Abstract
Natural fibres (NF) are becoming increasingly appealing to investigate due to their numerous applications, susceptibility, biodegradability, and ability to offer sustainable products that promote technical invention and a range of industrial applications. In this investigation, NF obtained from Bamboo fibres (BmF) was chemically modified. First, the NF was pre-treated with ethanol (C2H6O), followed by Potassium permanganate (KMnO4) in acetone solution at different circumstances concerning the treatment duration and chemical concentration. Utilizing both treated and untreated fibres, X-ray diffraction (XRD), Fourier transform infrared (FTIR), Thermo Gravimetric analysis (TGA), scanning electron microscopy (SEM), and mechanical characterization were utilized to determine the impact of this alteration on the crystallographic, thermal, macromolecular, morphological, and mechanical properties of the BmF. The XRD scrutiny unveiled significant modifications in the fibre’s crystalline characteristics. The partial elimination of wax, hemicellulose, and lignin was verified by FTIR examination. TGA analysis results demonstrated that the treated fibres were suitable for processing with low-temperature polymers. At optimal conditions, the tensile strength of treated single fibres exhibited 52.17% improved strength over pristine BmF, and surface roughness was attained according to SEM morphology. This study has demonstrated that at optimal treatment conditions, the treated BmF has the potential for usage as reinforcement in a variety of bio-composites for distinct industrial and advanced applications.
Introduction
NF has been a vital component of human society for generations, providing materials for textiles, construction, and other applications.1–5 Owing to their prospective uses in sustainable sectors and their environmental friendliness, these NF have lately attracted renewed study.6,7 With a diverse spectrum of characteristics, these NF are obtained from plants, minerals, and animals.
The biodegradability breathability, and comfort of plant-based fibres including jute, cotton, hemp, and flax are well-known attributes.7–9 NF offers various advantages over synthetic counterparts. They are biodegradable and renewable, reducing the environmental impact associated with disposal. NF is also less energy-intensive during production, contributing to lower carbon emissions. These fibres find applications in textiles, packaging, composites, construction, high-performance composites, and even medical fields.10–13 The processing of NF involves several stages, including harvesting, retting (for plant fibres), cleaning, and spinning.14,15 Mechanical, chemical, and enzymatic retting methods are employed to extract fibres from plant sources. Mechanical methods involve physically breaking down the plant tissues, whereas chemical techniques depend on dissolving the binding components with chemicals. One of the primary rationales for the resurgence of interest in NF is their eco-friendliness.14,16,17 Synthetic fibres, such as Polyester, Rayon, Nylon, Acrylic and Aramid, are derived from petrochemicals and contribute to microplastic pollution. NF, on the other hand, decomposes naturally without releasing harmful substances. The cultivation of NF can also positively affect soil quality and carbon sequestration, further enhancing their environmental credentials. NF treatments can improve surface roughness, minimize moisture absorption mechanisms, remove or minimize surface contaminants, and modify the surface chemically. Additionally, the amount of independent hydroxyl groups in cellulose is reduced, which lessens the contrariness of the molecules in the cellulose and improves their conformity with the matrices.18–20
To enhance NF properties and performance, researchers have explored different pre-treatment methods. Ethanol pre-treatment is one such technique that offers advantages in improving fibre characteristics and compatibility with matrices. The process aims to remove impurities, waxes, and lignin present on the fibre surface, enhancing fiber-matrix adhesion and mechanical properties.21,22 Ethanol is chosen for its propensity to dissolve both non-polar and polar compounds, effectively cleaning and modifying fibre surfaces.23–25
In comparison to other well-known natural fibers like jute, coir, sisal, straw, and banana, BmF offer higher specific strength and stiffness in plastic materials, which is why their anatomical characteristics, ultrastructure, and plant breakage process are generating increasing interest in them. 11 Permanganate treatment of BmF holds promise for enhancing its properties and expanding its range of applications. The treatment affects the surface roughness, wettability, mechanical and properties with implications in absorbent, textiles, and composites materials.26–28 Permanganate treatment significantly modifies the characteristics of NF, impacting colour, surface properties, and chemical composition. The permanganate treatment can be employed to modify a range of NF, including banana, cotton, bamboo, jute, linen, plantain, and hemp.27,29–31 BmF has emerged as a remarkable and sustainable alternative to traditional textile materials, offering a range of unique properties and environmental benefits. BmF has garnered considerable attention as an eco-friendly and sustainable alternative to traditional textile materials owing to their advancement and minimal environmental effect.32–35 BmF finds applications in various products such as clothing, packaging, architecture, and construction.25–34
In this study, ethanol pre-treated permanganate BmF was prepared at different permanganate concentrations and duration. The primary objectives of these actions are to isolate the technical fibres by removal of non-structural debris from the BmF bundles and evaluate the impact of these changes on the fibre’s crystallographic, thermal, macromolecular, morphological, and characteristics. The mechanical properties via tensile strength were also evaluated. KMnO4 and ethanol were selected as treatments for this study due to their effective surface modification properties. KMnO4 is a potent oxidizing agent that introduces hydroxyl and carboxyl functional groups onto the fibres surface, improving interfacial adhesion with the polymer matrix. Ethanol acts as a solvent to remove surface impurities, waxes, and residual contaminants, ensuring better chemical interaction. Alternative treatments such as NaOH 36 can enhance surface roughness but may weaken the fibres at high concentrations, while silane coupling agents improve compatibility by forming siloxane linkages. Acid treatments (e.g., HCl, H2SO4) can remove amorphous components but risk excessive degradation, whereas enzyme-based therapies offer an eco-friendlier approach with controlled surface modification. Considering these factors, KMnO4 and ethanol were chosen for their balance between effectiveness and minimal fibres damage.
Materials and methods
Bamboo fibres were supplied by Go Green Products, Valasaravakkam, Chennai. Tamil Nadu, India (Figure 1). KMnO4, ACS reagent, ≥99.0%, Acetone ACS reagent, ≥99.9%, and absolute ethanol were purchased from Sigma-Aldrich (Merck). Bamboo fibres (a) Pristine BmF (b) Treated BmF samples.
Sample preparation and permanganate treatment
Treatment parameters for BmF.

Characterization
The consequence of the chemical modification on the crystallographic structures of the BmF was investigated via XRD, engaging a Empyrean X-Ray diffractometer. The angle of diffraction was scanned from 5 ° to 90 °. There are several methods available to determine crystallinity index (CI).37–41 Some of these methods include the Segal method,
42
Ruland –Vonk method,
43
and Rietveld refinement method.
44
del Cerro, et al., (2020), reported CI is calculated by deconvolution (pseudo-Voigt fitting) of the 1D X-ray diffractograms.
45
In the XRD analysis, X-ray spectra were fitted using Origin software. Equation (1) is utilized to calculate the crystallinity index (CI), which was suggested by Segal et al., (1959). After subtracting the background signal, the height ratio between the crystalline peak’s intensity and the overall intensity was used to compute the CI as previously describe.
40
The Debye-Scherrer equation was utilized to calculate the crystallite size from equation (2).46,47

The variables λ, CuKα = 1.5418 Å, β is the full width at half maximum (rad) of the X-ray diffraction peaks, θ is the Bragg angle of the XRD peak, and K is a constant, usually equal to 0.89.46,47
Chemical compositions were identified by FTIR spectroscopy (PerkinElmer Spectrum version 10.03.02). density was determined using a pycnometer. TGA investigation was performed using a thermal analyser (NETZSCH STA 449F4) at a range of 20°C/20.0(K/min)/500°C. A SEM microscope (TESCAN VEGA3 XMU) was exploited to study the surface morphology of the BmF. An MTS tensile testing apparatus was employed to do single fibres tensile testing.
Results and discussion
XRD analysis
Figure 2 exhibits the XRD spectra of treated and untreated BmF. The diffraction peaks with Miller indices were located at the 2θ of 14.91°, (1-10), 16.31°, (110), 22. 35° (200) and 34.65° (004). These are diffraction peak regions for cellulose I.45,48 However, peaks 14.91° and 16.31° are so broad and close that they overlap.
49
The crystallinity index, crystallite size, and density are shown in Table 2. It was revealed that the concentration and treatment time of KMnO4 affect the CI. Similar studies have reported that this might be owing to the elimination of the native wax, surface impurities and amorphous part of the BmF, which improves the proportion of the crystalline portion.50,51 Xrd spectral of treated and untreated BmF. Physical and crystallographic parameters of treated and untreated BmF.

However, the highest value for crystallinity index, crystallite size, and bulk density for 0.01 % concentration was observed at 2 min treatment, while at 0.05 % concentration, 1 min treatment gave the highest, and for 0.10 % concentration, 1 min of treatment gave the highest values. This can be ascribed to the partial removal of cellulosic constituents alongside lignin at different concentrations and times.50–52
FTIR analysis
FTIR data of treated and untreated BmF are revealed in Figure 3. The broadband exhibited at 3321 cm-1 and 1637 cm-1, is assigned to the intricate vibrational stretching of hydrogen-bonded hydroxyl groups (O-H), linked to free, intermolecular, and intramolecular bound hydroxyl groups.
53
The peak at approximately 2900 cm-1 is attributed to the C-H Stretching vibration of the alkane group in carbohydrates and lignin existing in cellulosic BmF.54–56 Robust adsorption caused by nitrile (C-N) stretching was observed between 2200 cm-1 to 2335 cm-1.56,57 Significant changes in this absorption band after treatment can be observed. The Carbonyl C = O Stretching characteristic bands of hemicellulose in NF are noticed at around 1739 cm-1. Peak 1625 cm-1 – 1637 cm-1 represents the C = O bonds on hemicellulose. The increase of this peak intensity after treatment suggests the introduction of an ester group in the fibers after treatment.
58
The peak between 1500 cm-1 -1400 cm-1 is attributed as the characteristic peak of lignin in NF. This peak was observed to reduce after treatments. This is an indication of effective modification of the lignin presence in the fibres.59–61 The band located between 1430 cm−1 and 1490 cm−1 signified the presence of OH groups and the OH stretching caused by moisture present in BmF. The intensity of these peaks was observed to reduce after treatment, indicating the treatment was effective in reducing the moisture content thus improving the hydrophobicity of the BmF.60–63 The C-O Stretching C-vibration of the acetyl group in lignin was observed at 1242 cm-1 to reduce in treated BmF, indicating the partial elimination of lignin from the BmF surface. The most prominent peak in all BmF was observed at 1033 cm-1, this peak is ascribed to the aromatic ring’s C-H and C-O groups’ bending vibration in the polysaccharides found in NF. The increases in this peak intensity after treatment are an indication of the chemical reaction between the fibres and permanganate compound.
12
The absorption band at 896 cm-1 and 649 cm-1 is attributed to C-OH bending. It showed a decrease in the alcohol groups’ major C–O stretching and deformation in cellulose and the aromatic hydrogen groups’ significant C–OH out-of-plane bending in lignin.64,65 The variations in these peaks’ intensities show that the treatment was successful in modulating the BmF. The variations in these peaks’ intensities suggest that this procedure has effectively modulated the BmF. Ftir spectral of treated and untreated BmF.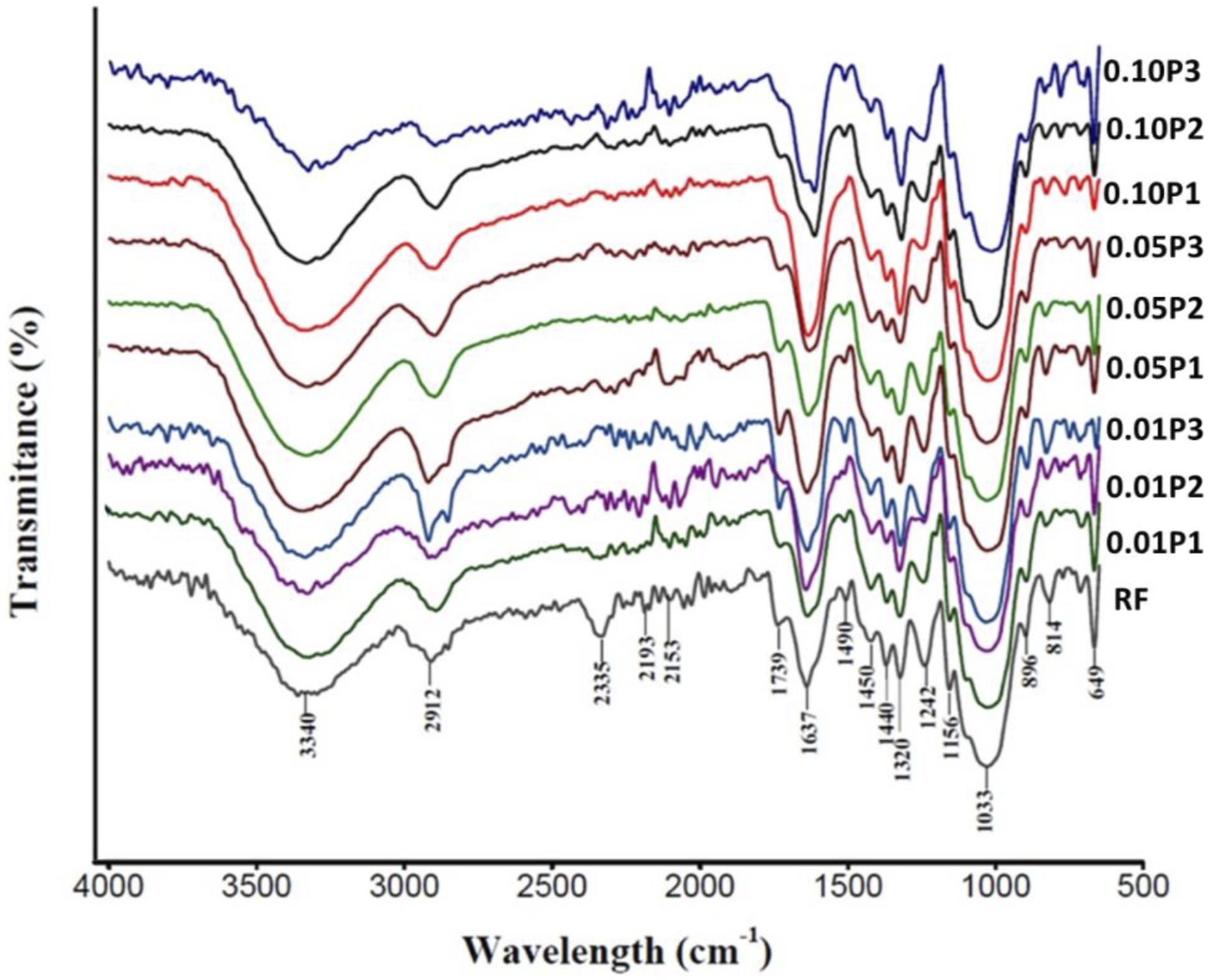
Thermo Gravimetric analysis
To explore the thermal stability of treated and untreated BmF at different concentrations and times, TGA was utilized. The TGA and derivatives curves (DTG) for untreated and treated BmF are demonstrated in Figures 4(a)–6(a). Three decomposition stages were noticed in the TGA spectral, the first stage (26°C −70°C) was owing to primary changes occasioned by moisture evaporation on the BmF surfaces. The second decomposition stage (220°C – 350°C) showed a significant weight loss. The degradation of hemicellulose and cellulosic portions occasioned this. Hemicellulose has been reported to begin breaking down at 220°C and kept going till 300°C.66–68 The onset of the second decomposition stage differs for each group of treatments. As observed in Figure 4, second-stage degradation started at 230°C, while in Figure 5 started at 220°C, and in Figure 6 onset was observed at 222°C. The dissimilarities in hemicellulose, cellulose, and lignin’s chemical structures after chemical modification, cause them to break down at varying temperatures. Comparable outcomes have been documented.66,69 The final stage degradation (350°C −500°C) resulted from cellulose and lignin decomposition. As attested by previous studies, thermal analysis of cellulose degradation began at 310°C and continued until 500°C.
70
When the temperature rose above 380°C, almost all the cellulose decomposed, and there were few solid leftovers. Analysis of 0.01 % KMnO4 BmF at different treatment times (a) TGA (b) DTG. Analysis of 0.05 % KMnO4 BmF at different treatment times (a) TGA (b) DTG. Dtg analysis of 0.10 % KMnO4 BmF at different treatment times (a) TGA (b).
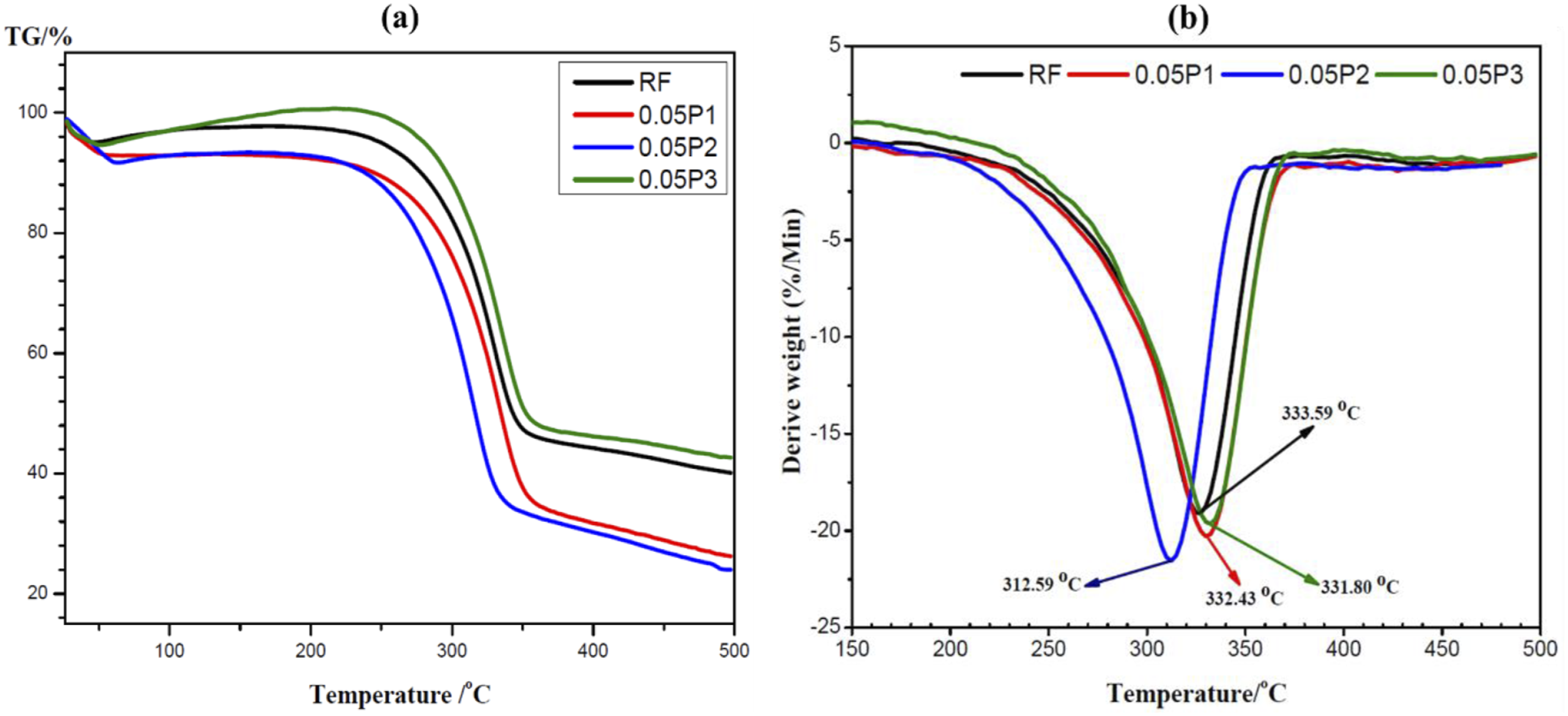

DTG analysis of treated and untreated BmF.

Tensile strength of BmF
The ultimate tensile strength (UTS) of single filament BmF was performed in triplicate, and the average data for each sample are displayed in Figure 7. The UTS of treated BmF showed improvement from untreated ones as the concentration and treatment time increased. This is attributed to removing natural and artificial impurities and effective oxidation of the fibres surface after treatments. As observed, when treatment time increased from 1 to 3 min, there were corresponding increases in UTS at 0.01 to 0.05% treatment concentration. However, as the treatment concentration increased to 0.1%, degradation in UTS was observed. This was consistent with results obtained from previous studies, which were attributed to delignification caused by excessive treatment of the fibres at high concentrations or prolonged treatment time of KMnO4.10,75–78 Maximum tensile strength at 0.01 % concentration was obtained after 2 min of treatment time, maximum UTS at 0.05 % concentration was obtained after 1 min of treatment, and at 0.10 % concentration, maximum UTS was obtained after 1 min of treatment. This was attributed to the effective improvement of the strength-bearing cellulose after treatment.75,76 These findings collaborate also with observations in XRD and thermal analysis in this study. However, higher concentrations and prolonged treatment of the fibres with KMnO4 cause excess delignification of the fibres structure and degrade its strengths.10,77,78 Ultimate tensile strength of treated and untreated BmF at different treatment times.
SEM analysis
Ethanol pre-treated permanganate BmF with improved thermal, micromolecular, and crystallographic structures from this study was selected for SEM morphology. The SEM morphological structure of selected BmF is exhibited in Figure 8. As observed, the smooth surface of the untreated BmF has presumably been encrusted with waxes and other contaminants. Displayed in Figures 8–11 is the optimal surface roughness achieved for each treatment concentration level. It was observed that BmF’s surface roughness amplified at different concentrations and times. This can be ascribed to the partial removal of lignin, hemicellulose, and other surface contaminants after permanganate treatment, as reported in previous studies.10,79,80 The BmF treated with 0.05 % concentration for 2 min exhibited the highest level of surface roughness (Figure 10), which could be attributed to the significant elimination of contaminants and partial extirpation of hemicellulose from the BmF, resulting in noticeable fibrous area. High fibres surface roughness has been reported as a potential for usage as reinforcement in a variety of bio-composites.29,75,77,79 SEM image of RF BmF. SEM image of 0.01P1 Treated BmF. SEM image of 0.05P2 Treated BmF. SEM image of 0.10P1 Treated BmF.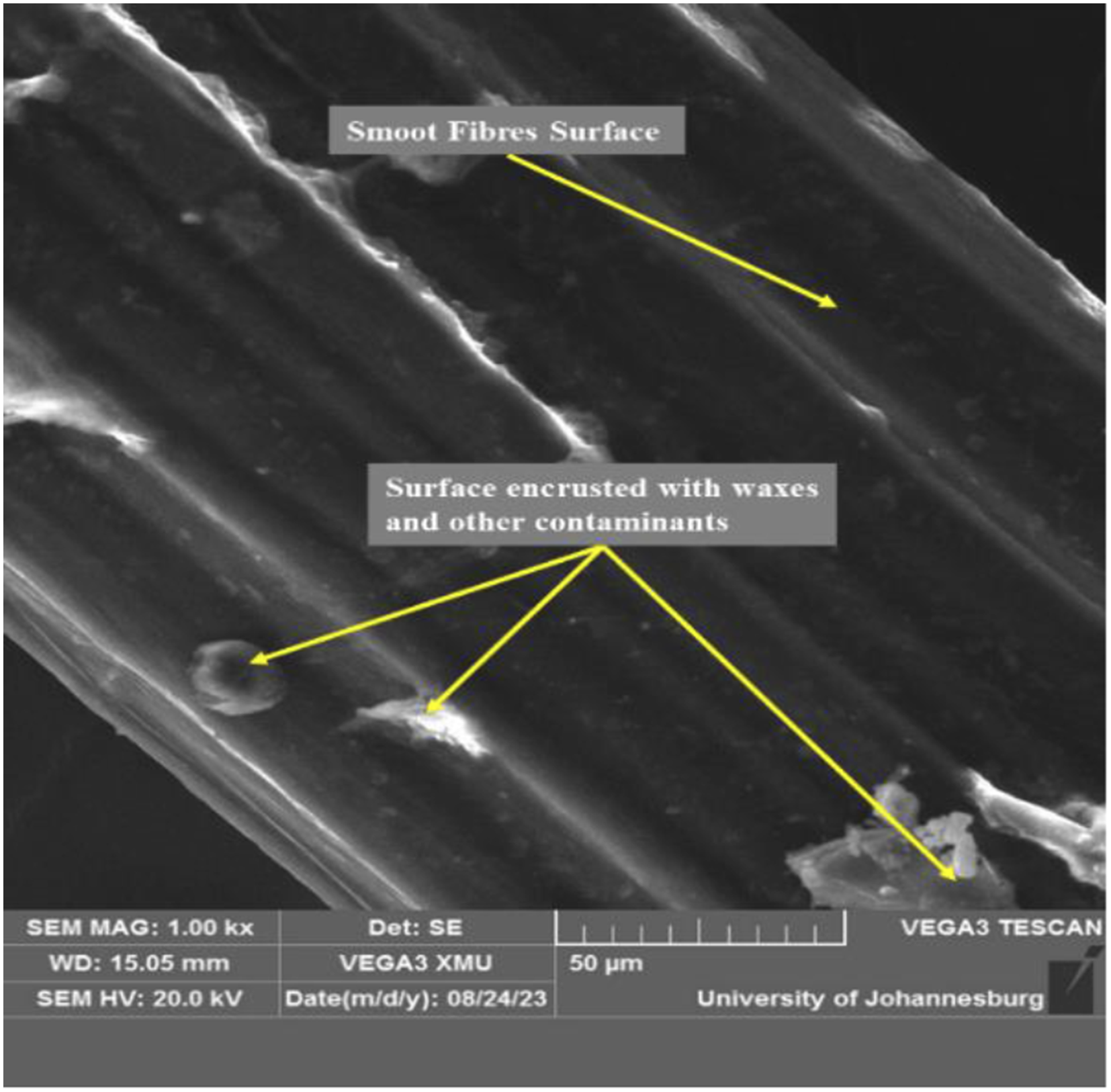



Conclusion
An investigation has been conducted to evaluate the suitability of pre-treating BmF with ethanol before applying KMnO4 treatment at varied concentrations and treatment durations. The chemical alteration impacted the BmF’s parameters, crystallographic structures, and macromolecular characteristics. Following treatment at ideal conditions of 0.05P2, improvements in bulk density, degree of crystallinity, crystallite size, and improved tensile strength were noted. The surface of the BmF altered following treatment, as evidenced by SEM micrographs, becoming rougher. FTIR spectra verified the treatment’s reduction of the hemicellulose and lignin groups existing in the BmF. Less thermal stability of these treated bamboo fibres suggests the treated fibres as a good choice for compounding with polymers as a fibre-reinforced composite that can be processed at a lower temperature. Therefore, these results demonstrate the potential of the analysed treated fibres for use as reinforcement in various bio-composites for industrial and advanced applications.
Footnotes
Acknowledgment
The authors would like to appreciate the support from the National Research Foundation (NRF) of South Africa and also the University of Johannesburg.
Consent for publication
All authors consent to the publication of this manuscript.
Author contributions
Conceptualization: Mamookho Elizabeth Makhatha Methodology: Mamookho Elizabeth Makhatha Patrick Ehi Imoisili. Formal analysis and investigation: Mamookho Elizabeth Makhatha Patrick Ehi Imoisili; Writing - original draft preparation: Patrick Ehi Imoisili; Writing - review and editing: Mamookho Elizabeth Makhatha and Tien-Chien Jen, Funding acquisition: Mamookho Elizabeth Makhatha; Resources: Mamookho Elizabeth Makhatha Patrick Ehi Imoisili, Tien-Chien Jen. Supervision: Mamookho Elizabeth Makhatha and Tien-Chien Jen.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The corresponding author will provide raw data upon request. Data will be made available on request.
