Abstract
Objectives
To investigate the risk of malignancy following stereotactic breast biopsy of calcifications classified as Breast Imaging Reporting and Data System (BI-RADS) 3, 4, and 5.
Methods
The study included women with pure calcifications (not associated with masses or architectural distortions) who underwent stereotactic breast biopsy at the Dutch Cancer Institute between January 2011 and October 2013. Suspicious calcifications (Breast Imaging Reporting and Data System 3, 4, or 5) detected on mammography were biopsied. All lesions were assessed by breast radiologists and classified according to the BI-RADS lexicon.
Results
Overall, 473 patients underwent 497 stereotactic breast biopsies. Sixty-six percent (326/497) of calcifications were classified B4, 30% (148/497) B3, and 4% (23/497) B5. Of the 226 (45%) malignant lesions, there were 182 pure ductal carcinoma in situ, 22 mixed ductal carcinoma in situ and invasive carcinomas (ductal or lobular), 21 pure invasive carcinomas, and one angiosarcoma. Malignancy was found in 32% (95% confidence interval [CI] 0.24 to 0.39) of B3, 49% (95% CI 0.43 to 0.54) of B4, and 83% (95% CI 0.61 to 0.95) of B5 calcifications.
Conclusions
Considering the high predictive value for malignancy in B3 calcifications, we propose that these lesions should be classified as suspicious (B4), especially in a screening setting.
Introduction
Breast cancer is the most common malignant disease and cause of death among women (522,000 deaths in 2012). 1 Despite increasing incidence, 1 survival has improved in the last two decades,1,2 with rates of up to 80% reported in Western Europe, North America, Japan, and Australia, due to a combination of more effective treatments, better supportive care, and earlier detection. 3
Mammography is currently the best imaging tool for early detection of breast cancer in women with average lifetime risk of developing this disease. The effectiveness of mammographic screening in reducing breast cancer mortality has been proven in many randomized trials.4–6 Before mammographic screening, ductal carcinoma in situ (DCIS) represented only 2.1% of all treated breast carcinomas. 7 Now, 20% of breast cancers are diagnosed in the pre-invasive stage.8,9
BI-RADS classification system. a

BI-RADS: Breast Imaging Reporting and Data System.
Excluding lesions that need additional imaging investigation (B0) and those with known biopsy proven malignancy (B6).
The increased detectability of calcifications observed in digital technique makes it essential that these lesions can be reliably classified as either benign or malignant, to avoid unnecessary biopsies and unnecessary treatment. However, it has been difficult for radiologists to properly differentiate between benign and malignant calcifications, especially those classified as B3.14–18 Even the use of additional magnetic resonance imaging tools (e.g. 3T imaging, kinetic analysis, diffusion-weighted imaging) has not resulted in higher sensitivity in classifying suspicious breast calcifications.19,20 Image-detected calcifications must have an appropriate risk stratification, to prevent both malignancy detection failure and unnecessary treatment delay. Such accurate classification is unlikely to be achieved in day-to-day clinical practice, as it depends on many aspects, including the observer interpretation.18,21–24
The study aims to investigate the risk of malignancy following 497 stereotactic biopsies of pure calcifications (not associated with masses or architectural distortions) classified as BI-RADS 3, 4, and 5 at the Dutch Cancer Institute.
Methods
All women who underwent stereotactic breast biopsy procedures at the Dutch Cancer Institute between January 2011 and October 2013 were included in the study. Only suspicious calcifications detected on mammography were biopsied. According to the Dutch Cancer Institute policy, calcifications are considered suspicious when classified as B3, B4, or B5. This policy differs from the original BI-RADS lexicon guidelines in which only calcifications classified as B4 or B5 are considered suspicious and should receive a biopsy; calcifications classified as B3 are considered probably benign and should undergo a short-term follow-up with patients returning after 6 months for additional imaging analysis. If the lesion remains stable, a second 6-month follow-up is indicated. After that, a third 12-month follow-up is made and, assuming stability, the patient may return to the biannual screening program. However, since 2010 the Dutch National Breast screening program recommends that calcifications that originally would be classified as B3 should be immediately reclassified as B4 and undergo further investigation. This procedure was adopted because, in the Netherlands, mammographic screening only happens once every 2 years and it is, therefore, not possible to follow-up these B3 patients once every 6 months. Consequently, in the screening setting, all B3 calcifications are immediately considered suspicious and reclassified as B4, and are referred for further investigation in the clinical setting. During the investigation in the clinical setting, the medical assistant may decide to follow-up these lesions, and ask for additional imaging investigation (such as ultrasound) or a direct biopsy of the lesion. At the Dutch Cancer Institute (clinical assessment setting and not screening), the policy is to consider all B3 calcifications as suspicious and submit these lesions to biopsy (no follow-up policy is applied in these cases).
Patients with calcifications associated with masses or architectural distortion were excluded from this study; only pure calcifications were included. Patients with calcifications not identified in stereotactic biopsy were also excluded (these patients underwent different diagnostic procedures for further investigation). All stereotactic biopsies were performed by a breast radiologist using a 9-gauge vacuum-assisted device under local anesthesia. On average, four to eight samples were obtained for each biopsy.
Digital mammographic examinations were performed with a LORAD Selenia (Hologic, Bedford, MA, USA) full-field digital mammography unit. Standard craniocaudal and mediolateral oblique mammograms as well as magnification views were obtained for all patients.
All lesions were assessed in routine care by dedicated breast radiologists and classified according to the BI-RADS lexicon, 10 which proposes that calcifications should be classified according to their probability of malignancy (Table 1). According to the BI-RADS, round and punctate calcifications when in isolated clusters are classified as B3, with a positive predictive value (PPV) between 0 and 2%. Punctate calcifications with segmental and linear distribution are classified as B4a (PPV approximately between 2% and 10%), coarse heterogeneous and amorphous as B4b (PPV between 10% and 50%), fine pleomorphic as B4c (PPV approximately between 50% and 95%), and fine linear or linear branching as B5 (PPV above 95%). In our study, all calcifications were classified according to these BI-RADS guidelines. The mammographic lesions were assessed by 11 breast radiologists.
Histopathological findings as described in the original reports were considered the standard of reference. Calcifications associated with DCIS and (or) invasive cancer were considered malignant. Lobular carcinoma in situ, classical type, was considered benign, once this would not have any treatment implications. The remaining calcifications that showed no signs of malignancy in histopathological analysis were also considered benign.
This retrospective study was approved by the Institutional Review Board of the Dutch Cancer Institute. Neither informed consent nor patient approval was required for the review of medical records (protocol no. 6713).
Results
Between January 2011 and October 2013, 496 women underwent 520 stereotactic breast biopsy procedures for calcifications classified as BI-RADS 3, 4, or 5. We excluded six patients with calcifications not identified in the stereotactic biopsy specimens, and 17 patients with calcifications associated with masses or architectural distortion. This research included 497 pure calcifications from 473 patients. In 24 patients, two or more foci of calcifications were found on mammography; 13 patients presented with bilateral lesions, and 11 with two or more ipsilateral lesions. The mean age of included patients was 55 (range 32–74).
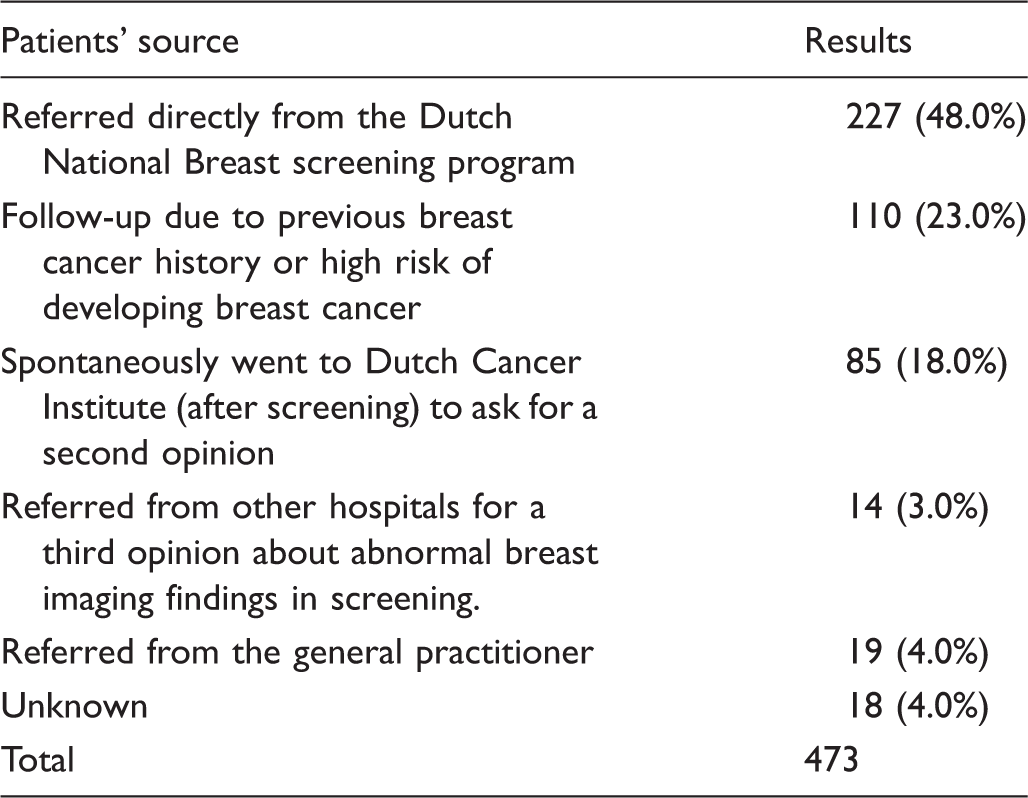
Patients referred to the Dutch Cancer Institute due to calcifications found in mammography (n = 473).
Histopathological results of suspicious calcifications conform BI-RADS classification (n = 497).
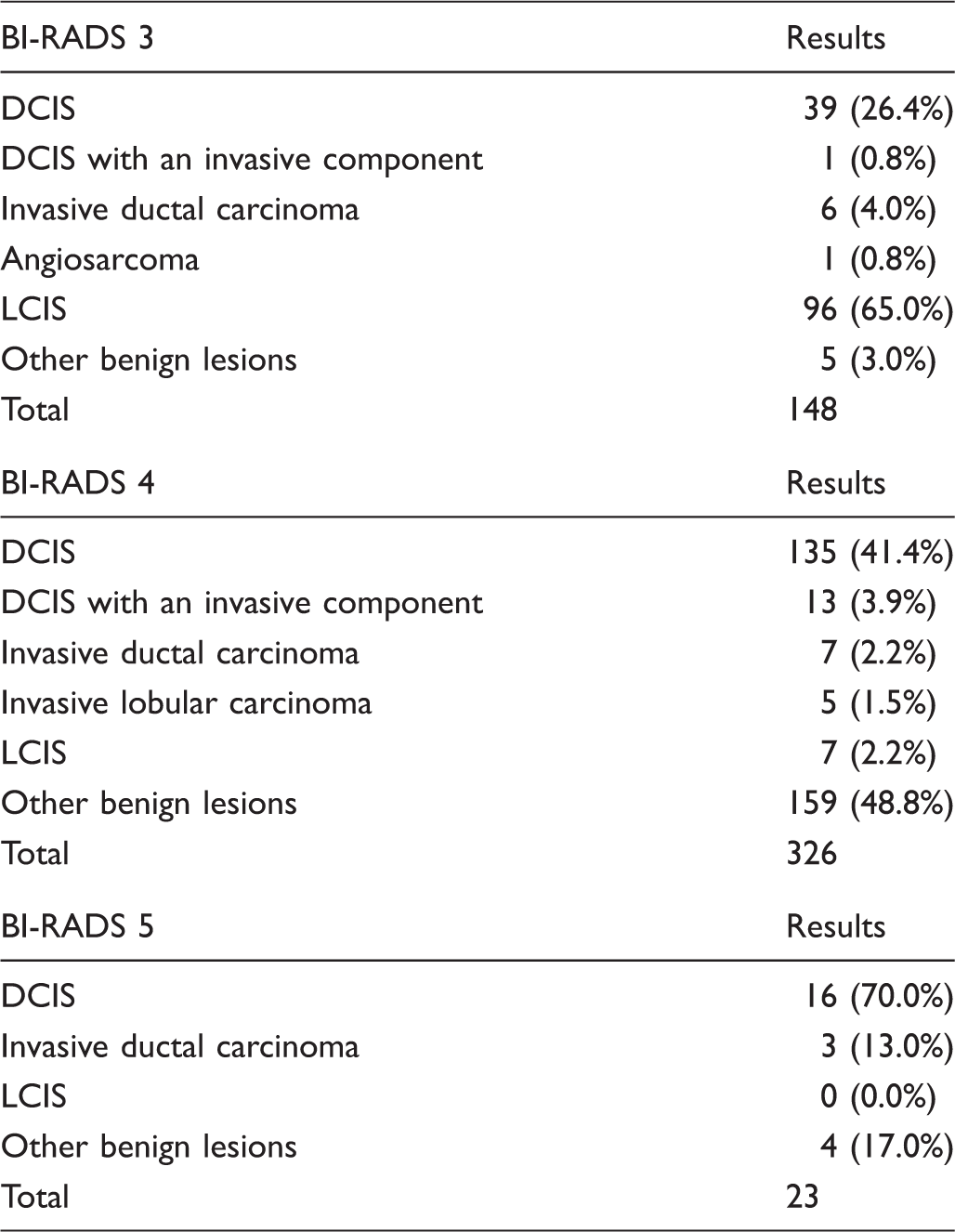
BI-RADS: Breast Imaging Reporting and Data System; DCIS: ductal carcinoma in situ; LCIS: Lobular carcinoma in situ.
Of the breast lesions in patients referred directly from the Dutch National Breast screening program, 45% (108/241; 95% CI 0.38 to 0.51) were malignant; 51% (130/256; 95% CI 0.44 to 0.57) of the breast lesions from the remaining group showed malignancy. The incidence of malignancy in BI-RADS groups 3, 4, or 5 did not vary significantly according to the type of referral.
Of the 497 calcifications, 30% (148) were classified as B3, 66 % (326) as B4, and 4% (23) as B5. Malignancy was found in 32% (47/148; 95% CI 0.24 to 0.39) of B3, 49% (160/326; 95% CI 0.43 to 0.54) of B4, and 83% (19/23; CI 0.61 to 0.95) of B5 calcifications. Invasive cancer was found in 17% (8/47) of B3, 16% (25/160) of B4, and in 16% (3/19) of B5 calcifications. Figure 1 shows examples of calcifications classified as B3 that revealed malignancy after histopathological analysis.
Regional punctate calcifications classified as BI-RADS 3. The final histopathological analysis showed a DCIS grade 3 (a) and a DCIS grade 2 (b). BI-RADS: Breast Imaging Reporting and Data System; DCIS: ductal carcinoma in situ.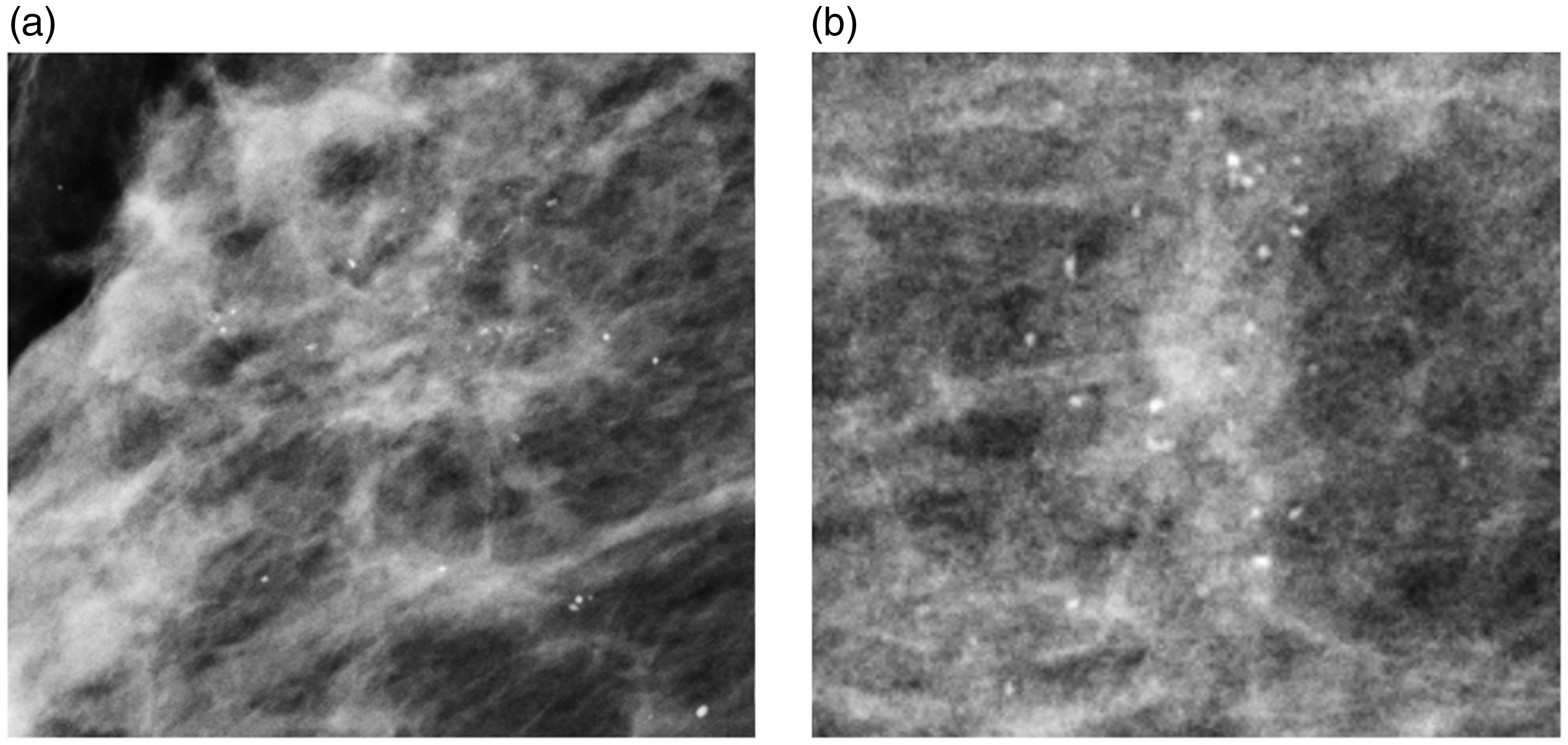
Discussion
In this study, we divided our sample into two groups: patients from the Dutch National Breast screening program, and patients with calcifications detected in a clinical setting (outside the screening program). The PPV for malignancy in B3 lesions was similar in both groups.
For B4 lesions, the PPV was as expected. According to the BI-RADS lexicon, a B4 abnormality can vary from >2% to an almost 95% likelihood of malignancy, 10 and B5 calcifications have a probability of malignancy higher than 95%. The PPV among our patients with B5 lesions was high (83%), but still slightly lower than the expected 95% rate. 10 This inconsistency may be due to the small number of patients included in this category.
Calcifications classified as B3 present a real challenge in clinical practice. The BI-RADS lexicon reports that the probability of malignancy of a B3 lesion is less than 2% and a follow-up mammography after 6 months is advised. 10 However, we found a remarkably high PPV of malignancy among B3 calcifications. Similar results to ours have been previously described in everyday clinical practice.14–17,25 In an evaluation of needle core biopsy results of patients from the German mammography screening with breast lesions classified as B3, Hoffmann et al. 15 found a PPV of 24% for B3 calcifications and recommended that all B3 lesions should undergo biopsy investigation. Other studies showed high incidence of malignancy among B3 calcifications. Kraal et al. 16 correlated the results of 236 stereotactic biopsies of pure breast calcifications with the initial BI-RADS classification and found a PPV of 16% in lesions originally classified as B3. Craft et al. 14 observed the outcome of 235 women attending a breast screening program who were recalled for assessment of calcifications, and examined the incidence of breast carcinoma detected during the following 5 years. Among B3 calcifications, an incidence of 12.7% of malignancy was observed. Kettritz et al. 17 performed 78 stereotactic vacuum-assisted breast biopsies of calcifications classified as B3, and found that 19% of these lesions were malignant.
Mammographic screening is recommended in The Netherlands once every 2 years, and in the United Kingdom once every 3 years. Since its first edition, the BI-RADS recommends that if a B3 lesion is found on mammography, patients should return after 6 months for additional imaging analysis. If the lesion remains stable, a second 6-month follow-up is indicated. After that, a third 12-month follow-up is made and, assuming stability, the patient may return to the biannual screening program. In clinical practice, the follow-up of B3 lesions depends on the preference of the assistant physician, the availability of biopsy procedures, and the patient’s wishes. 26 This policy is difficult to put into practice, especially in countries with large population screening programs. In the context of public health care, the financial and planning costs might be an obstacle to implement such a short-term follow-up. In the Netherlands, approximately 1 million women join the breast screening program every year, and in Australia and England 1.5 million 27 and 2 million, 28 respectively. Between 1% and 15% of all patients attending breast cancer screening programs worldwide are recalled for further assessment,29–32 and pure calcifications classified as B3 account for approximately 8% of all patient recalls.15,27,33–36
In light of these figures, the Dutch National Breast screening program has abandoned the use of B3 for calcifications; instead, B3 lesions are directly classified as B4 and all of them should undergo further investigation.28,37,38 The most recent edition of the BI-RADS suggests avoiding the use of the B3 category in large population screening programs. 10
In the context of private medicine, a short-term follow-up for B3 lesions could be affordable. Nevertheless, previous studies have documented a low compliance to the recommended follow-up.39,40 Helvie et al. 40 found compliance rates for low-suspicion lesions of 60% at 2 years and only 47% at 3 years. 40 In a clinical trial studying B3 lesions, Baum et al. 39 found that only 71% of the participants returned within the first 10 months for follow-up.
Moreover, the interobserver variability among breast radiologists has to be considered, as morphology and distribution of calcifications are the most important features to define the final BI-RADS classification. Lazarus et al. 22 found excellent agreement among breast radiologists when assessing the presence of calcifications on mammography (κ = 0.94), but agreement concerning morphology and distribution was only fair (κ = 0.32). Gulsun et al. 23 found κ = 0.31 for morphology and κ = 0.29 for distribution. Nascimento et al. 21 had similar results in a group of 115 patients (κ = 0.36 for morphology and κ = 0.24 for distribution). Pijnappel et al. 18 found a moderate (κ = 0.59) agreement regarding B3 lesions in the Dutch National Breast screening program. In a second study, the same authors observed a moderate agreement among radiologists, and concluded that a consistent and reproducible classification of calcifications using the BI-RADS lexicon is hardly possible, especially when considering calcifications classified as BI-RADS 3 or 4. 18 This study also revealed that, if the BI-RADS guidelines were followed for all B3 calcifications, diagnosis in 18% of malignancies would have been delayed, which could result in a worse prognosis for these patients. 18 In our study, 32% of the malignancies in B3 lesions would have been missed using the BI-RADS guidelines.
The short-term follow-up is also no guarantee of not missing a malignancy, as many malignant calcifications might take more than 2 years to present visible modifications.41–43 Lesions characterized by central necrosis tend to grow rapidly and the calcifications will also change accordingly. On the other hand, low-grade lesions lacking central necrosis change slowly, and the calcifications will reflect this process, presenting subtle or even no modifications. 41 Lev-Toaff et al. 43 showed that malignant calcifications may remain stable for up to 63 months, and so malignant calcifications might go unnoticed in a time frame of 2 years of follow-up.41,42,44
One of the limitations of this study is the retrospective design, which can incur selection biases. Notwithstanding this limitation, our findings are consistent with several previous studies,14–17,25 in the majority of which the involved radiologists were not performing as per usual practice. They were aware of their participation in the investigations and were therefore probably more cautious while reviewing the mammographic images. Another limitation is the fact that approximately half of the patients (246) were referred to the Dutch Cancer Institute from a variety of centers. All these patients had calcifications that were originally classified as B4 by the Dutch National Breast screening program. In 85 cases, these calcifications were downgraded to B3 during the clinical assessment, and these patients decided by themselves to ask for a second opinion at the Dutch Cancer Institute (after re-evaluation, 24 cases remained as B3 and 61 were reclassified as B4). This difference may be explained by the fact that the re-evaluation at the Dutch Cancer Institute was performed only by experienced breast radiologists, which does not always occur in clinical assessment. In 143 cases, patients were referred from other centers for a second opinion or due to previous breast cancer history (or high risk of developing breast cancer). In 18 cases, it was not possible to determine the reason why patients were referred for further investigation. All 246 patients were assessed at the Dutch Cancer Institute within 6 months of the first diagnosis during screening. Although these cases may not be representative of calcifications found in the population, we observed that the results in this group of 246 patients were very similar to the 227 cases originating directly from screening setting (and also similar to other studies described above). One of the advantages of our research is that the radiologists involved performed the BI-RADS classification according to their usual daily practice. Another advantage is the fact that almost half of the population was referred directly from the Dutch National Breast screening program, facilitating the generalizability of our results.
Conclusion
The most recent edition of the BI-RADS suggests avoiding the use of the B3 category in large population screening programs. We go beyond that: Considering the interobserver variability, costs (and low compliance) to short-term follow-up, and the high predictive value for malignancy in B3 calcifications, we believe that the use of this category for pure calcifications should not only be avoided but eliminated in the screening setting and in daily clinical practice. In a screening setting, where patients are seen once every 2 years (or more often, depending on the country), it is not possible to perform a short-term follow-up due to costs and difficult logistics. In addition, it has been observed in clinical practice that even pure B3 calcifications (round and punctate in isolated clusters) have high rates of malignancy (although their characteristics might not present visual modifications during the 2-year imaging follow-up). Abolishing the B3 category for calcifications could improve patient care and avoid delays in both diagnosis and treatment. Pure calcifications (not associated with a mass or architectural distortion) that originally would be classified as B3 should be directly classified as suspicious (B4), to avoid missing a malignant lesion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
