Abstract
This study evaluates the effectiveness of a developed surface-modified carbon black, both alone and combined with nano-silica, as a hybrid filler for butyl rubber (IIR) compounds. The modification process, followed by coupling agent treatment, influenced crucial curing properties, including torque, cross-link density, viscosity, stiffness, and curing time. Scanning electron microscopy (SEM) analysis revealed improved filler dispersion and enhanced filler-rubber compatibility due to surface modification. The ∆Torque for compounds containing modified carbon black surpassed others by 13%. Mechanical properties such as tensile strength, elongation at break, modulus, tearing strength, and hardness were significantly influenced by filler type and surface modification. The harness (Shore A) increased to the value of 45 after surface amendment. Dynamic mechanical analysis (DMA) provided insights into storage modulus, loss modulus, and tan δ, showing the impact of filler type and surface modification. The utilization of coupling agent-modified carbon black decreased
Keywords
Introduction
Butyl rubber, also known as isobutylene-isoprene rubber (IIR), a rich history dating back to the early 20th century. 1 In 1931, at Standard Oil (now ExxonMobil), chemists William J. Sparks and Frank J. Soday reported the discovery of this remarkable elastomer while searching for synthetic gasoline production methods. Their accidental polymerization of isobutylene yielded a highly gas-resistant rubber, marking a groundbreaking moment in the scientific and industrial landscape. Butyl rubber possesses a unique chemical structure that differentiates it from other common elastomers like natural rubber (NR) and styrene-butadiene rubber (SBR). It is a copolymer composed primarily of isobutylene (around 98%) and a small amount of isoprene (approximately 2%). This specific makeup grants butyl rubber its distinctive properties. 2 The majority of the isobutylene units in the chain consist of two methyl groups attached to every other carbon atom, creating a hindered structure. This steric hindrance restricts the chain mobility, leading to decreased crystallinity and increased gas impermeability compared to its counterparts. The limited presence of isoprene units, known for their double bonds, contributes to the crosslinking of rubber chains during curing and vulcanization. 3 Butyl rubber with exceptional air retention is the material of choice for inner tubes in tires. Its impermeability to gases prevents air from slowly escaping, maintaining tire pressure. The resistance of butyl rubber to chemicals and fluids, accompanied by excellent flexibility, makes it ideal for seals and gaskets in various applications. It is commonly used in automotive components, appliances, plumbing systems, and even pharmaceutical packaging due to its biocompatibility. 4
In scientific literature, isobutylene-isoprene or butyl rubber is frequently reinforced with various fillers to enhance its properties and performance, as well as to mitigate the overall cost of the rubber compound. Among the common fillers employed in IIR compounds are carbon black (CB) and silica. CB stands out as the predominant filler in the rubber industry, with nearly 90% of global carbon black production allocated to the tire industry.5,6 The benefits of CB over alternative fillers in rubber compounds encompass its cost-effectiveness, favorable processability, and satisfactory abrasion resistance. Additionally, CB can augment several mechanical, electrical, and thermal properties of butyl IIR compounds. Nonetheless, CB exhibits certain limitations, such as inadequate dispersion within the rubber matrix, leading to numerous defects in the rubber compound during usage. Silica serves as another filler in rubber formulations, enhancing the dynamic properties of the rubber compound, particularly in reducing rolling resistance and providing excellent traction on wet and icy surfaces.
However, silica is comparatively expensive and exhibits poor wear characteristics due to inadequate dispersion in rubber compounds.7–11 The utilization of nano-silica as a filler in rubber has garnered significant attention owing to its superior properties.12–14 A recent advancement in rubber technology involves the adoption of hybrid fillers, which entail combinations of two or more distinct fillers. 15 Such hybrid filler systems have demonstrated the capability to yield synergistic effects, thereby enhancing the interactions between rubber and fillers. Consequently, they contribute to superior reinforcement and dispersion compared to single filler systems. Moreover, hybrid fillers offer the potential to reduce the requisite filler quantities for achieving desired properties, thereby mitigating both the cost and environmental impacts associated with rubber compounds.16,17 Considering these benefits, the proposition of a hybrid filler system comprising carbon black (CB) and nano-silica emerges as a promising strategy to capitalize on the respective advantages of both fillers while mitigating their drawbacks.
The combination of CB and silica along with rubbers such as IR, 17 NR and modified NR,18–22 NR/BR,23,24 NR/NBR, 25 NR/SBR, 26 and SBR and modified SBR16,27–31 has received considerable attention in the scientific community, as evidenced by previous studies and reports.
To the best of the authors' knowledge, the utilization of silane-modified carbon black resulting from the formation of covalent bonds between commercial CB and a silane coupling agent has not been previously explored or documented. Previous work by the authors has involved the surface modification of commercial carbon black using a silane coupling agent. 32 Building upon this foundation, the authors have investigated the structure of the produced silane-modified CB (referred to as Modified-CB-Oxi) filler using various characteristic techniques. Based on their findings, the authors suggest that Modified-CB-Oxi holds promise as a highly efficient reinforcement filler in rubber compounds. This study aims to examine the impact of commercial CB, modified CBs, and nano-silica, both individually and in combination, on the curing, mechanical, and dynamic properties of rubber compounds filled with isobutylene-isoprene rubber (IIR). Additionally, the morphology and dispersion of the fillers within the rubber compounds were evaluated. The results and ensuing discussion center on the suitability of IIR rubber compounds for application in the formulation of tire inner liners.
Experimental
Materials
In this investigation, the primary raw material employed was isobutylene-isoprene rubber (IIR), specifically IIR-1675N, characterized by a range of 1.4-1.8 mol% unsaturation and a Mooney viscosity of ML (1 + 4)125°C falling between 46 and 56. This particular variant of butyl rubber was procured from SIBUR, Russia. The reinforcing fillers utilized included commercial carbon black N660, sourced from Pars Carbon Ltd, Saveh, Iran. This carbon black, serving as the primary filler, is a general-purpose furnace carbon black (99.9% purity). Additionally, nano-silica, obtained as ULTRASIL® (99.9% purity) from Evonik Industries AG, Germany, was employed as the secondary filler. In addition to the raw rubber and fillers, various other components were integrated into the rubber formulation as curing chemicals. These comprised TBBS (N-tert-butyl-2-benzothiazyl sulfenamide), an accelerator obtained from Flexsys, Belgium; nano zinc oxide (99.8%), functioning as an activator, sourced from Robin Arman Nano Technology Co., Iran; stearic acid, serving as another activator, procured from Anchor Chemical Ltd, UK; and elemental sulfur, a curing chemical acquired from Solvay Barium Strontium, Hannover, Germany. Furthermore, toluene (Laboratory quality with 99.8% purity), provided by Mojalali, Iran, was utilized as a solvent specifically for the measurement of physical properties, including crosslink density and bound rubber content. Bis(triethoxysilylpropyl) tetrasulfide (TESPT), coupling agent provided by Evonik Industries AG, Germany.
Sample preparation and curing of the rubber compounds
Surface functionalization of CB
Based upon the methodology outlined in previous research,
32
the initial stage involved subjecting carbon black to oxidation by immersing it in a solution containing citric acid monohydrate and distilled water, aided by ultrasonic vibration (Sono Swiss, Switzerland, 280 W and 50/60 Hz). This process, conducted to generate modified carbon black (CB-Oxi), aimed to chemically graft functional groups such as hydroxyl (–OH) and carboxyl (–COOH) onto the surface of commercial carbon black (CB, N660). The introduction of these functional groups was anticipated to facilitate subsequent reactions with the silane coupling agent. Following oxidation, CB-Oxi was dispersed in ethanol using an ultrasonic bath for 30 min before the introduction of the coupling agent (TESPT) into the solution. The reflux process was continued for 3 h at 65°C. Upon completion of the grafting reaction, the solution was allowed to cool to room temperature. The treated CB-Oxi was then separated using a centrifuge (HS 18,500, Farzaneh Arman Co., Iran, 230 V, 50 Hz) and underwent three wash cycles with ethanol. Subsequently, the TESPT-functionalized CB-Oxi was dried at 50°C for 12 h, denoted as “Modified-CB-Oxi". A similar procedure was applied to untreated commercial carbon black (CB) to generate another modified carbon black variant, designated as “Modified-CB” (Scheme 1). The procedure for preparation of Modified-CB-Oxi/IIR compounds.
Mixing and preparation of rubber compounds
Formulation of butyl rubber (IIR) compounds with different fillers (phr).


The proposed mechanism for reactions between Modified-CB-Oxi and IIR rubber.
Characterization methods
Measurement of the cure properties
The cure characteristics, including the cure rate index (CRI), optimum cure time (t90), scorch time (ts2), and ∆torque (the difference between maximum and minimum torque), which serve as an indication of crosslink density changes in the butyl rubber compound, were assessed using a modulated disc rheometer (MDR, SMD-200B, Santam Co, Iran). The tests were conducted at a temperature of 160°C and a test frequency of 1.7 Hz. The CRI value was determined utilizing equation (1)33,34
Morphology assessment of the IIR compounds
The dispersion of fillers and the morphology of rubber compounds were evaluated using a scanning electron microscope (SEM, LEO 1450VP, Germany) with a resolution of 2.5 nm at 20 kV. Vulcanizate samples, with a thickness of 2 mm, were fractured in liquid nitrogen, and the resulting fracture surface was coated with a layer of gold via sputtering treatment prior to SEM observation.
Measurement of the Mechanical properties
The tensile properties, including tensile strength, elongation at break, initial modulus, and yield modulus of the butyl rubber (IIR) compounds, were determined via uniaxial tension testing utilizing a SANTAM STM-20 mechanical testing machine. Standard dumbbell-shaped test specimens, measuring 95 mm in length with a central neck of 26 mm length and 2.3 mm width, were employed for the tests. The experiments were conducted at room temperature with a crosshead speed of 500 mm/min. 35,36 Additionally, a tear test was conducted on at least three die C specimens in accordance with ASTM D624. 37 Both tensile and tear tests were repeated at least three times for each sample, and the median values were recorded. Furthermore, the hardness of the cured cylindrical samples, with dimensions of 12.5 mm thickness and 29.0 mm diameter, was assessed following the ASTM D2240 standard test method using a shore A type durometer. The measurement duration for shore A hardness was set at 5 s, and the hardness was recorded at room temperature. This procedure was repeated at five different positions on each sample, and again, the median value was reported.38,39
TGA analysis
Thermogravimetric analysis (TGA) stands as an effective technique for assessing the thermal stability of rubber compounds. 40 TGA was conducted utilizing a TGA-STA6000 instrument from Perkin Elmer, USA. The analysis was performed at a temperature range of 25 to 600°C at a heating rate of 10°C/min under a nitrogen atmosphere.
Dynamic mechanical thermal analysis
Dynamic mechanical thermal analysis (DMTA) was employed to assess the damping factor (tan δ = E″/E′), elastic modulus (E′), and loss modulus (E″) of the compounds. The testing was performed using a NETZSCH DMA 242 instrument from Germany. All measurements were executed at a frequency of 1 Hz, with a temperature sweep ranging from −150 to 80°C at a heating rate of 5°C/min.
Measurement of crosslink density, bound rubber, and swelling ratio
The crosslink density (CLD) of the vulcanized isobutylene-isoprene rubber (IIR) compounds was assessed in accordance with ASTM D6814.
41
The cured rubber compounds underwent swelling in toluene at room temperature for 72 h until equilibrium swelling was attained. Subsequently, the swollen rubber was extracted from the solvent, and any excess solvent present on the rubber’s surface was carefully removed prior to weighing. The crosslink density (CLD) was determined using the Flory–Rehner equation (2)33,42:

The density of toluene is 0.867 g/cm3. The swelling ratio of the IIR-cured compound was also calculated using equation (4)33,34:
The bound rubber, which indicates the percentage of rubber bound by filler, was determined using toluene as the solvent, similar to the method employed for CLD and swelling ratio measurements. For this determination, samples sized “20 × 20 × 2 mm” were placed in 50 mL of the solvent in labeled bottles and allowed to swell for 3 days at room temperature. The weight of the samples was measured daily until equilibrium was achieved. Subsequently, the solvent was removed, and the samples were dried in air for 9 h followed by further drying in an oven at 85°C for 24 h before re-weighing. The samples were then allowed to rest for an additional 24 h at room temperature. Equations (5)8,46 was utilized to calculate the bound rubber content (Rb).
Measurement of thermodynamic parameters of IIR compounds
Similar to the test method described in the previous section, the Flory-Huggins equation was utilized to calculate thermodynamic parameters such as elastic Gibbs free energy (∆G) and conformational entropy (∆S). These parameters were determined using ,equations (6) and (7) respectively.

R is the universal gas constant, T is the absolute temperature, Vr is the volume fraction of swollen rubber, and X is the rubber solvent interaction parameter. In equation (7), it is assumed that there is no change in the internal energy of the rubber network during stretching.45,48–50
Results and discussion
The curing properties of IIR compounds
Curing characteristics of prepared IIR compounds with different fillers.

The maximum torque (
The delta torque (∆Torque), denoted as the difference between the maximum ( The comparison between Δ torque for nano-silica/CB (A6), nano-silica/Modified-CB (A7), nano-silica/Modified-CB-Oxi (A8), and nano-silica/CB-Oxi (A9) filled IIR compounds.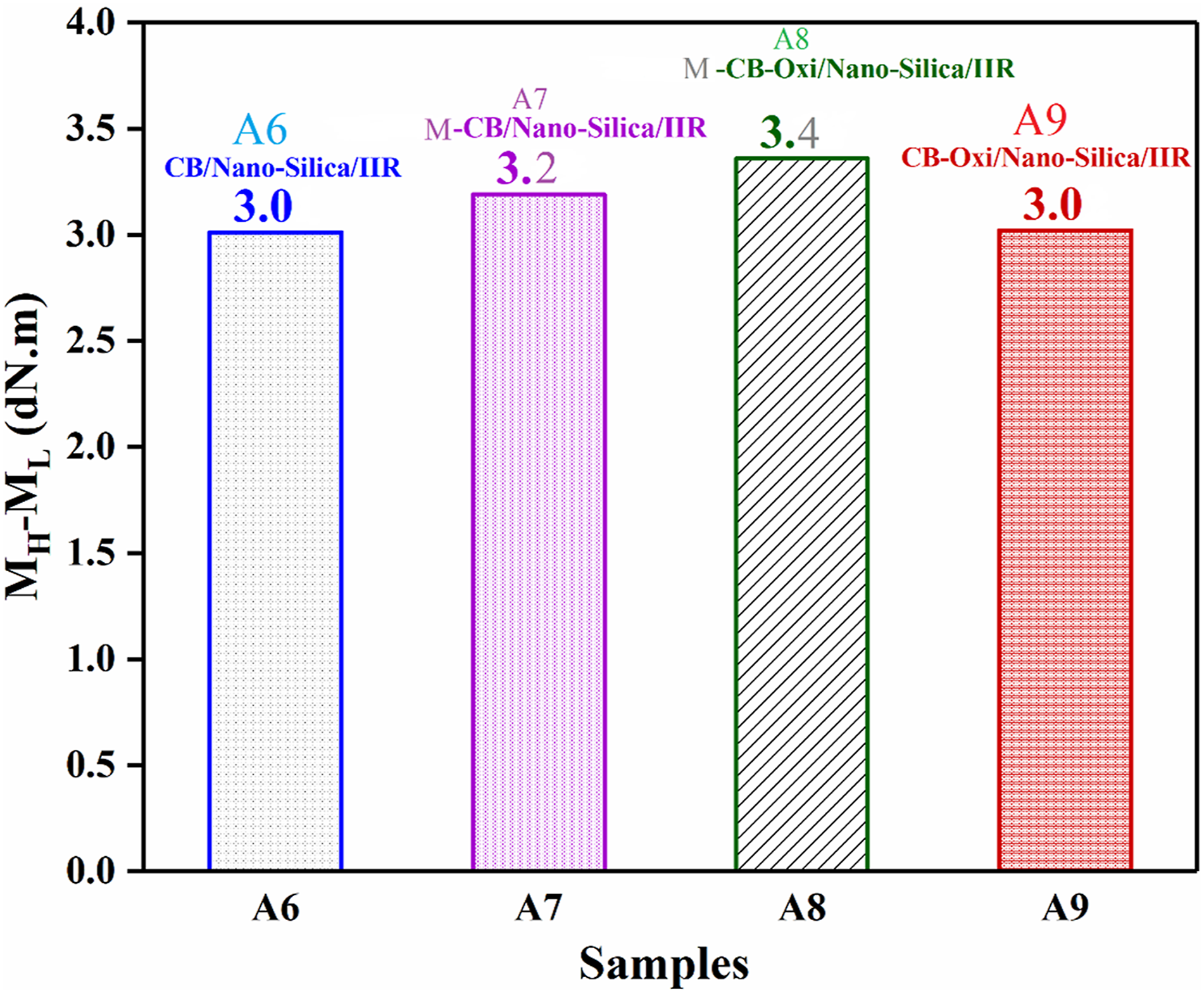
Crosslink Density, swelling ratio, and bound rubber of prepared butyl rubber compounds with different fillers.

The optimum curing time (
Furthermore, the cure rate index (CRI) of the mentioned compounds exhibited remarkably different values. The CRI of A5 was significantly higher compared to the other compounds, reaching 25.4
In conclusion, the reported results demonstrate how surface modification of carbon black and the use of different fillers can influence various aspects of the rubber curing process, including pre-scorch time, optimum cure time, and cure rate. The specific effects depend on the type and degree of modification, filler type, and filler-rubber interactions. Additionally, the reported findings underscore the importance of carefully selecting and optimizing filler materials and surface modifications to tailor the curing process to achieve desired characteristics and performance outcomes in rubber-based applications.
The morphology study of IIR compounds
Figures 2 and 3 provide a comparative analysis of scanning electron microscopy (SEM) images illustrating the cross-sectional surfaces of cured butyl rubber (IIR) compounds employing selected single (Compounds A2, A4, and A5) and hybrid fillers (A6 and A8) at a magnification of 100 μm. In Figure 2, Compound A4 containing Modified-CB-Oxi particles exhibits a well-dispersed and homogeneously distributed morphology within the IIR matrix. This dispersion is ascribed to the formation of covalent bonds between the rubber macromolecules and the filler surface (carbon black), facilitated by the coupling agent TESPT. Such enhanced filler–rubber interaction in Compound A4 contributes to improved filler dispersion.9,13 Conversely, poor filler dispersion is evident in Compounds A2 and A5, filled with CB and CB-Oxi, respectively. This phenomenon is notably pronounced in Compound A5 due to the higher presence of polar functional groups, such as carboxyl and hydroxyl, on the carbon black surface. Large agglomerates of carbon black were observed in the SEM image of A5. As IIR is inherently non-polar, CB-Oxi particles tend to agglomerate, forming larger agglomerates rather than dispersing within the rubber matrix. Similarly, in Figure 3, a more favorable dispersion of Modified-CB-Oxi/nano-Silica in the rubber matrix (Compound A8) is observed compared to commercial CB/nano-Silica (A6). SEM images of these compounds reveal the presence of nano-silica agglomerates, further highlighting the importance of filler dispersion in influencing the properties of cured rubber compounds. A well-dispersed filler in a cured rubber compound is associated with enhanced mechanical and dynamic properties.9,34,53 The uniform distribution of filler particles facilitates efficient stress transfer from the rubber to the filler, resulting in increased strength, modulus, and tear resistance. Conversely, inadequate dispersion can yield adverse effects. Agglomerated filler particles serve as stress concentration sites, rendering the compound more susceptible to crack initiation and propagation, thereby deteriorating its mechanical and dynamic performance. The incorporation of the coupling agent TESPT in Compound A4 led to a significant enhancement in filler dispersion by establishing covalent bonds between the Modified-CB-Oxi and the IIR matrix. This demonstrates the potential of surface modification techniques to improve filler-rubber compatibility and achieve superior dispersion. Importantly, the findings from SEM images were consistent with the results obtained for most mechanical and dynamic properties, as detailed in The mechanical properties of IIR compounds and Dynamical properties of prepared IIR compounds. This coherence reinforces the critical role of filler dispersion in influencing the overall performance of cured rubber compounds. The SEM images for selected individual filler-filled rubber compounds, CB/IIR (A2), Modified-CB-Oxi/IIR (A4), and CB-Oxi/IIR (A5) at the magnitude of 100 μ The SEM images for selected hybrid filler-filled rubber compounds, CB/nano-Silica/IIR (A6) and Modified-CB-Oxi/nano-Silica/IIR (A8).

The mechanical properties of IIR compounds
Selected mechanical properties of the cured butyl rubber (IIR) compounds.

The elongation at break parameter provides valuable insights into a rubber compound’s flexibility, range of motion, compatibility, and resistance to fatigue, all of which are critical factors in material selection for diverse applications, ensuring dependable and enduring performance. Among Compounds A1 to A5, each formulated with individual fillers, A2 exhibited the highest elongation at break value at 576.3%, while A1 showed the lowest at 325.7%. Compound A5, consisting of CB-Oxi, demonstrated the highest elongation at break, recording a value of 560.2% after A2. The low value for A1 refers to incompatibility and low rubber-filler (nano-silica) interaction in the rubber matrix. In the case of compounds utilizing hybrid fillers (A6 to A9), Compounds A9 and A8, incorporating CB-Oxi/nano-silica and M-CB-Oxi/nano-silica, respectively, demonstrated the highest and lowest elongation at breaks, measuring 422.1% and 334.0%, respectively. The remaining compounds exhibited elongation at break values within the range of 346.7% (A7) to 393.7% (A6), dependent upon the type of carbon black utilized. These observed results indicate a dependency on filler-rubber interaction, the formation of filler networks, and the type of surface modification applied to the filler on elongation at the break of the IIR rubber compound.
In rubber mechanical testing, both initial modulus and yield modulus serve as indicators of stiffness, yet they denote distinct points in the material’s behavior. The initial modulus, characterized as the slope of the initial linear segment of the stress-strain curve, reflects the material’s stiffness during small deformations. Conversely, the yield modulus, defined as the stress at which the material undergoes permanent plastic deformation, signifies the junction at which the material shifts from elastic to plastic behavior. Beyond the yield point, the material fails to fully restore its original shape following deformation. The initial modulus of Compound A1 to A5, except Compound A1, with a value of 0.8 MPa, remained practically unchanged, with values ranging from 0.5 to 0.6 MPa. For compounds filled with hybrid fillers (Compounds A6 to A9), this parameter showed a modest change ranging from 0.5 for A6 and A9 to 0.7 MPa for A8. Approximately, a similar trend was observed for the yield modulus. Among individually filled rubbers, the lowest and highest values were attributed to Compounds A1, and A2 and A5, measuring 0.6 and 0.3 MPa, respectively. However, for compounds utilizing hybrid fillers, the lowest and highest values were found in Compounds A6 and A9, and A8, with values of 0.4 and 0.6 MPa, respectively. The observed lower values of both initial and yield modulus for compounds containing nano-silica, and CB and CB-Oxi compared to those of M-CB and M-CB-Oxi suggest a reduction in the overall stiffness of the rubber compounds, indicating the inherent incompatibility of these fillers with the rubber matrix, as previously described in SEM images. Additionally, this serves as evidence of the effectiveness of the coupling agent TESPT in improving both initial and yield modulus.
The tearing strength of rubber compounds is a critical property that directly influences their ability to resist punctures, cuts, and cracks, thereby impacting their functionality, safety, and reliability across various applications. Notably, Compound A1, formulated with nano-silica, exhibited the highest tearing strength, measuring 17.2 MPa, followed by A2 with a value of 16.0 MPa. Unexpectedly, the surface modification of carbon black had a detrimental effect on this parameter, reducing it to the range of 12.4 to 13.5 MPa. Additionally, among the hybrid filler compounds, Compounds A6 to A9, a similar trend was observed. Incorporating modification on the carbon black surface in combination with nano-silica had an adverse effect on tearing strength, with values ranging from 12.4 to 12.9 MPa. As observed from A6, the combination of CB and nano-silica created a synergistic effect, further reducing this mechanical property due to increasing destructive filler (CB)-filler (nano-silica) interaction.
Optimal hardness for a specific application depends on individual requirements and desired performance characteristics. A material might need a balance of hardness with other properties like flexibility or tear resistance. Table 4 compares the hardness (Shore A) of all prepared compounds. Among Compounds A1 to A5, A4 exhibited the maximum hardness at 45, while A5 showed the minimum hardness at 32, indicating the suitability of M-CB-Oxi as a filler in IIR rubber compound. Similarly, for hybrid fillers, a consistent trend was observed, with this parameter ranging between 39 and 45 for compounds filled with nano-silica and CB-Oxi, and M-CB-Oxi, respectively.
Dynamical properties of prepared IIR compounds
The dynamic properties of rubber compounds play an important role in determining their performance under varying and cyclical loads, contrasting with static properties that evaluate behavior under constant stress. Dynamic properties gauge how the material responds to oscillating forces or deformations, crucial for applications requiring specific behavior when subjected to vibrations, impacts, or repeated stretching and compression. Rubber compounds possessing appropriate dynamic properties, such as hysteresis and loss modulus, effectively absorb and dissipate vibrations, crucial for vehicle components like engine mounts, which mitigate noise and enhance ride comfort. Moreover, dynamic properties like the friction coefficient influence the grip and traction generated by rubber, critical for tires to maintain traction on diverse road surfaces, ensuring safe driving conditions. Dynamic Mechanical Analysis (DMA) emerges as a valuable tool for assessing the dynamic properties of rubber compounds in scientific literature. It allows for the comprehensive measurement of diverse dynamic properties, including storage and loss modulus, tan δ, and glass transition temperature ( Tan Dynamic mechanical properties of selected IIR compounds with different fillers (A6: CB/nano-silica and A8: Modified-CB-Oxi/nano-silica).

Considering ice and wet grip,11,53 a rubber compound’s higher tan δ at −20°C and 0°C generally indicates its potential for improved traction and performance on icy and wet surfaces. This elevated tan δ at these temperatures suggests enhanced energy dissipation within the rubber compound, leading to heightened frictional forces between the rubber surface, namely the tire tread, and the ice or wet surface. This heightened friction is conducive to better traction and grip. Nevertheless, a comprehensive assessment of tan δ data alongside other rubber properties, such as rubber hardness and article geometry such as tire tread patterns for tires is essential to gain a thorough understanding of a rubber compound’s suitability for winter conditions. As expected, Compound A8 exhibited a higher tan δ value at −20°C, measuring 1.16 compared to A6 with a value of 1.14. This observation indicates that the rubber compound filled with M-CB-Oxi is likely to offer improved ice grip. Unexpectedly, the tan δ at 0°C for A8 (0.74) was lower than that of A6 (0.82), implying a probable decrease in wet grip for A8 compared to A6.
If a rubber compound demonstrates a lower tan δ at +60°C, it indicates that the compound dissipates less energy, leading to reduced heat generation during rolling. This results in decreased rolling resistance, as less energy is expended in overcoming internal friction within the rubber-made articles, such as tires.11,53,55 As observed, Compound A8 exhibited a tan δ value of 0.12, which was lower than that of A6, which had a value of 0.15. This suggests lower rolling resistance for the former compound due to better filler dispersion within the rubber matrix compared to the latter compound. The above observations were consistent with the observed results for loss and storage modulus as indicated in Figure 5. Storage and loss modulus versus temperature for compounds A6 and A8 at 1 Hz. measurements were conducted at 1 Hz with a temperature sweep between −100 and 80°C at a heating rate of 5°C.min−1.
Thermal Stability and organic solvent resistance of prepared IIR compounds
Figure 6 depicts a comparative analysis of thermal stability between selected IIR Compounds A6 and A8. Thermal gravimetric analysis (TGA) was employed to investigate the thermal behavior of cured rubber compounds under a nitrogen atmosphere within a temperature range of 25 to 600°C, with a heating rate of 10°C/min. The findings reveal that degradation of IIR rubber occurred within the temperature range of 300 °C–430°C, with a peak loss rate observed at approximately 406°C. Furthermore, mass loss was noted within the temperature interval of 25 to 300°C, attributed to the decomposition of other light hydrocarbons present in the compound formulations. Notably, both A6 and A8 exhibited total mass losses of 86%, indicating no significant difference between the two. Moreover, there was no substantial mass loss observed above 430°C, suggesting the presence of unburned materials such as carbon black and zinc oxide.
56
In conclusion, the TGA thermographs of the studied compounds showed no significant disparity in terms of thermal stability. TGA thermograms for cured rubber compounds A6 and A8. TGA was performed in the range of 25 to 600°C at a heating rate of 10°C.min−1 under a nitrogen atmosphere.
To assess the resistance of the prepared rubber compounds to organic solvents, toluene was selected for immersion and swelling tests. As depicted in Table 3, the swelling ratios of Compounds A1 to A5, filled with individual fillers, varied between 1.6 for A1 and A4 to 2.1 for A2. Compound A5 exhibited a swelling value of 1.9, suggesting it was more susceptible to degradation by the organic solvent, in line with expectations. However, the treatment of carbon black (CB-Oxi) with TESPT to produce M-CB-Oxi notably improved the solvent resistance of the IIR rubber compound, as evidenced by the results for A4. Among the hybrid filler compounds, A6 to A9, no consistent trend was observed for this parameter, with the values remaining relatively unchanged when different types of carbon black were used along with nano-silica in the IIR rubber compound formulation. However, A8 exhibited the lowest swelling value of 1.6, indicating improved resistance to toluene compared to the other hybrid compounds, which had a swelling value of 1.8.
Bound rubber, a commonly utilized metric for assessing the interaction between filler and rubber chains, is presented in Table 3 for the IIR compounds. In the rubber compounds filled with a single filler, the bound rubber content of samples A1, A2, A3, A4, and A5 were recorded as 94.9%, 94.4%, 91.9%, 94.0%, and 93.1%, respectively. Interestingly, the silane modification of CB did not alter the bound rubber content significantly, maintaining nearly identical values across the CB-filled IIR compound, A2. Similarly, in the rubber compounds containing hybrid fillers, the bound rubber content ranged between 92.6% and 94.8%, exhibiting minimal variation. The sole exception was A6, which displayed the lowest bound rubber value of 92.6%, indicating exceptional destructive interaction between CB and nano-silica in this compound.
Thermodynamic study of IIR compounds
The thermodynamic properties of prepared IIR compounds with different fillers.

Conclusions
This investigation highlights the significant influence of surface modification of carbon black and the selection of different fillers on various curing properties essential for rubber compound processing and performance. Parameters such as minimum and maximum torque, delta torque, and cross-link density were notably affected by filler type and surface modification, impacting viscosity, stiffness, and curing time. Scanning electron microscopy (SEM) analysis provided visual evidence of how filler dispersion affects the morphology of cured rubber compounds. Surface modification of carbon black notably improved filler dispersion and enhanced filler-rubber compatibility, leading to improved mechanical properties. Tensile strength, elongation at break, modulus, tearing strength, and hardness were all influenced by filler type, surface modification, and filler-rubber interactions. The incorporation of nano-silica and surface-modified carbon black impacted the stiffness, flexibility, and tear resistance of the compounds. Dynamic mechanical analysis (DMA) revealed insights into dynamic properties such as storage modulus, loss modulus, and tan δ, showing how filler type and surface modification influence these characteristics. Glass transition temperature and tan δ at different temperatures provided further insights into the compounds' performance under various conditions, including ice and wet grip and rolling resistance. Thermal gravimetric analysis (TGA) indicated similar thermal stability across different compounds, while solvent resistance varied depending on filler type and surface modification. Surface modification notably improved solvent resistance, as evidenced by the observed swelling ratios. The bound rubber content reflected filler-rubber interaction, showing minimal variation across different compounds. The thermodynamic analysis highlighted the impact of filler type and surface modification on the elasticity and flexibility of rubber compounds, with surface-coupling agent-modified fillers mitigating detrimental effects observed with untreated fillers. In conclusion, precise selection and optimization of filler materials and surface modifications play a crucial role in tailoring rubber compound properties to meet specific performance requirements across diverse applications. Used surface modification not only enhanced filler dispersion and improved filler-rubber compatibility but also influenced curing properties, mechanical performance, dynamic behavior, thermal stability, solvent resistance, and thermodynamic parameters, ultimately shaping the overall quality and performance of rubber-based products.
Footnotes
Author contributions
Farzaneh Jaberi Mofrad, Ali Ahmadpour, and Saeed Ostad Movahed designed the experiments. Dr. Ali Ahmadpour prepared the manuscript with contributions from all co-authors. The authors applied the SDC approach for the sequence of authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the staff of the central laboratory, Ferdowsi University of Mashhad, Iran for their sincere support and cooperation (Grant no. 56010).
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
