Abstract
The phenomenon of crime is not ordinary; behind every criminal act lies a motive, and after every crime, there exists a trail or clue. These traces are crucial for investigators and forensic scientists in establishing the perpetrator and linking them to the crime. With the advancement of technology, the methods employed in committing crimes have also evolved, and the ready availability of poisons has made the act of killing distressingly simple. Poisons, whether organic or inorganic compounds originally intended for eradicating insects, pests, or rodents, are unfortunately misused to end human lives. In cases where poisons are employed, tracing the specific poison to determine the cause of death is imperative. However, when poisons are mixed with other substances, such as liquids, the task of tracing them becomes significantly challenging. One such medium where poisons can be surreptitiously introduced is beverages. The focus of this study is to trace poisons mixed in beverages containing citric acid and to ascertain their stability period, during which the presence of the poison can be detected—a critical timeframe within which poison traces can be identified. To conduct this study, pesticides including cypermethrin, chlorpyrifos, and phenthoate were chosen and mixed with beverages containing citric acid. Ultraviolet spectroscopy was employed to analyze the samples. The study’s findings indicate that, with the exception of cypermethrin, the stability of the other two pesticides, chlorpyrifos, and phenthoate, was compromised, leading to chemical changes and loss of stability over time.
Introduction
Crime is a universal phenomenon, yet the methods employed by criminals vary widely and are often unique to each individual case.1, 2 While understanding the motive behind a crime is crucial for its resolution, evidence holds paramount importance in convicting the perpetrator in a court of law. In this context, forensic science plays a pivotal role, with forensic toxicology being a crucial subsection that specializes in analyzing both organic and inorganic poison substances to determine the cause of death.3–6
In recent times, there has been a surge in reports of pesticides being mixed into soft drinks or beverages by perpetrators.7, 8 Pesticides are readily accessible substances that can be easily misused for nefarious purposes. A plethora of pesticides are available in the market, with organophosphate-based pesticides being among the most common.9–12 Therefore, for the this study, several organophosphate-based pesticides were selected:
These pesticides were chosen for their known effects on the nervous system and their potential lethality when ingested. 17
Objectives
The main aim of this is to determine the rate and extent of pesticide degradation in various types of acidic beverages.
Materials and Methods
The objective of this research is to assess the stability of pesticides, specifically cypermethrin, chlorpyrifos, and phenthoate, in carbonated beverages like 7UP and Sprite. These pesticides, known as organophosphate-based insecticides, are commonly utilized in agricultural practices. The choice of carbonated beverages such as 7UP and Sprite is motivated by their colorless nature, which facilitates the detection of any changes resulting from the addition of pesticides. Ethical clearance is obtained by IEC Order No. KARE/CEC/MOM/2023-24/02Dated:05-03-2024.
Before commencing the testing, the concentrated pesticides were appropriately diluted to ensure that the concentrations were within a manageable range for analysis. The dilution was carried out using a ratio of 10:90 with water, effectively reducing the concentration of the pesticides. Subsequently, the diluted pesticides were mixed with the beverage according to a specific ratio. The mixture consisted of 85% beverage, 10% citric acid, and 5% diluted pesticide. This formulation ensures a consistent composition for testing while also incorporating citric acid, which may influence the stability of the pesticides in the beverage. Following this preparation, the beverage was ready for testing, and daily observations were conducted over a period of 10 continuous days using ultraviolet (UV) spectroscopy. This methodology allows for a comprehensive examination of how these pesticides interact with carbonated beverages over time. By conducting daily assessments using UV spectroscopy, researchers can identify any degradation or changes in the pesticides’ concentrations, shedding light on their stability in this specific matrix.
Results and Discussion
Initially before analyzing the sample, a baseline reading was taken. This established the absorbance of the solvent or blank solution. The absorbance of the sample was then measured at specific wavelengths between 200 and 250. Any increase in absorbance compared to the baseline indicates absorption by the sample components.
Based on the absorbance of UV spectroscopy analysis conducted between wavelengths of 200 and 250 nm, the stability of the pesticides in the beverages was assessed as follows:
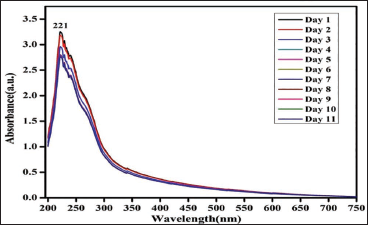
Cypermethrin (Figure 1):
Absorbance readings for cypermethrin did not show significant changes over the 10-day period compared to the baseline. This suggests that cypermethrin did not degrade in the beverages containing citric acid. The absence of notable changes indicates the stability of cypermethrin under the conditions tested.
18
Stability of Cypermethrin.
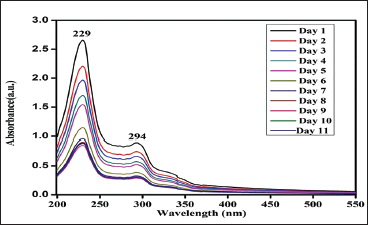
Phenthoate (Figure 2):
Phenthoate exhibited noticeable degradation over the first seven days of analysis, as evidenced by a decrease in absorbance compared to the baseline. However, after seven days, the degradation of phenthoate diminished, and the absorbance readings stabilized. This indicates that while phenthoate initially degraded in the beverages, its degradation slowed down and reached a stabilized state by the end of the 10-day period.
19
Stability of Phenthoate.
Chlorpyrifos (Figure 3): Chlorpyrifos showed a considerable amount of degradation on the initial day, as indicated by a decrease in absorbance compared to the baseline. However, the level of chlorpyrifos degradation gradually decreased over time, with absorbance readings dropping further. By the end of seven days, chlorpyrifos degradation diminished, suggesting that its degradation process slowed down and stabilized.20, 21
Stability of Chlorpyrifos.
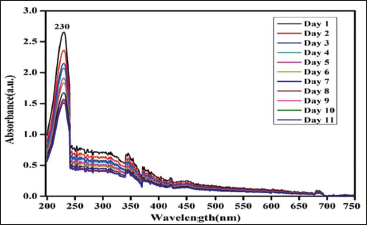
Overall, these findings suggest varying degrees of stability and degradation for the pesticides analyzed in the beverages containing citric acid. Cypermethrin showed stability, while phenthoate and chlorpyrifos exhibited degradation, albeit with different degradation profiles. These results provide valuable insights into the behavior of these pesticides in beverage matrices, which is essential for assessing their potential impact. 22
Discussion
Based on the stability assessment of the pesticides in the beverages over the 10-day period, it is evident that traces of the pesticides can be detected even after this duration. This finding suggests that if a deceased individual had consumed these beverages containing the pesticides within the past 10 days, it may be possible to detect traces of the pesticides in their body. 23
In forensic investigations involving suspected poisoning cases, analyzing body tissues or fluids for the presence of pesticides can provide crucial evidence to determine the cause of death. If the deceased had ingested beverages containing cypermethrin, phenthoate, or chlorpyrifos, the presence of these pesticides in post-mortem samples could indicate poisoning as the cause of death.24, 25
Analyzing extracts from the body tissues or fluids using appropriate analytical techniques, such as chromatography or mass spectrometry, can help detect and quantify the pesticides present. The results of these analyses, combined with other investigative findings, can contribute to determining the circumstances surrounding the individual’s death.26, 27
Conclusion
The accessibility of pesticides and insecticides in the market raises concerns about their potential misuse in criminal activities, including poisoning cases. In forensic investigations, the timely analysis of liquids found at crime scenes or recovered from victims’ bodies is critical for identifying potential poisons and determining the cause of death.
This study plays a crucial role in aiding forensic investigations by providing insights into the time frame during which specific chemical compounds, such as pesticides, can be reliably traced in beverages. By understanding the degradation patterns of these compounds, particularly in beverages containing citric acid, forensic analysts can better prioritize and expedite the analysis of liquid evidence. Identifying poisons and toxic substances is a primary task of toxicology divisions within forensic laboratories. The study’s findings help to establish a time frame within which these chemical compounds can be detected before degradation occurs. This understanding is essential for ensuring that forensic analyses are conducted within a critical window of time to maximize the chances of identifying the poison or toxic substance responsible for an individual’s illness or death.
Furthermore, the study underscores the importance of considering the law of progressive change in forensic investigations. As chemical compounds degrade over time or interact with other substances, the ability to detect and identify traces of poisons may become increasingly challenging. By recognizing the time-sensitive nature of toxicological analyses, forensic scientists can adjust their protocols and prioritize cases accordingly to optimize the chances of successful detection and identification of poisons.
Authors’ Contribution
Sanjana G, Krishnapriya R, Lavanya N and Vijjurothi Madhav Manikanta helped in acquisition of data or analysis and interpretation of data.
Rudrank Shukla was involved in drafting the article or revising it critically for important intellectual content.
M C Janaki helped in conception and design of the study and final approval of the version to be published.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study did not involve human participants or animal subjects. Therefore, ethical approval was not required in accordance with institutional and national guidelines.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable as this study did not involve human participants, and no personal data were collected.
