Abstract
Tissue engineering provides an innovative strategy for regenerating damaged tissues through the use of three-dimensional (3D) scaffolds, which can be fabricated using advanced techniques such as 3D printing. This approach enables the precise and reproducible design of scaffolds with critical architectural features, including interconnected porosity. One of the main challenges in tissue regeneration, particularly in the oral cavity, is bacterial contamination, which can trigger inflammatory responses and hinder healing. Therefore, scaffolds with inherent antimicrobial properties hold great clinical potential. However, the antimicrobial properties of chitosan vary depending on its molecular weight and the bacterial species tested. This study aimed to fabricate and characterize 3D-printed chitosan-based scaffolds and to assess their antimicrobial activity. Bioinks were prepared using high (CH-HMW) and low (CH-LMW) molecular weight chitosan, and their rheological properties were evaluated to confirm pseudoplastic behavior suitable for extrusion-based printing. Chitosan–alginate scaffolds were printed and characterized by scanning electron microscopy (SEM), and their antimicrobial efficacy was tested against ATCC reference strains: Escherichia coli, Streptococcus mutans, Staphylococcus aureus, and Enterococcus faecalis. The results indicated that 3% CH-HMW and 6% CH-LMW formulations exhibited suitable printability. Notably, 3% CH-HMW demonstrated significantly enhanced antimicrobial activity against all tested strains compared to 6% CH-LMW and the positive control. These findings highlight 3% CH-HMW as a promising material for clinical applications in oral bone regeneration, where antimicrobial performance is critical. Future studies should evaluate its biocompatibility and explore synergistic combinations with other materials to further improve scaffold functionality and tissue integration.
Introduction
Tissue regeneration and organ replacement have become an essential pillar of the 21st-century medicine, motivated by a need to provide solutions for diseases and traumas, such as bone malformations, premature loss of teeth, and aggressive treatments, which usually result in tissue and even organ loss. 1 To meet these needs, tissue engineering has emerged as an innovative alternative, focusing on the innovative development of materials and advanced techniques to restore the structure and function of the injured tissue.1,2 Tissue engineering is a multidisciplinary field that combines biological principles and engineering to maintain, restore, or regenerate tissue. 3 This field has progressed by incorporating materials science, enabling the in vitro production of functional tissues prepared and implanted in patients with missing or injured tissues. 4 However, the success of these treatments can become compromised by various limitations, such as immunogenicity, high morbidity at the donor site, and risk of infection. 4
One of the significant challenges in bone regeneration is the risk of infection by microorganism near the site of implantation, which can colonize implants and lead to treatment failure. 5 The oral cavity presents a diverse microbiota with bacteria such as Streptococcus mutans, Enterococcus faecalis, and others associated with periodontal disease, playing a key role in bone loss pathogenesis.6–8 E. faecalis is a facultative anaerobic bacterium generally associated with the gastrointestinal tract that can colonize dentin tubules and contribute to root canal treatment failure. Its capacity to survive in hostile environments, form biofilms with other microorganisms, and resistance to elimination complicates its eradication. 8
Likewise, S. mutans, known for its capacity to develop dental caries, has been isolated during oral bone regeneration, leading to treatment failure, due to its capacity to form biofilms and colonize used materials. 9 Additionally, Escherichia coli, a colonizer associated with periodontal disease, can contribute to procedure failure, due to its virulence factors and resistance genes that code for adhesins, responsible for bacteria adhesion to host tissue and potentially colonize materials employed in regeneration; hence, compromising treatment success. 10
To address these challenges, materials science has progressed in the design of bioactive, biocompatible, and biodegradable materials that also exhibit antimicrobial or bacteriostatic capabilities, thereby preventing bacteria colonization. 11 To obtain such materials, diverse techniques are used, among which 3D printing is a fundamental tool for creating 3D scaffolds that mimic biological tissue structure. Printing by extrusion is particularly relevant, since it uses pseudoplastic materials, which, given their capacity to flow under pressure, allow the formation of optimal structures for tissue regeneration. 12 These scaffolds can be fabricated with a variety of biomaterials, including natural and synthetic polymers, metals, and ceramics, each tailored to specific applications in tissue engineering. 13
Chitosan, a natural polymer derived from chitin, stands out as an excellent material for use in medical scaffolding applications, due to its key properties, including biocompatibility, biodegradability, and non-toxicity, which are fundamental in promoting wound healing and tissue regeneration. It acts as a healing agent, promoting cell proliferation, blood vessel formation, and extracellular matrix synthesis, and is involved in many cell types during healing processes. 14 This biopolymer’s antibacterial properties and immunomodulatory characteristics help control inflammation, which is critical in tissue repair settings. Its hydrophilic nature allows for maintenance of a humid environment, favoring cell movement and granulating tissue production. 14
Its chemical structure features repetitive units of N-acetyl-D-glucosamine (GlcNAc), like those found in cell wall glycosaminoglycans, making it ideal for creating cell scaffolds in tissue engineering. 14 It is employed in bone and cartilage regeneration, as well as in matrix manufacturing, supporting cell growth. Chitosan's 3D porous structure is key for its efficacy, and when it is inadequate, crosslinking agents are used to enhance its mechanical properties without compromising cell adhesion.10,15 However, chitosan inherently exhibits low compressive strength and limited structural stability, restricting its use in bone regeneration unless properly reinforced or crosslinked. Glutaraldehyde crosslinking has been widely reported to increase the mechanical stability of chitosan networks by creating covalent interchain linkages that reinforce scaffold rigidity.16,17 This is why crosslinking strategies are widely implemented to improve scaffold robustness for load-bearing or structural applications.
The antimicrobial activity of this molecule is carried out through different mechanisms. 10 Similarly, it is notable for its capacity to chelate metals in the outer structures of pathogens, which leads to the inhibition of toxin production. It also interacts with microbial DNA, affecting mRNA synthesis and subsequent enzymatic activity. 18
Despite chitosan’s advancements as a potential material with antimicrobial activities, no conclusive results regarding its activity against Gram-positive and Gram-negative bacteria exist. 11 Some studies suggest a greater susceptibility of Gram-positive bacteria to chitosan, whereas others report opposite effects. These inconsistencies may be related to the intrinsic factors of chitosan, such as its degree of deacetylation and molecular weight, as well as extrinsic conditions in the experimental environment. 11
Although the antimicrobial properties of chitosan have been widely described in the literature, most studies have evaluated non-printed chitosan films, coatings, or chemically modified derivatives under conditions that do not replicate the structural constraints of 3D-printed scaffolds. Limited information exists regarding how chitosan’s molecular weight influences the antimicrobial performance of crosslinked 3D-printed constructs, which is relevant for tissue engineering applications where pore architecture and crosslinking density affect bacterial interaction. These variations highlight the need to continue investigating to optimize its applications in antimicrobial therapies. Hence, the objective of this study was to generate, characterize, and determine the antimicrobial activity of 3D-printed scaffolds with high and low molecular weight chitosan against Escherichia coli, Streptococcus mutans, Staphylococcus aureus, and Enterococcus faecalis.
Materials and methods
Low and high molecular weight chitosan bioink fabrication
To fabricate chitosan bioinks, high-molecular-weight chitosan (CH-HMW, Chitosan high molecular weight CAS No. 9012-76-4) and low-molecular-weight chitosan (CH-LMW, Chitosan Low molecular weight CAS No. 9012-76-4) were purchased from Sigma-Aldrich. To this end, bioinks at 1%, 3% and 5% w/v CH-HMW and 3%, 6%, and 9% w/v CH-LMW were elaborated, respectively. All bioinks were dissolved in 1% glacial acetic acid (Glacial acetic acid 99.5%, CAS: 64-19-7, Sigma-Aldrich) and were mixed at 400 rpm for 24 h at room temperature. For the control bioink, 7% sodium alginate (ALG) (Alginic acid sodium salt from brown algae medium viscosity; CAS Number: 9005-38-3) was used, dissolved in PBS without agitation for 24 h. All bioinks were kept at 4°C for their further use and analysis.
Bioink’s rheological analysis
Rheological analyses of CH-HMW, CH-LMW, and ALG were conducted using a rotational rheometer (TA Instruments Rheolyst AR2000EX, New Castle, USA) with a 25 mm parallel plate. Samples were injected with a syringe onto a preheated plate at 37°C, creating a 1 mm gap. For shear rate evaluation, a shear rate between 0.1 and 300 s-1 was used. For the rotational recovery assay, bioink was submitted to a shear rate of 1 s-1 for 60 s, followed by a shear rate of 120 s-1 for 30 s. This process was performed three times for the higher range and twice for the lower range. For frequency sweep, a 1% frequency was used with frequencies ranging between 0.01 to 100 Hz. Each sample was analyzed in duplicate (n = 2).
CH-HMW, CH-LMW, and ALG 3D scaffold printing
To guarantee bioprintability, a homogenous mix with adequate viscosity and the capacity to maintain its shape after extrusion was selected. Therefore, for assay evaluation, only 3% CH-HMW and 6% CH-LMW, and 7% ALG met these criteria (see the result section). Following, 0.90 mm × 5.00 mm cylindrical constructs were printed with a 0.50 mm × 0.60 mm pore size and a layer height of 0.41 mm, using a mechanical extrusion bioprinter (Regemat 3D V1.0, Spain). Bioinks were placed in 3 cm printing cartridges that were extruded through 0.20″ nozzles. After their printing, their crosslinking was performed using 0.25% v/v glutaraldehyde in a 1:10 ratio (Glutaraldehyde, 25% Aqueous Solution CAS 111-30-8) for 5 min. To minimize cytotoxicity associated with glutaraldehyde, we used a very low concentration, a short exposure time (5 min), followed by three extensive washing steps in PBS to remove residual aldehyde groups. Similar low-dose crosslinking approaches have been reported to improve hydrogel stability while reducing cytotoxic risk. 19 Each sample was disinfected with UV light and stored at 4°C for later use. A total of three scaffolds per condition were printed for downstream analyses (n = 3).
Morphological studies of 3% CH-HMW, 6% CH-LMW, and 7% ALG bioprinted scaffolds
To visualize printed scaffold architecture, the samples were frozen at −80°C for 72 h and subsequently lyophilized for 24 h at −51°C using a Labconco FreeZone 2.5 lyophilizer (Kansas City, MO, USA). Dried samples were covered with gold for their visualization under a Zeiss electron microscope with an acceleration voltage of 25 kV (EVO HD15; Carl Zeiss, Oberkochen, Germany), at 60X and 800X magnification. For pore size evaluation, ImageJ (NIH, USA) was used. Pore size was quantified from three scaffolds per group and three representative images per scaffold (n = 3).
Evaluation of 3D construct antimicrobial activity against E. coli, S. mutans, S. aureus and E. faecalis
Strains of the ATCC bacterial collection (American Type Culture Collection) were used: E. coli (ATCC 12345), S. mutans (ATCC 25175), S. aureus (ATCC 25923), and E. faecalis (ATCC 29212). To evaluate viability, strains in their lyophilized state were reconstituted in Brain Heart Infusion (BHI) and incubated at 37°C for 18 to 48 h. To perform isolates, re-seedings were made from these cultures on BHI agar, which were incubated at 37°C for 18 to 48 h. To confirm their purity, following bacteria colony growth, Gram stains were performed, and biochemical tests were conducted to verify the bacterial isolates. For their antimicrobial evaluation, bacterial suspensions of the four isolated bacteria were prepared in a 0.85% saline solution, and their final concentration was adjusted to 1.5 × 108 CFU/mL according to the 0.5 McFarland standard. The antimicrobial activity of 3% CH-HMW, 6% CH-LMW, and 7% ALG scaffold was evaluated using the diffusion agar methodology (Kirby-Bauer method). To this end, 100 μL of each bacterial suspension was uniformly spread on Mueller-Hinton agar plates and allowed to dry. Following, 3% CH-HMW, 6% CH-LMW scaffolds were placed on the Mueller-Hinton agar. As a negative control, the 7% ALG scaffold was used, and ALG-chlorhexidine was used as a positive control. Scaffolds were disk-shaped and incubated at 37°C for 18 to 48 h. To determine antimicrobial activity after this time, inhibition halos, produced around the scaffolds, were measured in millimeters (mm) using a ruler. Antimicrobial testing was performed using three independent scaffolds per condition (n = 3).
Statistical analysis
For statistical analyses, the data’s normal distribution was first assessed. To determine significant differences among scaffolds and controls, a One-Way ANOVA was performed with a p < 0.05. To record and analyze inhibition halos, Excel software was used. In this manner, this approach facilitated the interpretation of inhibition zone results.
Results
Bioink morphology
From bioinks prepared based on three CH-HMW concentrations, 1%, 3% and 5%, concentrations at 1 and 5% were not used in subsequent experiments for the following reasons: 1% CH-HMW had a very low viscosity, and the 5% bioink did not allow for a homogenous mixture to be prepared. For the CH-LMW at 3%, 6% and 9%; however, 3% and 9% concentrations did not exhibit the properties required for bioprinting by extrusion. Collectively, only two bioinks were suitable for the following assays: 3% CH-HMW (Figure 1(A)) and 6% CH-LMW (Figure 1(B)). Hydrogels prepared with high-molecular-weight (CH-HMW) and low-molecular-weight (CH-LMW) chitosan: (a) 3% CH-HMW; (b) 6% CH-LMW.
Rheological behavior of CH-HMW, CH-LMW, and ALG bioinks
All evaluated bioinks exhibited pseudoplastic behavior, as evidenced by a decrease in viscosity with increasing shear rate (Figure 2(A)). Particularly, CH-HMW bioinks demonstrated a notable reduction in viscosity as the shear rate increased, reflecting an optimal behavior for its extrusion during the process of bioprinting. Rheological analysis of the bioinks. (a) Pseudoplastic flow behavior; (b) Rotational recovery test; (c) Frequency sweep showing storage modulus (G′) and loss modulus (G″); (d) Tan δ values. Each sample was analyzed in duplicate (n = 2). Abbreviations: CH-HMW: high-molecular-weight chitosan; CH-LMW: low-molecular-weight chitosan; ALG: alginate.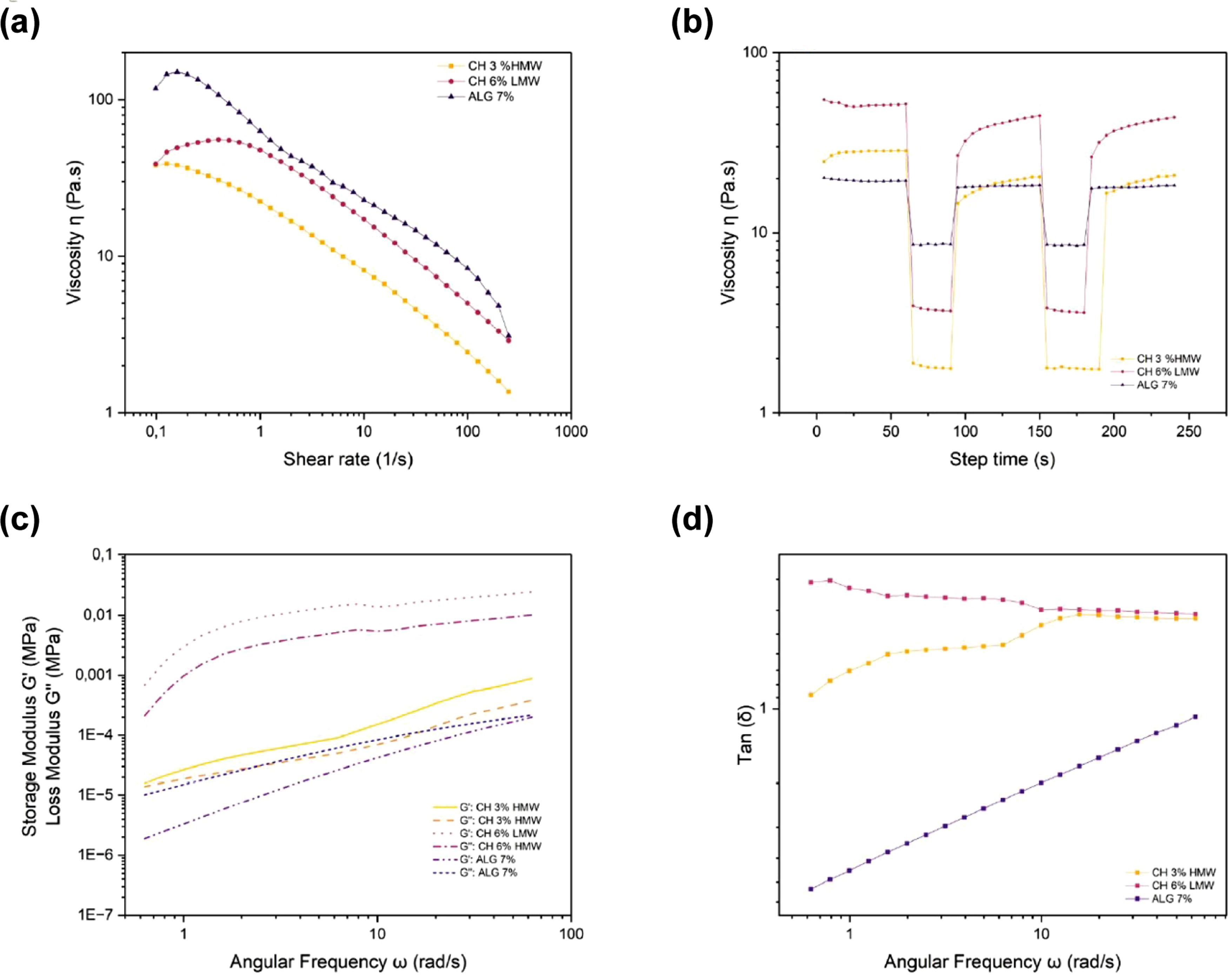
The rheological analysis also determined the minimal stress required for hydrogels to flow. 3% CH-HMW and 6% CH-LMW, presented elastic limits of 79 Pa and 95 Pa, respectively, whereas the 7% ALG hydrogel revealed a lower limit of 25 Pa, suggesting less resistance to flow and a greater ease for deformation under the applied stress (Figure 2(B)).
For the rotational recovery, the 7% ALG recovered 44% of its initial viscosity in 30 s, demonstrating a good restructuring capacity. In contrast, 3% CH-HMW and 6% CH-LMW revealed a limited recovery of 10%, suggesting a lower capacity to return to their initial state after being submitted to stress (Figure 2(C)).
Lastly, hydrogel viscoelastic behavior was evaluated by a frequency sweep, measuring the loss modulus (G″), storage modulus (G′), and the loss factor (tan δ). The storage module G′ represents the elastic component of the material, reflected in its stored energy, whereas the loss module G″ quantifies the viscous components, i.e., the energy dissipated as heat. The loss factor, tan δ, corresponds to the ratio between G″ and G′ (tan δ = G″/G′), indicating the balance between the hydrogel’s viscous and elastic components. Figure 2(D) illustrates that the 7% ALG hydrogel clearly presented a different behavior in comparison with the other hydrogels, demonstrating a more stable viscoelastic equilibrium at higher frequencies. In contrast, 3% CH-HMW and 6% CH-LMW exhibited at high frequencies, a tan δ value greater than one, indicating a more solid behavior.
3D scaffold based on chitosan and alginate printing
The 7% ALG scaffold presented a notably uniform and well-defined structure, with high resolution and geometrical precision, standing out structurally against the two chitosan scaffolds utilized (Figure 3(A)). The 6% CH-LMW scaffold demonstrated a less defined structure with somewhat inferior geometrical structure (Figure 3(B)), although it was able to maintain its stability after the crosslinking process. In contrast, the 3% CH-HMW scaffold presented a more defined structure and better geometrical fidelity in comparison with the 6% CH-LMW scaffolds, although it was not able to achieve uniform pores. These results suggest that alginate offers structural advantages for construct bioprinting, especially in terms of resolution and stability. 3D-printed scaffolds. (a) Control scaffold composed of 7% alginate; (b) scaffold printed with 3% high-molecular-weight chitosan (CH-LMW); (c) scaffold printed with 6% low-molecular-weight chitosan (CH-LMW).
Scaffold architecture visualization
Bioprinted scaffold visualization was carried out using a scanning electron microscope (SEM) with magnifications at 60× and 800×. The 7% ALG construct demonstrated better properties, presenting a pore size of 2169 ± 100.59 µm × 2809 ± 150.34 µm and a smooth surface that remained unaltered after the lyophilization process (Figure 4(A), (D)). In contrast, the 6% CH-LMW scaffold exhibited less porosity, with pore sizes of 1050 ± 213.66 µm × 1853 ± 195.65 µm and a rougher surface featuring small holes, suggesting structural modification associated with lyophilization (Figure 4(B), (E)). Lastly, the 3% CH-HMW scaffold presented pore sizes of 3411 ± 300.65 µm × 1846 ± 169.35 µm, in addition to a surface with multiple porosities and irregular roughness, also altered by the lyophilization process before SEM observation (Figure 4(C), (F)). Scanning electron microscope observations revealed that both chitosan constructs, low as well as high molecular weight, did not maintain optimal conditions, significantly affected by the lyophilization process, compared to the control scaffold (Figure 4). Scanning electron microscopy (SEM) images of 3D-printed scaffolds. (a–c) Surface morphology at 60X magnification: (a) 7% ALG scaffold, (b) 6% CH-LMW scaffold, (c) 3% CH-HMW scaffold. (d–f) Surface morphology at 800X magnification: (d) 7% ALG scaffold, (e) 6% CH-LMW scaffold, (f) 3% CH-HMW scaffold. Each sample was analyzed in triplicate (n = 3). Abbreviations: CH-HMW: high-molecular-weight chitosan; CH-LMW: low-molecular-weight chitosan; ALG: alginate.
Scaffold antimicrobial activity against the four bacteria strains studied
Antimicrobial activity results in four evaluated scaffolds. 7% ALG (negative control), ALG-CHX (positive control), 6% CH-LMW, and 3% CH-HMW.
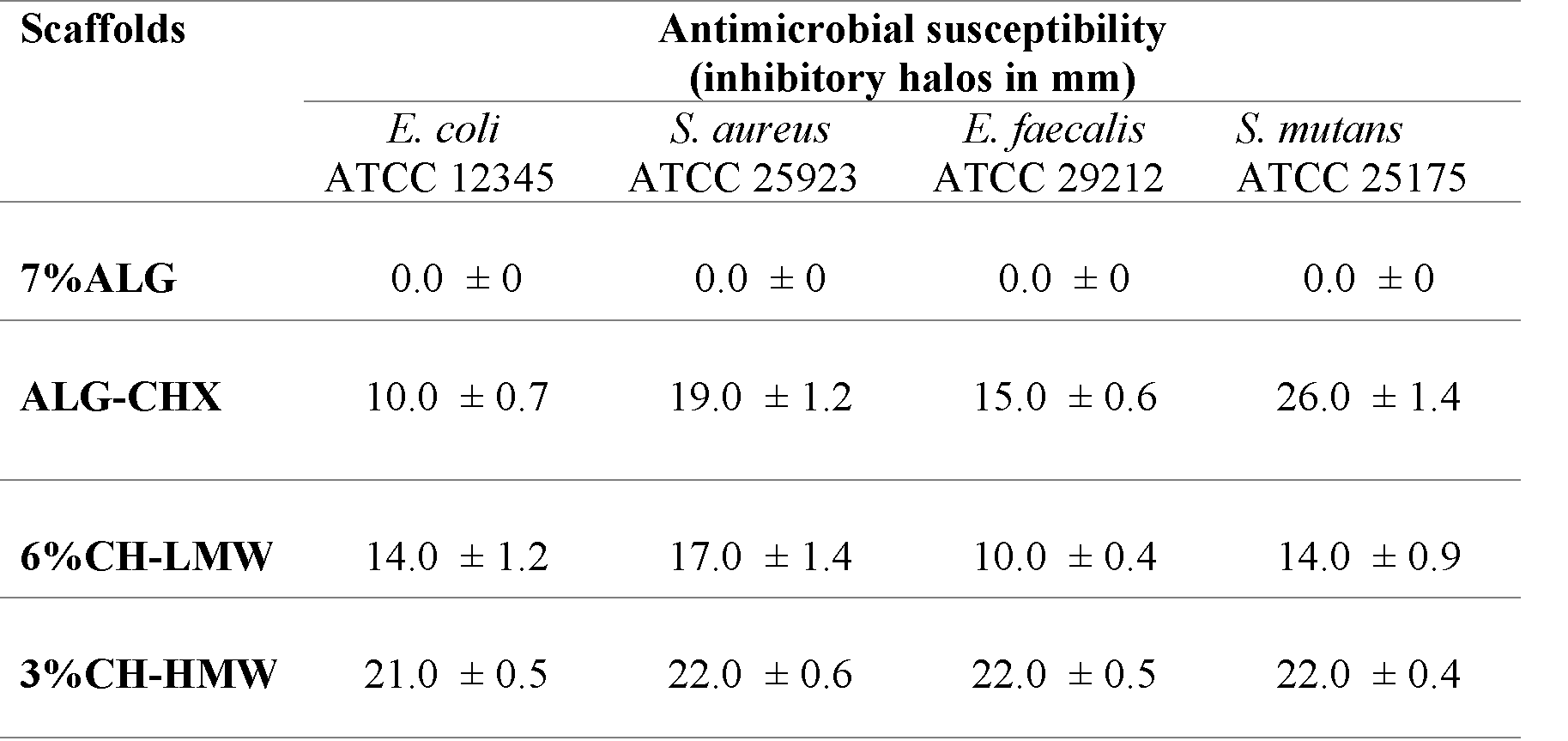
Abbreviations: ALG: alginate; CHX: chlorhexidine; CH-LMW: low-molecular-weight chitosan; CH-HMW: high-molecular-weight chitosan. All tests were made in triplicate (n = 3).

Antimicrobial susceptibility assay showing the effect of four scaffold types (1–4) against four bacterial strains (a–d). (a) Escherichia coli ATCC 12345, (b) Staphylococcus aureus ATCC 25923, (c) Enterococcus faecalis ATCC 29212, (d) Streptococcus mutans ATCC 25175. Scaffolds tested: (1) 7% alginate (negative control), (2) alginate with chlorhexidine (ALG-CHX, positive control), (3) 6% CH-LMW, (4) 3% CH-HMW. Abbreviations: ALG: alginate; CHX: chlorhexidine; CH-LMW: low-molecular-weight chitosan; CH-HMW: high-molecular-weight chitosan.
Discussion
This study revealed that 3% CH-HMW exceeded both 6% CH-LMW and ALG in antimicrobial activity. Therefore, it has great potential in tissue engineering applications to be used in scaffolds with antimicrobial activities. The high molecular weight and antimicrobial properties of chitosan can be beneficial in preventing infections during bone regeneration procedures in the oral cavity. In this environment, regenerations are naturally exposed to a great diversity of bacteria, where CH-HMW offers antimicrobial protection that could significantly improve treatment success by reducing the risk of infections. However, its implementation in bioprinting still presents challenges, due to certain mechanical and structural properties limitations, which underlines the need for adjustments in its formulation to meet the specific demands of biofabricated scaffolds.
Previous studies, such as Coşkun S et al., demonstrated that alginate and chitosan are highly effective materials for bioprinting, given their rheological and physicochemical properties that favor 3D printing of cellular structures. 20 Additionally, alginate facilitates precise printing, enhancing cell-matrix interactions critical for mesenchymal stem cell differentiation in osteogenic applications. 20 That same study demonstrated that alginate in combination with calcium chloride can be used for bioprinting osteogenic tissue grafts with significant therapeutic potential. Furthermore, chitosan hydrogels have been demonstrated to form stable networks with high shape fidelity and printing resolution, allowing for homogenous cell distribution within the matrix, a critical aspect for cell viability. Moreover, optimal printing conditions have been established for these bioinks, making them adequate for bone tissue engineering applications. 20 Both materials enable the creation of high-resolution and functional 3D structures, positioning them as promising options for clinical applications in tissue engineering. Scaffold capacity to maintain cell viability and facilitate effective biological interactions is fundamental for bioprinting success and the development of grafts, as it allows the creation of optimal environments for cell differentiation.
In this context, the results of our study demonstrate that chitosan’s scaffolds, high as well as low molecular weight, maintained their stability only in high pH solutions, which could affect the optimal environment required for such applications. In contrast, the chitosan-alginate scaffolds demonstrated greater versatility across different pH ranges, suggesting they could offer better conditions for biomedical applications in physiological environments. 21 Various factors can explain these differential environments; pure chitosan’s swelling and degradation are associated with protonation of amine/imine groups at the surface of the material, as well as the mechanical relaxation of its coiled chains. 16 To explore this interaction, chitosan was dissolved in 100% acetic acid at different concentrations; however, it was observed that at higher concentrations, low and high-molecular-weight chitosan, 9% and 5%, respectively, did not obtain a homogeneous mix after 24 h, which limited its use in this study.
On the other hand, it has been demonstrated that chitosan-alginate scaffold stability is due to chitosan’s amino group and alginate’s carboxyl group interactions, which prevent chitosan’s amino group protonation. Li Z et al. also confirmed the chitosan-alginate scaffold’s ability to maintain its integrity under different pH conditions, which is crucial for its application in biological environments, where pH variations can significantly influence the material’s performance. 21
Rheological analyses are fundamental to evaluate bioink and hydrogel printability, as they allow for characterization of flow and deformation properties, which are key for bioprinting. These analyses provide detailed viscoelastic properties regarding the bioink, evaluating parameters such as storage modulus (G′), loss modulus (G″), and loss factor (tan δ). These parameters are important to determine the material’s ability to maintain structural integrity during printing, allowing it to adjust to the desired geometry without collapsing. In this study, CH-HMW, CH-LMW, and ALG exhibited a non-Newtonian pseudoplastic behavior. This agrees with El-Hafian EA et al., 22 who described that chitosan in acetic acid solution demonstrated less shear stress thinning, and an augmented viscosity concomitant with increasing acetic acid concentrations. Hwang J et al., 23 also reported that an increase in chitosan concentration intensified the dependence of viscosity on the shear rate, resulting in a more pseudoplastic behavior. This phenomenon can be explained by the level of increased polymer crosslinking at greater polymer concentration, and the degree of individual chain movement, which is restricted by increased crosslinking. In our study, it was observed that when the shear rate was at zero, viscosity was directly proportional to the number of crosslinks in the polymer molecule, suggesting a greater viscosity at a zero value of shear rate, reflecting a more cohesive and crosslinked polymeric structure. Nevertheless, chitosan recovery was not optimal for both molecular types, attaining less than 10% its recovery. Additionally, chitosan’s samples presented the typical behavior of a weak gel, where storage modulus (G′) was greater than loss modulus for all frequency ranges, with a discrete dependency of both modules with respect to the frequency. At low frequencies, G″ was significantly lower than G′; however, with an increase in frequency, G″ rapidly augmented, with values close to G′. In contrast, alginate demonstrated the opposite behavior, which is in agreement with Capitani MI et al.’s findings. 24
Biofabrication of 3D scaffold printing faces various challenges. According to Schwab et al., 25 hydrogels and soft materials are key in bioink elements, presenting limitations due to their structural and viscoelastic stability. These properties make it difficult to create precise and porous constructs, required for effective applications in regenerative medicine. It has been established that bioprinting automatization has improved resolution and reproducibility; nonetheless, the system lacks real-time printing quality evaluation to monitor macro and micro scaffold morphology. 25
Additionally, when designing scaffolds, the following aspects must be considered: 1. Provide a base for cell adhesion, differentiation, proliferation, and cell migration, influenced by pore size and form, the scaffold’s porosity, and surface chemistry; 2. Have an adequate mechanical resistance, and 3. Take the shape of the anatomical part, which is to be replaced. According to Will J et al., 26 bone 3D scaffold porosity should be greater than 40 – 60% to facilitate rapid cell diffusion and nutrient flow, as well as cell transfer. Additionally, Ramos M et al. highlight the importance of pores in 3D collagen and chitosan scaffolds, pointing out that pore design influences key physical properties, such as humidity absorption. This capacity to retain water is essential for the stability and functionality of biological dressings in biomedical applications. 27 It has been demonstrated that scaffold morphology directly affects humidity absorption. High pore density and good interconnection improve this capacity, which is critical for material integration in biological tissues. 27 However, inherent limitations of non-bioink hydrogels make it difficult to effectively assess cell behavior and the viability of printed cells. Moreover, the use of a low-concentration glutaraldehyde solution (0.25% v/v, 1:10 ratio) has been widely reported to reinforce chitosan networks through covalent interchain linkages, thereby improving the mechanical stability and structural integrity of printed constructs.28,29
In our study, the 7% alginate construct presented 2169 ± 100.59 µm x 2809 ± 150.34 µm pores, whereas the 6% CH-LMW displayed a structure with a smaller pore size, 1050 ± 213.66 µm × 1853 ± 195.65 µm. On the other hand, the 3% CH-HMW had 3411 ± 300.65 µm × 1846 ± 169.35 µm pores, in addition to a surface with multiple square-shaped pores. In a similar study, Xu et al. 30 synthesized a novel bioink to generate scaffolds with structural pores that were at least 300 µm, utilizing a mix of Ti6Al4V powders in addition to PS polymer solvent, DCM, and NMP biosolvents. They established that the ink containing 77.5% volume Ti6Al4V powders and biosolvents optimized fabrication precision and reduced solidification time, with pore size ranging between 50 and 600 µm. Their in vitro cell studies revealed scaffolds with 100 µm pores favored adhesion and proliferation. Moreover, they demonstrated superior osteogenic capacities in comparison with a larger pore size. These findings redefine the optimal pore size range for bone implants and suggest a great potential for its application in orthopedics, with foreseen future investigations in animal models. 30
Khajehmohammadi M et al., 31 demonstrated that scaffolds with square pores exhibited greater rigidity and the capacity to support traction forces, making them suitable for applications in tissues requiring mechanical resistance. Furthermore, in their study, which examined different porosity percentages (40%, 50% and 60%) and pore shapes (square, star, and gyroid), scaffolds with square and gyroid pores at 50% porosity demonstrated better cell viability and proliferation, suggesting this range of porosity could be ideal for bone implants. Additionally, scaffold degradation was also highlighted as relevant and varies according to the pore shape, implying that designs could be optimized as a function of the repair time of the target tissue. Moreover, cell viability can also be affected by the scaffold’s geometry. As a case in point, star scaffolds have been demonstrated to be less favorable for cell proliferation, which raises questions about their use in tissue regeneration. This aspect is fundamental, since cell interaction within their 3D environment is crucial for successful regeneration. 26 In our study, a cylindrical scaffold was designed with a height of 0.31 mm and pore dimensions of 2.9 mm × 4.0 mm, aiming to optimize structural and functional properties for bone regeneration applications.
Perez-Puyana V et al., 32 investigated the creation of protein matrices from collagenous gelatin (CG), underlining glutaraldehyde’s role in crosslinking to improve the scaffold’s structure. Its use increased pore size and reinforced the structural network, although its concentration needs to be carefully controlled. Their study suggests a 1:10 proportion (0.1% glutaraldehyde and 1% CG) allows for optimal reinforcement without compromising cell viability, striking a strategic balance to maintain the scaffold’s viscoelastic properties. Considering these findings, in our study, we selected an even higher concentration of 0.25% glutaraldehyde to optimize crosslinking in the bioprinted construct. This concentration resulted in effective improvement of the structural stability of the scaffold, a crucial characteristic that guarantees its mechanical properties.
In the present study, it was observed that bioprinted structures based on chitosan lacked a regular surface and homogeneous pores, which could compromise their functionality. On the other hand, structures elaborated with alginate had pores of uniform size of approximately 2 mm × 2 mm, considered adequate for bone regeneration. Nonetheless, lack of structure uniformity, as evidenced by SEM images (Figure 4(B)–(F)), could negatively affect the optimal characteristics and mechanical resistance of the construct, impeding the scaffold from attaining its desired mechanical performance for bone regeneration applications.
Bioprinted constructs presented a similar behavior to that described by do Amaral Sobral PJ et al., 33 who observed that prepared samples after lyophilization appeared in the form of particles, similar to grains, with rough structures, fractured with irregular forms, and with a fibrous texture, without evidence of a continuous matrix, as was observed in our results. Likewise, do Amaral Sobral PJ et al. 33 noted that in samples prepared with a lower acetic acid concentration, some non-solubilized particles were identified, with a texture comparable to chitosan grains.
Antimicrobial properties of chitosan have been widely documented and are attributed to various mechanisms. According to Pereira-Malacara et al., 34 chitosan interferes with bacterial cell membrane and inhibits protein and nucleic acid synthesis. Additionally, Mawazi SM et al., 14 described how high molecular weight chitosan, due to its more extensive polymeric structure, forms robust 3D networks that improve that enhance the retention and controlled release of antimicrobial compounds, as well as metal ions, which is fundamental to its antimicrobial efficacy. Barbosa et al. 35 also highlight the importance of chitosan’s antimicrobial activity, suggesting that high molecular weight is associated with greater antimicrobial effectiveness.
In the present study, it was observed that 3% CH-HMW, compared to 6% CH-LMW, presented a significantly higher inhibitory activity against the four evaluated bacterial strains. These results are in agreement with reports in the literature that suggest CH-HMW has more extended and stable polymeric networks, which allow for a more effective interaction with bacteria. Additionally, physical properties, such as solubility, basic pH, and adsorption capacity, contribute to its adhesion to the microbial cell wall, which augments its antibacterial activity. 36
In this study, the lower antimicrobial activity exhibited by 6% CH-LMW was due to its shorter and less extensive molecular structure, which limits its capacity to form stable 3D networks. According to Rabea et al., 37 CH-LMW has a reduced capacity to form gels and retain antimicrobial compounds, thereby affecting its effectiveness. Kumar S et al., 38 argue that CH-HMW greater viscosity facilitates a more intense interaction with bacterial cell membranes, allowing for more effective injury to cell structure, altering bacterial metabolism.
The results evidenced in this study confirm 3% CH-HMW is significantly more effective than 6% CH-LMW in inhibiting bacterial growth. Structural differences between the two types of chitosan are fundamental in understanding their distinct antimicrobial efficacy. In addition, molecular weight and viscosity are important in regard to polymer functionality. Furthermore, to advance the development of CH-HMW biomaterials, it is critical to evaluate cell viability, ensuring they possess antimicrobial properties and promote cell adhesion and proliferation. Therefore, for bone regeneration, it is recommended to perform biocompatibility assays to confirm the capacity to support cell growth. Recent studies report that low glutaraldehyde concentrations combined with extensive washing can reduce potential cytotoxic effects. Several works using 0.25% GA in chitosan and chitosan–hydroxyapatite scaffolds showed that this concentration maintains cell viability at levels comparable to non-crosslinked controls, including osteoblasts, fibroblasts, and mesenchymal stem cells.39–43 However, when GA concentration increases to 0.5% or above, biocompatibility decreases significantly, with marked reductions in metabolic activity and viability reported across multiple cell types. These findings emphasize that GA-mediated crosslinking can improve the structural stability of chitosan matrices, but only within a narrow concentration window that requires careful control and extensive washing to remove unreacted aldehyde groups. Therefore, although our study employed a low GA dose (0.25% v/v) and rigorous washing, cytocompatibility must still be validated in future experiments. Moreover, it is essential to optimize the degree of chitosan’s crosslinking, as this factor significantly influences its stability, mechanical resistance, and antimicrobial activity. Future studies should therefore incorporate cytotoxicity and biocompatibility assays (MTT, Live/Dead) using osteoblast-like cells (SaOS-2) together with mechanical characterization to ensure scaffold safety and translational suitability. Lastly, explore combinations of CH-HMW with collagen, hydroxyapatite, or metal nanoparticles, which could enhance and facilitate its tissue integration, paving the way to new possibilities in bone regeneration and biomedical applications.
Conclusions
Collectively, this study demonstrated that CH-HMW bioprinted scaffolds offer a promising alternative for regenerating tissues in the oral cavity, specifically against bacteria such as E. coli, S. mutans, S. aureus, and E. faecalis. A comparison among CH-LMW, alginate, and alginate with chlorhexidine controls showed that 3% CH-HMW demonstrated the greatest antimicrobial activity, a key characteristic in clinical applications, where infections can compromise scaffold integration and treatment success. Hence, 3% CH-HMW is presented as a viable option for designing scaffolds that could improve clinical results by reducing the risk of infections and contribute to safer and more effective regenerative medicine. Although 3% CH-HMW showed the strongest antimicrobial behavior, its mechanical performance must also be addressed, and future studies should include mechanical evaluation and optimization to ensure adequate stability for bone regeneration. Lastly, this work responds to the growing demand for biomaterials that not only promote bone regeneration but also provide antimicrobial protection, a critical factor in oral tissue engineering.
Footnotes
Acknowledgments
Engineer Manuel Ocampo, School of Engineering, Pontificia Universidad Javeriana, Bogotá, Colombia, for SEM visualizations.
Ethical Consideration
This study received approval from the Pontificia Universidad Javeriana School of Dentistry Ethics Research Committee Act 006, 2017.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Pontificia Universidad Javeriana (ID 20551).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
