Abstract
This study aimed to prepare and characterize PAA/carbonized waste rubber composites. Composites containing different proportions of PAA, and carbonized waste rubber were prepared (0.5%, 1% and 2% carbonized waste rubber by weight of PAA). Structural characterization of the prepared composites was carried out using FT-IR and XRD techniques. Thermal stability was examined with the TGA/DTG technique. Their morphologies were investigated using SEM. It is evaluated that the thermal stability of PAA is increased by preparing the composites obtained from all the results and the obtained composites can be used in areas where the thermal management of the electronic product is required.
Introduction
Composite materials are mostly products of advanced technology and knowledge, have high added value and are industrially important. Although different matrices are used in composite materials, approximately 90% of them consist of polymer materials. As the usage areas of polymers have increased and diversified, the development of polymers that provide the mechanical, thermal, and electrical properties required by their use or bringing the materials to the desired properties with polymer additives has gained importance and the studies carried out in this direction have increased. The reasons why composite materials are more advantageous than traditional materials can be listed as features such as high specific strength, hardness and fatigue, which arise from the more versatile structural design of the new material. Nowadays, in formulating composites, even the limitations of polymers are being pointed out by scientists to adapt to positive environmental effects. In addition, composites are also known as materials containing reinforcement, that is, load-bearing material, with strong mechanical properties embedded in the weak matrix material. Thanks to the advantages they provide, composites are widely used in aircraft and ship manufacturing industries, especially in the automobile industry.1–3 Rubber material, which is very useful for humanity like other polymer materials, is used as the basic material of many applications due to its unique properties. It exhibits extraordinary behaviour thanks to its high elasticity, which can instantly return to its original shape and stretch up to several hundred percent. The extremely high elastic behaviour of rubber occurs thanks to the low Young’s modulus and the high yield stress resulting from the vulcanization process. Rubber is also of critical importance in sectors such as food processing, chemical production, refining, pharmaceuticals, electronics and automotive, as it is a material that is highly resistant to acids, alkalis and chemical solutions.4–7
Poly (acrylic acid) (PAA) is a widely used pH-sensitive polymer used as pharma-ceutical carriers, antifouling agents, flocculants, and super absorbents. 8 These acrylates are polymerized using a controlled free radical polymerization process and have a wide side chain range. When inorganic support materials are treated with acrylic acid, they can be incorporated into composite materials in the polymerization process. The polymer layer provides controlled thickness and functionality, improving some physical properties of composites such as wettability and abrasion. 9 It is especially used in wastewater treatment, mineral processing, metal ion recovery and soil quality improvement in agriculture.10–12
In recent years, important research has been carried out on the production of fuel, energy and carbon materials by applying thermochemical conversion methods to biomass. 13 Carbon materials have important applications such as energy storage, removal of pollutants and catalyst. Activated carbon is another carbon mate-rial. It does not have any structural formula, is porous, has a high surface area and has a high carbon content. The high cost of commercial activated carbons used industrially makes it important to obtain them from cheap biomass. Activated carbon is obtained by applying different physical, chemical, and thermal processes to fossil resources and biomass containing carbon. The type of raw material, activation type and reaction conditions are the factors that determine the properties of activated carbon. Activated carbons obtained from biomass are used in the removal of heavy metals from polluted water, CO2 capture, hydrogen storage, energy storage and catalyst applications.14–16 Some areas of use of waste tires are mentioned in the literature. In environmentally friendly disposal processes, waste tires are ground into rubber powder, 17 vulcanized rubber products are prepared at low cost and used as fillers, 18 asphalt modifications,19,20 concrete modifications, 21 plastic hardening processes, 22 polypropylene, 23 nylon, 24 polyester 25 shows promising results in the field of circular economy by using them in converting them into high-strength products, such as the inclusion of ground tire rubber in thermoplastic resins.
Zor et al. 26 used waste tire pyrolysis oil obtained because of pyrolysis and evaluated this material as a reinforcing element in nanocellulose-based nanocomposite films. They revealed that nanocellulose-based pyrolysis oil nanocomposite films with superior thermal properties and structural compatibility, demonstrated by characteristic results, can be used in future industrial applications such as pharmaceuticals, coatings and green packaging. Verma et al. 27 produced and characterized composites using pyrolysis oil obtained from the pyrolysis of tire waste and epoxy resin. The results showed that composites with lower density and higher tensile strength than pure epoxy could be produced, and the produced composites could be used in the automotive industry. This study will reveal the originality of many innovations such as the utilization of waste rubber, especially with PAA polymer, due to the limited applications of polymer-based materials and rubber composites, the use of many polymer/synthetic materials, synthetic and waste rubber materials in industry, and the transformation of composites into engineering materials with high added value.
In this study, it was aimed to produce PAA/carbonized waste rubber composites with the pyrolysis method to recycle waste tires. Thermal gravimetric analysis (TGA/DTG), FT-IR, XRD and scanning electron microscope (SEM) characterization processes have been performed.
Materials and methods
Materials
Activated Carbon materials were obtained by pyrolysis method under high pres-sure (at 600°C for 30 min; the heating rate was 100°C/min.) and supplied by ZBB GmbH. PAA was purchased from Sigma Aldrich.
Preparation of the composites
Design of experiments for composites.

Measurements and Characterization
TGA/DTA was performed using a Hitachi brand device (Tokyo, Japan) out under nitrogen atmosphere with a heating rate of 25°C per minute between 40 and 600°C. The morphology of the biocomposite was observed by field emission scanning electron microscopy (FESEM, mod. LEO 1525, Carl Zeiss SMT AG, Oberkochen, Germany). Samples were immersed in liquid nitrogen, then cut to obtain the cross section, and finally coated with gold (layer thickness 250 Å) using a sputter coater (mod. B7341, Agar Scientific, Stansted, UK). 28
X-ray diffraction (XRD) measurements were performed at room temperature on a PANalytical XRD diffractometer (Netherlands). Crystalline structural analysis of all samples was recorded on a diffractometer with CuKα radiation (λ = 1.5406 Å). The X-ray diffractometer was operated at a voltage of 40 kV with a current density of 40 mA. The scanning range is 2θ = 10° to 90° at a scanning speed of 0.071°/s. The peak height and Curve Fitting method used by Segal et al., (1959) 29 were used to determine the crystallinity index (CrI) and crystallinity value. The surface chemical composition of the composites was characterized by Perkin Elmer, FT-IR Spectrometer Frontier and the wavenumber was set in the range from 4000 cm−1 to 400 cm−1.
Results and discussion
Thermal properties of the composites
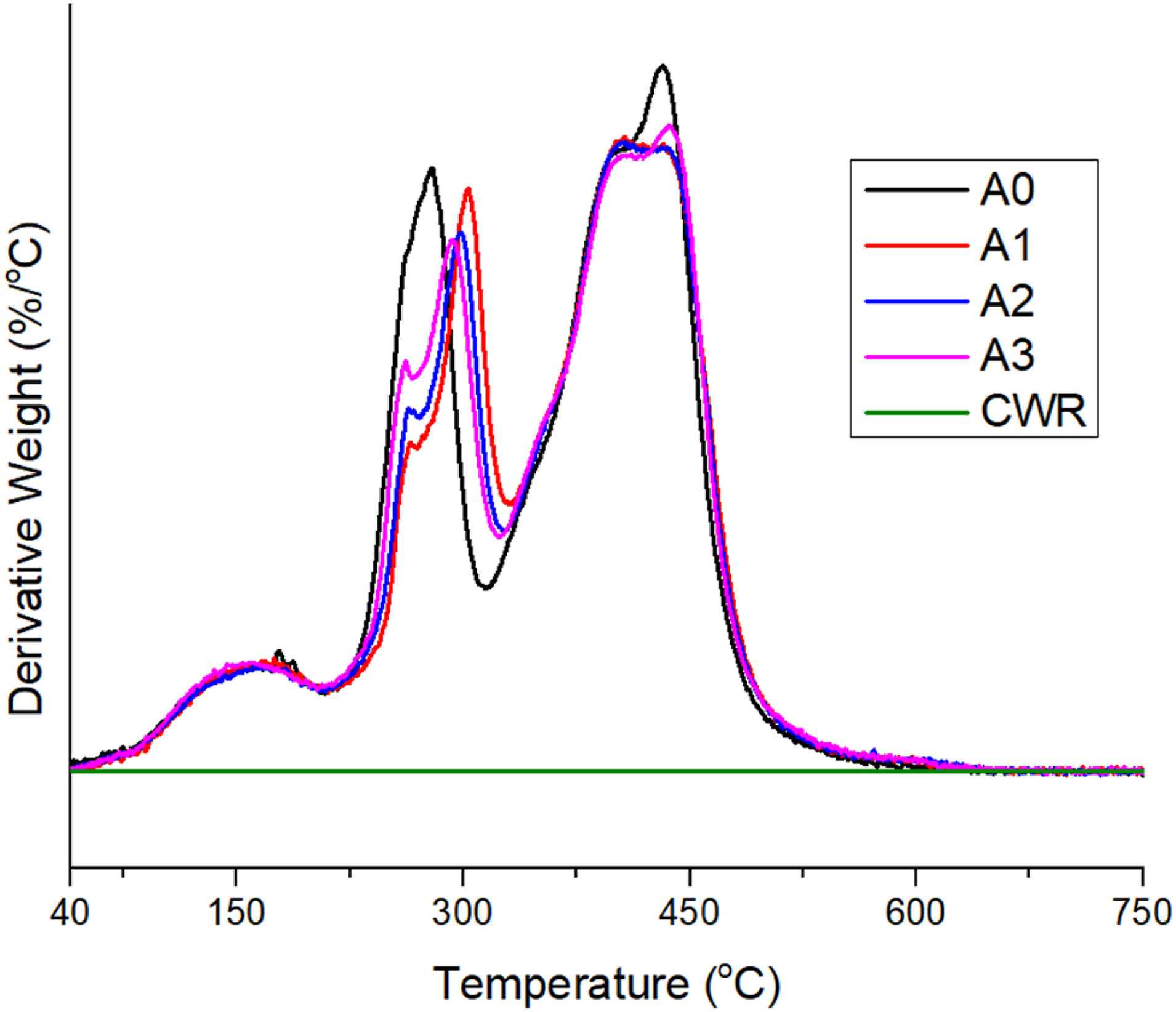
Thermal stability of the composites and neat CWR were determined by the TGA/DTG method (Figures 1 and 2). When the results obtained are examined, it is seen that the composites start to lose mass around 230°C. It has been determined that there are two different main decomposition temperatures. The first major degradation temperatures are around 300°C, and this value is due to the degradation of the carboxyl group of PAA. The second major decomposition occurs at approximately 420°C. This degradation is due to the degradation of carbon main chains.
30
As the amount of carbonized waste rubber increases in composites, a partial increase in thermal stability is observed. It is seen that the amount of ash remaining in the composites at 600°C is between 11% and 13%. As the amount of carbonized waste rubber in the composites increases, the amount of ash obviously increases Table 2. TGA graph of composites and neat CWR. DTG graph of composites and neat CWR. Thermal stability characteristics of the composites.

XRD of the composites
XRD pattern of the composites are shown in Figure 3. Two important 2θ peaks are seen in the XRD pattern. The first of these is approximately 33° and this value belongs to PAA. The other one is about 56° and this peak belongs to carbonized waste rubber. While there is no peak at 56° in the A0 sample because there is no carbonized waste rubber, it is seen in other samples.
31
The crystallinity of natural rubber decreases with the addition of PAA. XRD pattern of composites.
FT-IR of the composites
FTIR-ATR spectra of the composites are shown in Figure 4. The characteristic stretching band of the carboxylic group is observed at 1710 cm−1, the CH stretching band between 3000 and 2700 cm−1, the CH2 bending band at 1431 cm−1, and the C-O band at approximately 1162 cm−1 and 1230 cm−1. Similar results were reported in Hojjati et al. (2007)
32
study. FT-IR structure of composites.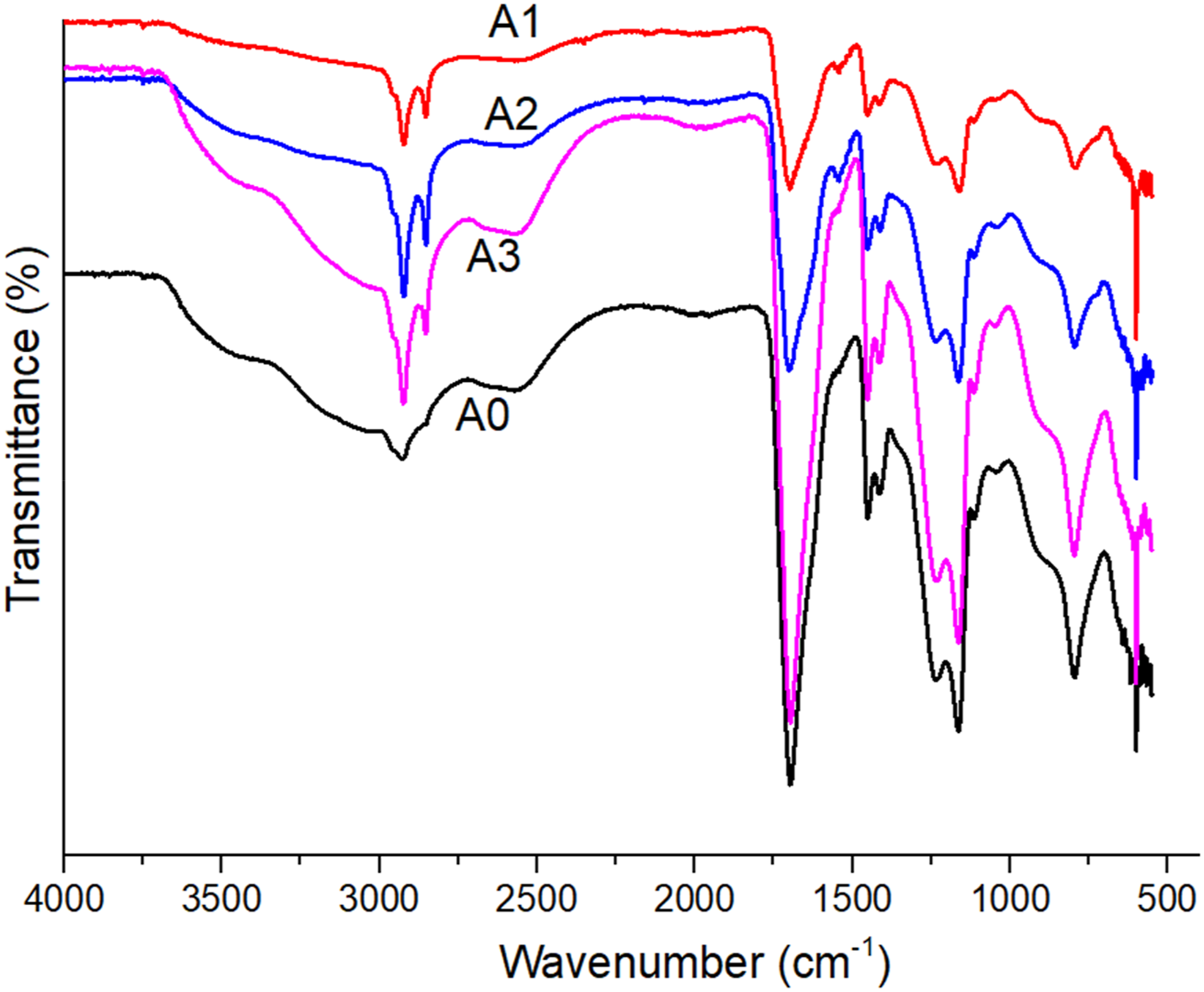
Morphologies of the composites
The morphological images of the composites are shown in Figure 5. The morphologies of the composites were determined by SEM technique. When SEM images are examined, all samples have a homogeneous and smooth morphology. These images show the harmonious combination of PAA and carbonized waste rubber. Morphological structure of composites.
Conclusion
In this study, it was aimed to prepare and characterize PAA/carbonized waste rubber composites. The results obtained are listed below. • FT-IR spectra prove that all composites were prepared successfully. • In the XRD spectra, peaks belonging to PAA and peaks belonging to carbonized waste rubber were characterized. • TGA results showed that as the amount of carbonized waste rubber increased, the thermal stability partially increased, and the char yield increased obviously. • From the SEM images, PAA and carbonized waste rubber are compatible with each other.
Thermal and morphological characterization processes of carbonized waste rubber added PAA composites obtained by the pyrolysis method were successfully carried out. The addition of carbonized waste rubber into the PAA matrix resulted in a significant increase in the thermal stability of these composite. The data obtained because of thermal analysis was supported by SEM analysis data. It is evaluated that the thermal stability of PAA is increased by preparing the composite obtained from all the results and the obtained composites can be used in areas where thermal resistance is required (bioelectronic devices and biological applications). As a result, when the structure-property relationship is examined from both macroscopic and microscopic perspectives for the visible systems of the composites, it is seen that PAA and activated carbon have a homogeneous structure. Moreover, this perspective will shed light on future studies in the characterization of simple components of holistic systems with complex structures such as porous structures and reinforced composites.
Footnotes
Acknowledgments
The author would like to thank Lawyer Ali Osman ODABAŞ for providing the technical infrastructure.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
