Abstract
Objectives
To assess screening quality metrics and to describe mortality rates eight years after redesign of breast cancer screening and diagnosis pathways, and the introduction of digital breast tomosynthesis.
Setting
Breast Unit of the Toledo Health Area in the region of Castilla-La Mancha (Spain).
Methods
We recorded screening metrics and mortality data following the introduction of digital breast tomosynthesis in 2011 for screening and diagnosis pathways. We then compared the mortality between Toledo Health Area and the rest of Castilla-La Mancha, where digital breast tomosynthesis is not available.
Results
All screening quality metrics improved following the introduction of digital breast tomosynthesis. The cancer detection rate significantly increased from 2.3 (95% confidence interval (CI): 1.9–3.6) to 4.5 per 1000 women (95% CI: 3.2–5.2) on average between the periods 2005–2009 and 2015–2018, while the recall rate significantly decreased from 7.0% (95% CI: 6.8%–8.2%) to 2.6% (95% CI: 2.0%–3.6%). Comparing breast cancer mortality rates for 2014–2018 in the Toledo Health Area with the rest of Castilla-La Mancha, which had similar cancer treatment access and management protocols but without digital breast tomosynthesis, the crude mortality rate was 17.79 (95% CI: 15.38 -20.19) vs. 24.76 per 100,000 (95% CI: 26.12–23.39), respectively. The cumulative risk of death was also significantly lower for the Toledo Health Area than for Castilla-La Mancha.
Conclusion
The introduction of digital breast tomosynthesis improved screening quality indicators. Breast cancer mortality simultaneously decreased with respect to the rest of Castilla-La Mancha. Further research is needed to assess the long-term results, and the role that the redesign may have played in reducing mortality.
Keywords
Introduction
Breast cancer accounts for up to 13.8% of deaths due to malignant tumors in women in Castilla-La Mancha (CLM, Spain), and it is the tumor with the highest incidence in this region. 1 The autonomous region of CLM is part of the publicly funded Spanish National Healthcare System, and it is structured as eight healthcare areas, one of which is the Toledo Health Area (THA), with a catchment area of almost 500,000 people.
The breast cancer screening program was established in the THA in 1991, according to the European Guidelines for Quality Assurance in Breast Cancer Screening and Diagnosis. 2 However, certain limitations were observed over time. The population-based screening was outsourced to private clinics, who carried it out in agreement with the regional public healthcare system. However, this yielded a series of issues. First, the communication between the private clinics and the hospital was largely inadequate. Digital images and complete reports could not be shared between the screening program (hosted at clinics) and the diagnosis pathway (hosted at public hospitals). Thus, recalls and unnecessary further investigations were common. Moreover, imaging technology was more powerful at hospitals than at clinics, and staff in hospitals were more highly specialized. Clinics also had limited flexibility when it came to scheduling appointments, and recall delays were sometimes long, generating more anxiety in women. Finally, women generally showed more confidence in the Breast Unit of the Hospital Virgen de la Salud (Toledo). As a result, many of them booked their routine checkups directly at the hospital, bypassing the official screening program. This increased the workload of the Breast Unit and altered the population-based screening statistics. 3
Consequently, in 2011, a task force was formed to redesign the program workflow in order to obtain more efficient and completely public management. 3 This included the implementation of digital breast tomosynthesis (DBT) scans. In the present study, we describe and discuss breast cancer mortality outcomes in this region eight years after this redesign of the breast screening program and diagnostic circuit.
Materials and methods
Characteristics of the redesigned screening and diagnosis pathway
The screening program is aimed at asymptomatic women between 45 and 69 years old. The subject undergoes mammography on day 1, the images are read on day 2, and the results are sent out to the patient and their general practitioner on day 3. This is possible thanks to pre-downloading of the subject’s images the night after the mammography, which are then available at the radiologist’s reading station on day 2, without any delay. In addition, imaging reports are prepared with software that was especially designed for our department and has been almost completely automated. Thus, the radiologist can fill out the report form with just a few clicks of the mouse, and the resulting letter is automatically prepared and sent out. All these efforts were aimed at reducing women’s anxiety associated with waiting for the results, as far as possible. 3
Women with suspicious findings are recalled to undergo the necessary complementary tests at the Diagnostic Radiology Unit. 3 Thus, this Unit was established as the link between the breast screening program pathway and the pathological diagnosis pathway, where a “one-appointment” strategy was introduced. The radiologist schedules all necessary diagnostic tests needed for each woman in just one day. In our redesigned patient flow, the radiologist is the key person responsible for a subject from the first appointment until final diagnosis. 3 This protocol is unique in our region, as other healthcare areas are organized on the basis of diagnostic tests. In the latter case, in general, the subject undergoes mammography and, depending on the findings, may be recalled for further testing by another radiologist, a gynecologist or even a surgeon, with longer diagnostic time and no overall key person in charge. It also implies busier and more complicated schedules for the radiology department, and the risk of more heterogeneity in the diagnosis.
Technological improvements were also implemented, such as the use of DBT (Selenia Dimensions system, Hologic) with synthesized digital mammography, as well as computer-aided double reading of images with arbitration. A pool of five radiologists were dedicated exclusively to breast pathology. 3
Assessment of redesign and reorganization outcomes
In order to determine the results after reorganization of the breast screening program in the THA, we assessed:
The quality metrics of the screening program before and after reorganization and The mortality trend since reorganization of the screening program.
Data sources were the Toledo Breast Unit computer system, the Cancer Registry of the CLM Autonomous Region, the Spanish Ministry of Health Death Registry and the Spanish National Institute of Statistics.
Confidence intervals (CI) and statistical significance were calculated by the exact method. All analyses were carried out using SPSS 23 and Stata 14.
Breast cancer screening metrics
Breast cancer screening indicators were estimated based on the CLM Healthcare Service (SESCAM) and CLM Health Department Registry. Indicators were estimated using their own statistics application:
Participation rate (%): the percentage of subjects who actually underwent screening out of the total who received an invitation. Recall rate (%): the percentage of subjects with Breast Image Reporting and Data System scores of 0, 4 and 5 out of the total number of exams carried out in the study period. Cancer detection rates for tumors <10 mm (%): percentage of invasive cancers detected with diameter <10 mm out of the total number of invasive cancers detected. Percentage of tumors with diameter ≥2 cm. Ductal carcinoma in situ (DCIS) rate (%): percentage of DCIS out of all detected cancers. Cancer detection rate (out of 1000 women): total number of screening-detected cancers out of the total number of exams carried out in the study period.
All indexes were compared between the pre- and post-redesign periods (2005–2009 and 2011–2015, respectively).
The interval cancer rate was also estimated for the period 2011–2015. However, it was not compared with the pre-redesign period because this figure was not available. Interval cancers were defined as those diagnosed within 12 months of a negative screening exam, since the THA policy is to review interval cancers annually.
Breast cancer mortality at eight years
Mortality data have been prospectively collected since the redesign of the screening program. In this paper, we present the mortality trend estimated over the first eight years after redesign in the THA as compared to the trend in the rest of the region in the same period using Poisson regression models. The other seven healthcare areas of CLM are geographically close to the THA and share historical, cultural and social elements, as well as comparable access to oncologic treatments and similar cancer management protocols. However, none of them has yet implemented our redesigned screening workflow, and they continue to use full field mammography as an imaging technique. Thus, the rest of CLM was used as a control.
The crude and adjusted breast cancer average annual mortality rates in the eight healthcare areas of CLM were analyzed. Finally, also for the period 2014–2018, we estimated:
The crude mortality rate; Standardized mortality ratio; Cumulative death risk and Potential years of life lost (PYLL) ratio.
Additionally, the breast cancer mortality trends according to age group for a period before redesign (2002–2009) and a period after redesign (2011–2018) were compared for Toledo alone.
Results
Quality indicators
The participation rate in the program increased, surpassing 70%, the minimum acceptable threshold, as indicated by the European Guidelines. 2 All assessed quality indicators are presented in Table 1 as mean values over the seven years before (2002–2009) and after (2011–2018) redesign.
Mean metrics of the screening program before and after redesign, with reference quality standards.
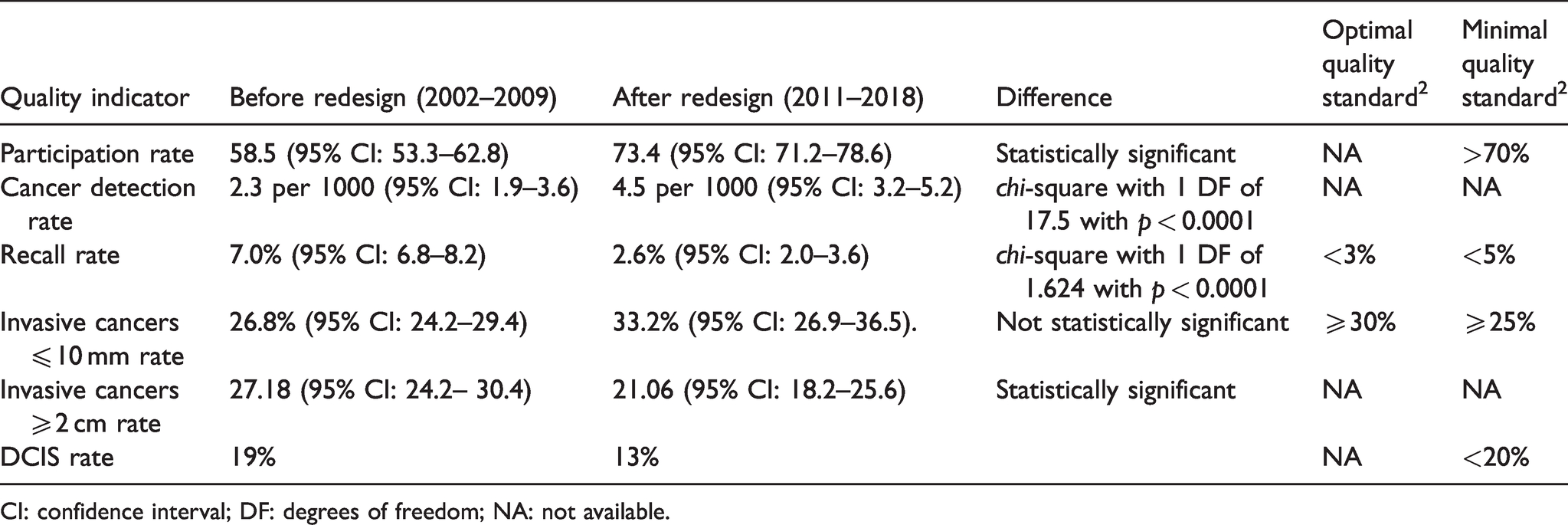
CI: confidence interval; DF: degrees of freedom; NA: not available.
Although the interval cancer rate was not available for the period before redesign, it was 0.34 per 1000 women in the first year after reorganization and decreased to 0.17 per 1000 women in the following years, remaining stable over time.
Mortality
The crude and adjusted mortality rates due to breast cancer in the THA and the rest of the region over the 2014–2018 period are presented in Figure 1(a) and (b), respectively. Both rates were lower in the THA than in any other health area in CLM. Using the THA mortality rate in the five-year period 2009–2013 (which is when the screening program was being redesigned) as baseline risk, the relative breast cancer death risk in the 2014–2018 period, i.e. after redesign was fully established, was 0.84 (95% CI: 0.64–1.10, p < 0.05) in the THA and 0.93 (95% CI 0.82–1.04, p < 0.05) in the rest of CLM. This corresponds to a 16% decline in mortality in the THA vs. 7.4% in the rest of CLM. The relative risk of death in the THA vs. CLM in 2009–2013 was 0.81 (95% CI: 0.67–0.996, p = 0.046), while in 2014–2018 it was 0.73 (95% CI: 0.59–0.90, p = 0.003). The standardized mortality ratio in the THA indicates that breast cancer deaths were approximately 20% lower than expected in the area, and the lowest in the region, being significantly lower than in the rest of CLM (Figure 1(b)).
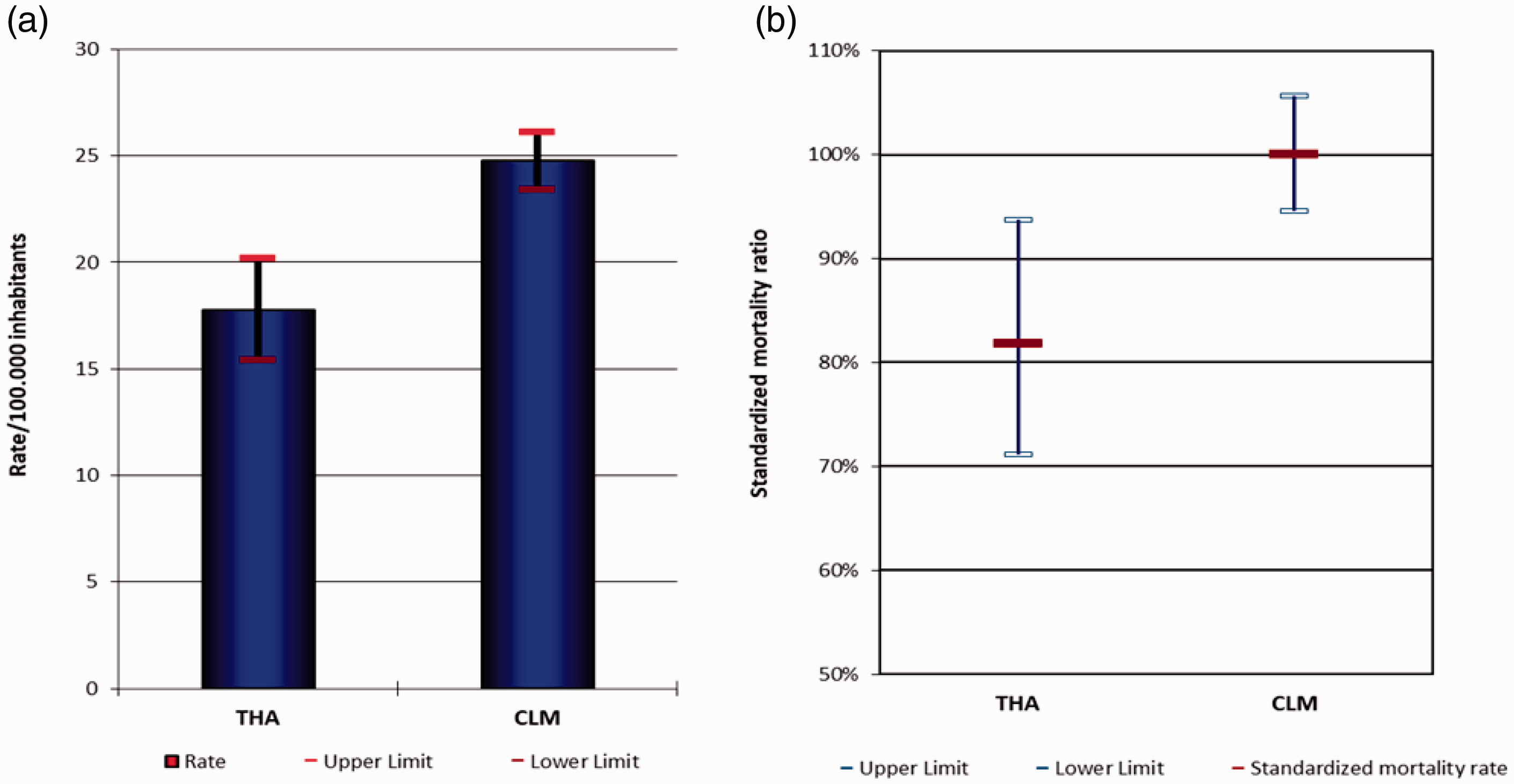
Breast cancer crude mortality rate (a) and standardized mortality ratio (b) in the THA compared to the rest of CLM, over the 2014–2018 period.
The cumulative risk of death (0–69 years) in the THA was significantly lower than in the rest of CLM over the 2014–2018 period (Figure 2). The PYLL rate indicates that, in the THA, approximately 25% fewer years of life were lost to breast cancer than in the rest of CLM in the 2014–2018 period (Figure 3). Additionally, the mortality rate per age group in the THA was lower in the period after redesigning the screening program (2011–2018) than before (2002–2009), especially in the age range 55–75 years, which is the life interval when the benefit of screening is most evident (Figure 4).
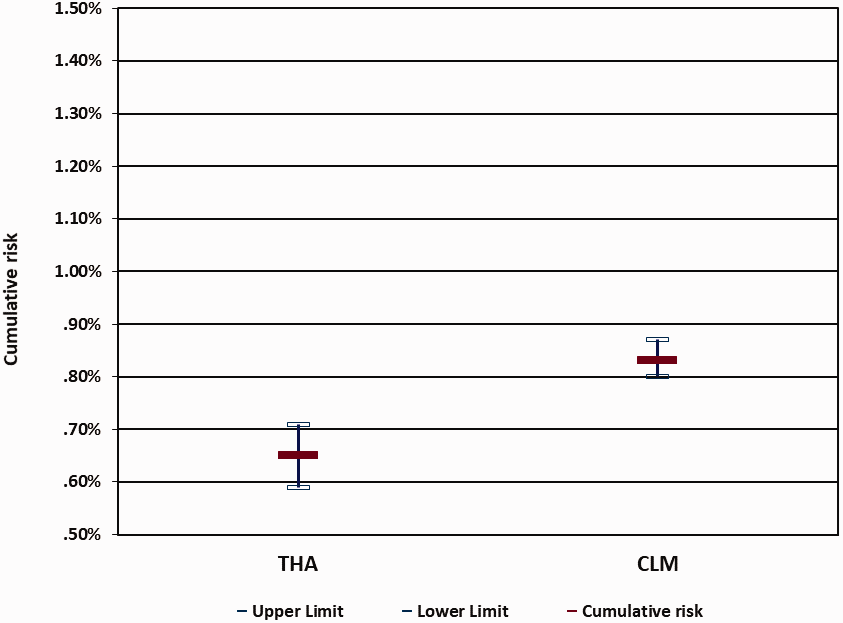
Cumulative risk of breast cancer death in the THA compared to the rest of CLM, over the 2014–2018 period.
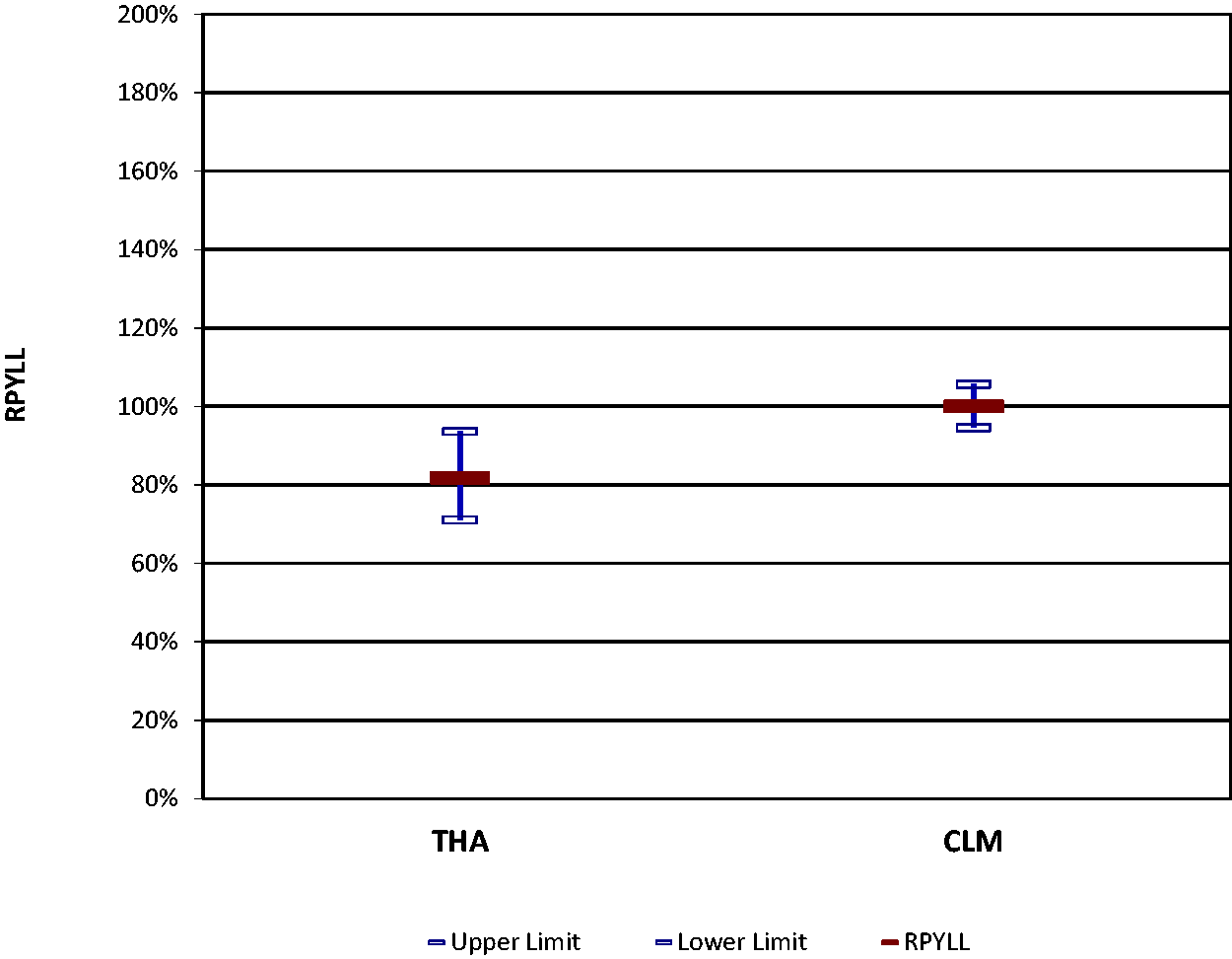
Rate of potential years of life lost (RPYLL) to breast cancer in the THA, compared to the rest of CLM over the 2014–2018 period.
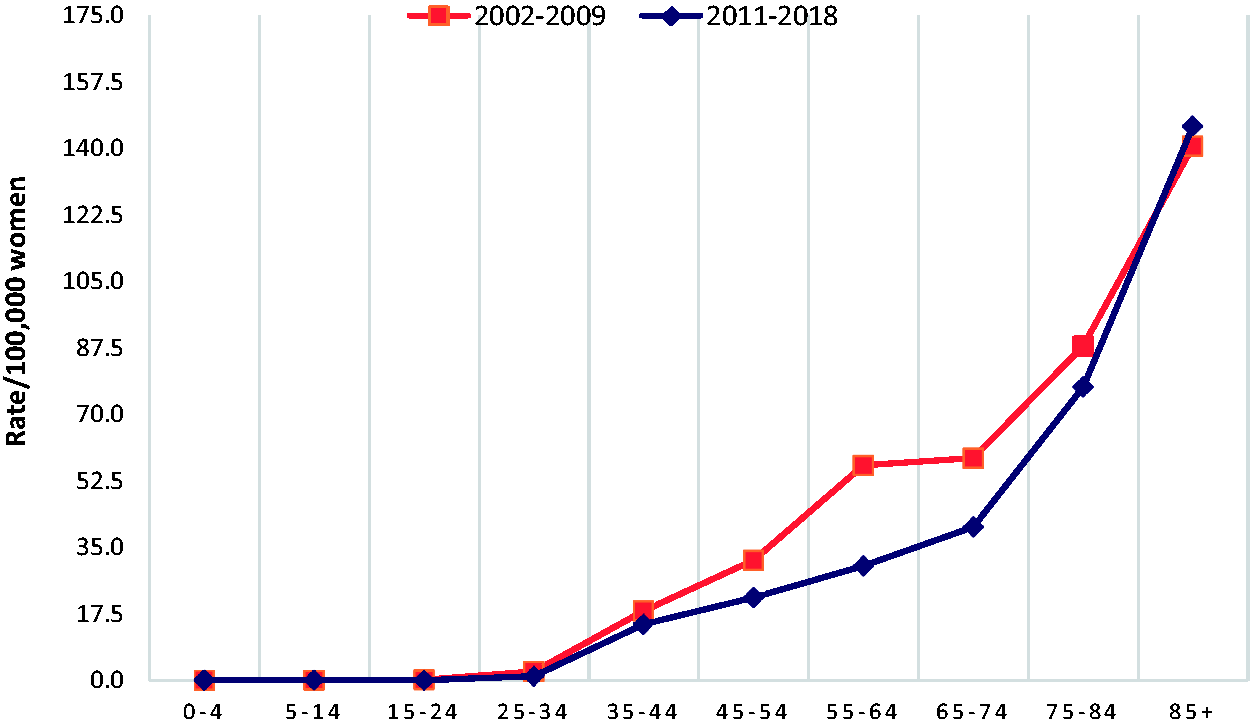
Comparison of the breast cancer specific mortality rate according to age group in the THA before (2002–2009) and after (2011–2018) redesigning screening.
Discussion
We present herein the results obtained following the redesign of our breast cancer screening and diagnosis circuits, where one of the most notable changes was the introduction of DBT. The aim was to make these protocols more efficient and completely publicly managed.
The redesign has yielded a significant improvement in screening quality metrics, which were already within the standard values established by the European Guidelines. 2 In particular, the cancer detection rate significantly increased from 2.3 (95% CI: 1.9–3.6) to 4.5 (95% CI: 3.2–5.2) per 1000 women on average between the time intervals of 2005–2009 and 2015–2018, while the recall rate significantly decreased from 7.0% (95% CI: 6.8–8.2%) to 2.6% (95% CI: 2.0–3.6%). Additionally, comparing breast cancer mortality rates for 2014–2018 in the THA with the rest of CLM, with similar cancer treatment access and management protocols but without DBT, the crude mortality rate was 17.79 (95% CI: 20.19–15.38) vs. 24.76 per 100,000 (95% CI: 26.12–23.39). The cumulative risk of death was also significantly lower for the THA than for CLM.
One of the main criticisms directed at breast cancer screening programs is the risk of overdiagnosis and overtreatment. In particular, it is not completely clear whether all DCIS will progress over time and should therefore be treated as an invasive cancer precursor. 4 In fact, there are some studies in which less than 50% of DCIS left untreated progressed over a woman’s lifetime. 5 Thus, these types of lesions pose a clinical and ethical dilemma. Fortunately, DBT has been shown to significantly increase the detection of invasive cancer, but not of DCIS. 6 Indeed, since the introduction of DBT in our screening procedures, our rate of DCIS detection has decreased and has been constantly well under the acceptability threshold of 20% recommended in the European Guidelines. 2
Interval cancers are difficult to compare between programs across Europe, as the recommended screening periodicity is between 1 and 3 years, 7 and different countries have adopted different screening frequencies. 8 Furthermore, despite the Guidelines, 2 there is still no homogeneity in the criteria used for interval cancer assessment in clinical practice. Nevertheless, interval cancers could be an interesting outcome for follow-up. Previous studies published on DBT vs. digital mammography data indicate that, despite the increase in screen-detected breast cancers, there is no clear decrease in interval cancers with the introduction of DBT.9,10 Since interval cancer data before the introduction of this modality are not available for our area, we cannot say whether they improved; however, the interval cancer rate decreased one year after introduction and has remained steady for the last seven years.
Breast screening detractors often argue that these programs do not significantly reduce breast cancer mortality and, therefore, should be avoided for women with moderate cancer risk. However, these criticisms are mainly associated with the widely debated Canadian National Breast Screening Study, 11 which was vehemently criticized for flawed methodology. Moreover, the Canadian study only targeted screening in women between 40 and 49 years of age, which only partially overlaps with our lower age limit for screening, which is 45 years, as recommended by the European Guidelines. 7 Furthermore, a meta-analysis of randomized clinical trials in various countries has demonstrated a 20–30% benefit in mortality associated with screening programs. 12
A systematic review of observational studies conducted in Europe identified a decrease in breast cancer mortality of 38%–48% in women who underwent breast screening vs. 25%–31% in women who were invited to screening but never attended. 13 Furthermore, a long-term population-based study carried out in Australia showed that screened women had a 39% reduction in breast mortality compared to unscreened women (Hazard Ratio [HR] = 0.61, 95% CI: 0.55–0.68) over 16 years of follow-up. 14 Finally, it has been demonstrated that the cancer detection setting (symptomatic vs. screening) is an independent risk factor for breast cancer death. In fact, the adjusted hazard ratio of dying from breast cancer was twice as high for women with symptomatic breast cancer than for screen-detected cancer. 15 However, ours appears to be the first study describing mortality decline after a screening redesign, as well as the first study of eight-year mortality data recorded since the introduction of DBT.
Our data show a decline in mortality in the THA in the 2011–2018 period, compared to the 2002–2009 period. The two time intervals begin nine years apart, so many factors may have influenced the mortality rate, including improvements in treatment efficacy, e.g. with the introduction of targeted therapies 16 and management protocols, as well as advances in technology. While it is certainly difficult to determine the specific role that our redesign of the screening process may have played in breast cancer mortality, it is notable that the main mortality reduction in our area is more marked in the age interval most affected by breast cancer screening, namely between 55 and 74 years.
According to a theoretical model designed to estimate the impact of the introduction of new and more effective treatments over time on the mortality benefits of screening, screening clearly reduces the absolute mortality, and in fact more patients diagnosed in advanced stages can be successfully treated than before. However, the relative mortality benefit offered by screening remains unchanged. 17
A study on the reduction of breast cancer mortality considering the year of implementation of national screening programs has been recently published for 14 European countries, including Spain. According to this data analysis, while a downward trend in breast cancer mortality was already evident before the beginning of systematic screening programs, they seem to have accelerated the process, further reducing annual mortality by –1.5% to –5.4% in the 50–59 age group, –0.2% to –8.1% in the 60–69 age group and by 0% to –7.1% in the 70–79 age group, depending on the country, within three years of implementation of the mammography screening program. 18 Thus, we could hypothesize that quality improvements in screening programs also improves these rates, however, slightly.
Our analysis also shows a reduction in breast cancer-specific mortality in our healthcare area with respect to the rest of the region. This should be interpreted considering that all healthcare areas of our region have access to the same breast cancer treatments and share similar management protocols, while the THA was the only area that redesigned its screening and diagnostic pathways. Thus, comparing the rates found in THA with other areas of CLM reduces the effect of differences between cancer treatments over time, which may be affecting the mortality time trend. A study carried out in Denmark 19 used a similar approach, comparing breast cancer mortality between the health area of Funen and the rest of Denmark upon introduction of breast cancer screening in Funen. This was among the first counties to introduce systematic screening, while most of Denmark only introduced it in 2008. This study shows a 26% reduction in mortality in the Funen area in women targeted by screening vs. the rest of Denmark.
As shown by the quality indicators measured in the present study, redesigning our screening program has improved its performance, by introducing appropriate technological advances and a multidisciplinary approach to diagnosis and management, analyzing and remodeling inefficient workflow steps, improving communication between different healthcare levels, and changing the role of the radiologist and the Diagnostic Radiology Unit. Nonetheless, it seems that further improvement in breast cancer management could be achieved by introducing risk stratification for breast cancer screening, which is the subject of current debate.20,21
We hypothesize that at least part of the reduction in mortality is a result of the redesign of our screening/diagnostic pathways. In particular, earlier detection of invasive lesions is certainly due to the use of DBT, especially in dense breasts, resulting in earlier and less aggressive treatment, with better prognosis. Nevertheless, this is only part of the story: the redesign of the screening program has fostered more participation in the program itself. This in turn may have reduced the number of cancers detected in the pathologic pathway, which, as we know, is an independent risk factor for cancer death. 15 Our approach also improves continuity of care: the same person is responsible for each case from the moment they enter the diagnostic pathway until final diagnosis. He or she also presents the case to the multidisciplinary group, which will collectively decide which therapeutic options to present to the patient. Thus, there is reduced variability in therapeutic approach, too.
Limitations
The number of deaths by age was derived from the CLM Death Register, which receives the death certificate issued by the doctor certifying the patient’s death. This may lead to two problems. First, there may be a mismatch between the actual cause of death and the one reported on the certificate, which may alter the mortality count. However, in the case of breast cancer, this problem is marginal, with a correlation between the Death Register and clinical records of about 90%. 22 Second, there may be variability in the sequence of events reported by the doctors as causing the death. Again, this problem is minimized because, in CLM, the central unit of the Registry of Deaths reviews each certificate, checking the sequence of all events that could have resulted in death, according to WHO guidelines, 23 and thus achieving a certain degree of homogenization.
Comparison of the breast cancer mortality trend among different territorial units may be influenced by the incidence of breast cancer in each territory, among other factors. However, in our case, this effect is limited, because the different healthcare areas that we compared are geographically very close, belonging to the same region and sometimes even to the same province, with similar breast cancer incidence. Moreover, all of them are managed by the same regional healthcare service, which distributes the resources in a uniform manner. We also wanted to approach this problem by performing analyses adjusted for possible confounding factors, but this was not viable, due to lack of data for the other healthcare areas.
Finally, we did not directly analyze the impact on mortality of the introduction of new treatments or changes in the management protocols. As the aim of our study was simply to examine the mortality trend after reorganization, we assumed that changes in treatment or management would not impact significantly on mortality differences when using the rest of CLM as a control. With this approach, we cannot establish a cause–effect relationship between screening/diagnosis and mortality. However, our intent is mainly descriptive.
Conclusion
Redesigning the breast cancer screening and diagnostic program in the THA has yielded an improvement in screening quality indicators. Simultaneously, in the years following redesign, breast cancer mortality has decreased in our healthcare area, as compared to the time before redesign, and with respect to the rest of CLM in the same time period. Further research is needed to assess the long-term results of the newly structured program and the role it may have played in reducing mortality.
Footnotes
Authors' Note
Authors are pleased to inform that the accession state of the above-mentioned article has changed to Open Access.
Acknowledgements
The authors acknowledge Maria Giovanna Ferrario and the Medical Statistics Consulting team (Valencia, Spain) for their collaboration in writing this manuscript. All authors contributed with intellectual material and reviewed it critically before approving the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Uniquely the Open Access fee of this paper was funded by Hologic Spain, this study is otherwise independent from any other external funding.
