Abstract
Objective
To determine the occurrence of clinical peripheral artery disease in a cohort of middle-aged and older persons who, though they initially had no symptoms of peripheral artery disease, had blood pressure levels in the arteries of their ankles and feet that were at least 10% lower than those in the arteries of their arms.
Methods
We analyzed data obtained in the Multi-Ethnic Study of Atherosclerosis, in which lower and upper extremity blood pressures were assessed in over 6000 Americans aged 45–84 and the ratio of these (the ankle–brachial index) was calculated. During a median follow-up of 13 years, the incidence of symptomatic peripheral artery disease (identified through annual questionnaires, review of hospital records, and notations of peripheral artery disease found in data obtained from the Center for Medicare Services) was compared between persons whose ankle–brachial index was ⩽9 and persons with higher ankle–brachial index values.
Results
The incidence of clinical peripheral artery disease was 23.0 per 1000 person-years among the 172 participants whose baseline ankle–brachial index was <0.9, compared with 2.0 per 1000 person-years in those with a higher ratio. The incidence of clinical peripheral artery disease rose steadily with decreasing ankle–brachial index below 0.9. The excess risk associated with a low ankle–brachial index was present in persons with and without the other measured risk factors for peripheral artery disease (cigarette smoking, hyperten sion, diabetes, and obesity).
Conclusions
Even in persons asymptomatic for peripheral artery disease, those with a low ankle–brachial index are at an appreciable risk of the development of manifestations of peripheral arterial insufficiency.
Introduction
One of the manifestations of peripheral arteriosclerosis is insufficient arterial flow to the lower extremities. Known as peripheral artery disease (PAD), this reduced flow can be asymptomatic, but in its more severe form can lead to exertional leg pain, leg pain at rest, and gangrene. Ascertainment of the presence of reduced arterial flow to the legs can be readily accomplished by determining the degree to which blood pressure in the feet and ankles is lower than that in the upper extremities. The disparity between lower and upper extremity blood pressure is commonly described as a ratio, the ankle–brachial index (ABI), with values <0.9 suggesting the presence of circulatory impairment.
A low ABI predicts (to some extent) the probability of the development of clinical heart or cerebrovascular disease, beyond the predictive ability of the cardiovascular disease risk factors with which a low ABI is associated (e.g. cigarette smoking, diabetes).
1
The measurement of the ABI of persons with no symptoms of PAD as a means of identifying those at high risk for interventions to prevent arteriosclerotic heart and cerebrovascular disease (interventions such as smoking cessation, or blood pressure and serum lipid control) has been considered for broad-scale use, but at present is not widely advocated.
2
Bearing on the decision to obtain a screening ABI would be the impact of such screening (and the treatment to which it might lead) on the development of the symptomatic manifestations of PAD, but this has never been evaluated in a formal comparison of screened and unscreened persons. In the absence of this sort of direct comparison, data that bear on the magnitude of the efficacy of screening can be obtained by documenting:
The proportion of asymptomatic individuals who test positive for a low ABI; The likelihood that test positivity predicts adverse clinical outcomes, in this case the development of leg pain on exertion, rest pain, and/or gangrene; and The degree to which the likelihood of progression to clinical outcomes differs according to the presence of exposures and characteristics that influence the incidence of PAD, such as cigarette smoking and diabetes.
The present analysis addresses these questions using the experience of persons enrolled in the Multi-Ethnic Study of Atherosclerosis (MESA). We seek to provide additional information relevant to the decision to screen middle-aged and elderly persons by means of the ABI.
Methods
The MESA takes place in six locations of the United States: Baltimore city and Baltimore County, Maryland; Chicago, Illinois; Forsyth County, North Carolina; Los Angeles County, California; Northern Manhattan and the Bronx, New York; and St. Paul, Minnesota. From July 2000 to August 2002, the participating institutions recruited residents of these communities aged 45–84 who were white, African-American, Hispanic, or Chinese. All participants were free of clinically apparent cardiovascular disease (physician diagnosed heart attack, angina, stroke, transient ischemic attack or heart failure, current atrial fibrillation, taking nitroglycerin, or having undergone angioplasty, coronary artery bypass graft, valve replacement, pacemaker or defibrillator implantation, or any surgery on the heart or arteries). Other exclusions included pregnancy, active treatment for cancer, weight >300 lb, cognitive inability as judged by the interviewer, living in a nursing home or on the waiting list for a nursing home, plans to leave the community within five years, language barrier (those who speak other than English, Spanish, Cantonese, or Mandarin), CT scan of the chest within past year, or any serious medical condition that would prevent long-term participation. Each site recruited an approximately equal number of men and women, according to prespecified age and race/ethnicity proportions. All participants gave informed consent, and the study protocol was approved by the institutional review board at each study site. Details regarding the means of recruitment have previously been published. 3
At the time of enrollment in the study, a baseline exam was performed, which included the assessment of each participant’s ABI, anthropometric characteristics, and other exposures and characteristics potentially relevant to the occurrence of cardiovascular disease (e.g. smoking). At approximately two-year intervals, four additional exams have taken place, with a reassessment of ABI at Exam 3 (2004–2005) and Exam 5 (2010–2012). Using a handheld Doppler instrument with a 5 mHz probe, systolic blood pressure was measured in both brachial arteries, as well as in both dorsalis pedis and posterior tibial arteries. The ABI was calculated for left and right sides separately as the maximum systolic pressure in the dorsalis pedis and posterior tibial artery divided by the average of the left and right brachial systolic pressures. If the right and left brachial pressures differed by more than 10 mmHg, the higher value was used in the ABI calculation. For purposes of our analyses, we characterized each participant by the lowest of the two calculated ABIs. A subset of 384 MESA participants had replicate ABI measurements that showed excellent reproducibility. The overall intraclass correlation coefficient (ICC) from these measurements was 0.932, while the intra-technician ICC was 0.953, and the inter-technician ICC was 0.918.
Information on demographics, smoking, and medications were collected via questionnaire at the initial examination. Height and weight were measured with participants wearing light clothing and no shoes. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. Resting blood pressure was measured three times in the seated position, and the average of the last two measurements was used in analysis. Hypertension was defined as diastolic blood pressure >=90 mmHg, systolic blood pressure >=140 mmHg, or use of antihypertensive medications. Diabetes was defined as fasting glucose >125 mg/dl or taking diabetes medication. Current smoking was defined as answering yes to the question “Have you smoked cigarettes during the last 30 days?” For those answering no, former smoking was defined as answering yes to the question “Have you smoked at least 100 cigarettes in your lifetime?”
At intervals of 9–12 months, a telephone interviewer sought to contact each participant to obtain information regarding interim hospital admissions and outpatient diagnoses of (or procedures performed for) cardiovascular disease. Relevant to the present analysis, cohort members specifically were asked if a doctor or healthcare professional had told them that they had PAD, intermittent claudication, or leg pain from a blockage of the arteries. In addition, for cohort members aged 65 and over, data from the Center for Medicare Services (CMS) were reviewed for a diagnosis of PAD. Finally, MESA personnel obtained and abstracted records of hospitalizations that possibly or probably resulted from cardiovascular events. Two study physicians reviewed this information and adjudicated diagnoses of cardiovascular disease and dates of onset.
For purposes of the present investigation, clinical PAD was defined as: (1) A diagnosis made by the physician reviewers as “definite” or “probable” PAD. At a minimum, this required the presence of appropriate symptomatology; or (2) a report during a telephone follow-up interview that a physician or other health professional had told the participant that he/she had PAD, intermittent claudication, or leg pain from a blockage of the arteries; or (3) a CMS notation of PAD (code 443.9).
Because of our interest in the incidence of clinical PAD in persons eligible to be screened using a measurement of his/her ABI, in the primary analysis we sought to restrict the analysis to participants: (1) who had not previously been diagnosed as having PAD and (2) who had no evidence of as-yet-undiagnosed PAD at the time of the ABI assessment. At baseline, participants were asked the question, “Do you ever get pain in either leg or buttock while walking?” If they responded affirmatively, they were then asked “Does this pain ever begin when you are standing still or sitting?” We excluded persons who answered “yes” to the first question and “no” to the second, since this pattern was most compatible with the presence of intermittent claudication. We acknowledge that some persons who were excluded may well have had a nonarterial basis for their symptoms, and that a very small number who had rest pain from arterial insufficiency were incorrectly included. Nonetheless, we believe that the experience of the remaining participants should provide a reasonably accurate basis for gauging the later incidence of clinical PAD in persons with no current symptoms of PAD.
We measured the incidence of clinical PAD over a median follow-up of 13 years in groups of participants defined by the presence or absence of a low ABI at baseline, and also within categories of severity of diminished ABI (0.80–0.89, 0.70–0.79, <0.70). Kaplan–Meier estimates of the five-year cumulative incidence of clinical PVD are reported, along with incidence rates per 1000 person-years at risk that accrued during the whole of the follow-up period. We used a linear regression model to explore the adjusted associations between abnormal ABI at baseline and incident PAD risk on an absolute (additive) risk scale. This model 4 considers the event indicator (incident PAD yes/no) divided by the square root of the time to event (or censoring) in years as the endpoint, each predictor multiplied by the square root of time, and an additional term for the square root of follow-up time itself. No intercept is included in the model, and robust standard errors are used. The beta coefficients in the model are interpreted as the absolute annual increment in risk associated with a one unit increment in each predictor.
In addition, we constructed a risk model for incident PAD using known risk factors (age, gender, BMI, current smoking, diabetes, hypertension) and ABI, the latter represented in five categories to capture the nonlinear relation between ABI and risk. To evaluate discrimination we calculated Harrell’s C statistic, 5 and to assess calibration we graphically compared observed with predicted risk by decile of risk.
Results
From the 6814 participants who completed the baseline MESA exam, we excluded 644 who reported exertional lower extremity pain. Also excluded were 149 whose medical records or CMS files indicated the presence of PAD prior to the baseline exam, 79 who did not complete the baseline ABI measurement, and 27 for whom there was no follow-up information. (The above categories are not mutually exclusive.) This left 5953 participants who comprised our analytic sample.
There were 168 participants who met at least one of our criteria for the development of clinical PAD during follow-up. Of these, the majority (119/168 = 71%) were identified by means of annual phone contact, at which time they indicated that their healthcare provider had told them that they had this condition. For 38 of these 119 cases, there was additional evidence of PAD found during adjudication of records of hospitalized patients or from diagnoses made in CMS records.
An ABI <0.9 was found in 172 (2.9%) persons in the analytic sample. Most (91) had an ABI between 0.80 and 0.89; 43 and 38 persons, respectively, had an ABI between 0.70–0.79 and <0.70. Of the 168 persons who went on to develop clinical PAD, 32 had an ABI <0.9 (sensitivity = 19.0%). Of the 5785 who did not have clinical evidence of PAD during follow-up, 5645 had an ABI of 0.9 or greater (false positive proportion = 2.4%). For an ABI cutoff of 0.8, the corresponding sensitivity was 23/168 = 13.7%, and the corresponding false positive proportion was 58/5785 = 1.0%.
The incidence of clinical PAD in persons with an ABI <0.9 was 23.0 per 1000 person-years, 11.5 times the rate of 2.0 per 1000 person-years in participants whose ABI exceeded this value (Table 1). The size of the increased risk rose as the measured ABI fell (see Figure 1). As of the end of the first five years of follow-up, the cumulative incidence of clinical PAD was approximately 40% in persons whose ABI was <0.70, 25% in those whose ABI was between 0.7 and 0.79, and 10% in those whose ABI was between 0.80 and 0.89. MESA participants with a low ABI had a higher prevalence of risk factors for PAD than did other participants, but even after adjusting for these disparities, the difference in the rate of clinical PAD between persons with and without an ABI < 0.9 was 19.7 per 1000 person-years (95% confidence interval = 11.4–28.0 per 1000 person-years).
ABI at baseline in relation to the occurrence of subsequent clinical PAD.
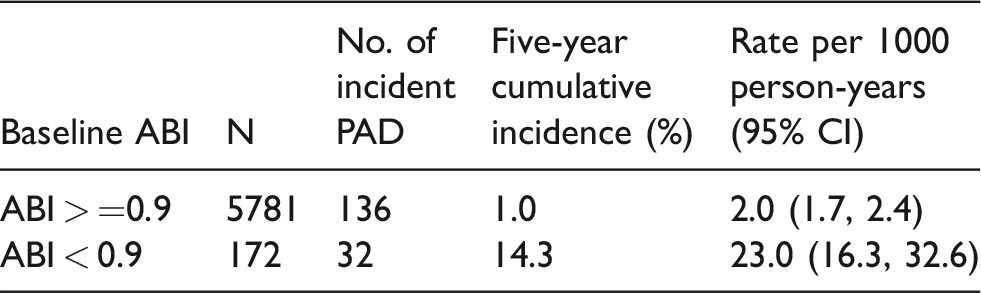
ABI: ankle–brachial index; CI: confidence interval; PAD: peripheral artery disease.
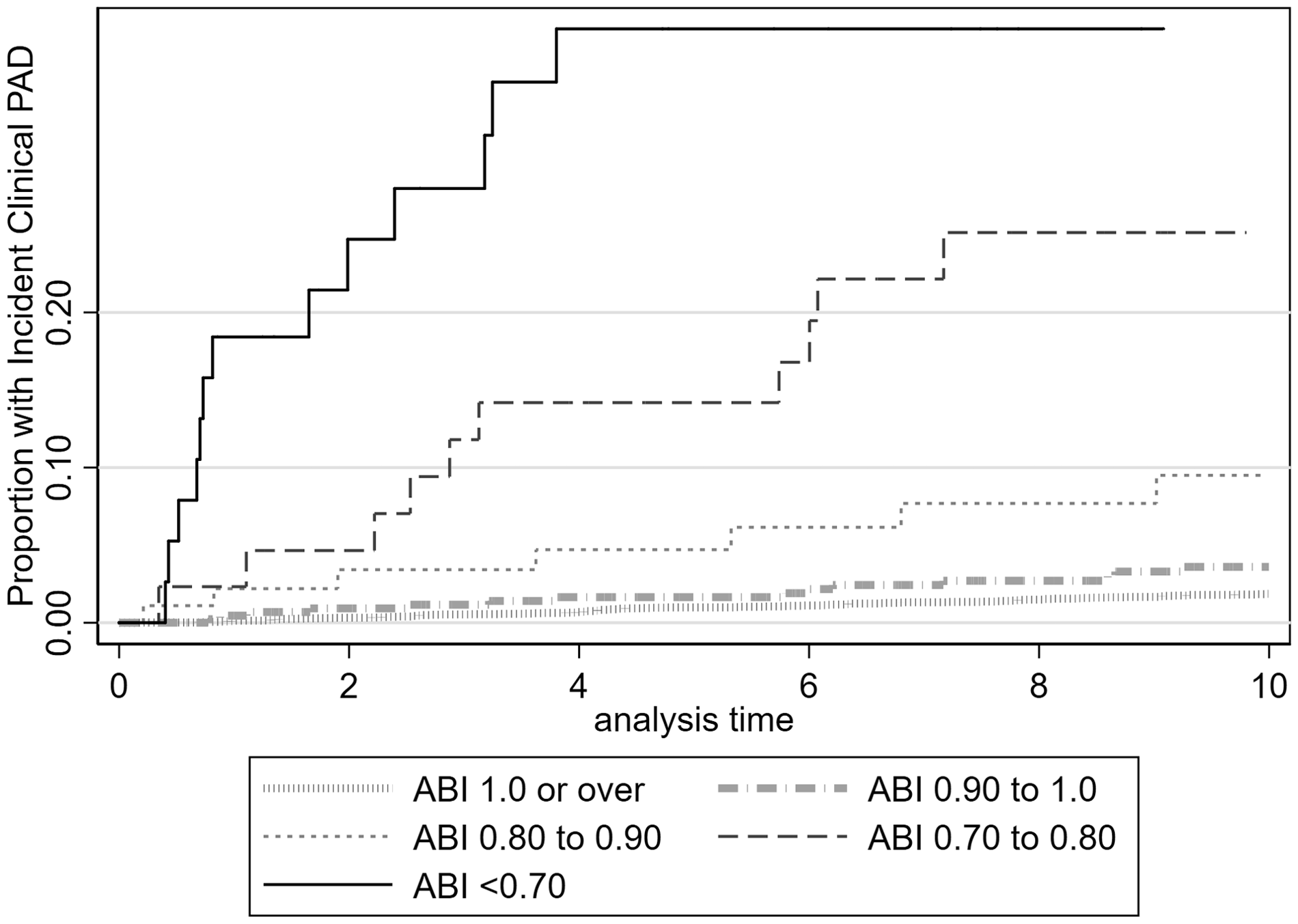
Cumulative incidence of clinical PAD over time in relation to baseline ABI. ABI: ankle–brachial index; PAD: peripheral artery disease.
Beyond a low ABI, the strongest predictors of the development of clinical PAD are cigarette smoking, hypertension, diabetes, and a BMI of at least 30 kg/m2. There was a substantial difference in the rate of PAD associated with an ABI <0.9 both in smokers and nonsmokers, hypertensive and normotensive individuals, diabetics and nondiabetics, and in obese and nonobese persons (Table 2).
Difference in the rate of PAD associated with an ABI < 0.9 within subsets of participants defined by other risk factors.

ABI: ankle–brachial index; CI: confidence interval; PAD: peripheral artery disease.
aStratified models adjusted for age, gender, race/ethnicity, and the other three risk factors shown in the table.
Figure 2 plots the observed versus the predicted 10-year risk of PAD, according to the level of predicted risk. There were 153 participants with a predicted 10-year risk of more than 10%; all of them had an ABI <0.9 at baseline. The observed incidence among these participants was 10%. Harrell’s C statistic for a model with (0.734) and without (0.707) baseline ABI indicates that ABI is an important contributor to this prediction.
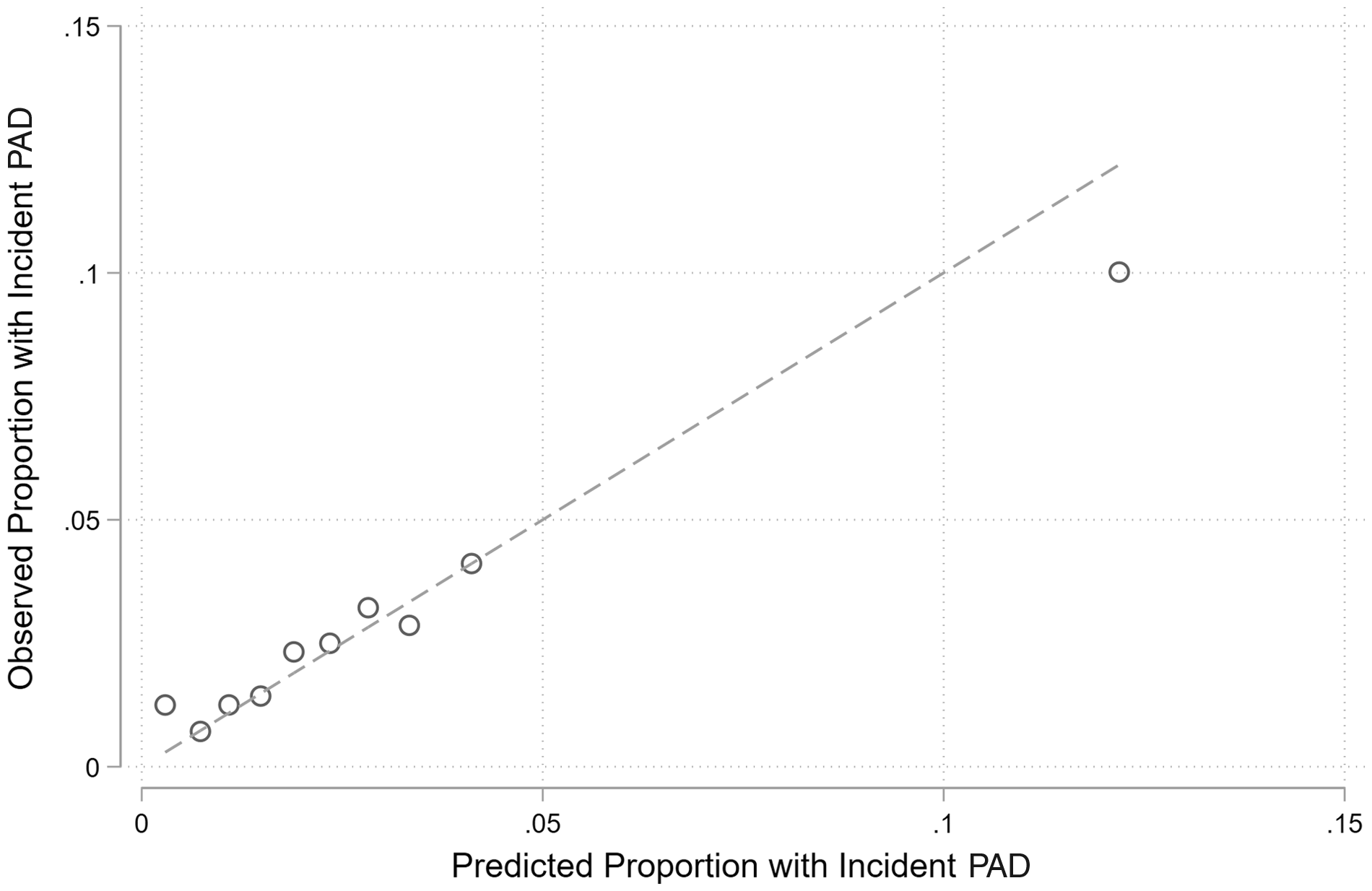
Observed versus predicted 10-year risk of incident PAD by decile of predicted risk.PAD: peripheral artery disease.
Discussion
This analysis was limited by the imperfect accuracy of the means by which the development of clinical PAD was ascertained. To identify events that occurred during follow-up, we chose not to use participant reports of symptoms compatible with the presence of intermittent claudication. We felt that while most persons who genuinely had intermittent claudication would indicate the presence of the symptoms, many persons with a nonarterial basis for them (e.g. arthritic pain) would do so as well. Because of this concern, we relied on the presence of a clinical diagnosis of PAD during follow-up. The diagnosis could have been identified in CMS files, made by MESA adjudicators when reviewing data on hospitalized MESA participants, or told to the participant by his/her physician and then reported by that participant during an annual follow-up questionnaire. However, because the results of the baseline ABI had been provided to the same physicians, it could be that the PAD diagnosis that was told to some of the patients by their physician was based not on the occurrence of symptoms of PAD, but only on the MESA-reported abnormal ABI. This would lead to a falsely high estimate of the occurrence of clinical PAD in persons with a low baseline ABI. However, it appears that this potential bias did not materialize to any appreciable extent. First, the fraction of persons with a low ABI whose study diagnosis of PAD was based solely on telephone follow-up (11/32 = 34.4%) actually was lower than the fraction among persons with a normal ABI (70/136 = 51.5%). Second, there was a large gradient of reported PAD among MESA participants with a low ABI, with the highest incidence seen in those with the lowest ABI values. Thus, it is likely that a large part of the association observed between a low ABI and the development of clinical PAD is genuine. This belief is reinforced by the results of an additional analysis, in which persons whose only indication of clinical PAD had been a follow-up questionnaire mention of a physician diagnosis were required also to have ambulatory leg pain before being categorized as having developed clinical PAD. In that analysis, the rates of PAD among persons with a baseline ABI <0.9 and >=0.9 were 18.0 per 1000 person-years and 1.3 per 1000 person-years, respectively. The difference between these (16.7 per 1000 person-years) is only slightly smaller than that observed using more liberal criteria for the development of clinical PAD (19.7 per 1000 person-years).
To our knowledge, there has not previously been an examination of the incidence of clinical PAD in relation to ABI among asymptomatic individuals. Our results suggest that: (a) an abnormally low ABI (<0.9) is present in about 3% of middle-aged and older Americans who do not have a prior diagnosis of PAD and have no symptoms of this condition (and also do not have another clinically manifest form of cardiovascular disease); and (b) in these persons, a low ABI predicts a sharply increased incidence of clinical PAD, both in relative and absolute terms, during the ensuing decade, particularly in persons whose ABI is extremely low. But could knowledge of a person’s low ABI lead to an improved prognosis vis-à-vis the health of his/her lower extremities? There are no data directly bearing on this question. Based on the results of studies of the treatment of patients with symptomatic PAD, there are reasons to believe that most currently available medications would not be an effective form of treatment. 6 In contrast, a supervised exercise program; 7 initiation of statin therapy, 8 together with the PCSK9 inhibitor, evolocumab; 9 and for cigarette smokers, a smoking cessation intervention10,11 might be helpful.
It could be argued that because, in middle-aged and older adults, exercise and smoking cessation are desirable to promote health irrespective of ABI, results of a person’s ABI screening exam ought to be of little importance. However, it is plausible (but not yet evaluated) that in persons who are sedentary or who smoke cigarettes, knowledge of their diminished peripheral arterial circulation, and the high risk of lower extremity symptomatology associated with this, would make them particularly receptive to exercise and/or smoking cessation interventions. Nonetheless, the impact of knowledge of an abnormal screening ABI on an individual’s adoption of behaviors relevant to arterial pathology (of the lower extremity and other parts of the body) would need to be assessed before a scientifically based recommendation for or against ABI screening could be made.
Conclusion
Persons with a low ABI who are free of symptoms of PAD have an appreciable risk of the development of manifestations of peripheral arterial insufficiency.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a contract from the National Heart, Lung, and Blood Institute – HHSN2 6820150003I.
