Abstract
Objectives
To evaluate experiences and reactions after receiving a mailed, unsolicited human papillomavirus self-sampling kit and identify psychosocial correlates of using kits.
Methods
Survey participants were underscreened women aged 30–64 who were mailed human papillomavirus kits as part of a pragmatic trial at Kaiser Permanente Washington, a United States integrated health care system. Six months after the mailing, we invited kit returners and non-returners to complete a web survey that measured psychosocial factors (e.g. cervical cancer/human papillomavirus knowledge, attitudes toward screening), experiences, and reactions to kits. We compared responses between kit returners and non-returners.
Results
Comparing 116 kit returners (272 invited) and 119 non-returners (1083 invited), we found no clinically significant differences in psychosocial factors. Overall, survey respondents showed knowledge gaps in human papillomavirus natural history (82% did not know human papillomavirus infection can clear on its own) and interpreting human papillomavirus test results (37% did not know a human papillomavirus-negative result indicates low cancer risk). Kit returners found kits convenient and easy to use (>90%). The most common reason for non-return was low confidence in ability to correctly use a kit, although many non-returners (49%) indicated that they would consider future use. Women reported low trust in human papillomavirus testing to identify women at high risk for cervical cancer (52% in returners, 42% in non-returners).
Conclusions
Screening programs could improve uptake and acceptability of human papillomavirus self-sampling through outreach materials that emphasize the high efficacy of human papillomavirus testing for cervical cancer screening and educate patients about how to interpret results.
Keywords
Introduction
Women who never or rarely attend Pap screening are at increased risk for cervical cancer.1–3 Pap screening barriers include sociodemographic factors (e.g. race/ethnicity), 4 poor health status,5,6 logistical difficulties,7,8 embarrassment,8,9 and fear of abnormal results.8,9 Recently expanded United States cervical cancer screening guidelines include primary human papillomavirus (HPV) screening (i.e. HPV alone) as an additional recommended option for women aged 30–65. 10 With primary HPV screening, samples can be self-collected, with comparable accuracy to clinician-collected samples. 11 HPV self-sampling (HPV-SS) is an emerging option that may address known Pap screening barriers.
Numerous studies have shown that women find HPV-SS acceptable, 12 and mailing HPV-SS kits directly to underscreened women in an organized screening program increases screening rates, compared with traditional invitations or reminders for Pap screening. 11 However, little is known about women’s reactions to receiving unsolicited HPV-SS kits in the mail, their preferred screening options, future screening intentions after receiving a kit, and how much their willingness to use an HPV-SS kit may be affected by psychosocial factors such as HPV/cervical cancer knowledge, trust in HPV-SS results, or trust in their physician.13–15 A better understanding of these factors can help healthcare systems to optimize mailing HPV-SS kits as a cervical screening outreach strategy.16–19
Our objectives were to: (1) measure potential psychosocial correlates of HPV-SS uptake among underscreened women who were randomized to receive a mailed HPV-SS kit as part of a pragmatic trial within a United States healthcare system; (2) compare correlates between women who returned and did not return HPV-SS kits; (3) characterize experiences with HPV-SS kit use; (4) identify reasons for non-return; and (5) characterize women’s reactions to receiving kits, including screening preferences, future intentions, and trust in HPV-SS.
Methods
We conducted this study among women randomized to the intervention arm of the Home-Based Options to Make screening Easier (HOME) pragmatic trial (ClinicalTrials.gov ID:NCT02005510). 20 HOME evaluated whether direct mailing of HPV-SS kits to underscreened women increases cervical cancer screening uptake and cervical pre-cancer detection/treatment, compared with usual care. From 2014 to 2017, 16,590 underscreened women (>3.4 years since last Pap) aged 30–64 with a primary care provider at Kaiser Permanente Washington (KPWA, a large integrated healthcare system in Washington State) were randomized to a control arm (usual care consisting of annual patient reminders to attend Pap screening and ad-hoc outreach by clinics), or an intervention arm (usual care plus a mailed, unsolicited HPV-SS kit and reminders). Kits included an invitation letter, a research information sheet, instructions, two Dacron-tipped swabs, a collection tube, and a pre-paid return envelope to the KPWA laboratory. HPV test results were entered into the electronic health record (EHR) and provided to women’s primary care teams for appropriate follow-up. The study protocol was reviewed and approved by the KPWA Institutional Review Board.
From January to July 2015, we mailed survey invitations six months after trial randomization. We used the EHR to identify and recruit women in two groups, based on kit return status (hereafter called “kit returners” and “non-returners”). The target sample size (100 per group; 200 total) was determined based on study resources. We estimated 80% power to detect between-group mean scale score differences as small as 0.4 standard deviations. We excluded women who opted out of EHR review after receiving a kit, were undergoing diagnostic follow-up, or were invited to participate in a qualitative interview after an HPV-positive kit result. 19 Women were invited to complete a 5–10-min web survey about their experience with a “health screening kit” mailed six months prior. The letter included a survey URL, personalized access code, cash incentive, and a toll-free number to request a paper survey or opt out. We randomized women 1:1 to one of two cash incentives: $5 pre-incentive only, or $2 pre-incentive plus $10 post-incentive after survey completion. A paper version was mailed if the web survey was not completed within six weeks. Invitations were mailed weekly until we reached the target sample size. In total, 1355 invitations were mailed, 272 to kit returners and 1083 to non-returners. The web survey was hosted on Qualtrics (Provo, UT).
We assessed sociodemographic characteristics, health status, length of health plan enrollment, and time since last Pap through EHR data. Supplementary Figure 1 outlines the order of survey constructs. Unless otherwise noted, item responses used a five-point Likert scale format. We adapted items from validated questionnaires when possible.21–29
The first section of the survey measured constructs hypothesized to be correlates of underscreening. These included knowledge of HPV/cervical cancer (seven items; yes/no response scale), 21 perceived risk of HPV infection and cervical cancer (two items), 30 perceived barriers to Pap testing (physical, emotional, and structural issues, 11 items),22,23 and trust in physician recommendation for new health technologies and medical care (two items).
The second section used a skip pattern based on whether the woman remembered (1) receiving a kit and (2) using the kit and returning it. A picture of the kit was included as a memory aid. Women who did not remember receiving a kit were skipped to the end. We asked kit returners about their experiences (ten items, including eight from a previous study 25 and two new items) with the kit itself (instructions, ease of use), including physical (pain, discomfort) and emotional (embarrassment) responses, and feelings about the HPV-SS modality (trust, confidence, convenience). Exploratory factor analysis 31 supported a single factor (eigenvalue = 4.47; all factor loadings >0.3). Cronbach’s alpha was 0.88, indicating high internal consistency. We assessed reasons for not returning a kit with seven statements covering similar domains to kit returner experience questions based on items from a previous study. 25 We also identified additional reasons among open-ended responses. Two investigators (CM and RLW) independently reviewed each response before reaching a consensus.
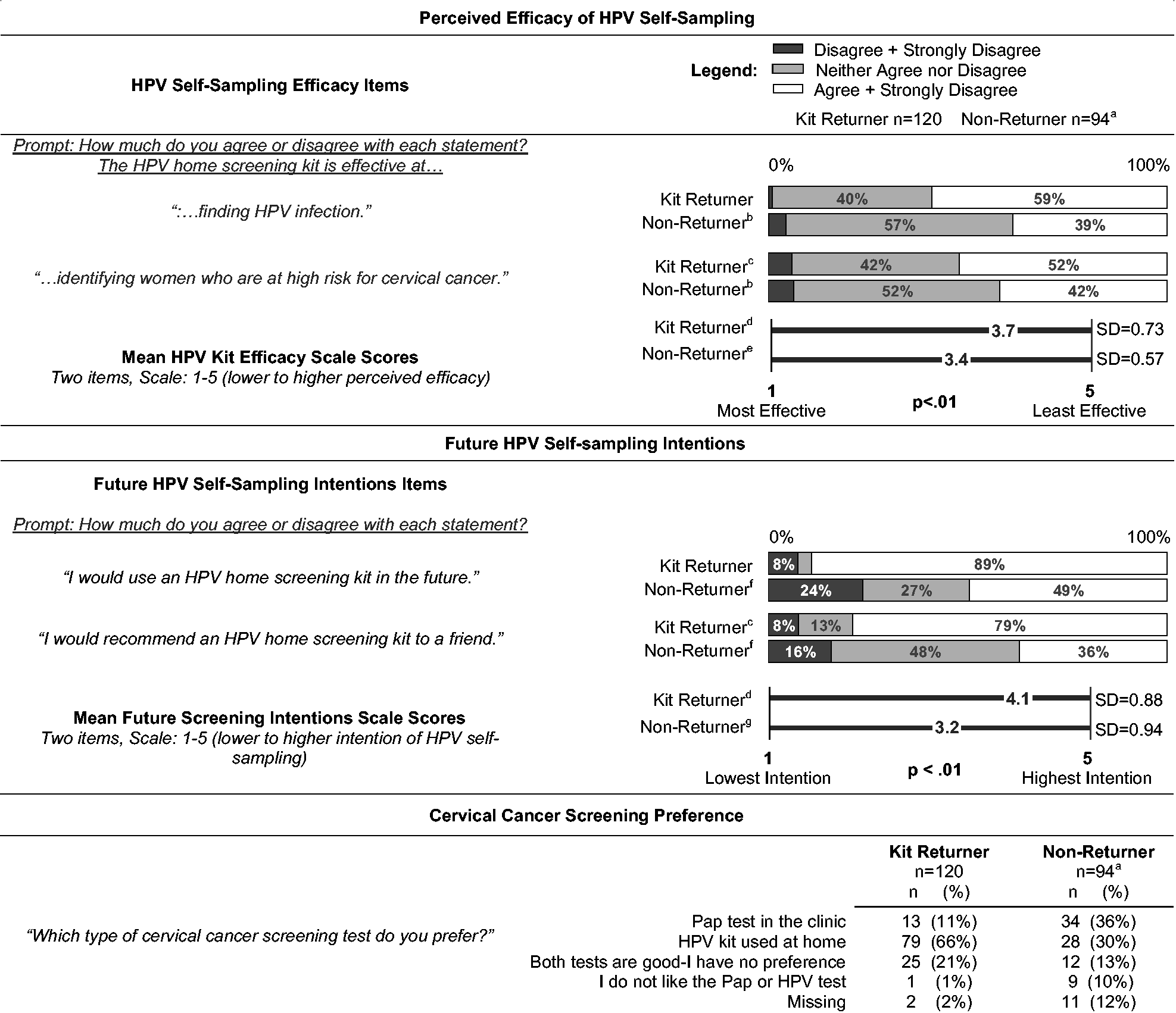
The third section asked women who remembered receiving a kit about three constructs: perceived efficacy of HPV-SS kits to detect HPV infection and women at risk of developing cervical cancer (two items), intentions to use kits in the future or recommend to others (two items), 25 and screening preference (one item),29 with four response options: Pap, HPV-SS kits, both (no preference), or neither.
Using chi-square tests, we compared personal characteristics between survey responders and non-responders by kit return, and personal characteristics between kit returners and non-returners who completed the survey. For personal characteristics comparisons, we used the EHR to define kit return. For the following comparisons of psychosocial correlates, we used women’s self-reported status to define kit returners and non-returners, because we were interested in women’s subjective experiences based on their own recall. To compare HPV/cervical cancer knowledge between kit returners and non-returners, we combined responses into an additive index (higher score indicates more correct responses), and compared scores by group using a chi-square test. For women with one (n = 17) or two (n = 6) missing or “prefer not to answer” responses, those responses were coded as incorrect. Women with more than two missing or “prefer not to answer” knowledge items were dropped from the index (n = 2). We estimated mean scale scores to compare perceived risk of HPV/cervical cancer and physician trust by kit return, and used two-sided t-tests to test for differences. Because our perceived barriers to Pap screening questions encompassed different structural and emotional barriers, rather than a single underlying theoretical construct, we evaluated each item’s association with kit return individually. We compared item responses by kit return using chi-square tests or Fisher’s exact test for comparisons with cell sizes <5. To compare HPV-SS reactions, we compared mean scores for perceived efficacy of HPV-SS kits and future HPV-SS intentions by kit return using t-tests. We used chi-square tests to identify significant differences in reported screening preference between kit returners and non-returners.
Analyses were conducted using Stata 15 (College Station, TX).
Results
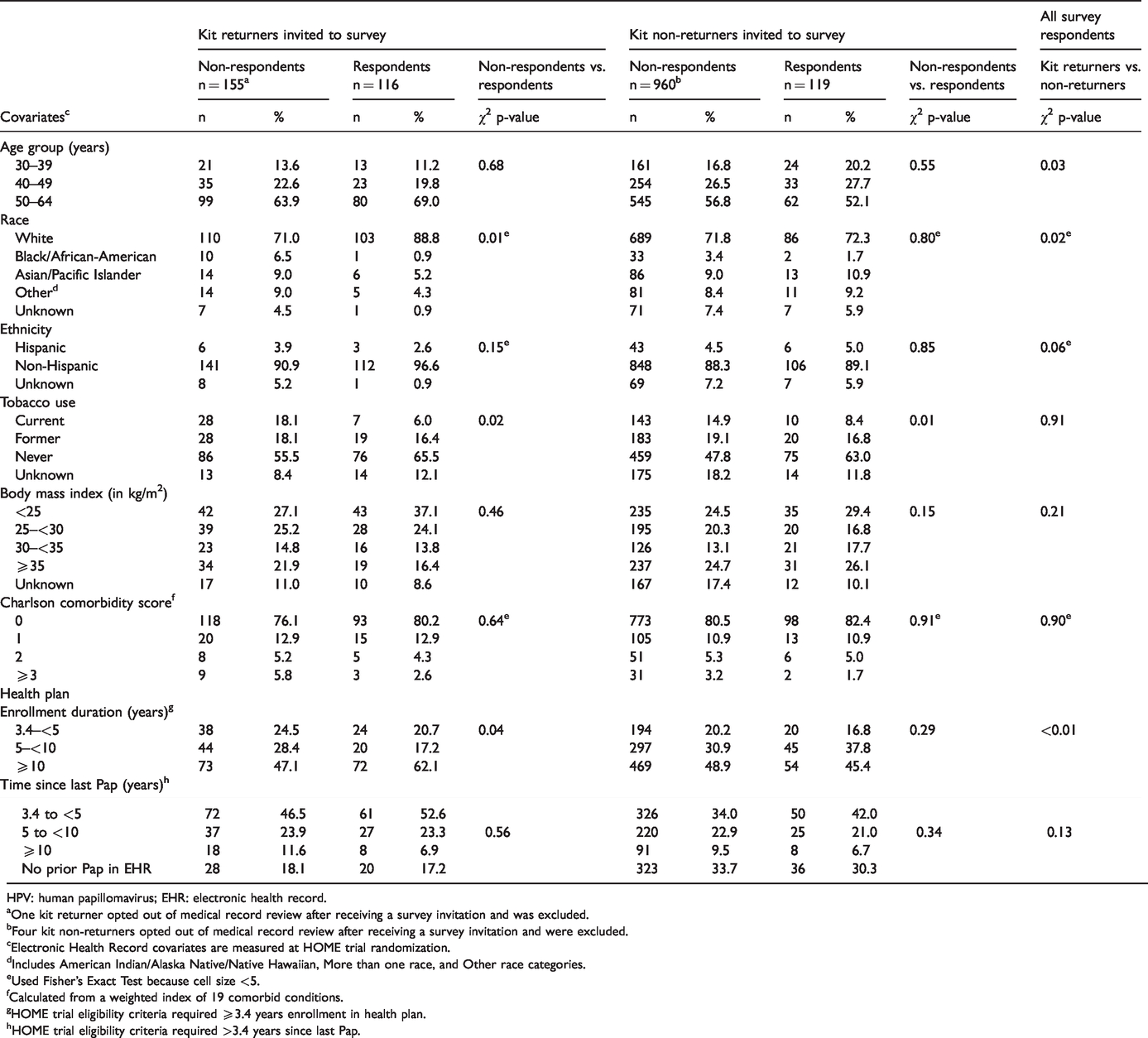
Survey response was 43% among kit returners (116/272) and 11% among non-returners (119/1083). One returner and four non-returners opted out of EHR review after receiving a survey invitation, and were excluded from EHR comparisons. Among returners, survey respondents were more likely to be White, non-smokers, and have a longer duration of KPWA enrollment than non-respondents (Table 1). Among non-returners, survey respondents were more likely to be non-smokers than non-respondents. Overall, most survey respondents were non-Hispanic, White, and aged 50–64.
Sociodemographic, health status, and cervical cancer screening history of underscreened women in a U.S. healthcare system who were invited to complete a survey six months after receiving an unsolicited mailed HPV kit, by kit return and survey response status.
HPV: human papillomavirus; EHR: electronic health record.
One kit returner opted out of medical record review after receiving a survey invitation and was excluded.
Four kit non-returners opted out of medical record review after receiving a survey invitation and were excluded.
Electronic Health Record covariates are measured at HOME trial randomization.
Includes American Indian/Alaska Native/Native Hawaiian, More than one race, and Other race categories.
Used Fisher’s Exact Test because cell size <5.
Calculated from a weighted index of 19 comorbid conditions.
HOME trial eligibility criteria required ≥3.4 years enrollment in health plan.
HOME trial eligibility criteria required >3.4 years since last Pap.
Self-reported kit return was mostly concordant with EHR data. Five women reported returning a kit with no record in the EHR, and one woman reported not returning a kit despite one being documented in the EHR. The following comparisons use self-report to define kit return for a total of 120 kit returners and 115 non-returners:
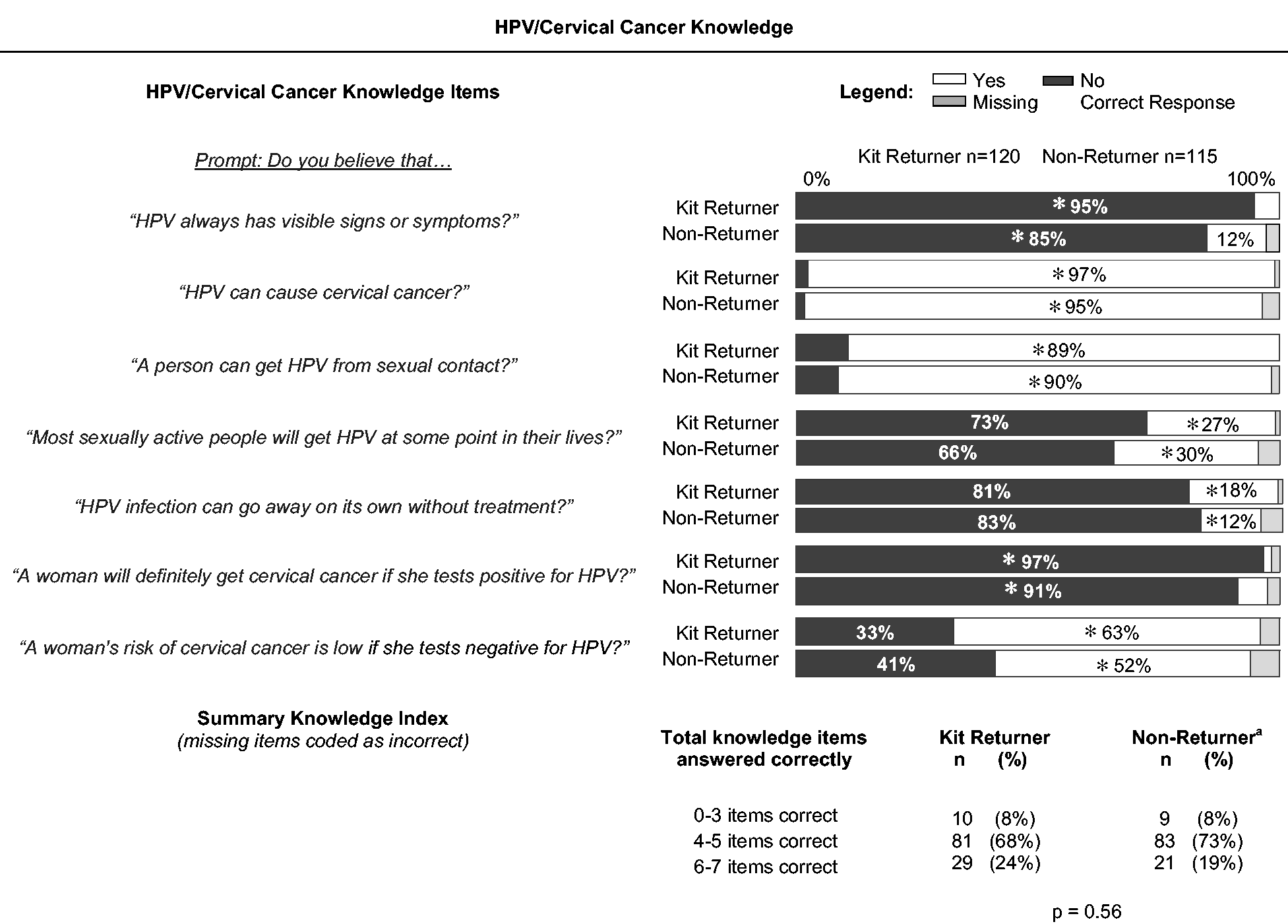
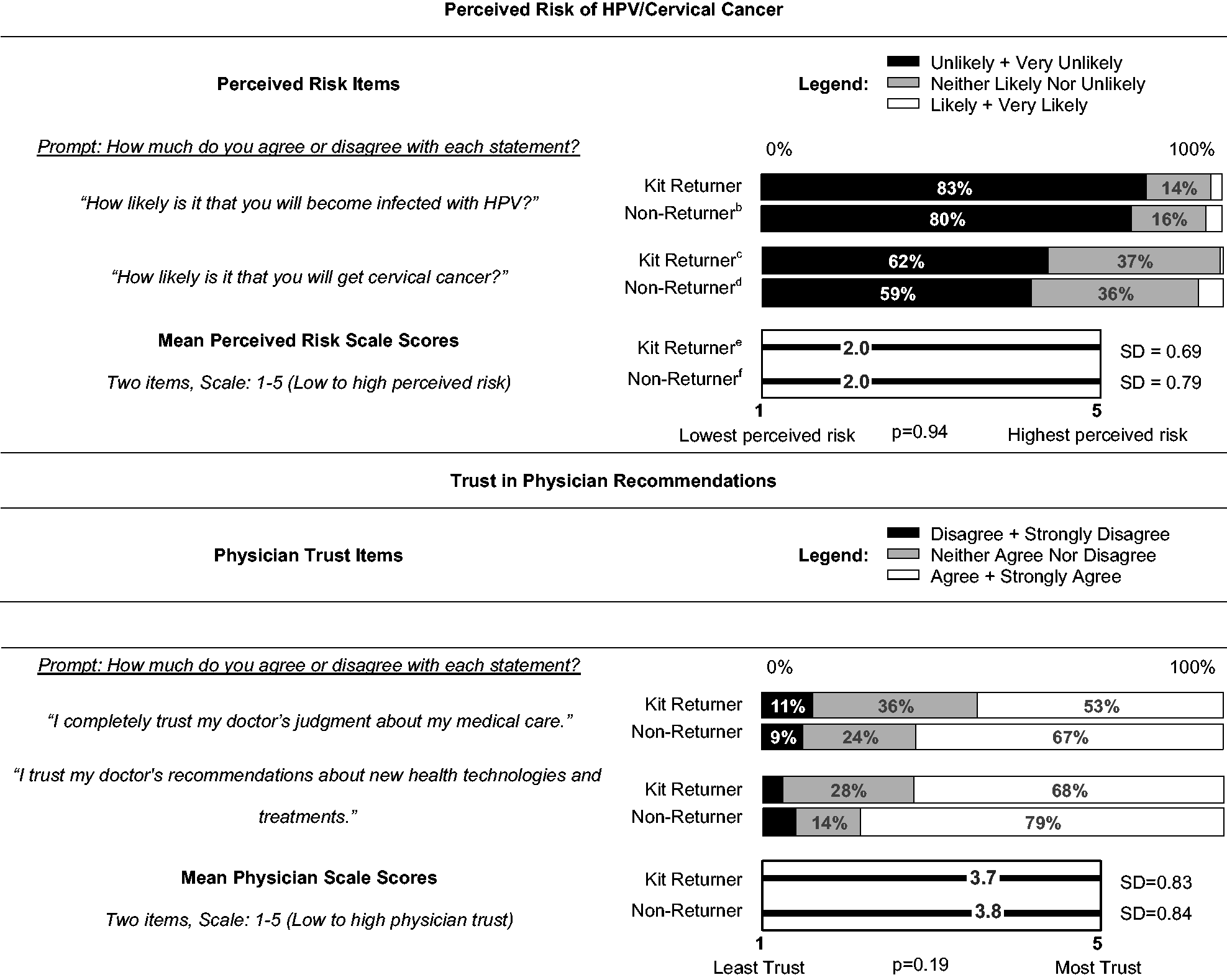
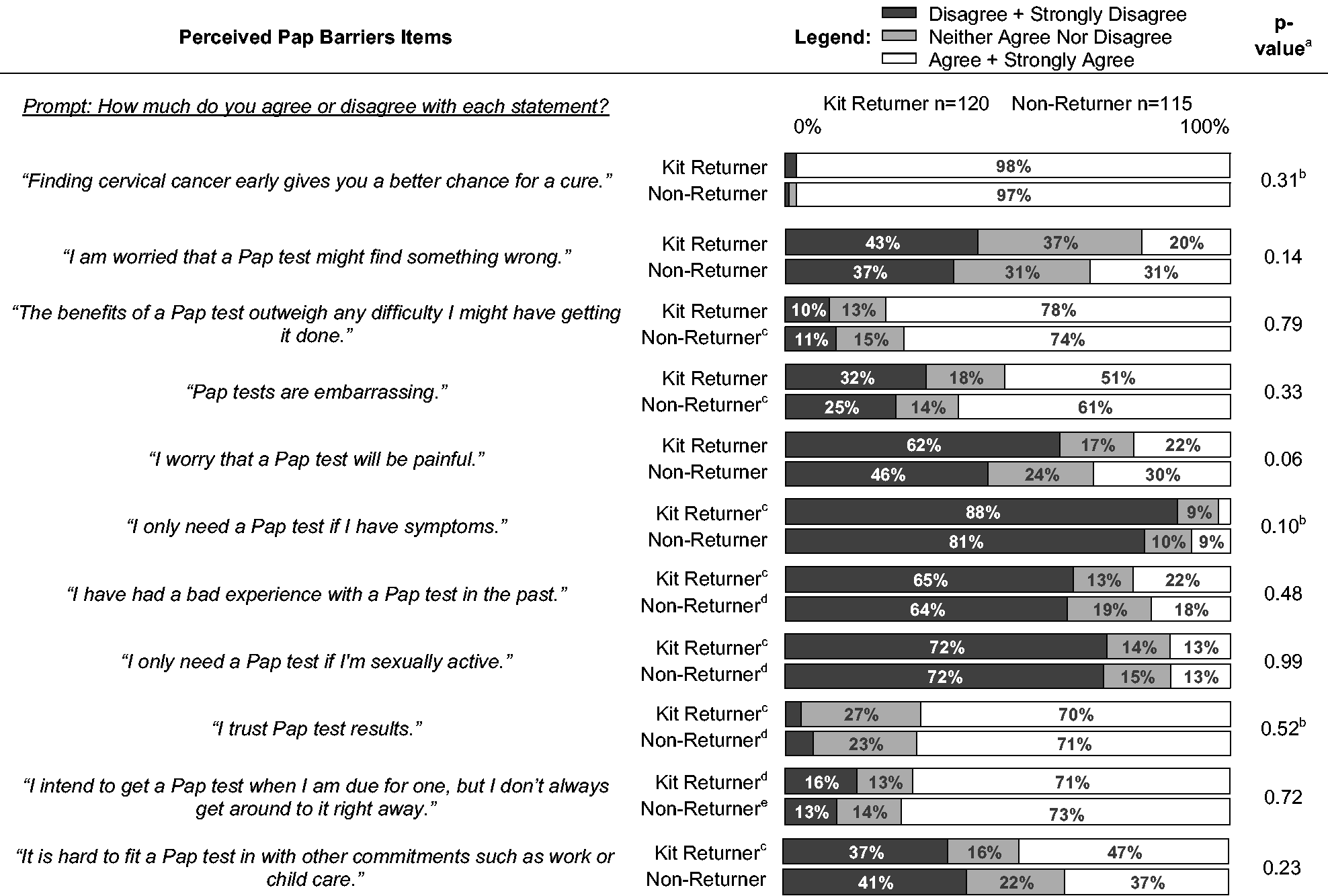
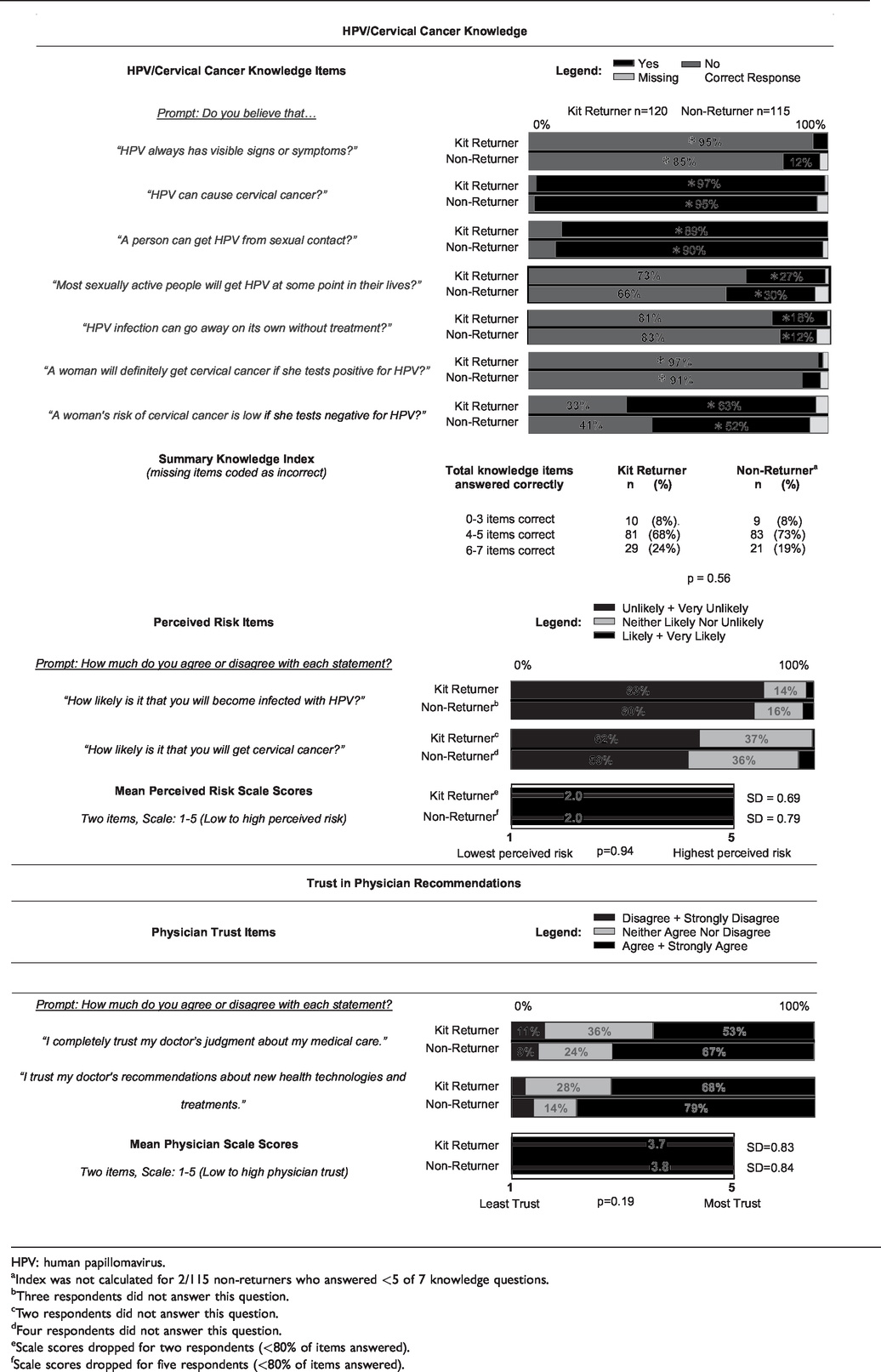
Knowledge: Knowledge scores were similar between kit returners and non-returners (Table 2). Most (89%) knew HPV is sexually transmitted and can be asymptomatic (90%), were aware of the link between HPV and cervical cancer (96%), and knew that a positive HPV test does not necessarily indicate cancer (94%). However, most women did not know HPV infection can resolve without treatment (82%), or that most sexually active women will get HPV (69%), and 37% were not aware that HPV-negative tests indicate low cervical cancer risk. Kit returners were more likely than non-returners to know that HPV infection can be asymptomatic (95% vs. 85%), and that an HPV-negative test indicates low cancer risk (63% vs. 52%). Perceived risk: Overall, respondents perceived themselves at low risk for cervical cancer, and even lower risk for future HPV infection; there was no difference by kit-return (p = 0.94) (see Table 2). Physician trust: Most women trusted their provider’s medical judgment (60%) and recommendations about new technologies (73%) (see Table 2). Kit returners were slightly less likely than non-returners to say that they trusted their doctor’s judgment (53% vs. 67%) or recommendations about new technologies and treatments (68% vs. 79%). Perceived Pap barriers: There were few differences between kit returners and non-returners. Most women believed Pap screening is needed in the absence of symptoms (85%) or sexual activity (72%), and agreed that the benefits of screening outweigh the difficulties (76%) (see Table 3). Over half (56%) found Pap tests embarrassing. Most said they intended to get a Pap test despite not always getting around to it (72%), and 42% reported difficulties fitting Pap tests into their schedule. More kit non-returners than returners worried about Pap results (31% vs. 20%), found Pap tests embarrassing (61% vs. 51%), and worried that Pap tests are painful (30% vs. 22%).
Scores and item responses for potential correlates of HPV self-sampling, by self-reported kit return.
HPV: human papillomavirus.
Index was not calculated for 2/115 non-returners who answered <5 of 7 knowledge questions.
Three respondents did not answer this question.
Two respondents did not answer this question.
Four respondents did not answer this question.
Scale scores dropped for two respondents (<80% of items answered).
Scale scores dropped for five respondents (<80% of items answered).
Perceived Pap barriers item responses, by self-reported kit return.

Chi-squared test unless noted.
Fisher’s exact test (cell size <5).
One respondent did not answer this question.
Two respondents did not answer this question.
Three respondents did not answer this question.
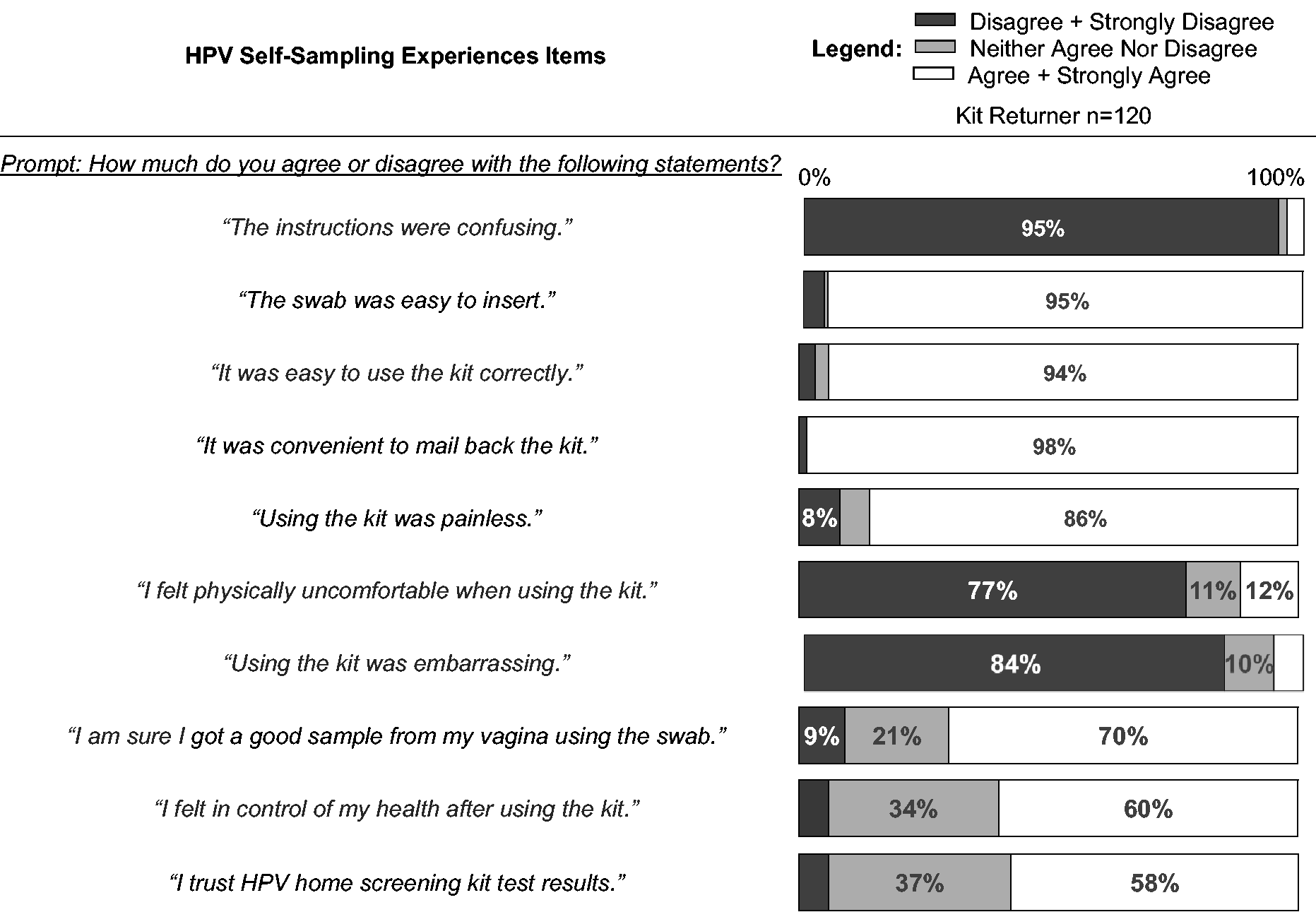
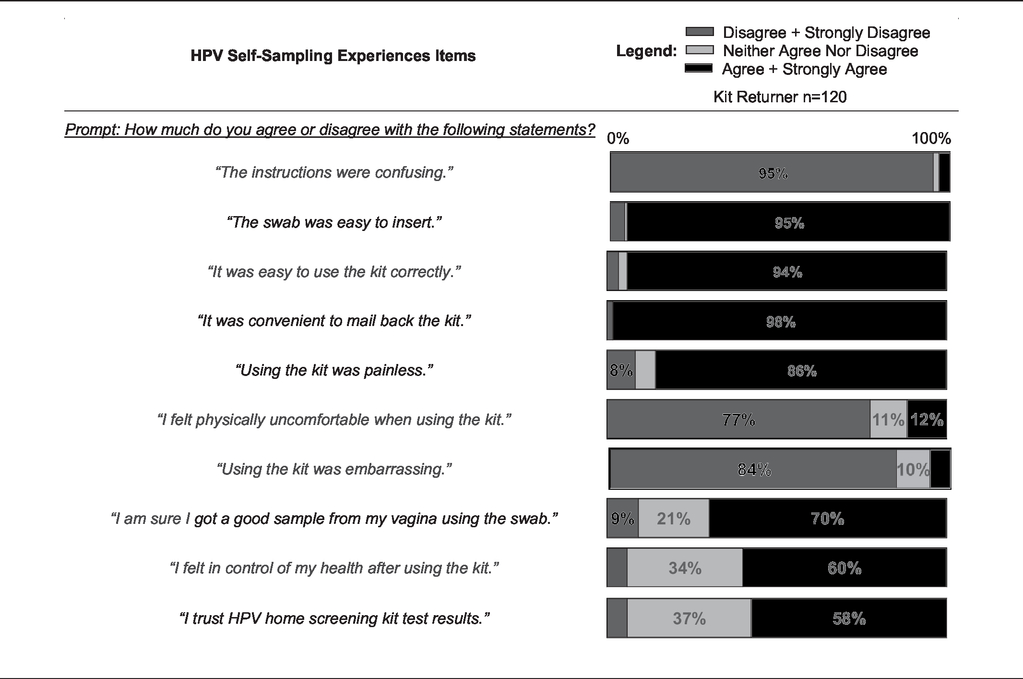
Kit returners were positive about their experiences (see Table 4). Most believed that the instructions were easy to follow (95%), the kits were easy to use (94%), convenient (98%), and painless (86%), and that swabs were easy to insert (95%). Fewer (70%) were confident they had obtained a good sample, and just over half (58%) said they trusted HPV-SS results.
Experiences with using HPV self-sampling kits.
Among non-returners, 94 (82%) said they remembered receiving a kit in the mail. Of these 94 women, 87 (93%) chose at least one of seven listed reasons for non-return and/or wrote in an open-ended response. Listed reasons included being unsure they could use the kit correctly (38%), not wanting to insert the swab (20%), being embarrassed to use the kit (14%), not trusting kit results (13%), not finding it convenient to use the kit (10%), finding the instructions confusing (9%), and fear that using the kit would be painful (9%). Additional reasons identified in open-ended responses included forgot/did not get around to it (17%), and low perceived risk due to sexual behavior (11%).
In assessing reactions to HPV-SS kits, the following items were considered. Perceived efficacy: Kit returners were significantly more confident than non-returners about HPV-SS efficacy (p < 0.01, Table 5). More than half of kit returners believed that kits effectively detected HPV infection or identified women at high risk for cervical cancer, compared with roughly 40% of non-returners. Future screening intentions: Most kit returners (89%) said that they would use a kit in the future and would recommend HPV-SS to a friend (79%) (see Table 5). Scores were higher among kit returners than among women who did not use the kit (p < 0.01), but 49% of non-returners expressed a willingness to try a kit in the future. Screening preferences: Most kit returners preferred HPV-SS to Pap tests, 66% preferred HPV-SS over Pap, and 21% stated that they liked both equally (see Table 5). Despite not having used a kit, 30% of non-returners preferred HPV-SS to Pap tests.
Scores and item responses for reactions to HPV self-sampling kits, by self-reported kit return.

HPV: human papillomavirus.a21 women who did not remember receiving a kit did not receive these questions.bThree respondents did not answer this question.cOne respondent did not answer this question.dScale scores dropped for one respondent (<80% of items answered).eScale scores dropped for three respondents (<80% of items answered).fSix respondents did not answer this question.gScale scores dropped for seven respondents (<80% of items answered).
Discussion
By surveying underscreened women who received a mailed, unsolicited HPV-SS kit in a pragmatic trial, we found that knowledge of and perceived risk for HPV and cervical cancer, physician trust, and perceived Pap screening barriers did not differ between kit returners and non-returners. Although knowledge about HPV and its association with cervical cancer was generally high, both groups showed some knowledge gaps about HPV testing and natural history. Kit users were highly positive about their experiences and would use them again, but were not as confident about trusting HPV-SS results. Non-returners lacked confidence in using HPV-SS kits and trust in the test, but half expressed willingness to use a kit in the future. To our knowledge, this is the first survey of women receiving unsolicited HPV-SS kits in a United States healthcare system.
As countries transition to primary HPV screening, HPV/cervical cancer knowledge has been identified as a crucial factor in women’s acceptance of HPV screening. 32 Women’s knowledge of the epidemiology and natural history of HPV was somewhat limited, as the majority of surveyed women did not know that most sexually active women will be infected with HPV over their lifetime, or that HPV can clear on its own. There was also a lack of clarity among surveyed women on the clinical relevance of a negative HPV test. The observed gaps in knowledge of HPV natural history and the meaning of negative HPV tests could have an adverse impact on reactions to HPV screening (including HPV-SS) in the future. Underscreened women in our study also reported low perceived risk of cervical cancer, consistent with other studies. 33 Kit returners and non-returners reported the same low level of perceived risk, suggesting that it is unlikely that perceived risk is driving choices in this population.
Surveys conducted in countries with organized screening programs found that the most powerful predictors of Pap underscreening 23 were structural/logistical factors (e.g. forgetting to make an appointment 15 and scheduling difficulties 13 ), and emotional factors (e.g. embarrassment).14,15 These factors were commonly cited by both kit returners and non-returners in our study.
Similar to other studies using convenience samples and in a trial context,12,14,15,34 kit returners were accepting of kits, found them to be convenient, and were not embarrassed to use them, suggesting that HPV-SS kits helped to address some logistical barriers to Pap screening. Physician trust was high among both kit returners and non-returners. When implementing their HPV-SS program, Australia emphasized continued clinician engagement.35,36 Our results indicate that physicians could be important as endorsers and educators in future efforts to increase HPV-SS kit uptake.
Significantly more kit returners than non-returners believed HPV-SS is efficacious. This difference could have influenced women’s decisions about using the kit. Compared with other studies of unsolicited mailed HPV-SS kits, women in our survey reported lower levels of trust and confidence. Two surveys conducted within large-scale trials of unsolicited mailed HPV-SS kits in Australia 14 and Finland 13 found that roughly 80% of kit returners believed that they had collected a sample correctly, compared with 70% in our study. The Finnish survey found 78% of kit returners trusted test results, 13 compared with 58% in our study. Nonetheless, most kit returners in our study indicated that they preferred HPV-SS to Pap screening, and would use and recommend kits in the future. Additionally, a relatively high proportion of non-returners also reported a preference for HPV-SS, and intended to self-sample in the future, indicating that many non-returners are still open to the idea of HPV-SS. Future research should focus on ways to increase women’s trust in HPV self-sampling kits.
In previous surveys of HPV-SS kit non-returners, the most common reasons for non-return were related to women’s screening eligibility (e.g. prior hysterectomy). Our use of the EHR facilitated identification and exclusion of women with recent Pap tests, hysterectomy, or pregnancy. Additional study strengths included recruiting non-returners, and asking a broad range of questions to enable robust comparisons with kit returners.
As with similar surveys,14,15 the response rate among non-returners was low (11%). We attempted to engage non-returners through mailed invitations, reminder calls, and cash incentives, but a large population of women remain unresponsive to Pap reminders, HPV-SS kits, or survey invitations. Responses from non-returners who participate in a survey may not be representative of women who are less engaged with the healthcare system. Open-ended responses revealed several additional reasons for non-return (e.g. forgetting and low perceived risk) that could be targeted in future outreach efforts. It is likely that we underestimated the frequency of these reasons by not including them as pre-specified choices. Women in our study were mostly non-Hispanic, White, and residing in urban areas. All were insured and received screening reminders. Therefore, our results cannot be generalized to all racial and ethnic groups, rural populations, or uninsured women. Individual-level data on socioeconomic barriers to screening, such as income 37 and education 38 were not available for our study. We waited six months post-randomization to mail survey invitations. This allowed us to purposively sample kit returners and non-returners, ensuring that recruitment did not interfere with the HOME trial’s primary outcome measures, and provided an adequate sample size among non-returners, but also meant subsequent (post-kit) experiences, such as receiving test results, knowledge-seeking, or inaccurate memory, could have influenced women’s responses. Our survey excluded most women with positive HPV-SS results, because they had been invited to participate in an interview to learn about their experiences. 19 Interview invitees with positive HPV-SS results were similar to kit returners invited to the survey with respect to age, race, and screening history. 19 Interviewees reported intense feelings and emotions upon receiving positive HPV-SS results. Including these women in the survey would possibly have resulted in lower overall levels of trust and confidence among kit returners, although when asked similar questions, interviewees reported similar levels of trust and preferences for HPV-SS to kit returners in our study. 19
Conclusions
Cervical cancer screening outreach efforts involving HPV-SS should emphasize the accuracy and reliability of self-collected samples, and educate women about the high screening efficacy of HPV testing. More research is needed on ways to increase trust and confidence in HPV testing and HPV-SS kits. Women’s trust in physicians suggests that physicians may play an important role in educating women and encouraging HPV-SS. The low survey response rate among kit non-returners highlights the need for continued research on new ways to engage underscreened women in screening and research. The heterogeneity in response to mailed HPV-SS kits reported in similar international trials 11 underscores the importance of research on barriers and facilitators to HPV-SS uptake. With several countries (including Australia35,36 and The Netherlands 39 ) now offering HPV-SS for underscreened women as part of their national cervical cancer screening programs, our results suggest potential targets for education and outreach.
Supplemental Material
MSC885994 Supplemetal Material - Supplemental material for Reactions of women underscreened for cervical cancer who received unsolicited human papillomavirus self-sampling kits
Supplemental material, MSC885994 Supplemetal Material for Reactions of women underscreened for cervical cancer who received unsolicited human papillomavirus self-sampling kits by Colin Malone, Jasmin A Tiro, Diana SM Buist, Tara Beatty, John Lin, Kilian Kimbel, Hongyuan Gao, Chris Thayer, Diana L Miglioretti and Rachel L Winer in Journal of Medical Screening
Footnotes
Acknowledgements
We greatly appreciate the dedicated work of Jenna Leonardo, Margie Wilcox, and Scott Caparelli at Kaiser Permanente Washington Health Research Institute for their significant contributions to administrating the web surveys.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute of the National Institutes of Health (grant number R01CA168598-01A1). Mr. Malone also received support from the National Cancer Institute [grant number R25CA092408] and from the National Center For Advancing Translational Sciences (grant number TL1 TR002318) of the National Institutes of Health. The National Institutes of Health were not involved in the study design, the collection, analysis, or interpretation of data, the writing of this report, or the decision to submit this report for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.