Abstract
Wound dressings used in medical applications serve as a protective barrier for wounds, creating an environment that promotes wound healing. The typical traditional wound dressings often present challenges, including weak barrier function and tendency to adhere to wounds. Therefore, there is a critical need to develop advanced wound dressing with strong antibacterial properties that can prevent the spread of infection and accelerate the healing process. To develop hydrogels with antibacterial properties, silver nanoparticles (AgNPs) derived from 1 mM AgNO3 using a green catalyst matcha green tea extract (camellia sinensis) were incorporated into chitosan and alginate This was accomplished by mixing silver nitrate and green tea extract to produce the nanoparticles, which were then added to the hydrogel using crosslinkers. The synthesized nanomaterials and hydrogels were characterized using UV-VIS spectroscopy, TEM, EDX, FTIR, and SEM techniques. Their antimicrobial efficiency has been evaluated through inhibition zone assays, antibiofilm evaluation against P.aeruginosa, E. coli, S. aureus, C.albicans, and A.niger, and minimum inhibitory concentration (MIC). It was found that, novel hydrogel, infused with AgNPs, exhibited superior water absorption and retention, ensuring a moist wound environment while effectively absorbing a substantial wound exudate. Additionally, the antimicrobial hydrogel demonstrated robust mechanical properties and potent antimicrobial activity. This study highlights the exceptional antibacterial properties of a newly developed natural polymer hydrogel incorporated with Ag nanoparticles with anti-inflammatory action. Hence, the silver-loaded hydrogel proves to be an ideal wound dressing material.

Introduction
The human skin is the largest organ in the body, it plays a crucial part in numerous dynamic processes that sustain homeostasis. Skin is made up of several cell types, acts as an excellent barrier to protect us from environmental insults like sun exposure, direct contact with chemicals, microbes, and physical damage. 1 As a result, it is frequently damaged. When there is damage to the skin and it cannot heal properly, the injury will result in numerous local or even systemic issues, including potentially fatal immune system disorders, bacterial infection, elevated metabolism, excessive protein and water loss, and increased metabolism. 2 Wound dressings can offer temporary protection to replace damaged skin. It can prevent or manage wound infections and offer an environment that is conducive to healing. Cotton balls and gauze are the most popular wound dressings; however, these conventional wound dressings have inherent flaws, like quick wound adhesion and inadequate barrier performance. 3
Wound healing, especially chronic wound cases, remain a great challenge in modern healthcare. Chronic wound care creates great barriers in most health care institutions; with costs of treatment estimated to be in the billions of dollars every year, across the globe. The perfect signs of Wound Dressings are capable of acting as a physical barrier to the external environment while at the same time it is to minimize the concentration of bacteria in the wound site and enable the exchange of gasses and moisture while at the same time promoting the reformation of new tissue. Other dressings used in the traditional system failed to satisfy these qualities, especially in chronic wounds where coagulation, proliferation, and maturation are slow due to inflammation and poor blood flow and infection from high bacterial bioburden.1,2 Generally, chronic wounds fail to proceed with their normal stages of healing due to some underlying causes such as infection, inflammation, or poor blood circulation. 4 Such kinds of wounds are quite susceptible to bacterial and fungal infections. 2 While conventional wound dressings are quite successful in covering and protecting wounds, they fail when it comes to the management of infections and establishing an optimum environment that facilitates the wound-healing process. Recent biomaterials advances have involved the use of materials such as hydrogels with incorporated antimicrobial agents, like AgNPs. 5 Adding to these agents like matcha green tea extract, antioxidants, and anti-inflammatory agents enhances the dressings. This combination not only copes with the infection problem but also favors a good environment for chronic wound healing, enhancing tissue regeneration and reducing inflammation. 6
Infections are one of the factors that limit wound healing, and these occur given the fact that there’s poor blood supply and the existence of pathogenic bacteria in the affected area.3,7 Since the skin barrier has also been impaired and exudate accumulates, it creates a perfect environment for bacterial growth. S. aureus is classified as a Gram-positive bacteria and is commonly found in various sites throughout the body that can be as small as skin rash infections, possibly pneumonia and bacteremia.8,9 The development of antibiotic resistance, particularly Methicillin-resistant Staphylococcus aureus (MRSA), has become a significant concern in global healthcare. 10 Additionally, Gram-negative bacteria such as Escherichia coli and Pseudomonas aeruginosa, along with opportunistic fungal pathogens like Candida albicans, pose serious threats to wound healing, especially when they form bacterial biofilms that complicate infections. Given the complexity of wound healing, which involves multiple phases including accelerated healing, infection control, and tissue repair, there is an urgent need for safe and effective wound healing agents.11,12
Recently, silver nanoparticles have become one of the leading approaches in injury investigation.1,6,11 AgNPs enhance the effectiveness of biomaterials against microbial attack through their antimicrobial properties. 12 Moreover, green-synthesized silver nanoparticles demonstrate multiple therapeutic benefits that make them particularly suitable for wound healing applications.13,14 Their biocompatible and environmentally friendly nature, combined with enhanced surface properties, enables precise and effective drug delivery to targeted areas with minimal harmful effects.15,16
Green-synthesized silver and gold nanoparticles have shown antinociceptive activity by reducing pain sensitivity. This has been demonstrated in animal studies using the acetic acid-induced writhing test in mice, where bioengineered AgNPs synthesized from plant extracts of Prunus domestica effectively reduced nociceptive pain.14,15 Additionally, these biologically engineered nanoparticles, synthesized by eco-friendly methods, exhibit anticoagulant effects that offer promising possibilities for cardiovascular disease prevention.5,6 Further expanding their therapeutic potential, AgNPs synthesized using Saccharomyces cerevisiae have demonstrated significant antidiabetic activities through their ability to control glucose levels.4,14,17
Hydrogels are three-dimensional networks of hydrophilic polymer chains that can absorb and retain large amounts of water while maintaining their structure.3,8,9 Hydrogel can be synthesized either from natural and/or synthetic polymer.2,4 Hydrogels are multifunctional and could be applicable for wound healing acceleration, because it has the potential for reducing inflammation and oxidative damage. 18 The gel extracted from the natural herbal sources showed a good antibacterial properties and wound healing applications. 19 Seaweed is a known natural source of sodium alginate, which is a biocompatible and biodegradable polymer. 20 Sodium alginate is a non-toxic, biodegradable natural polymer that can be obtained from seaweed and forms a gel layer on the surface of the mucous membrane and releases the drug gradually for a long time. 20 Similarly, sodium alginate hydrogels would also prevent microbial invasion of the wound site and ensure gaseous exchange together with the retention of moisture for optimum wound healing.21,22 Chitosan, a natural polymer derived from shellfish exoskeletons (crab, lobster, and shrimp), serves as an excellent backbone for natural hydrogel preparation.23,24 When fabricated into a bioactive hydrogel, chitosan nanofibers demonstrate significant antioxidant properties and enhances the wound healing process. 25 In addition, chitosan nanocomposite loaded with metallic nanoparticles; especially antibacterial nanoparticles make the candidate suitable for therapeutic application. 26 Moreover, biopolymers-based hydrogels cellulose-alginate hydrogels are also widely studied for biomedical applications particularly for wound healing.25–27
Over the last ten years, green synthesis methodologies-using plant extracts as reducing and stabilizing agents-have gained momentum as environmentally friendly techniques against the chemical synthesis of AgNPs. 23 Matcha green tea is extremely rich in polyphenols and may be considered as a natural source for synthesizing AgNPs by providing antimicrobial efficacy with biocompatibility. 6 Furthermore, matcha green tea extract (camellia sinensis) has some extra functionalities in facilitating the wound healing procedure meaning it has enhanced antioxidant and anti-inflammation features.28,29 Green tea extract was chosen as an excellent reducing and capping agent for silver nanoparticle synthesis due to its high content of catechins, which are compounds known for their potent antioxidant and antibacterial properties.28,29 Correspondingly the stated medicinal benefits of matcha green tea, the polyphenolic compounds that are present in matcha green tea extract have other health benefits, including facilitating rapid wound healing; they are anti-inflammatory, they support the process of tissue regeneration. 30 Moreover, the antimicrobial activity of Ag-NPs also depended upon the matcha green tea extract integrated with Na-alginate matrix. The chitosan-alginate hydrogel not only encapsulates but also controls the release of Ag-NPs at the injury site over an extended period.31,32
The presented dressing material based on matcha green tea extract silver nanoparticles (MGEAg-NPs) and natural hydrogel composed of (chitosan-alginate) is developing as a promising nanocomposite material owing to the improvement of the traditional Ag-NPs formulations. 33 In addition, enhancing the antioxidants and anti-inflammatory properties of the matcha green tea extract and sodium alginate may also stimulate the mechanism of wound healing.4,16,34
The current study aims to investigate the potential synergistic effects of silver nanoparticles (Ag-NPs) derived from matcha green tea when embedded in a novel natural hydrogel composed of alginate and chitosan. This study also assesses the effectiveness of this amalgamated hydrogel in controlling wound infections. Additionally, the physicochemical properties of the Ag NPs and the produced novel hydrogel, including size, shape, and composition, are characterized thoroughly.
Materials and methods
Materials
Chitosan (Ch) molecular weight 100–300K (Acros Organics, US), and alginic acid sodium salt from brown algae (Al) “sodium alginate” (Routh, Germany). N,N'-methylenebisacrylamide (NMB) (Fluka, Germany), Ammonium persulfate (APS) (Merck, Germany) were used as crosslinker and initiator, respectively. Matcha Green Tea (Imtenan company, Egypt). Silver nitrate ACS (AgNO3). Acetic acid, ethanol and calcium chloride, (El Nasr Pharmaceutical Chemicals Co., Egypt).
Test strains were obtained from Microbiology and Biotechnology Department, National Research Centre (NRC), Egypt. Five clinical microbial species were used in vitro antimicrobial tests after being identified by the VITEK 2® version 9.02 system. Gram-negative bacteria, such as P. aeruginosa (ATCC 27853), and E. coli (ATCC 25922), are examples of these species. Gram-positive Staphylococcus aureus (ATCC 23235). Both the yeast strain Candida albicans (ATCC 10231) and the mold strain Aspergillus niger (ATCC 16404) were used. All test strains (Oxoid, England) were maintained at −80°C in tryptic soy broth containing 20% glycerol. The bacteria were grown aerobically for 24 hours at 37°C after being spread out on blood agar.
Synthesis and characterization of silver nanoparticles
Green synthesis of silver nanoparticles using matcha green tea
For the preparation of the matcha green tea extract as illustrated in Figure 1, one g of dried matcha green tea powder (Camellia sinensis) was added to 50 mL of distilled water and stirred at 70°C for 5 minutes. Then, the mixture was allowed to cool to ambient temperature, and the extract was obtained using vacuum filtration. The green synthesis of Ag-NPs was carried out as follows: 50 mL of the matcha green tea extract was transferred to an Erlenmeyer flask and placed in an ice bath with continuous 200 r/min stirring. The extract was swirled and cooled to room temperature. An aqueous solution of 1 mM silver nitrate (AgNO3) was prepared. This solution was then added dropwise (1 drop/sec) into the cooled extract under moderate stirring.
35
The Ag-NPs solution was centrifuged at 10000 r/min for 20 minutes to separate the nanoparticles from silver ions and other particles. All free silver cations were removed in the supernatant fluid while the bluish pellet within the tube containing the Ag-NPs was retrieved and rinsed several times with distilled water to eliminate remaining free silver ions. A schematic process of silver nanoparticles synthesis.
Silver nanoparticles characterization
Ultraviolet-visible spectrophotometer
An ultraviolet-visible spectrophotometer (UV-VIS, Evolution 300, Thermo Scientific, USA) was used to verify the formation and stability of Ag-NPs. It measured the spectra of the SA-Ag solution at 300–500 nm.
Transmission electron microscopy
Transmission Electron Microscopy (TEM) (JEOL JEM-2100) with an accelerating voltage of 200 kV was used. Moreover, field emission scanning electron microscopy (FESEM, Quattro S, Thermo Scientific) was employed to examine the surface and morphology of materials that have been prepared. To prepare the samples, a droplet of colloid suspension was placed on a Formvar carbon-coated, 300-mesh copper grid (Ted Pella), and the droplet was allowed to evaporate at room temperature.
Synthesis and characterization of hydrogel
Synthesis of hydrogel
1 g chitosan was added to 70 mL of 1% (v/v) acetic acid solution and was stirred overnight at room temperature until a clear solution was obtained. Separately, 1 g of sodium alginate was added to 70 mL of distilled water and stirred for 5 hours at room temperature. Both chitosan and alginate solutions were added together and mixed for an extra 1 hour to produce chitosan-alginate blend. This blend has been divided into two equal parts. In the first part, 0.1 g NMB and 1 g of APS were added to the polymer blend solution, and the resulting hydrogel was named as Chitosan-Alginate (Ch-Al). In the second part, 10 mL Ag-NPs, 0.1 g of NMB, and 1 g of APS were added to the polymer blend solution, and the resulting hydrogel was named Ch-Al-AgNPs. The two samples were ultrasonicated for 30 min to remove air bubbles and then poured into a pottery dish. The resulting samples were completely dried at room temperature. After that, the samples were taken out and soaked in deionized water (change the water every 6 h/ 6 time for the removal of excess reagents and byproducts.
Characterization of hydrogel
Scanning electron microscope
The pore structure of the two hydrogel samples was observed using a scanning electron microscope (SEM, Quanta 250, FEI, USA) in order to assess the prepared sample’s percentage silver content. After applying liquid nitrogen to the hydrogel to create a cross-section, it was freeze-dried. Gold spraying was done on the samples before they were observed. Prior to imaging, the hydrogel samples were carefully dried using a critical-point dryer with liquid CO2. Following the drying process, the samples were sputter-coated with a thin layer of gold for 5 minutes for the enhancement of conductivity and improving image quality. The gold-coated samples were then examined using SEM model JC2000D2A from Zhongchuan Digital Equipment Co., Ltd., China, operating at an accelerating voltage of 10 kV.
Energy dispersive X-ray spectrometer
Energy Dispersive X-ray Spectrometers (EDX) were performed (QUANTA FEG250 - Thermo Fisher Scientific) for Ch-Al-AgNPs in order to assess the prepared sample’s percentage silver content.
Fourier transform infrared
Fourier Transform Infrared (FTIR) was utilized to identify chemical interactions within the hydrogel matrix. The spectra of the prepared samples were acquired using a Vertex 70-RAM II Bruker spectrometer (Bruker Analytical, Germany) FTIR spectra were collected at room temperature, ranging from 4500 to 500 cm−1.
Water absorption
Swelling studies were conducted by measuring the weight of the hydrogel discs after immersing them in distilled water for 24 hours at room temperature. Water absorption (WA) is generally used to describe the swelling behavior of the prepared hydrogels. The dry hydrogel was soaked in distilled water (DW). The swollen samples were allowed to drain using a plastic strainer for 10 minutes. Then, the swollen hydrogel was weighed after soaking.36,37 WA is given by Equation (1) as follows:
Mechanical properties
The mechanical properties of the prepared hydrogel films, tensile strength (TS) and percentage elongation at break (EAB), were determined using a texture analyzer (XT plus, Stable Micro Systems, Godalming, England) following the standard ASTM D882 method. For the present test, film strips (60 mm length and 7 mm width) were used. The equations (2) and (3) presented below were used to determine the values of the TS and EAB for the film samples.
F is the maximum force; A is the cross-sectional area of the film.
In vitro antimicrobial studies
Minimum inhibitory concentration determination
The prepared MGTAg-NPs was evaluated for their antimicrobial properties where the materials were analyzed using both quantitative (Green synthesized Silver Nanoparticles loaded on Hydrogel and attached culture growth) and qualitative (growth inhibition zone evaluation) techniques against all tested strains (P. aeruginosa, E. coli, S. aureus, Candida albicans and Aspergillus niger). The developed materials’ 1 cm × 1 cm inhibition zone fragments were sterilized using UV exposure for 30 minutes in order to assess the growth. Each sample’s sterile components were aseptically added to nutrient agar plates that had already been inoculated with 0.5 McFarland (1.5 × 10^8 colony forming units/ml) microbial suspensions made from the strains of microorganisms that had been tested. Using a sterile swab, the inoculation of microbial strains was carried out in accordance with the disc-diffusion antibiogram assay. To promote microbial growth, the inoculated Petri dishes containing the materials under test were incubated for 24 hours at 37°C. Development of growth following incubation. Growth inhibition was assessed after incubation. Following the period of incubation, the growth inhibition zone appeared.
Antimicrobial assessment
The antibacterial performance of the green synthesis of AgNPs incorporated hydrogel have been analyzed against specific selected microbial species by microbial growth inhibition tests. Cultures in nutrient broth with the hydrogel samples were made with respect to the microbial growth in attached (biofilm) as well as a desorbed population (planktonic). Sterile hydrogel pieces were grown on nutrient broth with microbial suspensions at a turbidity equivalent to 0.5 McFarland standard and incubated at 37°C for 24 hours for biofilm development. Microbial growth of the hydrogel samples was determined by absorbance values obtained on a spectrophotometer at 650 nm after incubation.
In this study, the minimum inhibitory concentration (MIC) was assessed by disc diffusion and well diffusion methods. Unfortunately, the data regarding growth analysis are quite tricky. Due to that, an appropriate form of agar, BHI Agar and Tryptic Soy agar for bacteria, SD agar for fungi, was incubated with microbial suspensions. Pesticidal discs or wells were prepared by impregnating various concentrations of the Ch-Al and Ch-Al-AgNPs hydrogels, including 300 and 400 μg/mL for fungi, and 1.0 and 2.0 mg/mL for bacteria. At 24-h incubation at 37°C the optical density of the microbial cultures determined the MIC representing the minimum inhibitory concentration of the compounds most effective against bacterial species.
For Bacterial and Fungal strains, SEM has been used for assessing the morphological changes of treated bacterial or fungal cultures treated by (MGEAg-NPs). Cut agar was used to prepare the strains, which were then fixed for at least 3 hours in 2.5% glutaraldehyde (100 mm phosphate buffer solution, pH 7.2) and 1 hour in 1% osmium tetroxide. A series of graduated ethanol (30, 50, 60, 70, 80, 90, 95, and 100%; each level applied twice for 15 min each time) and ethanol: isoamyl acetate (3:1, 1:1, 1:3, and 100% isoamyl acetate twice for 30 min) were used to dehydrate the agar blocks. The agar blocks were coated with a gold coater for 5 minutes after being dried with a critical-point drier using liquid CO2.
Biofilm assessment and statistical analysis
Using sterile Eppendorf tubes filled with 1 mL of sterile saline water, the Ch-Al and Ch-Al-AgNPs hydrogel previously prepared were placed in order to evaluate the microbial cells’ capacity to attach and form biofilms following a 24-h incubation period. Colony forming units (CFU)/mL values were determined by dilution and inoculation of the resulting suspensions on agar plates after the washed coatings with attached cells were again persuasively vortexed to separate the attached cells. With the Student’s T test, statistical significance was examined. Significant statistical values were defined as p values less than 0.05. The experimental data were analyzed using appropriate statistical methods, Version 19 of SPSS was utilized to perform the statistical analysis.
Results and discussion
Biopolymer-based hydrogels, especially those made from chitosan and alginate, are promising materials for wound dressings. Chitosan provides antibacterial properties, while alginate offers gel-forming and moisture retention capabilities. Together, they form a matrix that can absorb wound fluids, support tissue regeneration, and deliver antimicrobial agents to the wound site. This matrix can be further improved by incorporating silver nanoparticles. Characterization of the silver nanoparticles synthesized using matcha green tea and their incorporation into a hydrogel matrix comprising chitosan-alginate are presented in this paper. Consequently, the prepared Ch-Al-AgNPs hydrogel was characterized for its physicochemical properties, mechanical strength, water absorption capacity, and antimicrobial activity against common pathogens associated with wound infection, such as E. coli, P. aeruginosa, S. aureus, C. albicans, and A. niger.
Characterization of green AgNPs
UV-Vis spectroscopy
The UV-visible absorption spectrum of synthesized AgNPs displayed a characteristic surface plasmon resonance (SPR) peak at 405 nm (Figure 2(a))), indicative of the formation of spherical or quasi-spherical AgNPs. This absorption peak aligns with the reported wavelength range of 390–420 nm for AgNPs and confirms the successful synthesis of AgNPs using matcha green tea extract as a reducing agent. It is basically a phenomenon where the free electrons in certain metallic nanoparticles as Ag and Au particles oscillate in a particular direction due to the incidence of a specific wavelength of the E. M radiation.22,38 Nano silver Characterization (a) UV-visible absorption spectrum of AgNPs synthesized using matcha green tea, (b) TEM of green AgNPs.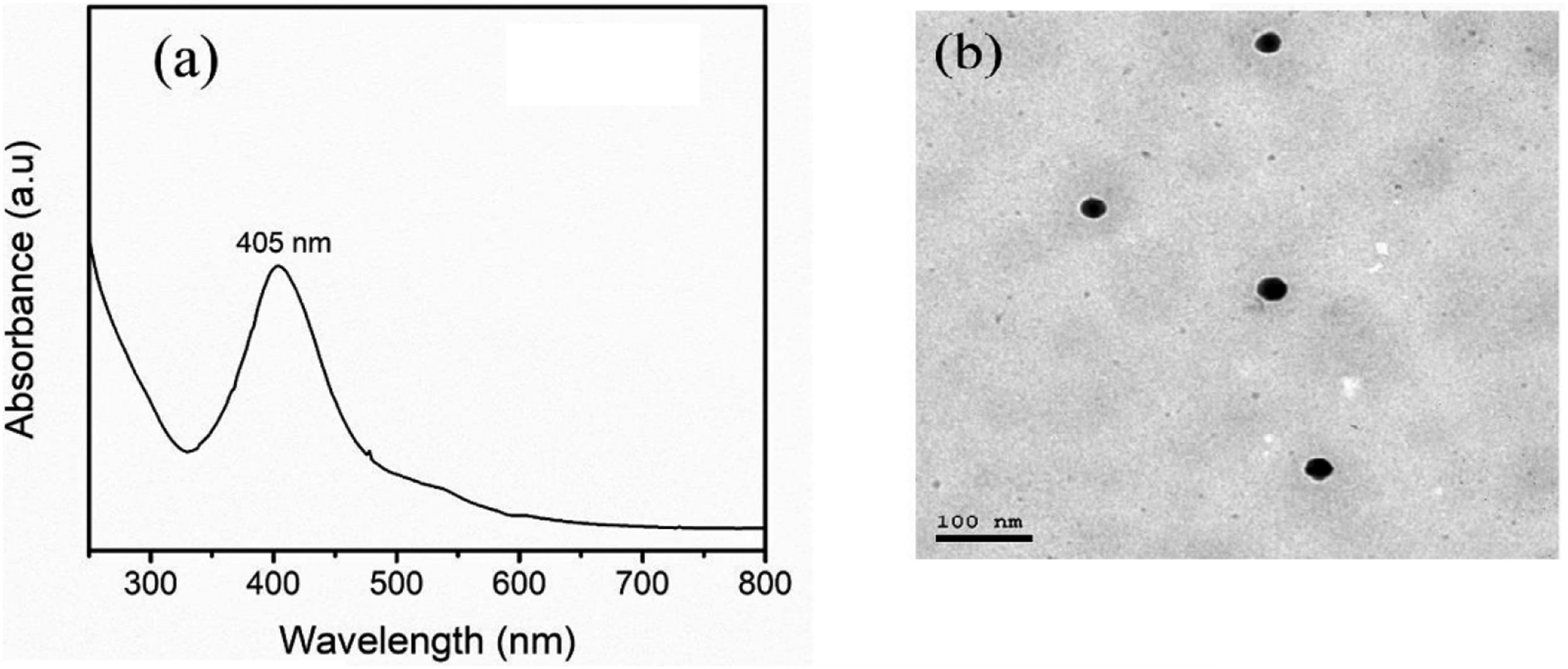
As per light reflection phenomenon of SPR, either light is absorbed or scattered hence resulting in the appearance of the absorption peak in the UV-Visible spectrum. The reflection coefficient increases and the SPR peak shifts depending on the size and form of nanoparticles, their chemical composition, and the dielectric constant of the environment. Among all the synthesized AgNPs, spherical NPs have the SPR peak at a wavelength of 390–420 nm due to the choice of the method and the condition used. From the spectral characteristic given above, the maximum SPR is seen at 405 nm which confirms the formation of spherical or quasi-spherical AgNPs while using the green tea extract as a reducer
Lack of peaks or humps in the UV-Vis spectrum also supports the lack of structural anisotropy or aggregation of nanoparticles. This essentially means that the green synthesis approach using matcha green tea extract lead to the formation of highly dispersed and uniform AgNPs. Matcha green tea extract probably by polyphenolic compounds may have exerted a vital influence on the decrease and stabilization of AgNPs by avoiding their aggregation and the preserve of spherical structured nanoparticles. This biocompatible and green reducing agent also reduces the usage of highly toxic chemical reductants in the entire synthesis process. The SPR peak shape of the synthesized AgNPs matches the recorded UV-Vis spectrum which is equal to their plasmonic capability for further uses in biosensing, SERS, or photocatalysis.23,29
Transmission electron microscopy
Transmission Electron Microscopy (TEM) analysis (Figure 2(b))) revealed well-dispersed AgNPs with sizes ranging from 20 to 40 nm. The image shows distinct nanoparticles dispersed,the TEM image confirms the successful synthesis of AgNPs and provides qualitative information about their presence, distribution, and general size range.
The dark spots or particles dispersed on the gray background represent individual or small clusters of AgNPs. The uniform distribution of the nanoparticles suggests their potential for consistent antimicrobial activity. These homogeneous distribution and absence of cluster formation similar to what is seen in the TEM images are suggestive of probable high overall antibacterial properties and higher biocompatibility of the synthesized AgNPs. Distribution of nanoparticles is also favorable for interacting with bacterial or fungal cells to ensure the maximum potential of the antimicrobial nanoparticles. Apart from the routinely used bright field TEM, HRTEM analysis was performed to determine the crystal structure and atomic planes of the AgNPs. From the observed HRTEM images however, clear lattice fringes were observed indicating that the synthesized AgNPs have a crystalline character as shown in the images below.5,22
Characterization of chitosan-alginate hydrogel embedded with AgNPs
Scanning electron microscope
SEM analysis (Figure 3(a)–(d))) revealed the SEM of chitosan, alginate, and the two prepared hydrogel film samples. Chitosan, which appears as irregular, crystalline-like particles or chunks.
39
Whereas alginate has a more amorphous and rougher surface morphology.
40
Meanwhile, Ch-Al exhibits a fibrous, interconnected network structure. The fibers or strands appear to be randomly oriented, forming a porous and open matrix. Ch-Al-AgNPs also displays a fibrous network structure but with some differences. The inclusion of AgNPs resulted in a more compact matrix with visible AgNPs clusters. These morphological changes indicate that the hydrogel’s structure was influenced by the presence of AgNPs. Hydrogel Characterization (a–d) SEM images of (a) chitosan, (b) alginate, (c) Ch-Al hydrogel, and (d) Ch-Al-AgNPs hydrogel; (e) EDX spectrum of Ch-Al-AgNPs hydrogel; (f) FTIR spectrum for Alginate, Chitosan, Ch-Alg, and Ch-Alg-AgNPs hydrogel; (g) Water absorption capacity of Ch-Al and Ch-Al-AgNPs hydrogel.
The results thoroughly characterize the nature of hydrogels, the used Transmission Electron Microscopy to visualize the presence of AgNPs within the Ch-Al matrix. Furthermore, Scanning Electron Microscopy has allowed the study of surface morphology and interference of nanoparticles with microbial cell walls. This feature makes Ch-Al-AgNPs suitable for use as a wound dressing because it provides an appropriate space for cell growth and migration as well as absorption of tissue exudates.4,8
Energy dispersive X-ray spectrometer for Ch-Al-AgNPs hydrogel
EDX spectra (Figure 3(e)) confirmed the elemental composition of the Ch-Al-AgNPs hydrogel. The presence of Ag was detected, at a low weight percentage (0.28%), verifying the successful embedding of AgNPs within the hydrogel. 22 Several peaks corresponding to different elements present in the sample. The prominent peaks are labeled as C K (carbon), O K (oxygen), Ag M (silver M-shell), and Ag Lα, Ag Lβ, and Ag Lβ2 (silver L-shell emission lines).
The weight percentage of carbon is (60.48%), and the atomic percentage is (67.22%). This high carbon content is likely due to the presence of chitosan and alginate polymers, which are primarily composed of carbon and oxygen. While the weight percentage of oxygen is (39.24%), and the atomic percentage is (32.74%). The presence of oxygen is expected due to hydroxyl, carboxyl, and other functional groups in the chitosan and alginate polymers. Finally, the weight percentage of silver is (0.28%), and the atomic percentage is (0.03%). The relatively low but detectable amount of silver confirms the presence of AgNPs in the hydrogel formulation. 22
FTIR spectroscopy
The FTIR spectra (Figure 3(f)) confirmed the incorporation of AgNPs within the Ch-Al matrix. Key peaks, such as the broad peak around 3400 cm−1 (O-H and N-H stretching) and peaks in the 1650–1500 cm−1 range (amide bands), which is due to stretching vibrations of the hydroxyl groups. The peaks around 1600 cm−1 and 1420 cm−1 may be attributed to the asymmetric and symmetric stretching vibrations of the carboxylate groups, respectively. 41 The peak at 1030 cm−1 may be associated with C-O-C stretching vibrations of the pyranose ring. Furthermore, the spectrum of Ch-Al shows the main characteristic peaks of both used polymers (chitosan and alginate), which confirms the presence of both polymers in the sample Ch-Al. The peak at 3400 cm−1 is due to the overlapping O-H and N-H stretching vibrations from both polymers. The peaks around 1600 cm−1 and 1420 cm−1 may be attributed to the carboxylate groups of alginates, while the peaks in the 1650–1500 cm−1 region are related to the amide I and amide II bands of chitosan. 23 Finally, the spectrum of Ch-Al-AgNPs is similar to that of Ch-Al, exhibiting the characteristic peaks of both chitosan and alginate. However, there are a few differences in both the peak intensity and the peak shape of certain peaks, which may be attributed to the presence of AgNPs and their potential interactions with the polymer’s matrix. 29
Water absorption
Water absorption tests (Figure 3(g)) demonstrated that the Ch-Al hydrogel exhibited a higher swelling ratio (35 g/g) compared to the Ch-Al-AgNPs hydrogel (30 g/g). The presence of AgNPs within the hydrogel matrix reduced the swelling capacity, likely due to steric hindrance and interactions between the nanoparticles and the polymer chains.4,41 This lower swelling ratio may offer advantages for wound healing by preventing excessive absorption of wound exudates. 4
Figure 3(g) illustrates the swelling behavior of the two hydrogel samples over 24 hrs. It can be noted that, for initial swelling rate, both hydrogel samples exhibit a rapid initial swelling rate during the first 5 hours. This initial speed swelling is due to the hydrophilic nature of the polymeric chains. The swelling behavior analysis revealed that the pristine chitosan-alginate (Ch-Al) hydrogel exhibited a higher maximum swelling ratio of approximately 35 g/g, whereas the incorporation of green-synthesized silver nanoparticles (AgNPs) into the hydrogel matrix (Ch-Al-AgNPs) resulted in a slightly lower maximum swelling ratio of around 30 g/g. Swelling equilibrium as after reaching their respective maximum swelling ratios, both samples appear to reach an equilibrium state, where the swelling ratio remains relatively constant over the remaining time period. 42
The lower swelling ratio observed for the Ch-Al-AgNPs can be attributed to the presence of Ag NPs within the hydrogel matrix. The incorporation of Ag NPs can lead to interactions with the polymeric chains, potentially altering the hydrophilicity and water absorption capacity of the hydrogel. Additionally, Ag NPs may create steric hindrance or obstruction within the hydrogel matrix, limiting the accessibility of water molecules to the polymer chains and reducing the overall swelling capacity.4,32
Furthermore, the lower swelling ratio of Ch-Al-AgNPs may be advantageous in certain wound healing applications, as it can provide controlled swelling and prevent excessive absorption of wound exudate. This controlled swelling can help maintain structural integrity and minimize potential disruption of the wound bed. Also, the presence of Ag NPs imparts antimicrobial properties to the hydrogel, aiding in combating wounds.
This swelling behavior of hydrogels is one important aspect that affects the function of delivering therapeutic agents. A high swelling ratio may enhance the diffusion of drugs through the hydrogel, thereby improving the drug bioavailability at an applied site. In the current study, a moderate swelling ratio recorded for the Chitosan-Alginate hydrogel is considered an optimum value for controlled release of the drug with the aim to provide long-lasting antimicrobial activity while reducing the risk of early drug release. The high swelling capacity reduction of the Ch-Al-AgNPs hydrogel with respect to the pristine Ch-Al hydrogel points to its useful modification in relation to wound-healing applications. This controlled swelling behavior will not allow excessive absorption of wound exudate, hence maintaining structural integrity for longer periods of time.
Mechanical properties
Mechanical properties of the prepared hydrogel films.

As illustrated in Table 1 sample Ch-Al exhibits a higher tensile strength of (25.4 MPa) compared to Ch-Al-AgNPs , which has a tensile strength of (18.2 MPa). Sample Ch-Al also displays a higher percentage elongation at break of (42.7%), while Sample Ch-Al-AgNPs has a lower value of (35.9%).
The mechanical properties of edible hydrogel films are crucial for their intended applications, such as wound healing, food packaging, or drug delivery. 4 The tensile strength and percentage elongation at break provide valuable information about the films’ mechanical behavior and performance. Sample Ch-Al demonstrates superior mechanical properties compared to Ch-Al-AgNPs, with a higher tensile strength and greater elongation at break. This suggests that Sample Ch-Al can withstand higher tensile forces before failure and has better stretchability or flexibility compared to sample Ch-Al-AgNPs.
The differences in mechanical properties between the two samples can be attributed to various factors, including the composition of the films, the types and proportions of polymers, plasticizers, and other additives used, as well as the processing conditions employed during film preparation. When compared to literature values for similar edible hydrogel films, the tensile strength of Sample Ch-Al (25.4 MPa) is within the range reported for chitosan-based films (15–30 MPa),22,35 while Sample Ch-Al-AgNPs (18.2 MPa) falls slightly below this range. The percentage elongation at break for both samples (42.7% for Ch-Al and 35.9% for Ch-Al-AgNPs ) is comparable to the values reported for edible films containing polysaccharides and plasticizers (30%–50%). 43
The addition of AgNPs has slightly compromised the mechanical properties of the Ch-Al-AgNPs hydrogel; however, these properties have remained within acceptable limits for eventual clinical use. This reduced tensile strength and flexibility may be a compromise with the excellent antimicrobial activity, which is, in fact, much needed to prevent infection of open wounds.
Drug delivery and swelling rate are directly impacted by the swelling ratio; therefore the mild swelling (30 g/g) of the Ch-Al-AgNPs hydrogel allows for controlled drug release , to maintain antibacterial activity and avoid early drug loss. Morover, AgNPs lower the bacterial growth , which may promote neovascularization by reducing inflammation and infection. Therefore, a balanced wound environment that promotes tissue regeneration is produced. The Ch-Al-AgNPs hydrogel’s product swelling ratio helps preserve structural integrity and prevent over-absorption. This regulated swelling provides a consistent healing environment and is matched with the requirements for wound dressings.
Assessment of the growth inhibition zone for antimicrobial activity
There exists evidence that bacteria and fungi are contributory causes of severe wound infections in the world today. Since the barrier function of the skin tissue is lost in injured skin, the area of tissue damage becomes highly vulnerable to microbial colonization and infection by pathogenic microorganisms considered as skin commensals such as Staphylococcus aureus. For the identification of the antimicrobial efficiency of the developed materials, the inhibition zone assay was used. 43
The antimicrobial activity of the hydrogels was assessed against several pathogenic microorganisms (Figure 4). The Ch-Al hydrogel alone showed minimal inhibitory effects, with no significant inhibition zones against E. coli, P. aeruginosa, S. aureus, C. albicans, and A.niger. However, the Ch-Al-AgNPs hydrogel exhibited substantial antimicrobial activity, with inhibition zones ranging from 12 mm for C. albicans to 31 mm for S. aureus (Table 2). The enhanced antibacterial activity of the Ch-Al-AgNPs hydrogel was attributed to the synergistic effect of the chitosan-alginate matrix and the AgNPs, which disrupted bacterial cell walls and membranes.4,31,46 (a) E. coli, P. aeruginosa, S. aureus, C. albicans and A. niger growth plates. (b) Effect of Ch-Al on the Bacterial and Fungal isolates (No detected inhibition Zone), (c) Effect of Ch-Al-AgNO3 on the Bacterial and Fungal isolates (Detected inhibition Zone). Inhibition zone diameter (mm) using the two hydrogel samples.

Hydrogels with potent antibacterial abilities that completely cover the wound and have a higher clinical application value because wounds exposed to the air are more likely to become infected with bacteria and additional delays in the healing process. The common bacteria found in wounds, E. Coli, S. aureus, and P. aeruginosa, C. albicans have been selected to evaluate the antibacterial effectiveness of the developed medical formed material. 44 Hydrogel with diameters of 10 mm were placed onto the surface of the agar containing the bacterial and fungal strains. After the samples were set upright on the agar plate for 10 minutes, the plates were incubated for 24 h at 37°C. The diameter of the inhibition zone on the agar plate was recorded. In the present work, the prepared Ch-Al and MGTAg-NPs Ch-Al-AgNPs hydrogel were evaluated for their antimicrobial activities against both Gram-Positive and Gram-Negative bacteria and fungi.
Figure 4 shows, in the top panel, fresh growth plates of E. coli, S. aureus, P. aeruginosa, C. albicans, and A. niger; in the middle panel, the inhibition zones tested with Ch-Al hydrogel for the same microorganism; and in the bottom panel, the inhibition zones tested with Ch-AL-AgNPs hydrogel’s for the same microorganism. As shown in Table 2 and Figure 4 middle panel revealed that after 24 hours incubation at 37°C, the used alginate-chitosan hydrogel alone without Ag-NPs didn’t explain proper inhibition zones for all types of the used microorganisms. There was no measurable impact of Ch-Al on Gram-positive (S. aureus), Gram-negative (E. coli and P. aeruginosa), and on both types of fungi (C. albicans and A. niger). The corresponding inhibition zones were 6 mm, 5 mm, 3 mm at the concentration of 2.0 mg/mL for E. Coli, P. aeruginosa, and S. aureus respectively. However, there was no measured diameter zone of inhibition for the concentration 1.0 mg/mL. The corresponding inhibition zones 2 mm, and 3 mm for C. albicans and A. niger respectively at the concentration of 400 ug/mL while the concentration of 300 ug/mL didn’t show an inhibition zone for both types of the Fungi used. The results agreed with the study as the antibacterial effect of the silver-free hydrogel is weak, and the addition of AgNPs significantly improves the antibacterial ability.3,4,33 The results included the long-term antibacterial efficacy. The inhibition zones did not decrease for extended periods but rather slightly increased, indicating sustained release and continuous antimicrobial activity. Even after incubation at 37°C for 72 hours, the Ch-AL-AgNPs hydrogel still exhibited distinct inhibition zones, up to 31 mm for S. aureus, confirming Long-term efficacy. Conclusively, the results obtained suggested that Ch-Al-AgNPs hydrogel has the potential for long-term protection against microbial infection in a clinical setting.
As shown in Table 2, the prepared Ch-Al-AgNPs exhibited diverse inhibition zones with diameters (31 mm) for S. aureus (26 mm) for E. coli. (20 mm) for P. aeruginosa (12 mm) for Candida albicans and (14 mm) for A. niger. The cationic group of the AgNPs may have contributed to the inhibition by increasing the hydrogel’s positive charge density. This would have allowed the bacteria to be more readily adsorbed and accumulated on the cell wall through interactions with hydrophobic and electrostatic forces. The disruption of the bacterial membrane and subsequent cytoplasmic leakage would then cause the bacteria to lyse. 45 The hydrogels’ silver nanoparticles showed concentration-dependent antibacterial activity against Gram-negative, Gram-positive Staphylococcus aureus and Klebsiella pneumoniae during the hydrogels’ antimicrobial testing.4,8,20
The biggest inhibition zone was observed for the S. aureus strain, where zone inhibition reached (31 mm) for the used Ch-Al-AgNPs sample followed by E. coli (26 mm) then P. aeruginosa (20 mm). In all the tested strains, the Ch-Al-AgNPs had the highest antimicrobial impact. The antibacterial ability of the Ch-Al-AgNPs hydrogel was significantly high which can be attributed to the synergistic antibacterial properties of green AgNPs and chitosan-alginate hydrogel. The green AgNPs could attach onto the bacteria wall and membrane, then damage the intracellular molecules and structures to kill the bacteria.
31
The antibacterial effect of Ch-Al-AgNPs hydrogel was higher than the Ch-Al hydrogel, indicating that Ch-Al-AgNPs has the potential to be used as an antibacterial control of the wound infection. The Ch-Al-AgNPs novel hydrogel will be a promising wound infection control to repair open wounds and will significantly decrease the possibility of bacterial and fungal infection as illustrated in Figure 5. The green AgNPs can be considered to be therapeutically useful in the reduction of microorganisms. Inhibition mechanism of the Ch-Al-AgNPs hydrogel on the cell wall of the Gram-positive cell wall bacteria, Gram-negative cell wall bacterial and fungal cell wall.
The silver-free hydrogel’s antibacterial activity is minimal, and adding AgNPs greatly strengthens the antibacterial capability.4,20 This suggests that the free hydrogel cannot meet the antibacterial requirements due to its limited antibacterial activity. The hydrogel containing silver was surrounded by an obvious inhibition zone. Additionally, the inhibition zone did not decrease over an extended period of time and in fact slightly grew, suggesting that the antibacterial effect could be maintained for a prolonged period.
SEM analysis of the treated bacterial and fungal cells revealed signs of cellular damage caused by AgNPs. While, untreated bacterial and fungal cells exhibited typical morphological features, such as a spherical, smooth cell wall and intact cell membrane structures. Furthermore, upon treatment with higher concentrations of the green AgNPs, the cells displayed clear membrane damage, including ruptures and the formation of pits. These membrane disruptions likely led to the leakage of intracellular components, eventually resulting in cell death, as observed in Figure 6. SEM of (a) E.coli , (b) S. aureus , (c) P. aeruginosa, (d) A. niger after treatment with green AgNPs.
Biofilm assessment
In order to evaluate the ability of microbial cells to develop biofilms on the developed materials, cells were incubated for 24 h in the presence of E. coli, P. aeruginosa, S. aureus, C. albicans, and A. niger after which the correlated results were recorded, as shown in Figure 7. The results demonstrated that all the tested Ch-Al-AgNPs had an inhibitory effect against E. coli, P. aeruginosa,S. aureus, C. albicans, and A. niger Compared with the Ch-Al free control. Ch-Al-AgNPs have an enhanced anti-biofilm effect on the Gram-negative E. coli, P. aeruginosa, Gram-positive S. aureus, and both fungal-tested strains. Biofilm formation in the presence of tested Ch-Al and Ch-Al-AgNPs hydrogel on the tested strains.
A correlation can be made between the presence of the novel Ch-Al hydrogel along with the abundance of AgNPs on the different used strains and the results after the antimicrobial activity. The results suggested that there is a synergistic effect between hydrogel and silver nanoparticles, as highlighted by the evaluation of the growth inhibition zone. Overall, the SEM results show that the presence of Matcha Green Tea (camellia sinensis) improves the silver nanoparticles distribution on the hydrogel. In this sense, the effect of Ch-Al-AgNPs against biofilm formation is increased. Silver nanoparticles formed uniformly on the substrate, and the enhanced antimicrobial and antibiofilm potential of the nanomodified dressings was validated. 7 The developed wound dressings based on essential oils, sodium alginate, and silver nanoparticles have the potential to effectively treat infections and could be a viable substitute for topical medications and antibiotics in wound care.4,20
Therefore, the integration of the AgNPs to reinforce biopolymer-based hydrogels could be one of the most promising approaches toward designing appropriate dressings with considerably improved antimicrobial action. The present work describes the synthesis of silver nanoparticles from an extract of matcha green tea and their subsequent incorporation into a chitosan-alginate hydrogel matrix.6,29 Such findings are useful in gaining information about the physicochemical properties, mechanical strength, and antimicrobial activity of Ch-Al-AgNPs hydrogel developed. 29
The development of appropriate materials for wound care that would combine mechanical durability with antimicrobial properties has become even more vital today in view of an increasingly growing incidence of infections caused by antibiotic-resistant pathogens. These results confirm the hypothesis that the Ch-Al-AgNPs hydrogel possesses superior characteristics, which are very conducive to successful wound healing: prevention of microbial colonization and support of tissue regeneration.
The results clearly indicated that green-synthesized AgNPs using matcha green tea were indeed incorporated into the chitosan-alginate hydrogel matrix and remarkably enhanced its antimicrobial efficacy. Additionally, confirmation of the generation of AgNPs with desired size and morphology was obtained by using UV-Vis spectroscopy and TEM analyses, while FTIR and EDX confirmed their interaction with the hydrogel matrix.6,29
Conclusions
The study successfully demonstrates the powerful antibacterial and antifungal characteristics of a novel natural polymer hydrogel integrated with AgNPs derived from matcha green tea. Comprehensive characterization methods, such as TEM, SEM, UV-VIS spectrophotometry, and FTIR spectroscopy, confirmed the synthesis and presence of the nanoparticles within the hydrogel matrix. The novel hydrogel developed to control pathogen cause wound infections was emphasized by the substantial inhibitory effects against a wide range of bacterial and fungal strains, including E. coli, P. aeruginosa, S. aureus, C. albicans, and A. niger that were demonstrated by the antimicrobial efficacy tests. The current study proved that this novel hydrogel containing AgNPs has a practical replacer for antimicrobial treatment therapy, improving wound dressing management practices and decreasing the possibility of different pathogens causing wound infection resistance.
Footnotes
Acknowledgments
The authors extend their appreciation to the Researchers Supporting Project number (RSP2025R506), King Saud University, Riyadh, Saudi Arabia for funding this work, and national research center (NRC), Egypt.
Author Contributions
Conceptualization; F.M.A. and D.M.; methodology; D.M., M.M.E.S. and D.K.E.D.; formal analysis; D.M, C.T and M.M.E.S., D.K.E.D. and M.H.F.; resources; F.M.A., M.M.E.S. and D.M.; writing—original draft preparation; M.H.F., D.M. and M.M.E.S.; writing—review and editing; D.M., C.T., R.W., and M.M.E.S.; supervision; F.M.A.; funding acquisition; F.M.A. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors extend their appreciation to the Researchers Supporting Project number (RSP2025R506), King Saud University, Riyadh, Saudi Arabia for funding this work.



