Abstract
Background
Maternal mental health during the peripartum period is critically important to the wellbeing of mothers and their infants. Numerous studies and clinical trials have focused on various aspects of interventions and treatments for perinatal mental health from the perspective of researchers and medical health professionals. However, less is known about women’s experiences of participating in perinatal mental health research, and the ethical issues that arise.
Aim
To systematically review the literature on the ethical issues that emerge from pregnant and/or postpartum women’s experiences of taking part in perinatal mental health-related research.
Methods
Systematic review of nine bibliographic databases, from inception to July 2021. Qualitative, quantitative and mixed method studies were included if they reported on ethical issues experienced by perinatal women. Research ethical issues encompassed any issue relating to women’s experiences of being offered study information, recruitment, consent, retention and respect for autonomy.
Titles, abstracts and full text screening, appraisal of the methodological quality of included studies, and data extraction, were conducted independently by two reviewers.
Ethical considerations
Ethical approval was not required for this systematic review.
Findings
A total of 9830 unique citations was retrieved. Six studies met the inclusion criteria. Studies were clinically and methodologically heterogenous, and only one was purposively designed to explore women’s experiences. The key finding was the establishment of trust between the researcher and participant in all stages of the research process. Findings are presented according to recruitment and consent processes, participation and retention, and study follow-up and completion.
Conclusion
The establishment of trust between the researcher and perinatal women leads to a dynamic with research ethical implications relevant to all stages of perinatal mental health-related research. Further research on the research ethical issues experienced by perinatal women is required because of the limited literature.
Introduction
Maternal mental health during the peripartum period is critically important to the wellbeing of mothers and their infants.1,2 However, many pregnant and postpartum women experience mental health issues and the prevalence, 3 impact 4 and economic costs 5 are well documented in the literature. For these reasons, empirical investigation into perinatal mental health is essential to support positive maternal mental health and neonatal outcomes.
Numerous studies and clinical trials have focused on various aspects of interventions and treatments for perinatal mental health issues from the perspective of researchers and medical health professionals,6–9 and others have focused on issues related to feasibility, recruitment, consent processes and the retention of research participants in clinical trials.10–12 These studies, however, generally focused on ways to enhance engagement from participants from the perspective of the research processes and/or the experiences and observations of researchers. There are relatively few studies that examined the experiences and perspectives of research participants themselves, 13 and fewer still on the experiences of pregnant women who are recruited and participate in perinatal mental health-related studies.
This is not entirely surprising, as studies show that there is a gender gap in clinical research, with women generally being enrolled and participating in research studies and clinical trials less often than men.14,15 Moreover, recruiting pregnant women poses an even greater challenge, not only because of reluctance from the women themselves, 16 but also due to specific external regulatory barriers to the recruitment of pregnant women into clinical research studies.17,18 Regulatory barriers on pregnant women’s participation in research have ethical implications for women’s equitable access to safe and effective treatments for both physical and mental health diagnoses. 19
Ethical issues pertaining to perinatal mental health research have also received commentary in the literature, specifically with regards to the exclusion of pregnant women from research, the arguably paternalistic classification of pregnant women as a vulnerable group by governmental and other organisations conducting research, issues related to the recruitment process and study design,19–22 and ethical concerns around conducting randomised control trials for medications used to treat psychosis, 23 major depression, bipolar-type affective disorder, schizophrenia or substance use disorders 24 during pregnancy. However, much less is known about the ethical issues that perinatal women may encounter during the experience of participating in clinical research in general, and especially in perinatal mental health research.
Objectives
This systematic review explored how pregnant and/or postpartum women with mental health issues experienced being recruited into and participating in mental health-related research, and the ethical issues that emerged from their experiences.
Methods
A protocol was registered in the International Prospective Register of Systematic Reviews, Prospero a priori (https://www.crd.york.ac.uk/prospero/) (CRD42021286128).
Search strategy
Nine electronic bibliographic databases were searched in July 2021. The databases CINAHL Complete (EBSCO), MEDLINE (EBSCO), PsycINFO (EBSCO), EMBASE (Elsevier), Global Index Medicus (WHO Library), Web of Science (Clarivate), Scielo (Clarivate) and Applied Social Sciences Index, and Abstracts (ASSIA) (Proquest) were searched from inception, and all languages were included and screened up to the full text screening stage. The full search conducted in EMBASE is detailed in supplement Appendix 1. Additional Grey Literature searches, including databases which require a simplified search structure, repositories, and important websites were also conducted in July 2021.
Inclusion and exclusion criteria
Research studies were included if they were related to perinatal women (pregnant and/or postpartum) aged ≥18 years who had been diagnosed with or self-identified as having a mental health issue and their experiences of taking part in mental health-related research from a research ethics perspective. Perinatal mental health issues encompassed any type of adverse mental health issue experienced by pregnant and/or postpartum women, including depression, anxiety, psychosis and phobia. Qualitative, quantitative and mixed method empirical research studies were included.
Articles were excluded if they were literature reviews, comments, letters, conference abstracts, posters, book reviews or dissertations, or did not report on ethical issues related to women’s experiences of taking part perinatal mental health research.
Research ethical issues were interpreted broadly and encompassed any issue relating to women’s experiences of being offered study information, the recruitment process, consenting to take part and how researchers respected women’s autonomy throughout the study.
Keywords were determined in accordance with the three main concepts within the review questions: (1) mental health in the peripartum period, (2) taking part or engagement in research and (3) women’s experiences. Additional keywords were included after the initial search strategy was drafted and tested. Search terms were combined with the Boolean operands “OR” and “AND” as appropriate, and adapted across databases.
Proximity searching was used due to the complexity of concepts one and two. For example, the concept “mental health” was searched using a proximity of two terms within the peri- and postpartum periods. The second concept focused on the uptake and recruitment of participants using a proximity of three terms in a research methodology. The third concept explored experiences.
The final search strategy underwent an independent Peer Review of Electronic Search Strategies (PRESS) review by a second medical Subject Librarian.
Screening and quality appraisal
The authors used the Covidence platform to screen citations using the inclusion and exclusion criteria. Two reviewers independently screened citations by title and abstract (DD and RE), and by full text (SH and KH).
Quality assessment
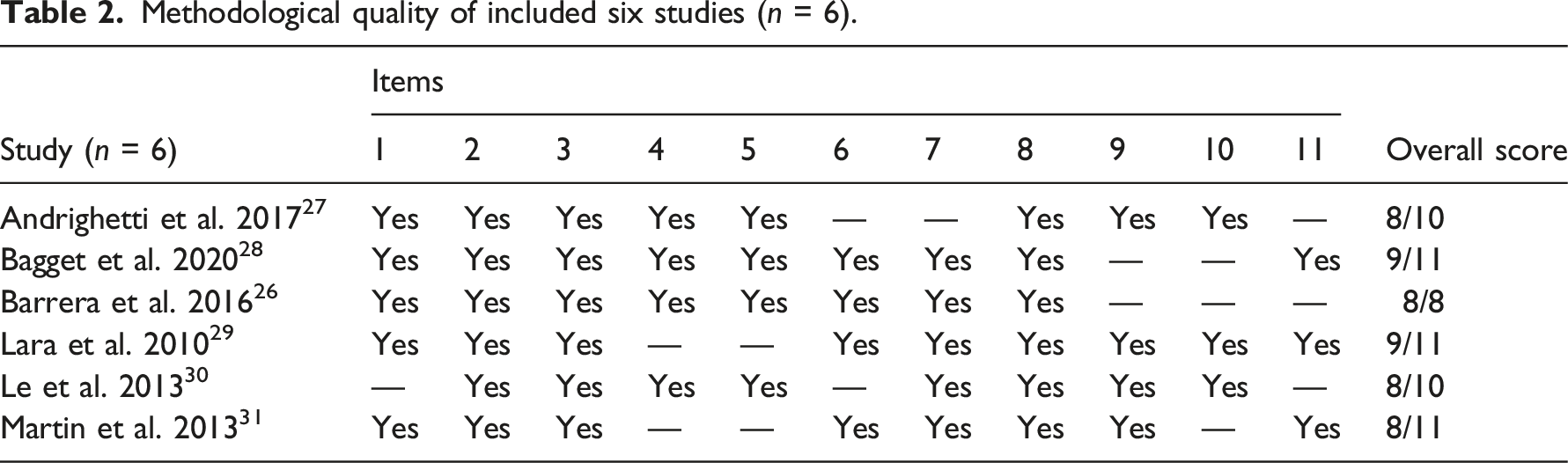
Methodological appraisal of included studies was independently conducted by two reviewers (AA and SU) using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Qualitative Studies (10 items), Analytical Cross-Sectional Studies (8 items) and Cohort Studies (11 items). These tools determine the extent to which a study has addressed the possibility of bias in its design, conduct and analysis, and/or assists in assessing the trustworthiness, relevance and results of published papers. The checklists are available at https://jbi.global/critical-appraisal-tools and are summarised in Supplemental Table 1. They consist of 8–11 items with four possible responses: yes (the criteria are clearly identifiable and assigned 1 point), no (the criteria failed to be applied appropriately), unclear (the criteria are not clearly identified in the report) and not applicable. Disagreements over assessment scores were resolved through discussion between the reviewers. A decision was made a priori to include all studies for data extraction regardless of the results of their methodological quality because even poorly conducted research can provide relevant accounts of the phenomenon being studied. 25
Data extraction
Data extraction was completed independently by two reviewers (IB and MdW) using a purposively designed data extraction form. The data extracted included: country, sample size, sample age range, characteristics of sample (i.e. pregnancy or postpartum and description of mental health issues) and study design (IB), and the ethical issues identified (MdW). For this purpose, a framework was developed according to the stages in the research process to glean a more acute insight into women’s experiences of participating in research and to broadly establish which ethical issues present themselves at each stage.
For this review, meta-synthesis was therefore planned a priori. However, due to the clinical and methodological heterogeneity of the included studies, the findings are presented narratively.
Findings
A total of 16,404 articles were retrieved. Results were exported into Endnote version 20 and de-duplicated using the Endnote 20 software. An additional manual deduplication was also executed. This resulted in 9824 unique citations which were screened using Covidence systematic review management software. An additional 146 unique citations were retrieved from other sources and also imported into Covidence. Six studies met the inclusion criteria and were included for data extraction and data analysis.
The screening and selection process is presented in Figure 1. Screening and selection process.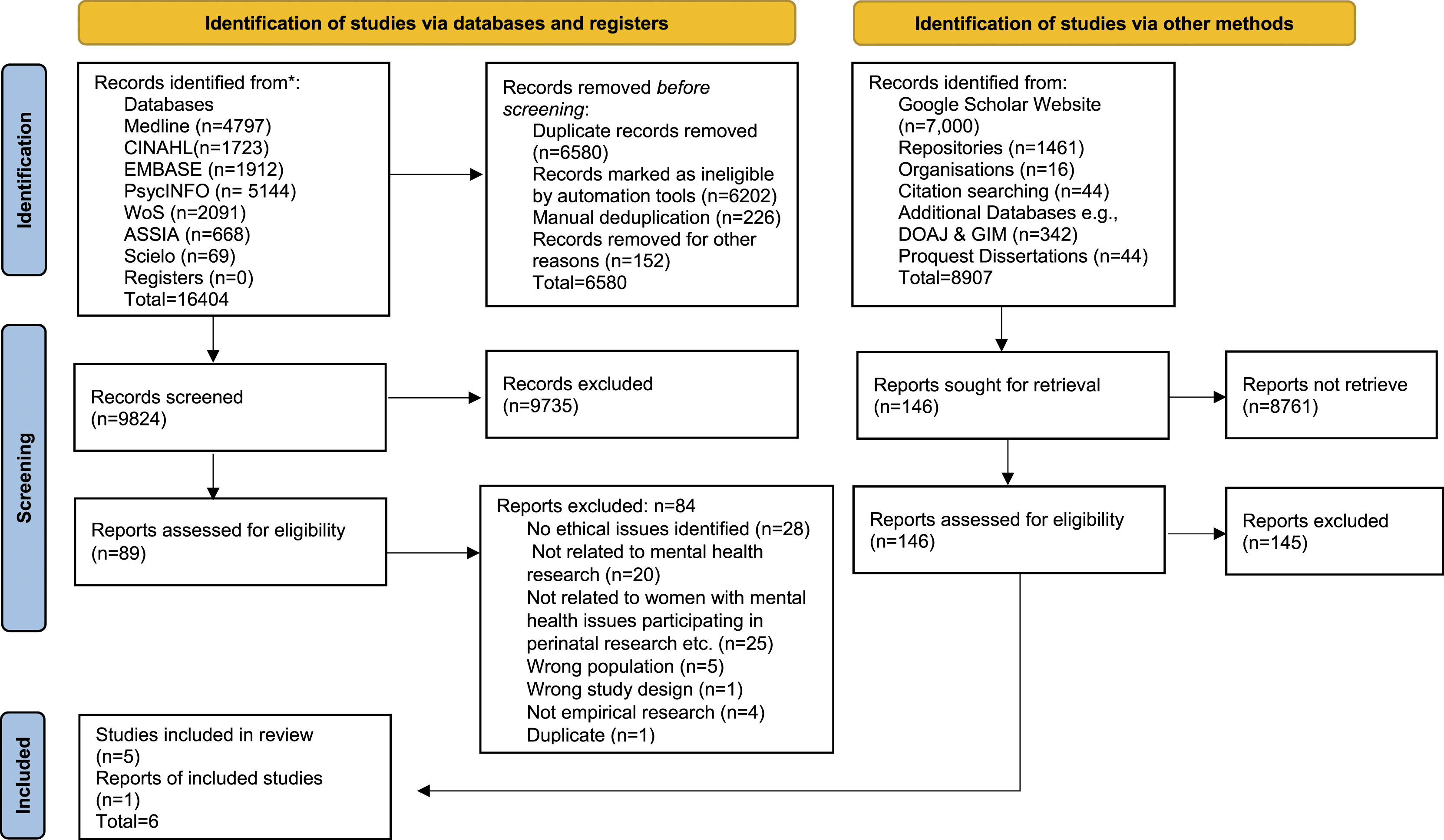
From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021; 372: n71. doi: 10.1136/bmj.n71. For more information, visit: https://www.prisma-statement.org/
Overview of the included studies
Characteristics of included studies.
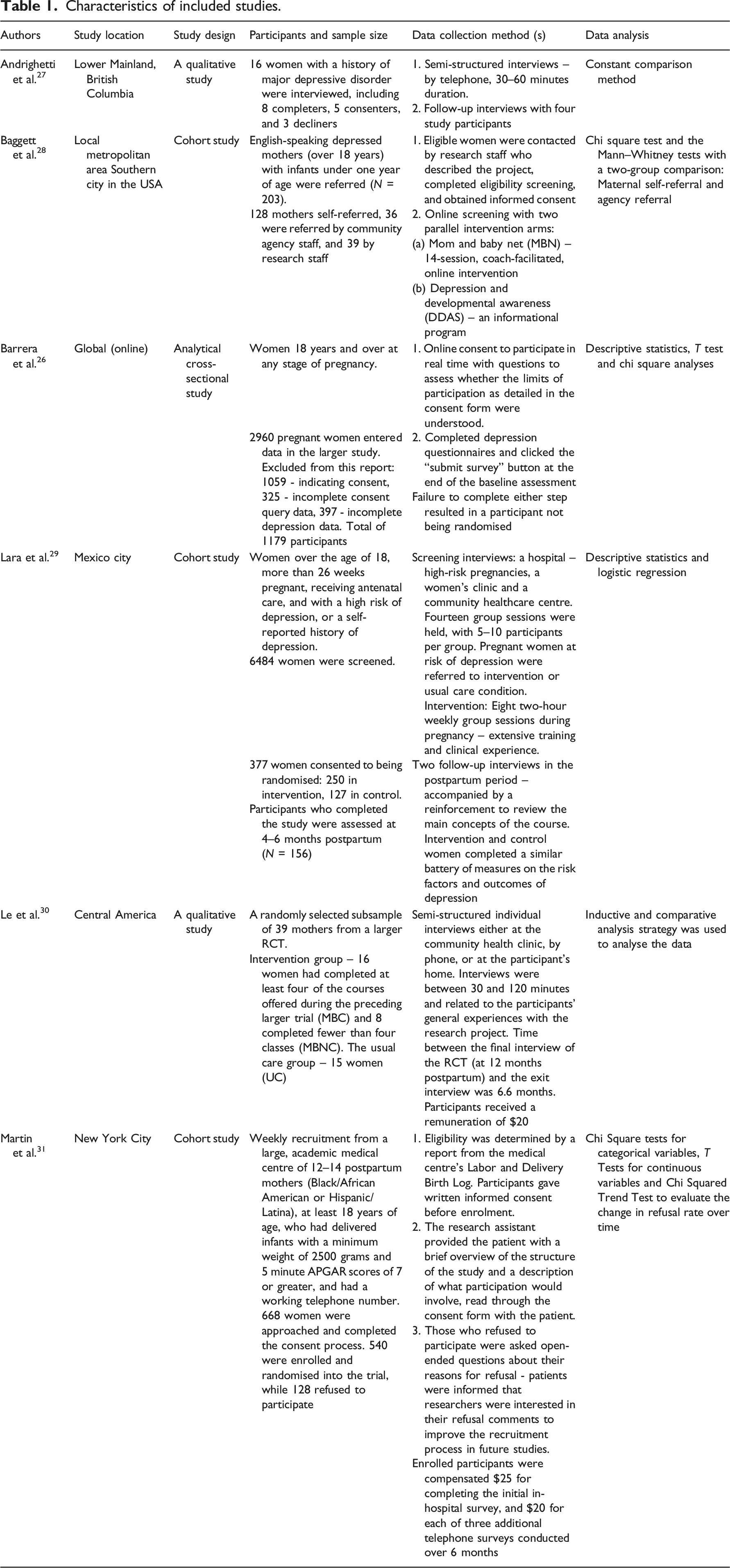
All six studies differed considerably in their aim and focus. Andrighetti et al. 27 explored women’s motivations and experiences during both the decision-making process and participation in a postpartum depression (PPD) research study, and was the only study to focus specifically on women’s experiences of taking part in research.
Baggett et al. 28 aimed to determine the differential success of three referral approaches (community agency staff referral, research staff referral and maternal self-referral) within a clinical trial of a mobile internet parenting intervention (application). Barrera et al. 23 focused on participants’ understanding of the informed consent form for an online PPD trial. Lara et al. 24 focused on retention in a longitudinal RCT, while Le et al. 25 conducted a follow-up study in order to understand the results of a completed longitudinal RCT which found that rates and levels of PPD in both the intervention (14%) and control (25%) groups were lower than expected. The final study by Martin et al. 26 implemented a recruitment strategy that used patient feedback to ascertain and understand the barriers to the recruitment and subsequent enrolment of Black and Latina women into a clinical trial.
Assessment of methodological quality
Methodological quality of included six studies (n = 6).
Summary of included studies and findings
Because each study had a unique focus, the key findings are presented narratively.
Andrighetti et al. 27 identified two conditions that needed to be met in order for a woman to consider participating in a PPD study: (1) she needed to have accepted her mental illness and its associated diagnostic labels, and (2) she needed to have an established sense of trust in the research team and institution conducting the study. Women agreed to participate if they viewed an institution as being reputable and involved with research that is both legitimate and conducted for the good of society, as opposed to being driven by profit. Women were motivated to participate if they perceived the research topic as being personally relevant to them. The second motivating factor was the perceived benefit of participation. Three categories of perceived benefit were identified by the participants: (1) they saw the study as gateway to gaining support in both the therapeutic sense and in the sense of getting better access to resources; (2) they saw their participation as an opportunity to learn; and (3) they understood that by having contributed to a study that carries value, they would have a sense of accomplishment which would improve their sense of self-worth. This then also ties in with the third motivational factor identified by the researchers, namely, wanting to give back to others with mental illness. In other words, there was an altruistic desire to contribute to society through their participation in a PPD study. The final motivational factor identified by the researchers was the accessibility of the study. Participants tend to favour interventions that are convenient and minimise their time commitment and travel-related costs.
Baggett et al.’s 28 study described three referral and recruitment processes used in their RCT trial of a mobile internet parenting intervention: agency staff referral, research staff referral and maternal self-referral. Women who self-referred were just as likely to consent to participating in the research trial as women who were referred by a gatekeeper. Moreover, women self-referred at more than 3.5 times the rate than women who were referred by a community agency or a member of the research team. The authors cited two possible reasons for this: busy agency staff may have reserved their efforts for women they saw as “the most vulnerable,” 28 (p. 10) and self-referral created a smooth path to entry for most women.
They also found potential participants who had greater resources were more likely to bypass gatekeeper systems and to access mental health services directly.
Barrera et al. 26 examined how well participants understood the online participation consent form of an RCT for the prevention of PPD against the backdrop of three ethically relevant questions: (1.) What was the overall understanding of the main aspects of participation (i.e. the risks and benefits, participation being voluntary)? (2.) Would participants misunderstand the nature of the study itself, i.e., that it is not aimed at providing therapy or prenatal care? (3.) Are there differences that can be traced back to demography and depression characteristics in response accuracy and initial commitment to participation? In their findings, the authors highlighted the fact that some women had misunderstood what the actual risks and benefits of the trial were. They suggested that the depressive state of the women who filled in the consent form may have contributed to their misunderstanding of the risks and benefits of the trial: “Participants meeting criteria for current MDE were more likely to incorrectly identify the purpose of the study as replacing current psychological treatment.... ” 26 (p.294).
Lara et al. 29 in their examination of predictors of retention, found that mothers who were at high risk of depression were more committed to participating in the research study. They concluded that interventions should target women who presented with risk factors for depression such as being single, being in a poor-quality partner relationship, having high anxiety levels and experiencing stressful life events. However, they also found that homemakers, defined by the authors as a vulnerable group, and mothers with low educational attainment, remained difficult to retain. They recommended that different retention strategies be implemented in accordance with the circumstances of the participants.
Le et al. 30 found that participants in both the intervention and the control groups drew benefits from participating in the research. The intervention group learned specific mood-management skills, while the control group experienced the study as a kind of “low-dose” intervention. They concluded that it is important to conduct qualitative studies in order to fully understand the quantitative outcomes of RCTs.
Finally, Martin et al. 31 found that employing a recruitment strategy that incorporated and responded to feedback resulted in improved recruitment of Black and Latina women in their clinical trial. They created a taxonomy of reasons for refusal throughout the 52-week recruitment period for their study. Eight primary reasons for refusal came to light: lack of interest, time commitment, feeling tired or in too much pain, relocation, feeling overwhelmed, issues around confidentiality, having been through childbirth before and not self-identifying as depressed. The most common reasons for refusing to participate were having a lack of interest or not wanting to accept the time commitment. The recruitment message was adjusted during the study period to convey the fact that participant protection was a priority, and to reflect a commitment to maintaining open and honest communication with recruits regarding the risks of participating in the research.
It should be noted that of the six studies, only the qualitative study by Andrighetti et al. 27 explicitly explored women’s experiences of participating in research, and the only study that explicitly set out to explore ethical concerns during the course of their research was the study conducted by Barrera et al. 26 The description of pregnant and peripartum women’s experiences of participating in the six studies included in this review, and the ethical issues that emerge from this, are therefore analysed meta-ethnographically as below.
Women’s experiences and their ethical implications
Andrighetti et al. 27 study set out to explore women’s motivations and experiences of deciding to and taking part in a PPD research study. The other five studies can be categorised in accordance with women’s experiences through the different stages of participating in a trial: the recruitment stage, including obtaining informed consent; the trial stage; and the post-trial stage, in the case of exit interviews and following up on participants after the conclusion of a trial. At each stage, the important ethical considerations that arose from the findings, and their implications for researchers, are identified.
Recruitment and giving informed consent
Three of the studies reviewed yielded some important insights into women’s experiences of the recruitment process and consenting to take part.
Andrighetti et al. 27 found that certain fundamental factors needed to be present for women to consider participating in PPD research, namely, personal acceptance of their mental health condition, trust in the research team or institution, perceived personal relevance, anticipated benefits like access to resources and improved self-worth, altruism and accessible study procedures. The authors inferred from the data that participating in a PPD study may help women better understand their mental illness experience and doing so is seen as receiving support. Therefore, having a personal connection with or experience of PPD seemed to increase a participant’s resonance with the study since they were more likely to feel that it was meaningful or valuable.
Andrighetti et al. 27 further suggested that the decision to participate was not predicated on a utilitarian cost-benefit decision either: even though women were promised a nominal fee for their participation at different stages of the trial, honoraria did not play much of a role in their decision to participate. They perceived this as a neutral factor – neither a motivator nor a barrier to participation. Moreover, the informed consent process and consent forms explicitly stated that there were no other direct benefits to participants. The stronger motivation to participate, therefore, was ‘making meaning, purpose and “good”’ out of the experience”. 27 (p. 556) It follows from this that the ethical and moral convictions of the women themselves were strong motivators in their participation, in the sense that they viewed their participation as being an act of altruism. This is also a finding that has been corroborated by numerous other studies into the motivators for participation in research.13,32,33
While the Andrighetti et al.’s 27 findings bring some interesting insights to light as regards women’s reasons for deciding to consent to participation, Martin et al. 31 drew attention to their reasons for refusal. Among the eight primary reasons for refusal that were identified, the most obvious ethical concern raised directly was participants’ concern about confidentiality.
Since refusal rates declined as recruitment messages were adjusted to reflect the concerns voiced by patients targeted for recruitment, the researchers concluded that it is critical to utilise recruitment approaches that respond to the identification of potential areas of concern for recruits.
From an ethics perspective, this approach allows for other ethical and moral concerns, such as concerns around data protection and respect for autonomy to become evident as well and to be addressed as soon as they arise.
Martin et al. 31 also noted that the ‘lack of interest’ refusal category they identified indicated that the recruitment message they had used did not reflect an understanding of the values of the prospective participants. From an ethical perspective, grasping the values of the participant population is of critical importance for guiding practices that are adopted throughout a clinical trial, beyond the recruitment stage. Doing so from the outset, as was done in this study, allows for better identification of ethical issues, in particular if they stem from an insensitivity to the values of the women involved. This would ostensibly allow for adjustments to be made along the entire trial period, as necessary.
Baggett et al. 28 found that gatekeeper referral systems that relied on agency and research staff referrals to their trial were not as successful as mother self-referrals. The two possible reasons for this were that agency staff may have specifically targeted the women they viewed as most vulnerable, and self-referral was the result of a convenient path to entry. They also found that potential participants with better resources were more inclined to bypass gatekeeper systems to access mental health services directly. While the authors make no mention of ethical issues, this finding is worth exploring in the context of certain ethical issues, if one allows for the hypothesis that mothers may be more likely to self-refer due to privacy and confidentiality concerns and a fear of stigmatisation. Another hypothesis is that the women who self-referred may not have understood the risks and benefits of participation as well as the ones who had these explained to them by a gatekeeper.
Barrera et al. 26 focused on the informed consent part of the online recruitment of participants into a PPD trial, without a gatekeeper guiding the process.
The authors of this study set out with the premise that operationalising ethical principles in the design and deployment of automated and online consent forms was a struggle since it is not readily apparent how to determine whether a study participant has properly understood the risks and benefits of participating.
Amongst the findings highlighted by the authors, was the fact that some women had misunderstood the actual risks and benefits of the trial, and that the depressive state of the women could have contributed to their misunderstanding of the risks and benefits of the trial. Their findings suggest that the informed consent form and the way in which it is presented and worded is closely linked to whether a participant understands how they will benefit from the trial, and consequently how motivated they would be to participate.
Considered alongside the findings from Andrighetti et al. 27 and Martin et al. 31 studies, both of which found that a lack of perceived benefit acted as a barrier to recruitment, it seems clear that the anticipated benefits of participation weigh heavily on the motivation to participate in a clinical trial. It should be noted that these anticipated benefits included an improved sense of self-worth and increased knowledge and access to support and resources to help manage their mental health. Monetary and other material incentives were not strongly considered as a benefit in either of these studies’ populations.
In light of this, and assuming that a strong cost-benefit calculus is indeed a key factor in deciding whether to consent or decline, it is imperative that the informed consent process not only leaves every participant with an accurate understanding of all the benefits and risks of participating, but also that it takes into consideration that this calculus may be characteristically different for women with mental health issues due to their illness, or for women from minority groups. A failure on the part of researchers to understand the values, perspectives and cost-for-benefit considerations of research participants may lead to therapeutic misconception. 34
After consent: participation and retention
During their investigation of the motivators for continued participation in their research, Andrighetti et al. 27 found that the same factors that motivated participants to enrol in the study were also the ones that motivated them to stay the course and not to drop out. The primary motivators boiled down to the establishment of trust and a rapport with the research team and seeing the anticipated benefits of participation realised through the course of the study. Moreover, the authors argued that their study slots into a growing body of research which suggests that participation in research can contribute to the improvement of the mental health of participants. They argued that it is possible that participating in PPD research studies is less stigmatising than accessing more traditional therapeutic interventions, even if the research is not intended to be therapeutic in and of itself. Research increasingly points to the benefits of sharing difficult experiences with a researcher and finding research-oriented interviews healing. They posit that this suggests that these kinds of studies may be prone to the Hawthorne effect 35 whereby the act of observation changes the outcome: ‘Specifically, being prompted to reflect on current psychiatric state, interacting with a researcher in the context of a trusting relationship, and/or feeling that one has the opportunity to meaningfully contribute altruistically all have the potential to protect mental health and thereby impact the expression of the phenomenon of interest’.27 (p.557)
The study conducted by Lara et al. 29 which focused on retention during an RCT to prevent PPD, had what the authors described as a low retention rate overall (41.7%). The researchers could not pin-point the reason for this as they lacked follow-up information, although some women cited health issues, miscarriages, time constraints and premature births as reasons for dropping out. The retention rate from randomisation to attendance of one or more sessions was also low (42.4%), meaning that a large number of participants had dropped out after signing the informed consent form but before commencing with the sessions. This was attributed to a cultural phenomenon. Mexican participants were prone to expressing their refusal to participate indirectly by not attending because they had difficulty doing so at the informed consent stage. From an ethical perspective, it could be surmised that removing the gatekeeper from the act of giving informed consent (but not from receiving information) would make it easier for women to refuse consent of their own volition, without feeling directly accountable to the person acting as gatekeeper. A further consideration is that the presence of a gatekeeper may have a coercive effect on a potential participant, whether intended or unintended. Hem et al. 36 note that researchers and gatekeepers are prone to justifying the recruitment of participants as being in the best interests of the participant and good for their well-being. In other words, beneficence as a justification for recruitment could result in a net effect of coercion, and this may account for some participants having difficulty refusing to consent to participation.
Future research could explore how an online approach like the one put forth by Barrera et al. 26 could be employed to overcome these issues.
Where high retention rates were reported during this study, it was amongst what the study’s authors describe as being ‘high-risk’, for example, single women, those who have difficult relationships with their partner, women with a history of mental health issues, and women with low educational attainment. The authors noted that this is consistent with the overall understanding in the field of mental health research that participants need to feel distress before they would consider participating in a prevention activity. Group session attendance was also high, and the researchers put it down to intensive efforts aimed at bolstering retention, especially in the form of establishing a good rapport with participants, as well as the fact that participants described marked improvement in their well-being. This then points to a parallel with the studies conducted by Andrighetti et al. 27 and Barrera et al. 26 in that the research team identified the establishment of trust between the researchers and the participants as a key factor in continued participation, and that research studies into perinatal mental health issues tend to have a significant therapeutic impact.
Study completion and follow-up
Of the six studies included in our review, only one focused primarily on the conclusion of a trial and its aftermath. When Le et al. 30 were confronted with unexpected results from their RCT, they sought to better understand the trial participants’ overall experiences with the research project through qualitative exit interviews.
During these interviews, the participants described their relationships with the research team as a key aspect to their participation. Some referred to this relationship as friendship, while others went even further, ‘frequently describing their connections with team members as if they were kin’. 30 (p.840) This relationship between the researchers and the participants implies the presence of a sense of trust, which accords with the findings of the other studies included in the review. However, the researcher-participant dynamic described in this study goes a step further and is described in very intimate terms – as friendship or kinship. This raises the all-important ethical question as to the implications of this dynamic for both the researcher and the participant: that is, how close is too close and what are the parameters that guide the researcher-participant relationship? A possible explanation for the unexpected outcome of the study was that the researcher-participant relationship was blurred and potentially influenced the results.
Another finding that emerged from these exit interviews that accord with the findings of most of the studies in this review, was that the women who had participated in this RCT experienced significant therapeutic effects from participation. They reported developing a better awareness of their moods, which helped to normalise their experience with PPD and to feel less isolated. They also acquired mood-management skills and cognitive coping strategies. This, in turn, led to increased maternal efficacy and improved interpersonal relationships.
The authors of this study concluded that their disappointing results, that is, seeing little difference between the outcomes of the intervention and control groups in the RCT, were attributable to the fact that they had purposely gone to great lengths to build intimate, supportive relationships with the participants in both groups in order to prevent attrition. However, in addition to bolstering retention, this strategy may have become an intervention for the women in the control group. This, together with the findings of Andrighetti et al. Barrera et al. and Lara et al.27,26,29 then suggests that women overwhelmingly experience their participation in perinatal mental health research as beneficial to the management and reduction of their symptoms.
Discussion
Based on the limited findings from these six studies, it appears that some women indicated that they would likely take part in a research study even if they had not been made aware of the risks and benefits by a gatekeeper, or through some form of personal contact.26–28 However, retaining women who self-refer may prove to be problematic once the risks and benefits become clear to them. 28 One assumption for this could be therapeutic misconception. Another more problematic assumption could be that the state of depression could impair a woman’s ability to fully understand what the risks and benefits of consenting are at the time of giving consent, depending on the severity of her depressive state. In fact, Weissinger and Ulrich 37 point out that capacity to consent to research participation is an underrepresented issue in the literature around mental health research and should be given serious consideration when recruiting participants into a mental health study.
This idea that the state of depression undermines decisional capacity and therefore the capacity to consent to participation in research is also, controversially, often clearly connected with the idea of vulnerability in research ethics. 38
A key finding in the studies we reviewed, was that the women who tend to consent to participation in a research project such as these are more likely to feel like they need the most help with their mental health.27,26,29
This raises a pertinent question as to the perception of researchers as regards the decision-making capacity of peripartum women with symptoms of depression.
This relationship between vulnerability, actual decisional capacity and researcher perceptions of decisional capacity in peripartum women with mental health issues warrants unpacking:
Perinatal mental health researchers generally find themselves in a difficult situation as far as recruiting and retaining participants in research studies and clinical trials is concerned since pregnant women are often explicitly classed as a vulnerable group from a legal and policy perspective. In the United States, for example, under the Federal Policy for the Protection of Human Subjects, 45 C.F.R. Part 46, also known as the ‘Common Rule’, pregnant women are categorised as vulnerable along with children, prisoners, mentally disabled people and people with physical disabilities. 39 The exclusion of pregnant women from research trials is well documented. In fact, in 2013, it was found that 89% of industry-sponsored trials involving either a drug or a medical device in the United States excluded pregnant women as participants. 40 This is due in part to the impact of policies and regulations such as the Common Rule, but also to the perceived risks to participation on the part of pregnant women themselves, who worry about harm to them and their unborn offspring. 15
Pregnant women with a mental health condition pose an additional challenge for researchers, particularly with regards to regulatory and policy concerns around the issue of vulnerability. The United Kingdom’s Research Governance Framework for Health and Social Care, for example, explicitly labels mental illness as a characteristic source of vulnerability. 33 There is an argument to be made that classifications like these tend to perpetuate a subtle and pervasive stigmatisation of women with mental health issues and could therefore result in stereotyping and unfounded assumptions that can shape the attitudes of researchers towards participants. 38 One such an assumption concerns decisional capacity. Bracken-Roche et al. 38 argue that when vulnerability is considered in the context of mental health research, it is often attributed to a lack of decisional capacity. In other words, there is little consideration of the relational and contextual factors that could affect a research participant’s understanding or appreciation of information such as that which they would encounter in a consent form.38,37 In fact, there is evidence that while severe mental health problems, such as the kind that would require hospitalisation, do impact decisional capacity in individuals,37,41 those with more moderate forms of depression, anxiety and so forth perform quite well on their decision-making capacities related to research participation. 41
Two areas for future research reveal themselves in this discussion. The first pertains to researcher bias, which raises a number of ethical as well as practical concerns, not the least of which is that when researchers view vulnerability as an intrinsic characteristic of a research participant, it affects the researcher/participant relationship. This could lead to an asymmetry in the power dynamic as well as to suboptimal study design, or even inappropriate or exploitative enrolment of research participants. 38
The second pertains to the research participants themselves. Future research should investigate how well pregnant women with mental health issues understand consent forms - particularly automated, online forms. A central question would be how best to structure the process of obtaining informed consent in this type of research, while respecting the autonomy and privacy of the women being recruited, with the aim of establishing a standard of sorts for perinatal mental health-focused research: a standard that includes an assessment of capacity to give informed consent, and takes into account the relational factors (educational level, health literacy, support systems, sociocultural context, etc.) that may impact a woman’s ability to autonomously give informed consent.
Another noteworthy point of discussion that emerged from our review of the literature, was that the included studies overwhelmingly highlighted the establishment and maintenance of trust as a key factor in the successful recruitment and retention of women in perinatal mental health research.
Guillemin et al. 42 define trust as an interpersonal relationship characterised by the extent to which one person relies on another person or entity to act in certain ways. The person who bestows the trust is vulnerable in the sense that they are reliant on the integrity and benevolence of the person or institution being trusted.
In the clinical research environment, the establishment and maintenance of trust is rooted partly in the rapport that is established between the researcher and participant, particularly in qualitative studies. This raises several ethical considerations:
First, we need to consider the role of the researcher in this dynamic. How should research institutions or even researchers themselves go about assessing the possible presence of the aforementioned researcher bias stemming from institutionalised views on vulnerability and decisional capacity?
Furthermore, is it possible to maintain the distance perceived to be necessary in research studies and trials while remaining true to the ethical imperatives of caring (particularly for medical practitioners)? How far can or should a researcher go in narrowing or broadening that distance in perinatal mental health research in order to establish trust and rapport?
‘Building rapport gives the researcher access to these crucial narratives in the participant’s life, but what is the ethical responsibility of the researcher to the participant and the relationship that has been built?’. 43 (p.201) In other words, there is also a need to examine what obligations, if any, researchers have towards participants not just during a study, but also afterwards.
Another crucial factor to acknowledge is the latent power dynamic between researcher and participant. This dynamic is inherently skewed towards the researcher and gives rise to several ethical considerations.
One such consideration was perfectly expressed by one of the women in the Andrighetti et al. 27 (p.552) study: ‘I had the same interviewer each time… you spend that time with someone, and you get to know them a little bit and feel also a personal obligation to continue on and help her out’.
This sense of obligation towards the researcher, towards whom the balance of power is skewed, is ethically problematic in the sense that it is unclear whether such a sense of obligation stems from pure altruistic motives, an intrinsic sense of morality, a sense of mutual trust and respect, or from an exercise of influence over the participant by the researcher. Hem et al. 31 argue that in clinical practice there is a hierarchy of treatment pressures where persuasion occupies the lowest rung, followed by leverage, inducements and threats, and ending with compulsion. It is, the authors contend, useful for mental healthcare professionals to consider their capacity to influence their patients along this type of continuum. It may be appropriate to extend a similar type of continuum to the researcher/participant relationship in order to ascertain whether participants who are enrolling into or staying the course in a perinatal mental health study are being subjected to either intended or unintended forms of persuasion or coercion, to what effect, and whether the participant perceives any disrespect of their autonomy in any way.
Another related consideration pertains to the payment of honoraria or other forms of reward and remuneration for participation in this type of research, such as was utilised by the Martin et al. 31 and Le et al. 30 studies. It begs the question whether doing so falls onto the continuum as a subtle type of coercion in the form of persuasion, and then, if it is accepted, what the consequences are for the subsequent researcher/participant relationship, as well as for the participant’s experience of participating. There is some evidence to suggest that participants may not necessarily view the payment of honoraria as coercion because perceptions of coercion are influenced to a large degree by a participant’s belief about whether a researcher is treating the participant with respect, is honest, and is qualified. 44
In the Le et al. 30 (p. 840) study, the researchers mentioned that participants started to view their interviewers or the researchers as “friends.” This could be reflective of an unconscious effort on the part of participants to “rebalance the power difference inherent in the research relationship” 43 and to reassert a sense of autonomy.
It is our view that further research is needed to investigate the ethical implications of the researcher-participant relationship in perinatal mental health studies.
Limitations and strengths of the systematic review
A key strength of this review lies in the conduct of a methodologically robust systematic review. The absence of specific ‘ethics’ search terms in databases can be challenging, but this was overcome by the use of proximity searching which led to the retrieval of 9830 unique articles, and the interpretation of ethical issues broadly. Limitations include the absence of meta-synthesis because of the clinical and methodological heterogeneity, and the relative geographical and cultural homogeneity of the included studies. However, the fact that only six studies met the inclusion criteria clearly points to the need for empirical research on the ethical issues relating to the experiences of women from different cultural and demographic backgrounds with perinatal mental health issues who participate in research. Whilst our search strategy and search terms were designed to reflect and cover the spectrum of perinatal mental health issues, we acknowledge the dominant focus on PPD in the included studies. This limits the generalisability of the review’s findings and, again, points to the need for additional empirical research on the broader range of mental health issues experienced.
Conclusion
We highlight four main areas of ethical concern in this systematic review. The first pertains to researcher bias which can stem from institutional classifications of pregnant women as being ‘vulnerable’, and/or the view that depression may impact a woman’s decisional capacity. Consequences of such a bias could include an asymmetry in the researcher/participant power dynamic, suboptimal study design, or even inappropriate or exploitative enrolment of research participants.
The second area of concern pertains research participants and how well pregnant women with mental health issues understand automated, online consent. A central question is how to structure the process of obtaining online informed consent in perinatal mental health research while respecting the autonomy and privacy of the women being recruited. We suggest that it would be helpful to establish a standard in perinatal mental health research that would include an assessment of capacity to give informed consent and would the relational factors (educational level, health literacy, support systems, sociocultural context, etc.) that may impact a woman’s ability to autonomously give informed consent into account.
The third area of concern we highlighted relates to trust, which the included studies overwhelmingly highlighted as a fundamental factor in the recruitment and retention of peripartum women into research. Some key questions that arise from the discussion of establishing and maintaining researcher/participant trust in a perinatal mental health study are:
How should research institutions or researchers themselves go about assessing the possible presence of researcher bias?
Since perinatal mental health research studies rely on the establishment of a strong rapport with participants, is it possible to maintain the distance perceived to be necessary in research studies and trials while remaining true to the ethical imperatives of caring (particularly for medical practitioners)? How far can or should a researcher go in narrowing or broadening that distance in order to establish trust and rapport?
Lastly, the relationship between researcher and participant is already inherently skewed towards the researcher insofar as the latent power dynamic is concerned. This dynamic raises the possibility of a greater sense of obligation felt by participants towards researchers, and the spectre of intended or unintended persuasion or coercion exercised by researchers.
It is clear from this systematic review that the ethical issues that arise from women’s experiences of participating in research are underrepresented in the literature and warrant further exploration.
Supplemental Material
Supplemental Material - Systematic review of ethical issues in perinatal mental health research
Supplemental Material for Systematic review of ethical issues in perinatal mental health research by Mickie de Wet, Susan Hannon, Kathleen Hannon, Anna Axelin, Susanne Uusitalo, Irena Bartels, Jessica Eustace-Cook, Ramón Escuriet, Deirdre Daly in Nursing Ethics
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
