Abstract
Objectives
There is considerable interest in whether mammography screening leads to over-diagnosis of breast cancer. However self-selection into screening programmes may lead to risk differences that affect estimates of over-diagnosis. This study compares the breast cancer risk profiles of participants and non-participants of population-based mammography screening. Risk profiles are also compared between those who have and have not used private screening services.
Setting
This study involved 1162 women aged 40–84 who participated in the 2012 Health Omnibus, an annual face-to-face interview-based survey of a representative sample of the population in the state of South Australia.
Methods
Data were collected on participation in mammography screening, demographic characteristics and breast cancer risk factors (including reproductive, familial and lifestyle factors). Missing data were multiply imputed. Factors independently associated with ever having been screened were identified using multivariable logistic regression, for population-based and ad hoc, private mammography screening separately.
Results
Compared with non-participants, participants of population-based screening were more likely to have used hormone replacement therapy (odds ratio [OR] = 3.72), experienced breast biopsy or surgery (OR = 2.22), and be overweight or obese (OR = 1.57). They were less likely to be sufficiently active (OR = 0.57) or be born in a non-English speaking country (OR = 0.50) or aged under 50 (OR = 0.09). Women who were screened privately were more likely to have a family history of breast cancer (OR = 1.66) and have experienced breast biopsy or surgery (OR = 3.17) than those who had not.
Conclusions
South Australian women who participated in the population-based mammography screening have a slightly higher prevalence of breast cancer risk factors. This also applies to those who undertook private screening.
Introduction
While numerous studies have examined socioeconomic and psychosocial predictors of participation in mammography screening1–5, few have focused on whether breast cancer risk differs between participants and non-participants. Given the current debate about the extent of ‘over-diagnosis’ due to mammography screening (ie. the detection of cancers that would not have been diagnosed in a woman’s lifetime had she not participated in screening 6 ), it is important to determine whether women who attend mammography screening differ in their level of breast cancer risk from those who do not. Self-selection into screening programmes may affect estimates of over-diagnosis if breast cancer risk is different among screening participants compared with non-participants.
The extent to which breast cancer risk factor profiles differ between breast screening participants and non-participants is largely unknown and may vary by population. Data from the original mammography screening trials suggest that women who did not take up the invitation to attend screening had a higher risk of death from breast cancer. 7 However, the underlying risk of breast cancer among non-attenders was not investigated, and little information is available in current non-trial settings. Heightened concern about cancer among women with a family history of breast cancer or a personal history of benign breast disease may lead to greater participation and, consequently, higher breast cancer risk among screening participants.8,9 Several studies have identified socioeconomic differences in mammography screening uptake.10–12 Many established reproductive risk factors (eg. nulliparity, later age at first birth, later menopause) are more prevalent among affluent women 13 , who are more likely to undergo screening, again leading to higher breast cancer risk among screening participants. A recent Swedish study, however, found little evidence of associations between screening attendance and reproductive histories. 14 Conversely, other lifestyle risk factors associated with lower socioeconomic position (eg. high alcohol consumption, physical inactivity, obesity) may be more prevalent among non-participants15,16, leading to lower breast cancer risk among screening participants.
Local context is also important. Many studies on mammography participation have been conducted in the United States, where factors associated with socioeconomic differences may be more important due to the lack of organized population-based screening. 17 Socioeconomic disparities, however, were not evident in most other countries where organized population-wide screening programmes have been established.18,19
Australia introduced population-based mammography screening, BreastScreen Australia, in 1991, which offers free biennial mammograms targeting women aged 50–69. Women aged 40–49 and over 69 are eligible to attend, but are not been actively recruited. Participation has been reasonably stable at around 55–60% of women in the target age range. 20 However BreastScreen records indicate that approximately 20% of women aged 50 or older have never participated in the screening programme. Women may choose to access mammography screening through private imaging services, but little is known about the extent of screening outside the BreastScreen programme.
In this study we examine differences in breast cancer risk factor profiles among South Australian women who have ever participated in BreastScreen with those who have not. We also compare the risk factor profiles of women who have ever and never participated in screening mammography outside of BreastScreen.
Methods
Study population
Data on screening participation, breast cancer risk factors and demographic characteristics were collected from 1162 women aged 40–84, via the 2012 Health Omnibus Survey. The Health Omnibus is an annual face-to-face interview survey which uses a random stratified cluster sampling technique to recruit a representative sample of 3000 South Australian men and women from selected households. 21 The participation rate in 2012 was 70.4%. Survey methodology and research questions were approved by the University of Adelaide Human Research Ethics Committee.
Measures
Demographic factors included age, highest education level, household income, country of birth and place of residence. Socioeconomic position (SEP) was derived from participant’s residential postcode using the Index of Relative Socioeconomic Advantage and Disadvantage for 2006 22 , which represents the average socioeconomic status of people living within a specified postcode district. SEP was categorized into quintiles according to the distribution for South Australia.
Data on screening attendance and breast cancer risk factors were only asked of women aged 40 and over. Women were asked whether they had “ever had a screening mammogram at a BreastScreen clinic or in a BreastScreen mobile van” and whether they had “ever had a screening mammogram at a private clinic or other facility that was not run by BreastScreen”. Screening was defined as “a routine check for breast cancer when there were no specific problems or symptoms”.
Personal and behavioural risk factors included parity, age at menarche, menopause and first birth, type and duration of hormone replacement therapy (HRT) use, height, weight, level of physical activity, alcohol consumption, family history of breast cancer and personal experience of breast biopsy or surgery. Body mass index (BMI) was categorized as normal or underweight (< = 24.9 kg/m2), overweight (25–29.9 kg/m2) or obese (30+kg/m2), based on self-reported height and weight. Physical activity was assessed as the time per week spent walking, undertaking moderate activities (eg. lawn bowls, golf, gentle swimming), and undertaking vigorous activities (eg. tennis, jogging, cycling, keep fit classes), converted to metabolic equivalent time (MET-mins/week) according to IPAQ scoring protocol (short form). 23 Insufficient physical activity was classified as <600MET-mins/week. Alcohol consumption was assessed using two questions: “How often do you usually drink alcohol?” and “On a day that you drink how many standard drinks do you have?” with a visual cue-card for standard serving sizes. ‘At-risk consumption’ was classified as >7 standard drinks/week, based on recommendations for cancer prevention. 24 Family history of breast cancer was classified as one or more close female relatives ever having been diagnosed with breast cancer (including mother, sisters, daughters and genetically related aunts). Women were also asked whether they had “ever had a breast biopsy or other surgical procedure involving the breast for any reason”. This question was intended as a proxy measure for precancerous conditions (eg. hyperplasia); hence women with breast cancer were excluded from the positive response category.
Analysis
Analysis was restricted to women aged 40–84. Data were initially weighted by individuals’ inverse probability of selection according to area and household size, and reweighted to reflect the age profile for South Australian females, using the 2011 mid-year estimated residential population. 25
Due to the high rate of case-wise missingness (cases with missing data in one or more variables) we conducted analysis using both complete case and multiply imputed (MI) data. Imputation was undertaken using multiple imputation chain equations to impute 20 datasets, with 5 cycles of regression switching. 26 Our imputation model included all outcomes and covariates in our analytic models, as well as marital status and employment status which both predicted missingness. Logistic regression was used in imputation modelling for binary variables and truncated regression for continuous variables, with upper and lower bounds taking the highest and lowest values of survey responses. Education and income levels were modelled using ordinal logistic regression and age at menopause modelled using multinomial logistic regression (to include ‘pre-menopausal’). Validity of imputed data was checked using diagnostic plots and frequency tabulations. Analyses using MI data were restricted to cases with complete data on screening attendance. 27
Weighted cross tabulations were performed for each of the selected characteristics and risk factors, comparing women who had ever participated in BreastScreen with those who had never participated. Multivariable logistic regression was used to identify risk factors that were independently associated with BreastScreen participation, with all variables modelled simultaneously. Reproductive and lifestyle risk factors were modelled as dichotomous variables indicating high versus low risk categories. Similar analyses were undertaken to identify risk factors associated with participation in private mammography screening.
Three summary measures of breast cancer risk were also developed to allow comparison of overall risk among screening and non-screening groups. The first of these measures was the total number of risk factors from twelve possible factors, including insufficient physical activity, overweight or obese, tall stature, at-risk alcohol consumption, HRT use, prior biopsy/surgery, nulliparity, late age at first birth, one or more family members with breast cancer, early menarche, late menopause, and being in the highest SEP quintile. The second measure was a risk score based the Gail model 28 with the following modifications: hyperplasia was not included in the model (because data were not collected); women reporting any breast biopsy or surgery were categorized as having had one biopsy, with no option for multiple biopsies in the model. The third of these measures was a risk score derived from the recently published risk prediction model for breast cancer developed by Pfeiffer et al. 29 This prediction model includes parity, age at first birth, menopausal status, age at menopause, length of use of estrogen-progestin HRT, other HRT use, BMI, alcohol consumption, benign breast disease and family history of breast and ovarian cancer. Risk factors were categorized as described by the authors except that any biopsy/breast procedure was substituted for benign breast disease and family history of breast cancer was substituted for family history of breast and ovarian cancer. Risk scores were calculated for each individual based on published relative risks for each risk prediction model. We used linear regression with imputed data, adjusting for age, to compare both the risk scores and mean number of risk factors, according to screening participation.
All analyses were undertaken using Stata v12.0. 30
Results
Demographic and risk factor profile of participants in the 2012 South Australian Health Omnibus Survey (females aged 40–84).

No. and per cent in each subcategory derived from survey-weighted data.
Counts may not total due to rounding.
BreastScreen participation according to demographic characteristics and breast cancer risk factors (South Australian women aged 40–84).

counts may not total due to rounding
p-values for differences across subcategories derived from separate unadjusted logistic regression models with individual variables using MI data
adjusted odds ratio derived from weighted multivariate logistic regression analysis with MI data with all variables modelled simultaneously
includes premenopausal if aged <55
In multivariate analyses (Table 2), BreastScreen participants had lower odds of being born in a non-English speaking country (Odds Ratio [OR] = 0.50; 0.29–0.85), having a sufficient level of physical activity (OR = 0.57; 0.39–0.83) and being aged 40–49 (OR = 0.09; 0.06–0.15), and higher odds of being overweight and obese (OR = 1.57; 1.08–2.29). The strongest associations were for ever using HRT (OR = 3.72; 2.26–6.13) and having had a breast biopsy or surgery (OR = 2.22; 1.23–3.99). Results showed a curvilinear association with SEP, indicating lower participation among the lowest and highest SEP groups. No significant differences were found for household income, education level, height, age at first birth, and age at menopause, after simultaneous adjustment. Results from complete case analysis (not shown) differed only slightly from multiply imputed data, in that associations with body weight and country of birth were statistically significant in the latter but not the former analysis. In addition a larger effect size was observed for non-English speaking country of birth in the MI analysis.
Participation in private screening (outside of BreastScreen) according to demographic characteristics and breast cancer risk factors (South Australian women aged 40–84).

counts may not total due to rounding
p-values for differences across subcategories derived from separate unadjusted logistic regression models with individual variables using MI data
adjusted odds ratio derived from weighted multivariable logistic regression analysis with MI data
includes premenopausal if aged <55
Comparison of summary measures of breast cancer risk according to participation at BreastScreen or other private screening service.
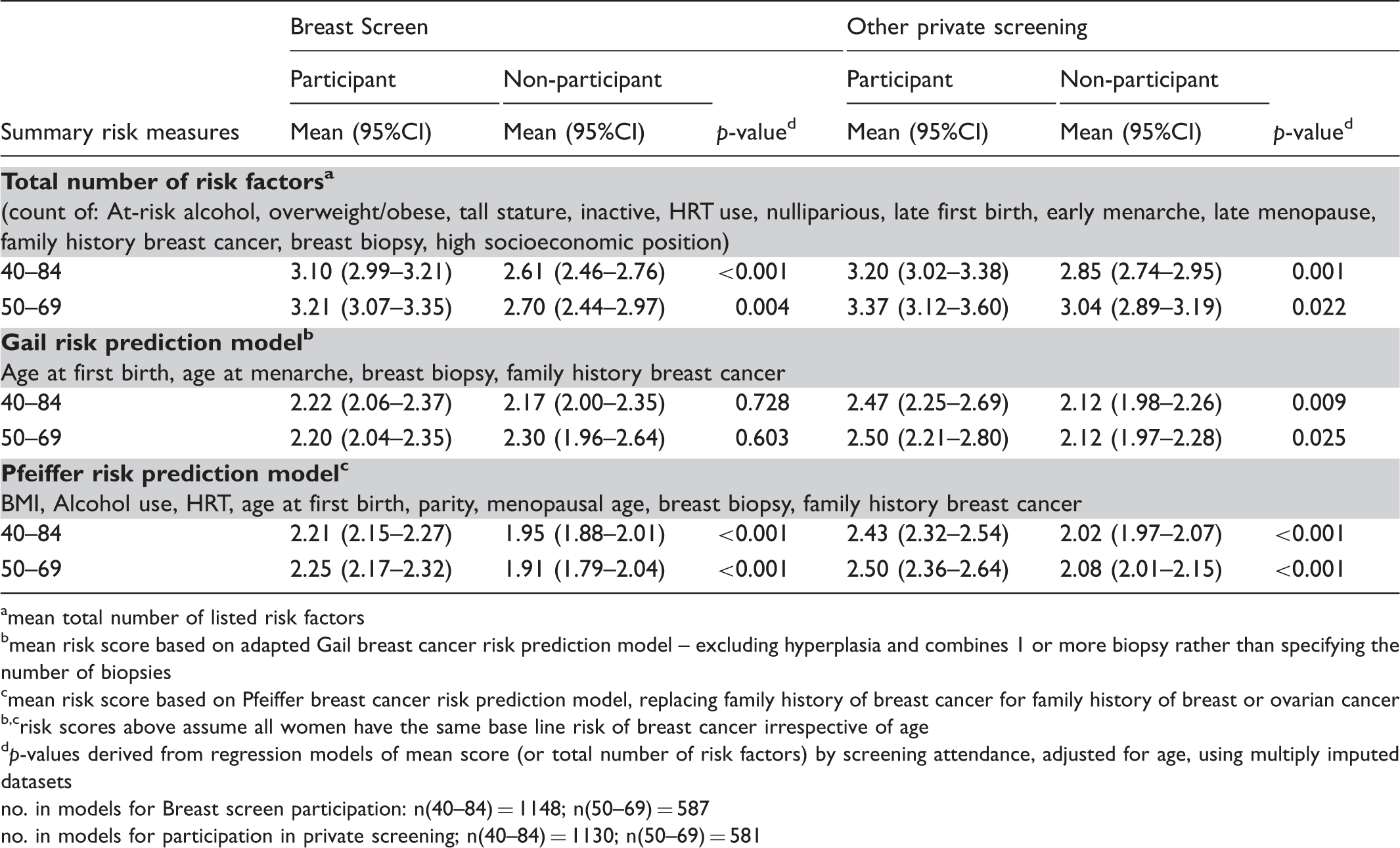
amean total number of listed risk factors
bmean risk score based on adapted Gail breast cancer risk prediction model – excluding hyperplasia and combines 1 or more biopsy rather than specifying the number of biopsies
cmean risk score based on Pfeiffer breast cancer risk prediction model, replacing family history of breast cancer for family history of breast or ovarian cancer
b,crisk scores above assume all women have the same base line risk of breast cancer irrespective of age
dp-values derived from regression models of mean score (or total number of risk factors) by screening attendance, adjusted for age, using multiply imputed datasets
no. in models for Breast screen participation: n(40–84) = 1148; n(50–69) = 587
no. in models for participation in private screening; n(40–84) = 1130; n(50–69) = 581
Discussion
This study indicates that, for South Australian women, breast cancer risk factor profiles differ between those who have and have not participated in population-based mammography screening. Participation was less likely among women younger than 50 who are not actively targeted by BreastScreen, those born in non-English speaking countries and those who were not sufficiently active, while the likelihood of having used HRT and of having experienced breast biopsy or surgery was much more likely among BreastScreen participants. Contrary to expectations, BreastScreen participants were more likely to have been overweight or obese, and no statistically significant differences were noted for family history of breast cancer.
The association between HRT use and BreastScreen participation may be due to increased contact with medical practitioners (to obtain or renew prescriptions). Due to concerns about increased risk of breast cancer many doctors may have recommended screening to patients using HRT. There is consistent evidence that a doctor’s recommendation is a strong predicator of screening attendance. 1
As observed in other studies 9 , a history of breast biopsy or surgery was associated with screening participation. However, determining the causal direction is difficult, given the cross-sectional design of this study. Women who attend BreastScreen would have a greater chance of biopsy due to investigation of abnormal mammographic lesions.
While our finding of increased participation among overweight and obese women contradicts other studies showing lower participation15,31,32, it is consistent with findings from a recent US study. 33 One explanation may be that lighter women with smaller breasts feel more confident in detecting breast abnormalities through self-examination and therefore see less benefit from attending mammography screening.
Also in contrast to other studies 9 , including a previous South Australian study 34 , we did not observe any association between family history of breast cancer and participation in population-based screening. Additional analyses using different post-hoc definitions of family history (eg. two or more close female relatives diagnosed at any age and/or one relative diagnosed before age 50) also failed to show any statistically significant differences, though odds were consistently elevated.
While there appears to be a relationship between income and having participated in the Breast Screen programme, this was only observed for univariate analysis. The association with income did not persist when controlling for other covariates in multivariate analysis. We believe the association with income is confounded by age. Older women will have had more opportunity to have ever participated in Breast Screening. They are also more likely to have lower household incomes given many will be pensioners or self-funded retirees with modest annual incomes.
Undergoing mammography screening outside of BreastScreen, however, was associated with family history of breast cancer, as well as higher education and higher SEP. There was also a strong association between private screening and breast biopsy/surgery. It is possible that some women confused screening and diagnostic mammography, which may explain the greater odds of biopsy/surgery among those who reported being screened privately. Alternatively those with prior breast problems may seek additional security by undertaking screening more frequently using both public and private services. This may also apply to women with a family history of breast cancer. Additionally women with some premalignant diagnoses, such as atypical ductal hyperplasia, may be discharged from population based screening programmes as a matter of policy, because their risk exceeds population level risks. The association with higher levels of education and SEP suggests those with sufficient financial resources may also seek additional security through private screening. We did not find evidence that BreastScreen non-participants were more (or less) likely undergo mammography elsewhere.
Our findings suggest that participants of population-based mammography screening have a slightly higher risk of developing breast cancer than non-participants. Both HRT use and being overweight are associated with a moderately increased risk of breast cancer.35,36 Likewise, increased likelihood of breast procedures may indicate a higher prevalence of premalignant abnormalities, and potentially greater risk of breast cancer. 37 Furthermore, women from non-English speaking countries (who participate less) tend to have lower risk of breast cancer. 38 Only the observed differences in physical activity levels would potentially lead to reduced risk among screening participants. 39
Comparison of the summary risk measures supports this conclusion. We found that BreastScreen participants, on average, had a greater number of breast cancer risk factors and a modestly elevated risk of developing breast cancer based on risk scores using the Pfeiffer’s risk prediction model. Risk differences were greater still in relation to participation in private mammography screening. While we did not find statistically significant differences in risk scores based on the Gail model, this is not unexpected, given that there was no difference between BreastScreen participants and non-participants for family history and age at first birth, on which the Gail model is largely based.
This study has a number of limitations. Measures of both screening attendance and risk factors may be inaccurate, due to our reliance on self-reported data. Recall and response bias may also have led to erroneous findings. Because BreastScreen is a well-recognized service, we believe that recall of attendance at BreastScreen would have been reasonably accurate. However participants may have over-reported attendance at private screening services if they did not distinguish between screening and diagnostic mammography. This study did not include all probable risk factors (eg. breast feeding, oral contraceptive use) or explore potential risk factors where evidence is less clear (eg. tobacco smoking, fat intake). Nor did it account for interaction between certain risk factors (eg. effect modification by BMI on HRT effects 40 ). Also the cross-sectional design does not allow causal inferences to be determined. This is particularly pertinent to disentangling whether increased breast procedures result from, or are a motivation for, screening participation. However, even when a history of biopsy/surgery is excluded from the calculation of the summary risk scores, we still observed significantly higher mean risk scores among women who have participated in Breast Screen using the Pfeiffer model and mean total count of risk factors.
Finally, our summary measures of risk were relatively crude. Using the total number of risk factors as an indicator of the risk profile does not account for the differing strength of association between various risk factors and the risk of developing breast cancer. Also due to the lack of data on women’s history of pre-malignant disease we used a modified version of the Gail model which did not included hyperplasia or multiple biopsy procedures. Furthermore, the recently published Pfeiffer risk prediction model has not been validated in an Australian population and we have applied it as if the background level of risk has not changed over time. Therefore results should be considered indicative rather than definitive.
The strengths of this study are that we collected a range of risk factor data from a randomly selected sample of women who are representative of the eligible screening population for screening, and used multiple imputation to increase precision and reduce potential biases due to the exclusion of a large proportion of cases with missing data.
Conclusion
Our findings suggest that South Australian women who participate in population-based screening have a slightly worse risk factor profile than those who do not. For the most part this can be attributed to greater HRT use among BreastScreen participants. These findings may not be generalizable to other settings, given variations in risk factor prevalence and/or organization of screening services in different countries. This highlights the need to understand differences in breast cancer risk profiles within the local context, especially in relation to evaluating benefits and risks of mammography screening including over-diagnosis.
Footnotes
Funding
This study was funded by the National Breast Cancer Foundation through a Novel Concept Award (#NC-11-53).
Conflict of interest
Authors have no conflict of interest.
Acknowledgements
We wish to thank Ms Lora Dal Grande and Mr Graeme Tucker for their assistance in relation to survey weighting. We also wish to acknowledge the contribution of women who took part in the 2012 HOS survey. Ms Beckmann is the recipient of a NHMRC PhD scholarship. Prof John Lynch is supported by a NHMRC Australian Fellowship.
