Korea reported that none of the DCs examined were error-free.11–17 According to a recent systematic review, the main causes of DC errors are a lack of training or poor training for certifiers, a lack of understanding among physicians about the significance of DCs, and the absence of quality control mechanisms. 2 This finding is strongly supported by studies from India, which identify a lack of formal training and knowledge, as well as work overload, as the primary causes of errors. These studies recommend formal training and the implementation of audit systems for quality control.16–21 To improve the quality of DCs, ongoing and targeted training for certifiers should be provided, quality assurance or control mechanisms should be established, and tracking mechanisms should be implemented to ensure the completion of accurate DCs. Moreover, standardizing DC forms to comply with WHO requirements and potentially using electronic DC forms are also suggested as feasible solutions.
In this study, the most common error was the omission of the time interval between symptom onset and death, followed by listing ill-defined UCODs. The study revealed that omission of the time interval between symptom onset and death was observed in 100% of the DCs. Similar findings have also been reported in studies conducted at Tikur Anbesa Specialized Hospital in Addis Ababa, Ethiopia, as well as in other countries, including Iran, India, and Egypt.10, 12, 22, 23 The potential reason for the exclusion of the time between the onset of symptoms and death may be attributed to the absence of a designated space for recording the time interval in the outdated DC format employed by healthcare facilities. Additionally, this could also be due to a lack of awareness regarding the importance of documenting this information. Although missing time intervals were considered minor errors in these studies, recording accurate time intervals is crucial for identifying UCOD and preventing sequencing errors.
Another prevalent error observed in this study was the documentation of ill-defined conditions as a COD. The mode and mechanism of death were frequently listed as ill-defined conditions in this study. The study also revealed that an incorrect sequence of events was present in 46.8% of the DCs. This error was also observed in previous studies conducted in India, Egypt, Iran, and Palestine.12, 20, 24, 25 Incorrect diagnosis, sequence, or incorrect completion of DC can lead to errors in mortality statistics, directly impacting the formulation of national health policy. Likewise, ill-defined conditions have limited value from a public health perspective.3, 5, 26 Hence, it is strongly advised to refrain from documenting ill-defined conditions in DCs and strive to offer more precise information whenever possible.
However, there are challenges in accurately certifying COD in medical practice.1–5 The accuracy of the MCCD can be influenced by factors such as certifiers’ knowledge and expertise in recognizing the series of events leading to death and their understanding of the concept of the UCOD. Nevertheless, medical students are rarely taught about the public health significance of accurate COD information, the concept of the UCOD, the sequence of events leading to death, and how to correctly complete the MCCD. Previous research and experiences from the Bloomberg Data for Health initiatives have suggested that the training provided to medical students on death certification is insufficient.2, 3, 5 It is imperative to introduce or enhance training on COD certification for medical students. Additionally, implementing measures such as continuous training and quality assurance can help improve the quality and reliability of DCs. Further research is needed to assess the quality of the MCCD in other health facilities and regions of Ethiopia and to evaluate the impact of interventions aimed at reducing errors in death certification.
The study showed that the use of abbreviations was the third most prevalent error in DCs, with 58.8% of the certificates containing this error. This finding is consistent with previous studies conducted in Ethiopia, India, and Bangladesh, which also reported high error rates related to the use of abbreviations.10, 23, 26 Abbreviations should not be used in DCs as they could result in misinterpretation of COD, incorrect coding, and erroneous statistics. 2 The error of illegible handwriting was found in 4.03% of the DCs in this study. This rate was lower than that of similar studies conducted in other countries.17, 22, 24, 25 To reduce errors such as the use of abbreviations, illegible handwriting, and omissions of information, digitalizing DC forms is an effective solution. In addition, the implementation of quality assurance and tracking systems that ensure the timely completion of incomplete DCs can be helpful. 2
In this study, most of the deceased individuals (66.7%) were treated by senior physicians, medical residents, and medical interns at different times during their hospital stay. Medical residents issued DCs for these individuals. Conversely, the remaining 33.3% of the deceased individuals received treatment from senior physicians, general practitioners, and interns, and their DCs were issued by general practitioners. Inaccuracies in death certification may arise from work overload because only residents or general practitioners are responsible for certification in the study facilities. 2 To address this issue, we recommend that DCs be completed by a physician who is familiar with the patient, even if this may lead to delayed completion of the DC. Additionally, medical certifiers are strongly encouraged to discuss DCs with the clinical care team. Countersigning DCs by senior medical staff, who are likely to possess greater knowledge and experience, is also crucial. Furthermore, our finding of an undetermined manner of death in 1.5% of the reviewed documents, which were not referred for autopsy, is concerning. When certifying a death, if the manner is classified as undetermined based on the information available, it is crucial to recognize that these cases may involve unnatural causes and should be referred for a postmortem examination. This practice is essential for accurately determining both the cause and manner of death, and all physicians should be made aware of this protocol.
Our study revealed a significant occurrence of DC errors in selected public hospitals in Addis Ababa, Ethiopia. The omission of the time interval between onset and death and the listing of ill-defined UCODs were the most common errors identified. To improve the accuracy of DCs, we recommend implementing ongoing targeted training for certifiers, establishing quality assurance mechanisms, and introducing tracking systems to ensure accurate completion of DCs. Additionally, healthcare institutions should adopt the internationally recommended DC format endorsed by the WHO to improve data consistency and reliability. Further research is needed to assess the quality of DC in other health facilities and regions of Ethiopia and to evaluate the impact of interventions aimed at reducing errors in death certification.
Footnotes
Abbreviations
DC: Death certificate
MCCD: Medical Certification of Cause of Death
COD: Cause of death
ICOD: Immediate cause of death
UCOD: Underlying cause of death
ICD: International Classification of Diseases
WHO: World Health Organization
SPSS: Statistical Package for Social Sciences
SPHMMC: St. Paul’s Hospital Millennium Medical College
RDDMH: Ras Desta Damtew Memorial Hospital.
Acknowledgements
The authors would like to express their sincere gratitude to St. Paul’s Millennium Medical College and Ras Desta Damtew Memorial Hospital for their support in the accomplishment of this study. We would also like to thank the data collectors (medical residents) for taking their precious time to collect the data.
Authors’ Contribution
NGB contributed to the conceptualization, data curation, methodology, formal analysis, investigation, resources, software, validation, visualization, and writing both the original draft and review and editing of the manuscript. BL and YW were involved in the conceptualization, data curation, methodology, formal analysis, investigation, supervision, validation, visualization, as well as review and editing of the manuscript. SAB participated in the conceptualization, data curation, methodology, formal analysis, investigation, validation, visualization, as well as writing both the original draft and review and editing of the manuscript. ASL contributed to the conceptualization, data curation, methodology, formal analysis, investigation, supervision, validation, visualization, and writing both the original draft and review and editing of the manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Ethical approval for the conduct of the study was obtained from the institutional review board (IRB) of Saint Paul’s Hospital Millennium Medical College (Reference number of PM 23/7). All methods were performed in accordance with relevant guidelines and regulations. All the information was treated anonymously and confidentially using unique identification codes rather than individual identifiers. This study received ethical approval from the Saint Paul’s Hospital Millennium Medical College IRB (Reference number of PM 23/7) on July 07, 2022. This is an IRB-approved retrospective study; all patient information was de-identified, and patient consent was not required. Patient data will not be shared with third parties.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
