Abstract
A new bio-based unsaturated polyester resin was blended with a bio-based reactive diluent and evaluated. Furan-based monomers were selected as the main monomers in both the resin and the bio-based reactive diluent to enhance thermomechanical properties and address solubility issues typically seen in bio-based resins and diluents. The resin was synthesized from 2,5-furan dicarboxylic acid, isosorbide, and glycerol, and the resulting polymer intermediate was then end-capped with methacrylic anhydride to introduce reactive sites for cross-linking reaction. The resin was then mixed with either different percentages of bio-based reactive diluent (2,5-bis(hydroxy-methyl) furan methacrylate) or with styrene to study the thermomechanical and rheological behavior of obtained resins. FT-IR, 13C-NMR, and 1H-NMR were used to determine the chemical structure of the bio-based reactive diluent. The thermomechanical properties of resin containing bio-based reactive diluent or styrene are characterized and compared by DMA, TGA, and DSC. The synthesized resin had good solubility in the bio-based reactive diluent but not in the styrene. The different mixtures of resin and bio-based reactive diluent showed glass transition temperatures ranging from 166 °C to 174 °C, which was higher than the commercial fossil-based unsaturated polyester resin used as a reference in this study. With thermal and mechanical properties comparable to commercial petroleum-based thermosets, these bio-based resins are promising candidates for high-performance composites, coatings, and other thermoset-based applications.
Keywords
Introduction
Thermoset unsaturated polyester resins are used in various applications, such as matrices in fiber-reinforced composites. They are valued for their easy processing and adjustable curing conditions, as well as their good thermomechanical properties, which include high strength and resistance to heat and chemicals.1,2 Currently, most commercial unsaturated polyesters (UPE) are made from prepolymers composed of fossil molecules like propylene glycol, maleic anhydride, isophthalic acid, phthalic anhydride, and terephthalic acid. The prepolymer is mixed in a reactive diluent, mainly styrene, which reacts in the curing to form covalent crosslinks between the prepolymers. There are some promising examples of using bio-based molecules to synthesize replacements for these fossil-based UPEs, but many of them still rely on styrene for cross-linking.3–7 However, styrene is volatile, fossil-based, and considered a hazardous air pollutant. Therefore, there is a need to develop fully bio-based and environmentally friendly UPE thermosets. Bio-based reactive diluents are an effective strategy in this regard, as they reduce volatile organic compound emissions by being incorporated into the polymer network rather than evaporating during processing. Their renewable origin supports reduced reliance on fossil resources and contributes to circular material flows. Recent research has focused on creating bio-based UPEs containing bio-based reactive diluents, demonstrating the possibility of achieving completely bio-based unsaturated polyester thermosets.8–13 The synthesis of these resins often involves the use of aliphatic monomers in the prepolymer or the reactive diluent structures, resulting in a resin with limited thermomechanical properties once cured. Therefore, the substitution of traditional monomers with bio-based alternatives presents a significant challenge. Furan-based monomers, known for their aromatic structure and the possibility to obtain from many different biomass resources, show promise as replacements for petroleum-derived phenyl components in commercial UPEs. These monomers are considered valuable due to their ability to enhance glass transition temperature (Tg) and mechanical strength in comparison to aliphatic monomers, thanks to their rigid and aromatic structures.14–17 Key furan derivatives such as (hydroxymethyl) furfural and furfural, are produced through the conversion of polysaccharides or pentose and hexose sugars. The United States Department of Energy has identified 2,5 furan dicarboxylic acid (FDCA) as a leading candidate among 12 molecules likely to drive the future green chemistry industry. 18
The adoption of furan derivatives in UPEs can be approached according to two main strategies: first, as a reactant in the synthesis of the UPE prepolymer, and second, as a reactant to synthesize a reactive diluent. According to the first approach, various bio-based UPEs were synthesized using furan-based monomers through the polycondensation of a mixture of diols and diacids. Sousa et al. synthesized FDCA-based UPE through noncatalyzed bulk polycondensation, resulting in a thermally stable crosslinked resin with high Tg (up to 104 °C) and storage moduli. They used 2-hydroxyethyl methacrylate as the reactive diluent instead of styrene. 19 Lomelí-Rodríguez et al. developed polyester resins for coil coatings by combining bio-based isosorbide (IS), FDCA, succinic acid (SA), and 1,5-pentanediol. Instead of focusing on detailed compositions and chemical characterization, the study aimed to evaluate the feasibility of bio-based coil coatings using common mechanical testing methods. 20 Suriano et al. developed a range of UPEs using different combinations of FDCA, itaconic acid, diols, and bio-based reactive diluents like butanediol dimethacrylate and dimethyl itaconate. After cross-linking, a Tg of 75 °C was achieved. 21 Hofmann et al. also presented UPEs based on FDCA and other bio-based building blocks with Tg reaching up to 102 °C. 10 Dai et al. created fully bio-based UPEs from FDCA, itaconic acid, SA, and 1,3-propanediol through direct polycondensation. The introduction of FDCA due to its rigid, aromatic structure significantly enhanced the modulus, flexural strength, and thermal properties of the cured resins, demonstrating the potential to replace fossil-based UPEs. 22 In another approach, researchers utilized furan-derived monomers to create reactive diluents for UPE or vinyl ester resins. Sadler and colleagues described the use of two furanic reactive diluents, furoic acid glycidyl methacrylate, and furfuryl methacrylate, which were synthesized from furoic acid glycidyl and furfuryl alcohol, respectively. Blends of vinyl ester resins with furoic acid glycidyl methacrylate and another furanic diluent exhibited lower Tg (82–100 °C) and higher viscosities (4-234 cP) compared to styrene-based vinyl esters. 23 Mixing furfural methacrylate (35%) diluent with high viscosity glycidyl methacrylate isosorbide significantly decreased viscosity to 2234 cP at 25 °C, with the cured specimen exhibiting a Tg of 101 °C. 24 Furthermore, a toughening agent hyperbranched acrylate and a bio-based diluent tetrahydrofuryl acrylate were integrated into UV-curable coating formulations of acrylated epoxidized soybean oil. The addition of hyperbranched acrylates enhanced the adhesion, mechanical properties, hardness, and thermomechanical characteristics of the UV-curable coating, while tetrahydrofuryl acrylate enhanced its flexibility. 25
A new approach involves the use of furan-based components in both the prepolymer and diluent, which has not been previously reported. By synthesizing UPE with furan-based monomers in both the prepolymer and the reactive diluent, a resin with good thermomechanical properties, a high Tg, and improved solubility can be achieved. Xu et al. synthesized a bio-based UPE containing 1,4 butanediol, succinic anhydride, isosorbide, and maleic anhydride. The resin was diluted with isosorbide methacrylate as a bio-based reactive diluent. Isosorbide was chosen as the main monomer in both the reactive diluent and UPE to improve the rigidity of the final product, increase thermal and mechanical properties, and improve the solubility of bio-based UPE and reactive diluent. 26
In a recent study, we introduced a bio-based branched UPE made from FDCA, isosorbide, and glycerol as a bio-based resin alternative. The resin had a Tg of 173 °C, showing potential high-temperature applications.. 27 Glass fiber reinforced composite made from this resin exhibited good mechanical properties. 28 However, the resin’s high viscosity and difficult processability for hand lay-up impregnation were noted as drawbacks. The high viscosity of the resin was mainly due to the branched structure of the resin resulting from glycerol (GLY) as a core molecule. 27 In such cases, viscosity can be adjusted with styrene or a bio-based reactive diluent.
This study aimed to synthesize a bio-based reactive diluent to blend with the prepolymer for viscosity reduction and enhanced processability. The study involved the first step of synthesizing branched low molecular weight polymer intermediates with FDCA to obtain a rigid skeletal and aromatic structure, with glycerol as a branching point and isosorbide for a unique bicyclic rigid structure to enhance thermomechanical properties. Methacrylic anhydride was then introduced as an end-cap to provide reactive cross-linking sites. 2,5-Bis(hydroxy-methyl) furan methacrylate was synthesized as a reactive diluent by the method mentioned by Chu et al. 29 The synthesized prepolymer was mixed in the second step with 40,50,60,65 % reactive diluent and properties were compared with the prepolymer mixed with 40,45,50% styrene. FT-IR, DMA, TGA, and DSC techniques were used to study the obtained UPEs.
Experimental
Materials
2,5-furan dicarboxylic acid (97%, Apollo Scientific), isosorbide (98%, TCI), glycerol (99%, Sigma-Aldrich) and tin (II)-2-ethyl hexanoate (95%, Sigma-Aldrich) catalyst, methacrylic anhydride (≥94%; Sigma-Aldrich), and hydroquinone (99% from Sigma-Aldrich) were utilized in the preparation of the prepolymer. The experiments were carried out under a nitrogen atmosphere (N2, 99.5%). Xylene (≥98.5%; Sigma-Aldrich)/isopropyl alcohol (99.8%; Sigma-Aldrich) mixture (1:1), potassium hydroxide (≥85%; Sigma-Aldrich) solution in absolute ethanol (VWR Chemicals), 0.02 M, was used for acid number titration, with phenolphthalein (98.5% Acros, 1% solution in ethanol) serving as an indicator. Potassium hydrogen phthalate (99.95%; Sigma-Aldrich) in distilled water was used as a standard solution. The synthesized resin was mixed with or a reactive diluent made from 2,5-bis(hydroxy-methyl) furan (Apollo Scientific), 4-styrene (99%, Sigma-Aldrich) dimethylaminopyridine (DMAP, 99%, Apollo Scientific), and methacrylic anhydride. The resin was cross-linked using Luperox® A75 (75%, remainder water, Sigma-Aldrich) as the initiator for free radical initiation. CRYSTIC 115 NT 30 (Scott Bader) orthophthalic polyester resin containing 40 wt% styrene was used as a reference resin.
Synthesis of FDCA-based UPE
The resin was synthesized following the procedure outlined in our previous publication.
27
To summarize, the resin was made using bulk polymerization in two stages. Initially, glycerol (0.033 mol), FDCA (0.1 mol), and isosorbide (0.11 mol) were combined in a round-bottom flask with a mechanical stirrer, condenser, nitrogen inlet, and thermometer. Tin (II)-2-ethyl hexanoate (0.2% w) was then added, and the mixture was heated to 200 °C until the reactants were completely mixed (the complete homogenization of acid and diols and formation of a transparent mixture, about 15 hours after starting the reaction). The reaction progress was monitored for 48 hours until a constant acid value was achieved. In the second stage, the reaction solution was cooled to 110 °C, and hydroquinone (0.3 w % of total monomers mass) was added as an inhibitor. Methacrylic anhydride (0.11 mol) was then added slowly, and the temperature was maintained at 110 °C for 4 hours. Figure 1 shows the schematic synthesis of resin. Reaction scheme for the synthesis of resin.
Synthesis of 2,5-bis(hydroxy-methyl) furan methacrylate
2,5-Bis(hydroxy-methyl) furan methacrylate was synthesized following the procedure described by Chu et al.
29
(Figure 2). In a 100 mL three-necked flask with an addition funnel, condenser, thermometer, and magnetic stirring bar, 2,5-bis(hydroxymethyl) furan (10 g) and DMAP (0.954 g) were placed. The flask was heated to 80 °C using an oil bath to melt 2,5-bis(hydroxy-methyl) furan and DMAP. Methacrylic anhydride (25.26 g) was then added dropwise over 1 hour. The reaction continued at 80 °C for 24 hours under stirring. The product, a combination of 2,5-Bis(hydroxy-methyl) furan methacrylate and methacrylic acid, was collected and utilized as a bio-based reactive diluent. Reaction scheme for the preparation of reactive diluent.
Curing of the resin
The curing method included blending the UPE resin with 40,50,60,65 wt% reactive diluent or 40,45,50 wt-% styrene and then mixing it with 2.0 wt% of Luperox® P peroxide. Subsequently, the mixture was poured into a Teflon mold and placed in a 140 °C preheated oven for 60 min. The cured specimens were removed from the mold and cooled down to room temperature for further characterization.
Characterization techniques
Titration
Titration was used to monitor the progress of the condensation reaction by evaluating the percent conversion of carboxyl groups during the synthesis process. A specimen weighing 0.1-0.2 grams was collected, diluted with a 1:1 mixture of xylene and isopropyl alcohol, and titrated with a 0.02 M KOH solution in absolute ethanol. The titration used 1% phenolphthalein in ethanol as an indicator to measure the amount of potassium hydroxide consumed.
Rheological properties
The viscosity of the resins was analyzed using a Brookfield instrument (DV-E viscometer) with spindle 31 at rpm range from 6-100 at 25 °C, and the values were recorded in Pa.s.
Nuclear magnetic resonance
The samples were dissolved in CDCl3 and analyzed using a Bruker AVANCE III Spectrometer with a cryogenic probe head, operating at 800 MHz. The 13C NMR data was obtained using a 30° excitation pulse and a 10-second relaxation delay. These experimental parameters were chosen to yield semi-quantitative 13C NMR spectra. The 1H NMR spectrum was obtained using 30 degree excitation pulse and 2.5 s relaxation delay.
Fourier transform infrared spectroscopy
Fourier Transform Infrared Spectroscopy was carried out using a Nicolet 6700 FT-IR spectrometer to analyze the samples at different reaction stages and the cured resin. The spectra were recorded between 4,000 and 400 cm-1 after 64 scans with a 4 cm-1 band resolution.
Dynamic mechanical analysis
Dynamic Mechanical Analysis was performed on cured resin mixtures using a Q800 instrument from TA Instruments. Specimens (35 mm length, 2 mm thickness, and 5 mm width) were heated in a single cantilever bending clamp from -25 °C to 200 °C in an inert atmosphere at a heating rate of 2 °C per minute, with an amplitude and frequency set at 15 μm and 1 Hz, respectively.
Thermogravimetric analysis
Thermogravimetric Analysis was conducted using a TGA instrument (Q500 from TA Instrument) to explore the thermal stability of specimens. A small, cured resin specimen (8-15 mg) underwent heating from room temperature to 600 °C at a rate of 10 °C/min under a nitrogen purge stream.
Differential scanning calorimetry
Differential Scanning Calorimetry measurements were carried out using a TA Instrument DSC Q 1000 device on samples (5-15 mg) heated from 0 to 200 °C at a rate of 10 °C/min in aluminum pans. Both cured and uncured (mixed with 2% peroxide) resin samples underwent analysis to investigate the efficiency of the crosslinking reaction under a nitrogen atmosphere.
Results and discussion
Resin viscosity
Viscosity of the resin mixtures and diluents.

1Not possible to determine due to the high viscosity.

A mixture of FDCA/IS/GLY resin with left: 60% styrene, right: 60% reactive diluent.

A mixture of FDCA/IS/GLY resin with 45% styrene.
Fourier-transform infrared spectroscopy
This technique was used to confirm the structure of the synthesized reactive diluent (Figure 5), the end-functionalization of the resin with methacrylic anhydride (Figure S1, S2, see supplementary materials), and the curing reaction of the resins (Figure S3, S4, see supplementary materials). The complete reaction of 2,5-bis(hydroxy-methyl) furan and methacrylic anhydride was confirmed by the absence of a hydroxy group peak which can be seen for 2,5-bis(hydroxymethyl) furan (3226-3312 cm−1) and the formation of a new ester group (emerging peak of C=O at 1717 cm−1 for 2,5-Bis(hydroxy-methyl) furan methacrylate and 1693 cm−1 for methacrylic acid) as well as peaks at 1634 cm−1 and 939 cm−1 corresponding to the C=C stretching and =CH2 bending respectively (Figure 5). The IR spectrum of FDCA/IS/GLY resin was similar to the previous study.
27
FDCA/IS/GLY peaks covered the reactive diluent peaks due to similarity in structure especially for functional groups like C=O and C=C but styrene peaks (The unsaturated C-H stretching peaks for benzene ring are at 3081 cm−1, 3058 cm−1, and 3026 cm−1, ring bending peak at 694 cm−1 and aromatic C-H band at 773 cm−1) were obvious in the different mixtures of resin with styrene. The =CH2 bond at 905 cm−1 for resin/styrene and carbon-carbon double bond peaks at 1634 cm−1 for both resin/reactive diluent and resin/styrene mixture disappeared as the produced resins were cured by a free radical reaction. FTIR results for (a) 2,5-bis(hydroxy-methyl) furan, (b) reactive diluent.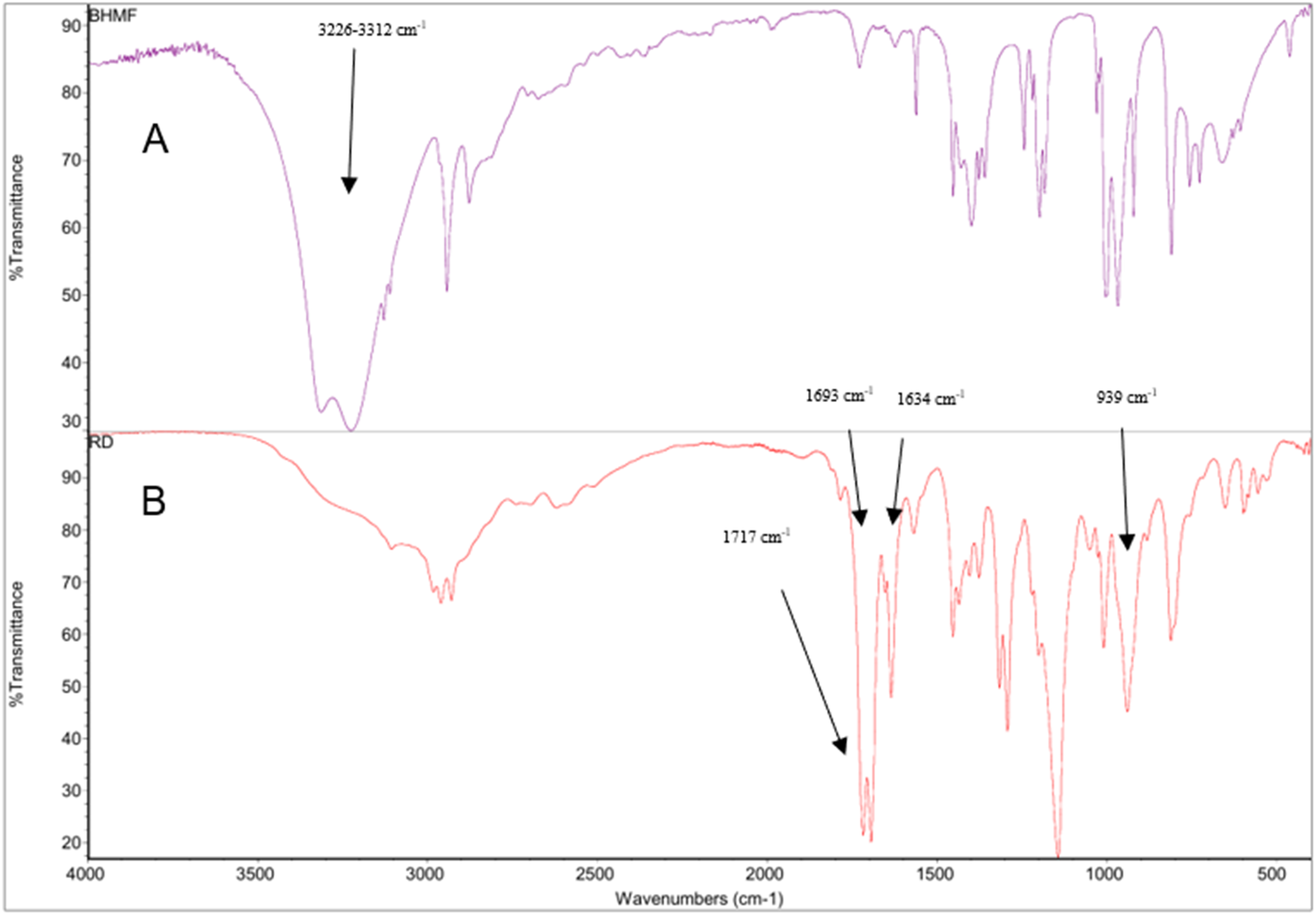
Nuclear magnetic resonance
In the NMR spectra (Figure S5, S6, see supplementary materials), it is possible to identify the two main components, methacrylic acid and 2,5-bis(hydroxy-methyl) furan methacrylate, in approximately 2:1 molar concentration, respectively.
Methacrylic acid, 1H NMR (CDCl3 =7,27, 298 K) d: 10.0 (bm, OH), 6.12 (pentuplet about 1 Hz, vinyl), 5.54 (pentuplet about 1 Hz), 1.86 (m, CH3); 13C NMR d: 172.37 (COOH), 136.11 (s, C-2), 126.68 (t, vinyl CH2), 17.87 (q, CH3).
2,5-Bis(hydroxy-methyl) furan methacrylate, 1H NMR (CDCl3 =7,27, 298 K) d: 6.32 (s, H-3,4), 6.05 and 5.50 (vinyl), 1.86 (m, CH3); 13C NMR d: 166.90 (COO), 150.12 (C-2,5), 135.86 (s, Methacryl C-2), 126.04 (t, vinyl CH2), 111.38 (d, 1JCH=175.8 Hz, C-3,4), 58.24 (t, 1JCH=149.5 Hz, CH2O), 18.15 (q, 1JCH=128.3 Hz, CH3).
Dynamic mechanical analysis
The storage modulus, loss modulus, and the glass transition temperatures of different resins.

*The difference between results from the DMA test in the previous paper 27 and here is due to styrene content. The commercial resin had a styrene content of 30% (reported as 30-40% in the datasheet), and the styrene content was adjusted to 40% here.
**The difference between results from the DMA test in the previous paper 27 and here is due to a different curing procedure.
Differential scanning calorimetry
DSC results for cured and uncured resins.

DSC curve for uncured resins.
Thermogravimetric analysis
TGA results for cured resins.

Conclusions
This study aimed to synthesize a bio-based reactive diluent to blend with the prepolymer to reduce viscosity and enhance processability. A range of bio-based unsaturated polyester resins was formulated using furan-based monomers as the rigid core constituent in both the resin and diluent. The bio-based resin (FDCA/IS/GLY) was created from 2,5-furan dicarboxylic acid, isosorbide, and glycerol, and then end-capped with methacrylic anhydride. By blending FDCA/IS/GLY resin with varying amounts of a bio-based reactive diluent (2,5-bis(hydroxy-methyl) furan methacrylate), a bio-based UPE was obtained. The thermomechanical properties of the fully bio-based resin mixed with the bio-based reactive diluent were compared to a partially bio-based resin made up of FDCA/IS/GLY resin and styrene as a diluent. Viscosity measurements demonstrated that the FDCA/IS/GLY resin had better compatibility with the bio-based reactive diluent compared to styrene. Thermomechanical analyses showed that the thermosets made with the bio-based reactive diluent exhibited superior properties, with a 10% weight loss occurring at temperatures up to 371 °C, glass transition temperatures ranging from 164 °C to 176 °C, and storage moduli up to 4292 MPa. The incorporation of furan-based monomers in both the resin and diluent resulted in improved thermomechanical properties compared to a traditional fossil-based unsaturated polyester resin, highlighting the potential of these bio-based thermosets as sustainable alternatives to petroleum-based UPEs. Based on the results, UPE formulations containing 60% and 65% of the bio-based reactive diluent met the requirements for viscosity and thermomechanical properties as a thermoset resin used in the industry. The synthesized resin and diluent show promise, and future research should focus on producing composites by reinforcing the resin and reactive diluent with fibers such as glass fibers or natural fibers.
Supplemental material
Supplemental Material - Synthesis of a bio-based unsaturated polyester resin from 2,5-furan dicarboxylic acid and isosorbide blended with a bio-based reactive diluent synthesized from 2,5-furandimethanol
Supplemental Material for Synthesis of a bio-based unsaturated polyester resin from 2,5-furan dicarboxylic acid and isosorbide blended with a bio-based reactive diluent synthesized from 2,5-furandimethanol by Samira Akbari, Tõnis Pehk, Ivo Heinmaa, Mikael Skrifvars, Sunil Kumar Ramamoorthy and Dan Åkesson in Polymers and Polymer Composites.
Footnotes
Acknowledgment
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The NMR tests were supported by the Estonian Research Council grant No. PRG1702.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
