Abstract
Facial bone fractures in facial injuries resulting from severe trauma can be treated using plate and screw systems. Depending on the location of the fracture, the fracture lines are brought end to end and fixed with plates and screws. Metallic implants are at risk of endogenous infection and there are inherent problems associated with their use, such as stress shielding phenomena, pain, and local irritation. After the bone has healed, a second surgery is needed to remove the metallic fixation. Considering these problems, it is important to use biodegradable plates instead of metal plates. Biodegradable fracture fixation plates degrade slowly in the human body, causing less pain and stress, and there is no need for a second procedure to remove the synthetic material. As these plates decompose, the load will also decrease. There are no toxic and mutagenic effects in an absorbable implant. However, there are some problems with the use of these implants, such as inflammatory response, rapid loss of initial implant strength, higher fracture rates, insufficient hardness of the implants, and weakness compared to metallic implants. In this study, biodegradable plates were designed for use in maxillofacial bone injuries, and treatment plates were prepared using extrusion and injection molding. Different proportions of trimethylene carbonate (TMC), poly-L-lactate (LPLA), poly(D,L-lactate) (DLPLA), polyglycolate (PGA) and beta-tricalcium phosphate (β-TCP) are mixed by extrusion to produce composites by injection molding. Plates were obtained. The degradation, morphological, thermal, and mechanical behaviors of the obtained products were examined.
Keywords
Introduction
The fixation method is an important consideration in the treatment of facial bone fractures. Traditionally, titanium plates have been used for the fixation of most facial bone fractures because these metal plates allow rigid fixation with low infection rates. However, due to common complaints about titanium fixation, resorbable plates are increasingly used in the fixation of fractured facial bones.1,2 There are inherent disadvantages such as susceptibility to corrosion, infection, stress shielding, plate palpability, temperature sensitivity, interference with advanced imaging techniques, possible growth restrictions, and removal of these plates due to implant migration associated with metallic plates.3,4 Despite their relatively high cost, resorbable plates provide poorer fixation due to biological degradation over time, which promotes bone regeneration and allows cranial or facial bone growth in children. 5 The development of biodegradable plates has the potential to offer clinicians the ideal fixation system that is small, biocompatible, adaptable, allows sufficient stability to achieve bone union, and is subsequently resorbed in a timely manner.6,7
In general, resorbable plates used for the fixation of fractures are created using polyglycolic acid (PGA), polylactic acid (PLA), polydioxane (PDS), or combinations of these materials. PGA has a crystalline structure and is therefore stronger than PLA; However, its strength decreases within 6 weeks and the material is rapidly absorbed within 3–12 months. PLA is hydrophobic, so it takes up to 5 years for the material to be absorbed.8,9 PLA and PGA are degraded by hydrolytic destruction of the polymer, followed by the metabolism of a single molecule of lactic or glycolic acid through the corresponding biochemical pathways and elimination primarily by respiration.10,11 The decrease in molecular weight is followed by a decrease in mechanical strength and subsequent mass loss. 12 LPLA has the longest half-life, followed by DLPLA and PGA, but degradation is also affected by molecular weight, crystallinity, thermal history, and implant geometry.13–16 It has also been observed that polymer implants with biodegradable polytrimethylene-carbonate (PTMC) have the advantage of being less brittle in use. 17 PTMC, the most important member of the polycarbonate family, is an amorphous elastomer that exhibits good mechanical properties, including high flexibility and tensile strength. 18 Ceramic materials such as poly(α-hydroxy esters), calcium phosphates (CaP), (hydroxyapatite (HA), β-tricalcium phosphate (β-TCP), biphasic CaP (BCP) are also used for facial bone plates. Hence, polymer-ceramic composites continue to be increasingly widely used and are believed to be more biocompatible than their pure polymeric counterparts because the alkaline absorption products of calcium phosphates can buffer the acidic products of hydrolytic degradation responsible for late inflammation around α-hydroxy ester implants 19
Each material to be used here has its advantages and disadvantages. For example, DLPLA is a medium-strength polymer that does not have bone-bonding capacity, while TCP ceramic has bone-bonding capacity but lacks mechanical strength, especially under tension exhibits brittle fracture behavior. 20 Thermoplastic polymers, such as PLA and PGA, soften and melt upon heating and a variety of implants can potentially be shaped using a wide range of techniques like injection molding, compression molding, and extrusion. 21 Due to different manufacturing processes, polymer implants produced by melt extrusion or injection molding may not have the same molecular weights, crystallinity, or microporous structures, and therefore may degrade at different rates. 22 Depending on the production process used, differences will appear in the product. Thus (As a consequence), each implant needs to be evaluated individually. 23 Polymer-ceramic plates can be produced by injection molding and extrusion molding. For example, granulated PLA polymer and TCP powder are mechanically mixed and injected into a mold at 160°C through a plastic injection machine. 24
Materials and methods
Materials
In this study, approximately; Trimethylene carbonate (TMC) weighing 105000 g/mol, polyglycolate (PGA) weighing 76000 g/mol, poly-L-lactate (LPLA) weighing 145000 g/mol, and poly(D,L-lactate) (DLPLA) weighing 112000 g/mol.) polymers, and β-tricalcium phosphate (β-TCP) ceramic granule raw materials were mixed in different proportions to obtain composite plates, and the produced composite plates were characterized. Especially the Ceramic-based Bioabsorbable β-TCP composite plate to be used for facial area treatment was composited with other products for the first time, and experiments were carried out.
Design and production of molds of designed biodegradable composite plates and mechanical test sample
Plates of certain shapes and sizes are designed to be used in the treatment of bone damage in the facial area. For mechanical tests, a composite test sample was designed in accordance with the TS EN ISO 527-2 international standard. Using the Solidworks 2017 Basic program, the following plates were designed in sizes suitable for facial area treatment. The designed plates, samples, and molds are shown in Figure 1. The molds produced are compatible with our existing devices and the mold-bench familiarization trials were carried out by our team. Our designs and produced molds and plates.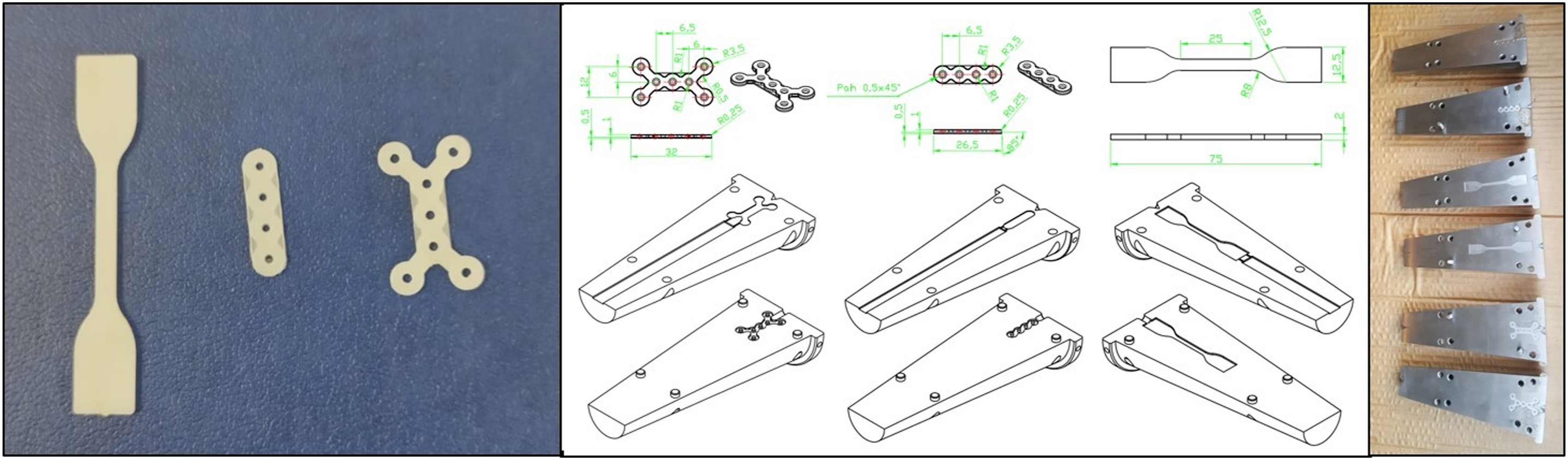
Production of biodegradable composite plates and mechanical test specimens
The product obtained composite mixtures and their ratios.

The composite groups were mixed in the extruder at 200°C and injected into the molds, whose temperature was set at 45°C, in the injection device with 3 bar pressure, again at 200°C, and the products were obtained. The 3-stage printing process was performed at 5-s intervals.
Characterization studies
All characterization studies were performed using products obtained from KP01, KP02, KP03, and KP04 composites which are shown in Table 1.
Mechanical testing
Structural characterizations of the polymer and ceramic materials we selected were carried out within the scope of this work package. In determining the tensile stress and elongation properties, tensile analysis was applied to 3 samples obtained from each of the 4 different composite mixtures shown in Table 1, which we prepared in 75 * 12.5 * 2 mm dimensions in accordance with the TS EN ISO 527-2 standard. In other words, a total of 12 tensile analyses were performed. Tensile analysis was performed with a Zwick/Roell Z020 device. Tensile tests were performed on tensile samples at room temperature. The tensile strength (σm) values obtained by the device were calculated using the maximum load value in the curves. According to the ISO 527-2 standard, the tensile strength value is defined as the “first local maximum” observed during the test. Therefore, tensile strength values for the samples were calculated manually. Elastic modulus was calculated by regression method between 0.05% and 0.25% deformation values. In the parameters, the preload is 0.5 N, the tensile modulus is 1 mm/min, the test speed is 50 mm/min, the grip-to-grip separation in the starting position is 45.00 mm, the gauge length is 20 mm, the temperature is room temperature.
Morphological analysis
The morphological analysis of the samples was performed using scanning electron microscopy (SEM) and micro-computed tomography (µ-CT).
SEM analysis was performed with a high-resolution (1.2 nm) QUANTA 400F Field Emission device and its morphology and surface structure were examined. The samples were coated with Gold – Palldium (Au – Pd) for SEM imaging.
Skyscan 1275 computerized microtomography scanner was used for scanning and structuring of samples in 2D and 3D for µ-CT. With a wide range of X-ray energies ranging from 20 to 100 kV, Skyscan 1275 can be used for sample sizes up to 96 mm diameter and 120 mm long, allowing for large samples including industrial large parts. All our samples comply with this dimensioning.
Thermal analyses
The thermal properties of composites were investigated using thermogravimetric assay (TGA) and differential scanning calorimetry (DSC).
TGA analysis was performed according to TS ISO EN 11358-1 standard. Briefly weight loss for samples was measured using a Perkin Elmer Pyris 1 TGA in a nitrogen environment at a temperature range of 25°C-600°C at a heating rate of 10°C/min.
DSC analyses were performed separately on 4 samples before and after gamma radiation using the Perkin Elmer Diamond Differential Scanning Calorimeter device. Temperature range of the device: −90°C to 550°C, temperature accuracy: ± 0.1°C, temperature resolution: ± 0.01°C, heating rate: 0.01°C to 500°C/min., calorimeter accuracy: < ± 1%, calorimeter resolution: < ± 0.1%, calorimeter sensitivity: 0.2 mW. Our DSC studies are carried out in a nitrogen environment between 20°C and 250°C, 10°C/min. speed.
X-ray diffraction analysis
X-ray diffraction (XRD) analyses were performed using Rigaku Ultima-IV X-ray diffractometer (Tokyo, Japan). The experiment is carried out using copper (Cu) source with Kα = λ = 1.54 Ang., 40 kV, 30 mA properties at a scanning speed of 1°/min, sampling step of 0.02°, scan axis of 2 theta, scan range of 20–90°. The structure of the target crystal can be understood by detecting the X-rays diffracted from the materials along with the analyses.
Deformation analysis
The samples shape memory performance in a hot water environment was examined using a hot water bath. For this purpose, the obtained samples were immersed in water at a temperature of 65°C and heated for 30 s, then deformed into a helix, and then they were dipped into water at a temperature of 10°C for 45 s, without removing the load, and their temporary shapes were given. The deformed product was again immersed in water at a temperature of 65°C and the recovered shape ability of the sample was monitored.
In vitro degradation
The in vitro degradation tests of composite plates were analysed according to percent weight loss. For this purpose, dry initial weights (w
i
) of composite plates were weighed and placed in 20 mL of lactated Ringer’s solution. The experiment was carried out using a thermoshaker (Gerhardt, Germany) at 37°C, at a speed of 30 r/min, for 30 days. At the end of 30 days, the samples were freeze-dried using the Labconco Freezone Lyophilizer System in our laboratory for 2 days. Their final weight (w
f
) was measured, and the % weight reduction of the plates was examined using equation (1).
In vitro cytotoxicity analysis
The biocompatibility test of the composite plates is done in accordance with ISO10993. The mouse fibroblast cell line L929 (NCTC clone 929, ATCC) was used for in vitro indirect cytotoxicity assessment with MTT assay according to the manufacturer’s instructions. The required chemicals for cell culture were obtained from Capricorn-Scientific, Germany. Briefly L929 cells were incubated using a medium prepared with Dulbecco’s Modified Eagle Medium low glucose (l-DMEM) (Capricorn, Germany) including %10 fetal bovine serum (FBS) (Capricorn, Germany, %1 l-glutamine, and %1 antibiotic-antimycotic solution. Cells were cultured in T75 flasks. When cell confluence was achieved, cells were passaged using trypsinization. At the same time, composite extracts were prepared by incubating sterilized bone plates with l-DMEM for 24 h using a CO2 incubator at 37⁰C and %95 moisture. For MTT analysis, cells were seeded to 24 well plates with a concentration of 2 × 104 cells/well and incubated with l-DMEM medium. After 24 h, the medium was changed with the composite extract. The composite extract was diluted to obtain concentrations of %75 (v/v), %50 (v/v), and %25 (v/v). After incubation, the cells were incubated with 60 µL MTT (2.5 mg/mL, a/h) and 600 µL base l-DMEM for 3 h. After MTT was reduced to formazan crystals, the medium was removed, and the formazan crystals were dissolved with dimethyl sulfoxide (Sigma, Germany) for microplate reading at 570 nm wavelength using 96 well plates. The control group was incubated with l-DMEM medium.
Results and discussion
Mechanical testing
Figure 2 shows 3 stress-strain curves for samples consisting of the mixtures shown in Table 1. With the use of DLPLA in the KP01 and KP02 groups, it was observed that the elongation at break was significantly higher than other groups, and the neck formations were clearly evident. In the KP02 and KP04 groups, a more fragile structure with less elongation at break that did not allow neck formation was observed due to the use of PGA. While the maximum voltages are close to each other in the KP01, KP02, and KP04 groups, there are more significant differences between the maximum voltage values in the KP03 group. Tensile analysis graphs of composite test samples.
Averages of elasticity modulus, ultimate tensile strength, and elongation at break for tensile groups.

The statistical analysis of elasticity modulus of bone plates showed statistically significant differences between KP01 – KP02 (p < .0001 ****), KP02 – KP03 (p < .0001 ****), and KP02 – KP04 (p < .0001), and statistically nonsignificant differences between KP01 – KP03, KP01-KP04, and KP03-KP04. The ultimate tensile strength of bone plates had statistically significant differences between KP01 – KP03, and KP01 – KP04 while the results showed statistically nonsignificant differences between KP01 – KP02, KP02 – KP03, KP02 – KP04, and KP03 – KP04. Lastly statistical analysis of elongation at break results revealed statistically nonsignificant difference between all sample groups.
Scanning electron microscopy analysis
Figure 3 shows the SEM images of the composite plates whose mixing ratios are shown in Table 1. Images obtained from the high-resolution (1.2 nm) QUANTA 400F Field Emission SEM device have 200x and 1000x zoom values. In order to perform imaging, the plates had to be cut into small pieces according to the chamber of the device. The shiny and particulate parts seen belong to the β-TCP ceramic phase. Dark regions are regions showing polymer structures. SEM imaging of composite facial bone plates.
A more homogeneous ceramic-polymer distribution is observed in KP01 and KP03. In the 200X zoomed image of KP02, it was observed that the ceramic phase was denser in the lower right corner, close to the hole in the plate. The 1000X image is focused on this area. In KP04, a slight accumulation of ceramic phase was observed in certain regions. TCP particles cause rheological changes in the melt in the mixture, changing both shear stress and shear rate at the interaction interface between the nozzle wall and flow materials, which can cause flow instabilities. 27
Thermogravimetric analysis
The weight loss for the 4 composite samples shown in TGA Table 1 was measured in a Perkin Elmer Pyris 1 TGA in a nitrogen environment at a temperature range of 25°C–600°C, at a heating rate of 10°C/min. Figure 4 shows the graphs of the analysis. In the analysis, the percentages of the remaining ceramic phase appear when the temperature values reach 600°C. In our mixtures, the ceramic raw material ratios in KP01, KP02, KP03, and KP04 were determined as 5%, 15%, 20%, and 24%, respectively. The TGA analysis results performed on the plates revealed the concentrations as 3.52%, 14.22%, 19.94%, and 24.70%, respectively. The results are quite consistent with the determined ceramic phase ratios. The accumulation of the ceramic phase in certain areas during extrusion and molding and the fragmentation of the plates caused by the device may have caused the small deviations seen in the ratios here. The faster weight loss seen in KP01 is due to the use of TMC at the highest rate among the groups, with 20% TMC. It has the lowest melting temperature (46°C) among TMC raw materials. The last reason for the weight loss in KP04 can be thought of as it contains very high β-TCP at 24% and LPLA at 46%. Thermograms of composite plates.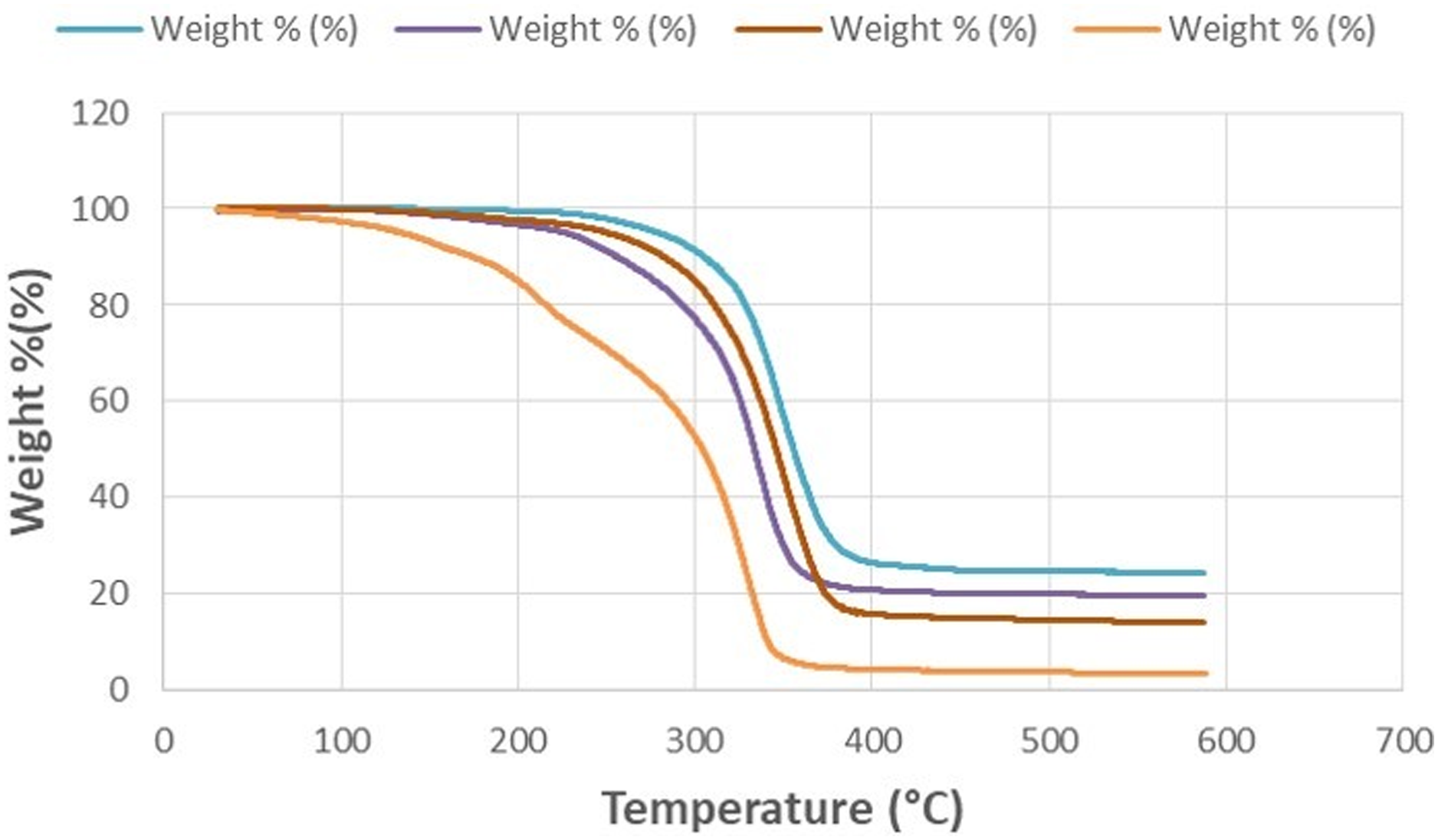
Micro-computed tomography (μ-CT) analysis
μ-CT studies were performed on plate pieces obtained from the mixtures shown in Table 1. Skyscan 1275 has been appropriately sized and analyzed. Figure 5 shows the images obtained from the wide upper surfaces, and front, top, and side surfaces of the plates. μ-CT images of the samples.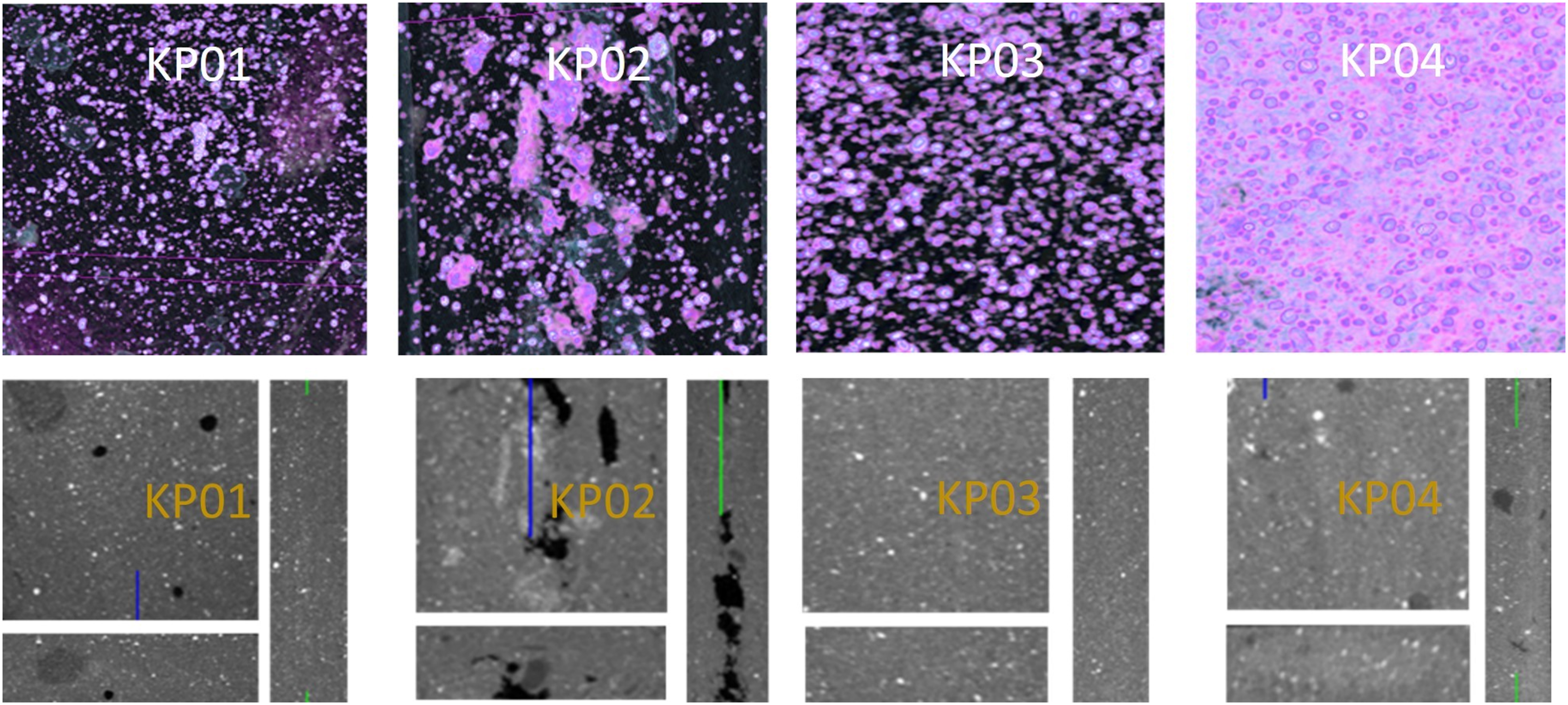
When the images in both figures are examined, it is observed that there are fewer gaps and a more homogeneous distribution in the KP03 and KP04 plates. It can be said that the gaps observed in KP01 give an almost homogeneous appearance. In KP02, an image with more gaps and less homogeneity was obtained. PGA, used at 45% in KP02, has superior mechanical strength compared to other biodegradable polymers.28,29 In this study, we observed that as the PGA ratio increased, the cracking and brittleness on the plate increased. It is thought that the increased use of PGA causes more voids in the composite structure.
X-ray diffraction analysis
XRD analysis was carried out on the Rigaku Ultima-IV X-ray Diffraction Device for the plates obtained from the 4 mixtures shown in Table 1. The measurement was made at a scanning speed of 1°/minute, 0.02° sampling step, 2 Theta measurement axes, 20-90° scanning range, using a copper (Cu) source, with Kα = λ = 1.54 Ang., 40 kV, 30 mA values. Figure 6 shows the visual of the analysis performed for composite plates. The analysis was specifically aimed to show calcium phosphate peaks. Xrd diffractogram of composite bone plates.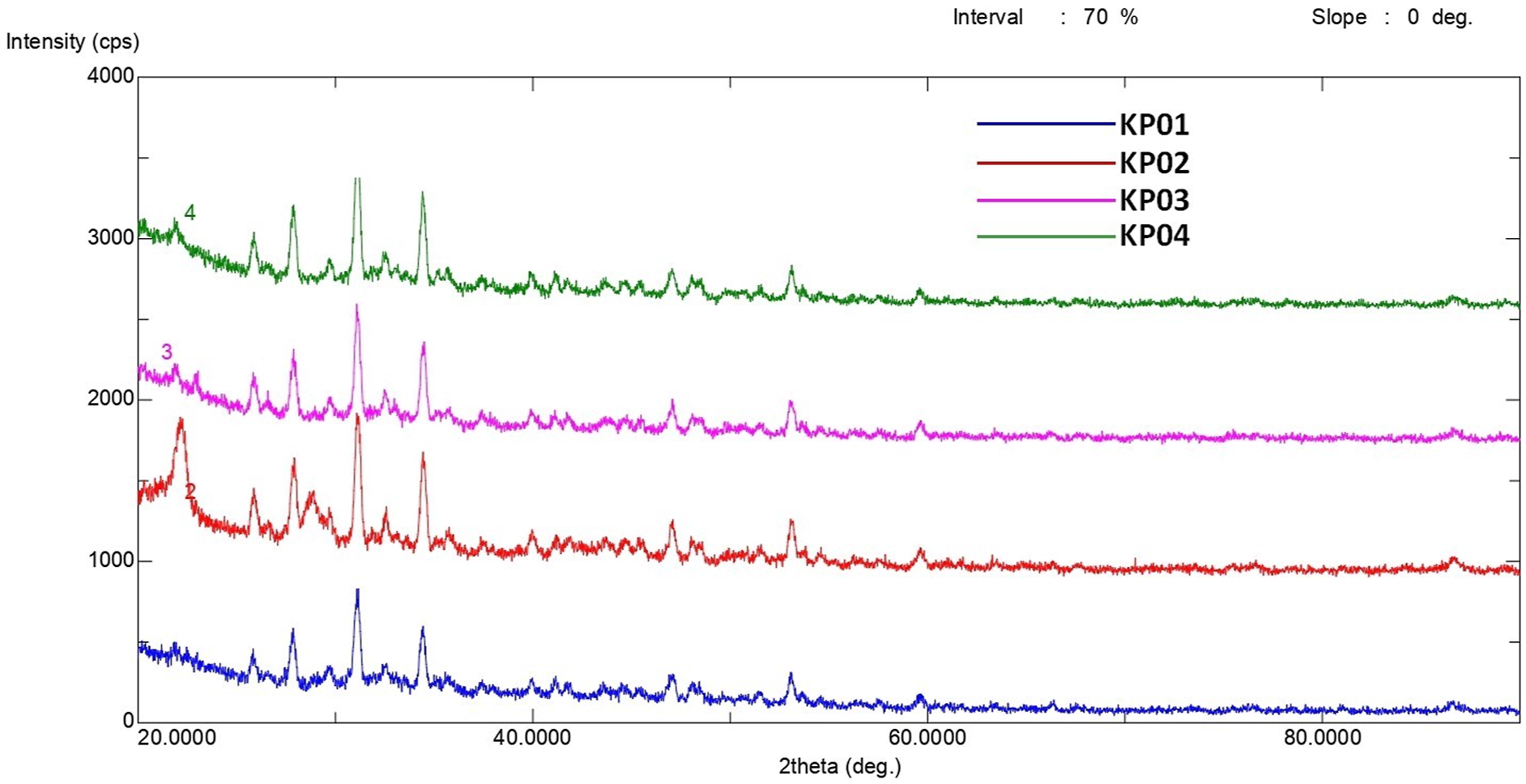
According to the results of the qualitative analysis, the analyzed samples contain Ca3(PO4)2 (Tri-Calcium Phosphate) groups. The Ca3(PO4)2 peaks shown in the Powder Diffraction File of Ca3(PO4)2 in the International Center for Diffraction Data matched the peaks of the XRD analysis of the plates we obtained. 30 The peaks were found to match completely.
Differential scanning calorimetry analysis
DSC analyses were performed separately on 4 samples before and after gamma radiation using the Perkin Elmer Diamond Differential Scanning Calorimeter device. By performing DSC analyses on the composite plates produced with the mixture ratios shown in Table 1, it was aimed to examine critical thermal parameters such as glass transition temperature and melting temperature and to reveal the character of the samples. In Figure 7, DSC analyses are given before gamma radiation (A) and after gamma radiation (B). In pre-irradiation (A) analyses, a distinct single peak was observed at 156.08°C in the KP01 plate as a result of the first heating. It is thought that the use of 20% TMC and 45% LPLA in KP01 causes this behavior. TMC has an amorphous and elastomeric structure, while LPLA has an amorphous structure, and crystallinity, and melting temperature peaks are not observed in amorphous structures in DSC analyses.
31
Therefore, no crystallinity peak was observed in KP01 during cooling. In the KP02 analysis, 38.35°C, 75.96°C, 143.44°C, 167.43°C, 192.70°C, 209.99°C. peaks were observed at the first heating. This composite structure has a behavior close to semi-crystalline and the melting and glass transition temperature peaks of the different polymers in it
32
. During KP02 cooling, the crystallinity peak appears at 146.27°C. In KP03, peaks were observed at 83.30°C and 156.44°C during the first heating. The absence of a distinct peak during cooling indicates the amorphous behavior of the KP03 structure. In KP04, many peaks appeared during the first heating, similar to KP02. It is thought that, unlike the KP01 and KP03 groups, their PGA doping causes this semi-crystalline-like behavior. DSC analyses before (A) and after (B) gamma radiation.
Similar behavior was observed in the peak images in the groups during the first heating after gamma radiation (Figure 7(A)). The least change was observed in the KP02 group. In the cooling zone, the groups maintained their structure and the least change was observed in the KP02 group.
In vitro degradation test
In vitro degradation experiments of 12 plates obtained from 4 different mixtures, the ratios of which are given in Table 1., were carried out by monitoring the decrease in molecular weight in lactated Ringer’s solution at 37°C, which is body temperature, in a continuous shaking system. Figure 8. shows initial weight status (wi) of KP01, KP02, KP03, and KP04 and the percentage weight loss at the end of 30 days (wf). In vitro degradation test results. The comparison of initial weight and final weight of composite bone plates. The statistical analysis showed that the weight reduction was statistically nonsignificant for KP01, KP02, and KP03 groups, while KP02 showed statistically significant (p < .05 *) (A) and % weight loss of bone composite plates. The statistical analysis showed statistically significant difference for KP01 – KP02 (p < .0001 ****), KP01 – KP03 (p < .001 ***), KP01 – KP04 (p < .001 ***), KP02 – KP03 (p < .0001 ****), KP02 – KP04 (p < .0001 ****) (B).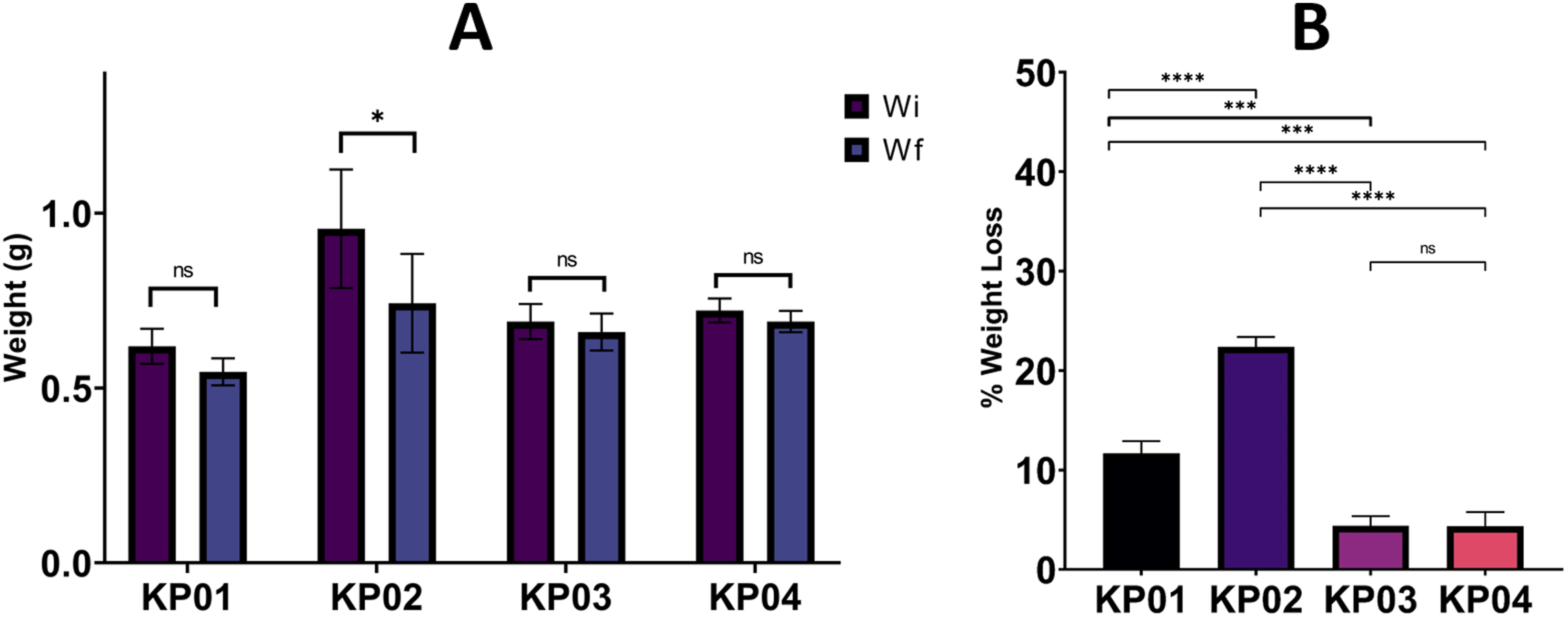
When we examine the KP01 plate group, weight decreases in the range of approximately 11%∼13% during the degradation process (30 days). When compared to other groups, KP01 is the second group whose degradation occurs the fastest. We think the fact that the TMC rate, which is the second raw material with the fastest degradation time among our raw materials mentioned in the literature and has the highest concentration of 20%, is effective in this. The fastest weight reductions were observed in the KP02 group, in the range of approximately 21.5%∼22.5%. The reason for this rapid degradation is thought to be due to the high concentration of PGA, which has the fastest degradation time among raw materials and its concentration is much higher than other groups. The KP02 plate group has proven its suitability for bone treatments in infants and children that show rapid development and recovery in terms of degradation rate. Groups KP03 and KP04 have similar degradation weight rates. Both groups contain 20% and 24% β-TCP and 45% and 46% LPLA, respectively. However, two separate components were used: 25% DLPLA in KP03 and 18% PGA in KP04. Despite the use of PGA in KP04, it is thought that the 24% β-TCP ratio slows down the degradation rate.
In vitro cytotoxicity analysis
The biocompatibility of bone plates was investigated with MTT assay (Figure 9(A)). Cell viability was determined as the percentage of living cells compared with the control group. All the bone plates showed non-cytotoxic properties. The cell viability for all the groups was found to be %100 indicating that the bone plates were biocompatible. Cell viability of composite plates (A) and effect of bone plate composition on cytotoxicity. The statistical analyses showed a statistically significant difference for %25 (v/v) extract cell viability. %50 (v/v), %75 (v/v), and %100 (v/v) extracts cell viability were statistically nonsignificant. (B). ⊥ indicates statistically nonsignificant groups.
The effect of the composition of bone plates on cytotoxicity is investigated by the MTT elusion method (Figure 9(B)). The statistically significant difference was only observed with %25 extraction groups. The cell viability for KP01, KP02, KP03, and KP04 were %89,4, %100, %81, and %100, respectively. For all groups and extracts cell viability was observed above %70 which means all compositions are noncytotoxic. Although KP01 and KP03 had the same components with different concentrations, the cell viability was above %70 hence the varying concentrations didn’t affect the biocompatibility. A similar result is also observed for KP02 and KP04 since they have the same components with varying concentrations too. There was statistically significant difference for %25 diluted extracts of KP01 – KP02 (p < .01 **), KP01 – KP04 (p < .01 **), KP02 – KP03 (p < .0001 ****), KP03 – KP04 (p < .0001 ****).
Deformation analysis
Four nose treatment plates and facial chin treatment plates produced using mixtures of KP01, KP02, KP03, and KP04 were selected, and their shape memory performance in a hot water environment was examined using a hot water bath. For this purpose, the obtained samples were immersed in water at a temperature of 65°C, heated for 30 s, and then deformed into a helix. Without removing the load, they were immersed in water at a temperature of 10°C and kept for 45 s, and their temporary shapes were given. The deformed product was again immersed in water at a temperature of 65°C and the recovered shape ability of the sample was monitored. Figure 10 shows the changes of the plates in the groups in hot water baths. Although less shape change was observed in the KP01 group than in the other groups, the products given temporary shape in cold water tended to return to their previous shape when thrown back to 65°C. All other groups showed a tendency to return to their initial shape when we immersed them in 65°C water and released them after being given their temporary shape at 10°C. Some slight cracks occurred in the KP02 group. It is thought that these cracks are caused by the PGA concentration we use. As a result of this experiment, the effect of TMC use on the shape change we targeted was clearly observed. Deformation tests of composite plates in hot water.
Conclusion
With the materials obtained, a buffering feature obtained from the ceramic additive has been added to the structure, and thus, a material has been developed that can prevent the necrosis problem caused by the decrease in ambient pH as a result of the degradation encountered in similar polymeric repair plates. It is thought that the regenerative potential of these materials is increased with the ceramic phase they contain. If these products are produced successfully, an innovative product will be developed to correct trauma fractures encountered with bone loss in the maxillofacial region. Deformable composite products were obtained in a 65°C water bath. In TGA analysis, ceramic-based β-TCP material showed more thermal resistance than polymers and contributed to increased thermal stability in the products. Especially the experiments carried out to develop a product by combining ceramic-based bioabsorbable β-TCP composite plates with other products in the study for the first time to be used for facial area treatment, have developed a new perspective for similar products.
Footnotes
Acknowledgments
This study was supported by the Scientific and Technological Research Council of Turkey (TÜBİTAK) with Grant Number 221M063. We thank BMT Group for equipment and material support. We would like to thank Corbion Company in Amsterdam for raw material support. This study is a part of Ilker Cengiz’s doctoral thesis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific and Technological Research Council of Turkey (TÜBİTAK) with Grant Number 221M063.
