Abstract
Owing to the requirement for environmental protection, water-based coatings have become a significant trend in the development of coatings. The water-based epoxy curing agent has also become a hot research topic. In this study, a B-A-B epoxy compound with a long hydrophobic alkyl chain connected to the intermediate nitrogen atom was synthesized at both ends of a molecule using ethylene glycol diglycidyl ether and 3,4-dimethoxyaniline as raw materials. Subsequently, the epoxy compound was sealed with triethylenetetramine. A nonionic, water-based epoxy resin curing agent with amino groups at both ends of the molecule was prepared. The target product structure was confirmed by infrared spectroscopy (IR), Mass spectrum (MS), and Nuclear magnetic resonance hydrogen spectroscopy (1H-NMR). Additionally, through three factors and three levels of orthogonal experimental design, the optimum experimental conditions were confirmed, the optimum yield was 82.83%. The thermogravimetric analysis (TGA) indicated that the monomer exhibited a significant weight loss in the temperature range of 320–450°C. The pencil hardness, flexibility, and impact resistance of the waterborne epoxy resin coating film prepared by the non-ionic water-based epoxy curing agent reached or exceeded those of similar products at China and foreign countries.
Keywords
Introduction
Research and development of epoxy resin curing agents are preparing a new type of curing agent that can improve its resistance to moisture, heat, toughening, and strengthening. It can improve insulation and flame retardancy, reduce viscosity, and improve processing performance.1–4 It can also improve the performance of the existing resin curing agent by modification and realize the high efficiency and multifunction of the curing agent through compounding technology.5–9 In recent years, numerous countries have promulgated environmental protection laws and regulations to limit volatile organic solvents (VOC), and water-based coating has become a crucial trend in the development of coatings.10–13 As a type of water-based epoxy curing agent, its properties have a vital influence on the physical and chemical properties of the film. Therefore, the development of a water-based epoxy curing agent has become a hot research topic.14–16
Water-based epoxy curing agents can be divided into two categories: type I and type II. Type I epoxy systems require their curing agents to have the functions of curing and emulsifying. The epoxy resin is a liquid or emulsion, and the curing agent is a water-soluble amine curing agent. The first appeared in the modified aliphatic amine water-soluble curing agents, such as Casmide360 and Casmide362, developed by the Thomas Swan Company, followed by the new type I water-based curing agent without requiring organic acid salt. 17 For example, the type I water-based epoxy curing agent by Stark et al. was obtained by the reaction of epoxy resin and excess m-benzo-diamine to obtain an end-epoxyamine additive, which was then reacted with end-carboxyl polyether alcohol to form amide-amine, and then obtained by sealing the ends. Zhang 18 had synthesized a novel self-emulsifiable water-based amine-terminated curing agent for epoxy resin based on glycidyl tertiary carboxylic ester through three steps of an addition reaction, capping reaction, and salification reaction of triethylene tetramine and liquid epoxy resin (E-44). Hu 19 had synthesized TETA-NPER-032 additives by the reaction of polyether polyol diglycidyl ether with triethylene tetraamine, and then further reacted it with liquid epoxy resin (EPON828) to synthesize a water-based epoxy curing agent. Zhao 20 had synthesized a new nonionic, self-emulsifying, water-based epoxy resin from bisphenol A epoxy resin E44 and p-methoxy polyethylene glycol benzylamine. Ni et al. 21 synthesized a water-based epoxy resin curing agent with both emulsifying and curing functions by using propylene glycol methyl ether as a solvent and ethylenetetramine, epoxy resin, and polyethylene glycol as raw materials.
The type II epoxy system is already in an emulsion state, so its curing agent only requires the function of a crosslinking agent, which is composed of solid epoxy resin with a relatively high-quality water-based epoxy curing agent. It is a new water-based epoxy resin system from the end of the 20th century into the 21st century. Type II water-based epoxy curing agents are mostly fatty amines with hydrophilic ends, epoxy-amine, and polyamide curing agents.22–24 Elmore 25 first added polyvinyl polyamine to epoxy resin (EP1001), then reacted with carboxylated polyether alcohol to prepare amide amine compounds, and finally the amide-amine water-based epoxy curing agent was obtained by sealing the monocyclic oxide compounds. Zou 26 used octadecylamine and ethylene glycol diglycidyl ether as raw materials in the first reaction and then sealed the end with triethylenetetramine in the second step. Finally, a nonionic water-based epoxy resin curing agent with a similar surfactant structure was prepared. Huang 27 used ethylene glycol, diglycidyl ether, and hexadecylamine as raw materials in the first reaction, and then sealed with diethylenetriamine. Finally, a non-ionic, self-emulsifying, water-based epoxy curing agent with a surfactant structure was synthesized.
In addition, Alice Mija,28–30 Jomin Thomas31,32 et al. reported the application of bio-based or vegetable oil epoxide, which could potentially compete with waterborne systems.
In this study, we synthesized a B-A-B type epoxy compound with a long hydrophobic alkyl chain connected to the intermediate nitrogen atom, using ethylene glycol diglycidyl ether and 3,4-dimethoxyaniline as raw materials. Subsequently, the epoxy compound was sealed with triethylenetetramine. A nonionic, water-based epoxy resin curing agent with amino groups at both ends of the molecule was prepared. The product structure was confirmed by infrared spectroscopy (IR), mass spectrum (MS), and nuclear magnetic resonance hydrogen spectroscopy (1H-NMR) detection. Additionally, through three factors and three levels of orthogonal experimental design, the optimum experimental conditions were confirmed. The thermogravimetric analysis (TGA) and the differential scanning calorimetry (DSC) techniques were used to investigate the thermodynamic properties of the target compounds. The pencil hardness, flexibility, and impact resistance of the waterborne epoxy resin coating film prepared by the non-ionic water-based epoxy curing agent reached or exceeded those of similar products at home and abroad.
Experimental section
Materials
Diglyceride glycol (SY-669) was obtained from the Changzhou Runxiang Chemical Company (China). 3,4-dioxyaniline was obtained from the Shanghai Sass Chemical Technology Company (China). Triethylene tetramine (TEDA) and anhydrous ethanol were obtained from the Chengdu Kelong Chemical Testing Factory (China).
Methods
The 1H-NMR spectra were recorded on a Bruker Advance-400 MHz spectrometer. Deuterium chloroform was used as the NMR solvent, and tetramethylsilane was used as the internal standard. The FT-IR spectral profiles were recorded on a Nicolet FTIR-380 instrument. The KBr salt slice method was used for recording the spectra. DSC measurements were performed using a Q1000DSC thermoanalyzer system (TA Instruments, Co., Ltd, USA; temperature range: 25 to 350°C) at a heating rate of 10°C/min under a nitrogen (N2) atmosphere. TGA tests were performed using a Q1000TGA thermoanalyzer system (TA Instruments, Co., Ltd, USA) at a heating rate of 20°C/min under an atmosphere of N2 in the temperature range of 50°C–600°C.
The Performance Test Method: The table dry time was determined based on GB/T1728-1979 (1989) “paint film putty film drying time determination method,” the hardness of coating was determined based on GB/T1730-1993 “paint film and hardness determination method,” the flexibility of paint film was determined based on GB/T1731-1993 “method for measuring flexibility of paint film” and the impact resistance of coating was determined based on GB/T1732-1993 “impact resistance method of paint film.”
Preparation of additional products of SY-669 and 3,4-dioxyaniline
Under the protection of dry nitrogen, glycol diglycidyl ether (B) was loaded into three flasks using anhydrous ethanol as a solvent. When warming to 65°C, the dissolved 3,4-dioxyaniline (A) of ethanol was transferred to the constant pressure drop funnel and slowly dripped into three flasks. After the titration was completed, the brown oily addition product (B-A-B) was obtained after the titration reaction had been ongoing for some time. The reaction equation is shown in Figure 1. Synthesis of nonionic water-based epoxy resin curing agent (C-B-A-B-C).
Preparation of a nonionic water-based epoxy resin curing agent by TEDA sealing end
Under the protection of dry nitrogen and using anhydrous ethanol as a solvent, the brown oily addition product (B-A-B) was loaded into three flasks with a constant pressure drop funnel. Warming to a constant value of 60°C, the speed of the mixer was 15r/s. The TEDA (C) dissolved in ethanol was then loaded into the constant pressure drop funnel, and the TEDA was slowly added when the system temperature reached 60°C. After the titration was completed, the target product of glycol diglycidyl ether (SY-669)-3,4-dimethoxyaniline-triethyltetraamine (TEDA) could be prepared (C-B-A-B-C) after the titration reaction. Subsequently, the obtained crude product was purifed through column chromatography to obtain a brown oily substance. The reaction equation is shown in Figure 1.
Results and discussion
Factors influencing the synthesis of B-A-B products
Effects of feeding methods
If the method of dripping ethylene glycol diglycidyl ether into 3,4-dimethoxyaniline, because more 3,4-dimethoxyaniline was in the solution, the epoxy groups at both ends of ethylene glycol diglycidyl ether molecules were opened to obtain A-B-A and other compounds rather than target products, so to obtain B-A-B target products, the 3,4-dimethoxyaniline would slowly be added to ethylene glycol diglycidyl ether (SY-669). Thus, the 3,4-dimethoxyaniline could be connected with two ethylene glycol diglycidyl ether (SY-669) molecules to achieve the B-A-B target structure.
Effects of temperature, raw material ratio, and reaction time
Since ethylene glycol diglycidyl ether (SY-669) was a bifunctional substance in this reaction, two reaction sites existed, and the hydrogen reaction between the epoxy group and 3,4-dimethoxyphenylamine was active. Therefore, it was easy to form low-relative molecular-weight polymers or even gels. To produce B-A-B compounds in which epoxy compounds are at both ends, the reaction should be performed at a lower temperature. Primary and secondary amine hydrogens on nitrogen atoms in 3,4-dimethoxyaniline were involved in the reaction to obtain B-A-B type compounds. So, the temperature must at least reach the reaction activity temperature of secondary amine hydrogen. Considering the increase of side reactions in organic reactions at a high temperature, the reaction temperature was set at 65°C. Under this temperature, the raw material ratio n (SY-669): n (3,4-dimethoxyaniline)=3:1 was more suitable, and the reaction time was 3–4 h.
Factors influencing the synthesis of target products by TEDA sealing end
Effects of feeding methods
Two epoxy groups existed in the addition product (B-A-B), and there were two reaction sites, and both epoxy groups were required to open rings. There were four reaction sites in the TEDA; however, two secondary amine nitrogen atoms in the TEDA molecule had low activity because of the existence of a great steric hindrance reaction. So, the main reaction sites of the TEDA were on two primary amines at both ends of the molecule. Only one primary amine reacted, and the other primary amine was retained. Therefore, to obtain a C-B-A-B-C target product with primary amine at both ends of the molecule, the material was added as follows: the addition product (B-A-B) was slowly added to the TEDA.
Effects of temperature, raw material ratio, and reaction time
The key point of this reaction was to take control of the reaction. To obtain C-B-A-B-C compounds, only the primary amine hydrogen at one end of the TEDA was required to participate in the reaction, and the secondary amine hydrogen did not react. Thus, the reaction temperature should not be excessive, and so 60°C was sufficient. The ratio of raw materials at this temperature: n (B-A-B): n (TEDA)=1:5 was more appropriate because of the characteristics of molecular structure; insufficient TEDA was easy to produce gel, resulting in crosslinking and excessive waste. The reaction time was 3–4 h.
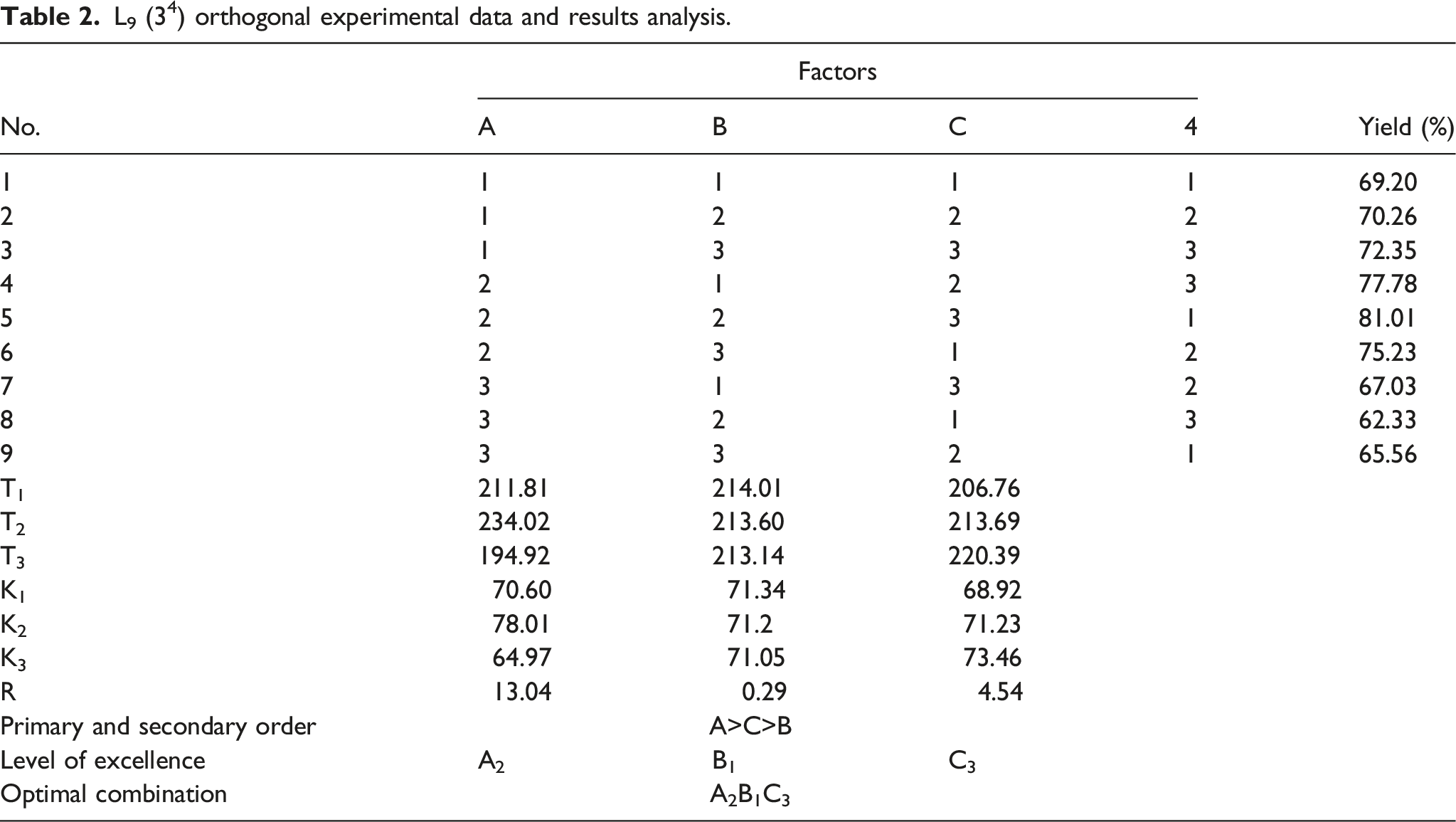
Study on the conditions of synthesis reaction by the orthogonal experiment
Three factors and three levels.

L9 (34) orthogonal experimental data and results analysis.
Characterization of products
The infrared spectrum of the raw material TEDA is shown in Figure 2. The 3354.57 cm−1 was the stretching characteristic peak of -NH2, which should show two peaks in theory; however, because of the association of -NH2, it presents a wide peak. The 2930 cm−1 and 2820 cm−1 were the C-H stretching vibration peaks. The N-H shear characteristic vibration peaks were 1660 cm−1 and 1576.52 cm−1, showing two peaks. The C-H (-CH2-) bending vibration peak was 1470.46 cm−1. IR spectra of type C-B-A-B-C compounds and TEDA.
A TEDA molecule was required to be connected at both ends of the B-A-B compound to react with the epoxy group in the B-A-B compound to obtain the final C-B-A-B-C compound. TEDA molecules open all the epoxy groups in the compounds, and only one of the primary amine hydrogens was involved in the reaction, while the other end of the primary amine did not participate in the reaction. Subsequently, the characteristic peaks of cyclooxygenase in the infrared spectra of C-B-A-B-C compounds should disappear and not appear, and the corresponding primary amine characteristic peaks in the TEDA should appear.
The infrared spectra of C-B-A-B-C compounds are shown in Figure 2. 3431.71 cm−1 were the overlapping peaks of - NH2 and - OH, which were a broad peak because of the association effect; 2915.95 cm−1 were the peaks of saturated C-H stretching vibration; 1519.30 cm−1 were the characteristic peaks of benzene ring skeleton vibration; 1350.89 cm−1 were the characteristic peaks of methyl vibration on Ph-OCH3; 1132.97 cm−1 were the characteristic peaks of the C-O-C bond vibration in ethylene glycol diglycidyl ether; and 1021.12 cm−1 were the characteristic peaks of Ph-O-C ether bond vibration. The characteristic peaks of the 911.20 cm−1 and 855.28 cm−1 epoxy groups in Figure 2 did not appear, indicating that all the epoxy groups of the B-A-B compounds were opened and that TEDA was involved in the reaction.
The 1H-NMR spectra of the type C-B-A-B-C compounds are shown in Figure 3. The chemical shifts and splitting patterns of the protons present in the compound were recorded: δ 6.77,6.35 ppm (Ph-H, -CH=CH-), 3.70 ppm (-C-O-H), 1.68 ppm (C-N-H), and 1.23 ppm (Ph-O-CH
3
). 1HNMR spectra of type C-B-A-B-C compounds.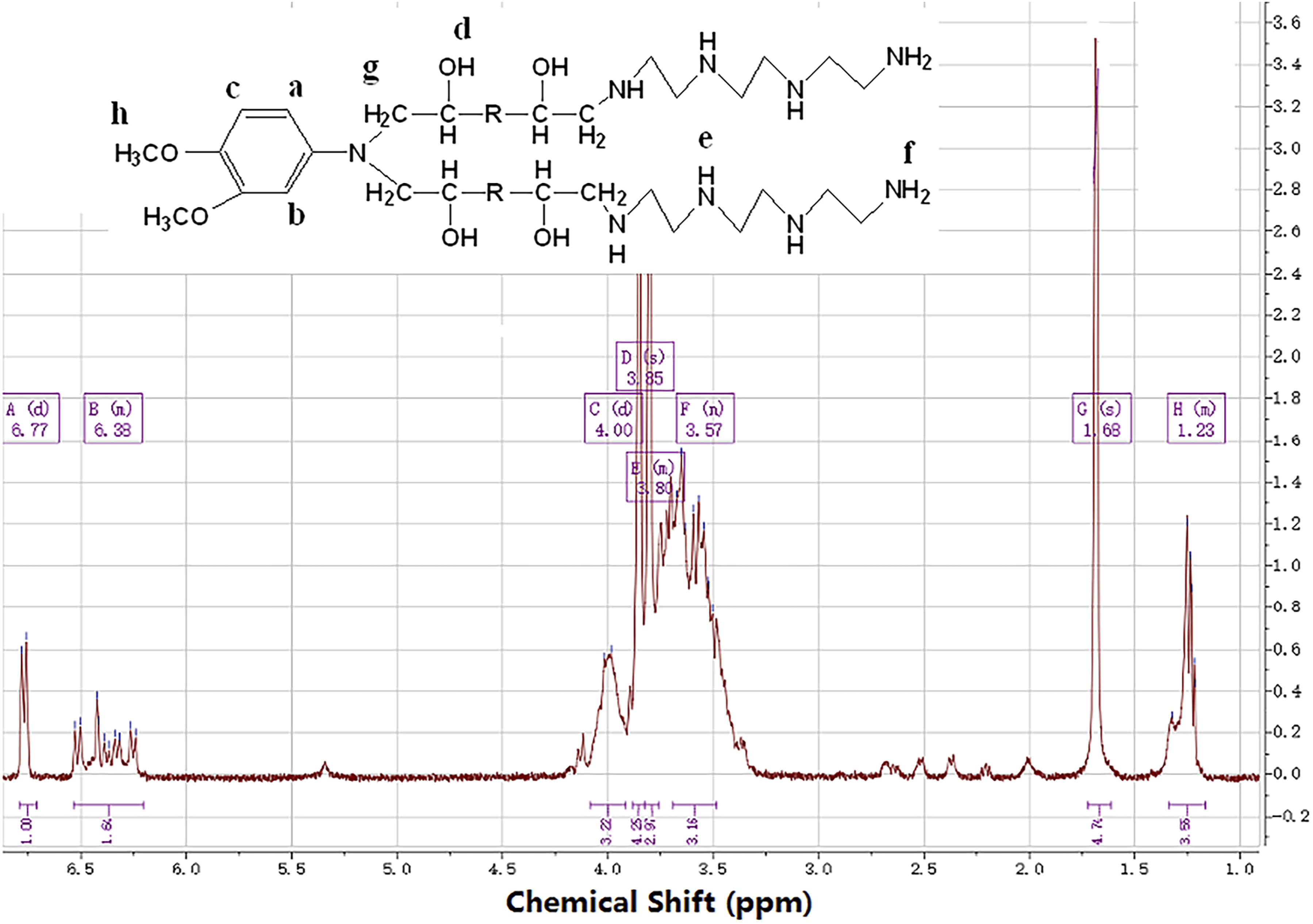
The theoretical relative molecular weight of C-B-A-B-C compounds was 793. No molecular ion peaks were found in Figure 4. The ion peak with a mass-charge ratio of 556.3 should be formed after the breaking of the C-C single bond on a B molecule structure in the C-B-A-B-C molecule. Mass Spectrometry of type C-B-A-B-C compounds.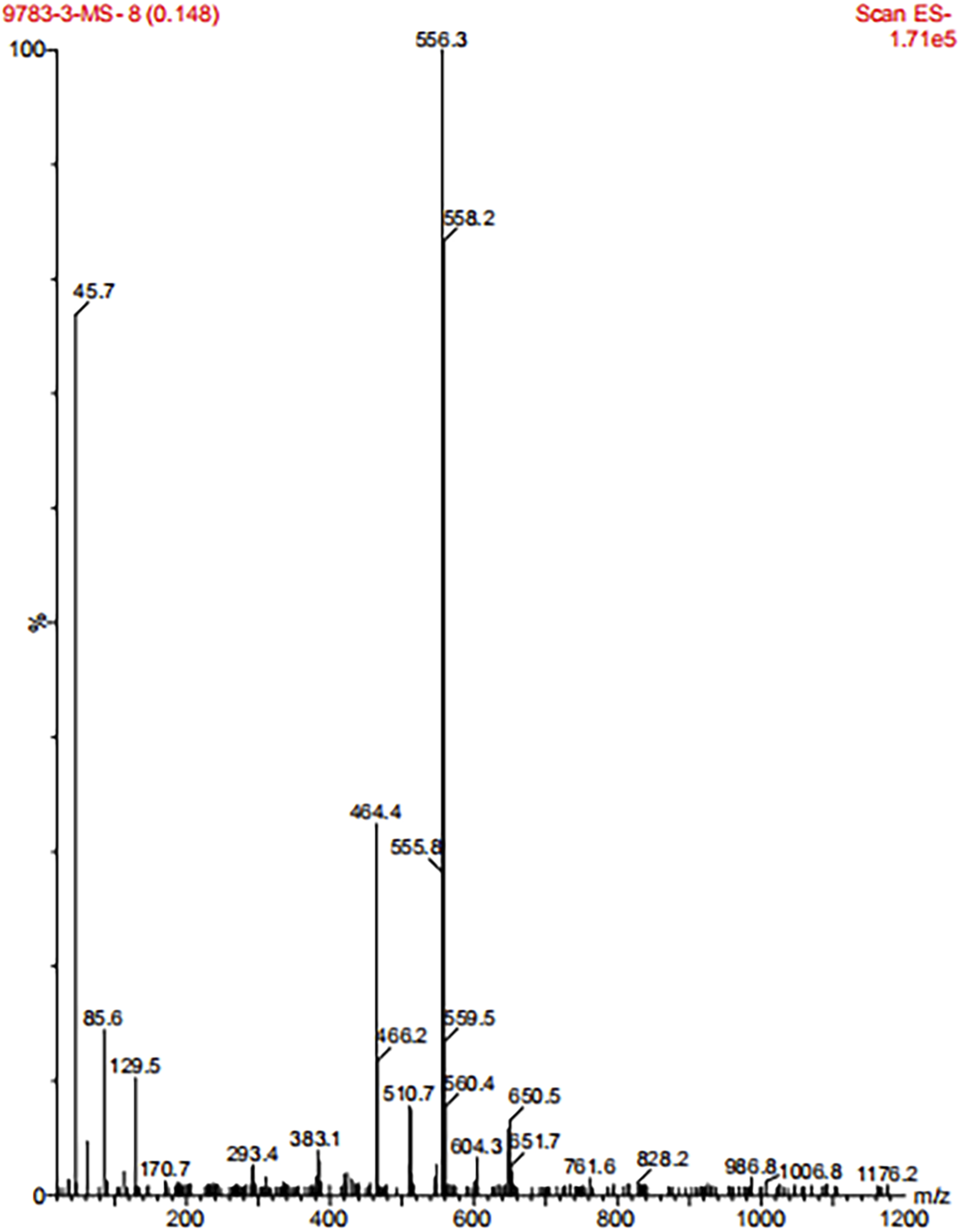
The comprehensive analysis of IR, 1HNMR, and MS of C-B-A-B-C compounds might determine that the C-B-A-B-C compounds were the target product.
Evaluation of the thermal properties of the product
The thermal properties of the target product, a C-B-A-B-C type compound, were evaluated using the TGA technique (Figure 5). The rate of weight loss was 5% at 253°C. This indicated the volatilization of small monomers or solvent molecules. The monomer exhibited a significant weight loss in the temperature range of 320–450°C. At 368°C, the weight loss rate was 50%. At 565°C, the weight loss rate was 94%. This indicates that the residual carbon rate was low at high temperatures. TGA spectra of C-B-A-B-C type compounds.
Figure 6 shows the DSC spectra of the target product, C-B-A-B-C type compounds; the heating rate was 20°C/min. As shown in the diagram, at 91.535°C, the heat of the reaction was 37.922 J/g; thus, it could be the volatilization of small molecules. At 143.251°C, an obvious endothermic peak existed; the heat of the reaction was 12.438 J/g, and at 296.656°C, there was an obvious exothermic peak of the curing reaction; the heat of the reaction was 68.010 J/g. It showed that the curing reaction temperature was relatively high, and the reaction heat was low. DSC spectra of C-B-A-B-C type compounds.
Filming properties
Comparison of the filming properties of different curing agents.

As shown in Table 3, the pencil hardness, flexibility, and impact resistance of the waterborne epoxy resin coating film prepared by the non-ionic waterborne epoxy curing agent reached or exceeded those of similar products at China and foreign countries. Moreover, because the long flexible alkyl chain was introduced in the curing agent, the brittle defects of the general epoxy resin cured film were improved, and the flexibility of the cured film reached 1 mm.
Conclusions
First, a B-A-B epoxy compound with a long hydrophobic alkyl chain connected to the intermediate nitrogen atom was synthesized at both ends of the molecule and had an epoxy base using SY-669 and 3,4-dimethoxyaniline as raw materials. Subsequently, the epoxy compound was sealed with TEDA, a new nonionic water-based epoxy resin curing agent with amino groups at both ends of the molecule was prepared. IR, MS, and 1HNMR detection confirmed this as the C-B-A-B-C product. Additionally, through three factors and three levels of orthogonal experimental design, the optimum experimental conditions were as follows: reaction temperature (T1:T2)=65°C:60°C, reaction time was 2.5 step by step, and the raw material ratio was n(3,4-dimethoxyaniline): n(ethylene glycol diglycidyl ether): n(triethylenetetramine)=1:3:4 (standard for 3,4-dimethoxyaniline). The optimum yield was 82.83%. TGA performance analysis indicated that the monomer exhibited a significant weight loss in the temperature range of 320–450°C. At 368°C and 565°C, the weight loss rates were 50% and 94%, respectively. This indicated that the residual carbon rate was low at high temperatures. DSC performance analysis demonstrated that the product had an obvious endothermic peak at 91.535°C, and the reaction heat was 37.922 J/g, which may correspond to the volatilization of small molecules. An obvious endothermic peak at 143.251°C existed, and the reaction heat was 12.438 J/g. An obvious exothermic peak at 296.656°C existed, and the reaction heat was 68.010 J/g, which indicates that the curing reaction temperature was high, and the reaction heat was low. The pencil hardness, flexibility, and impact resistance of the waterborne epoxy resin coating film prepared by the non-ionic water-based epoxy curing agent reached or exceeded those of similar products at China and foreign countries. Moreover, because the long flexible alkyl chain was introduced in the curing agent, the brittle defects of the general epoxy resin cured film were improved, and the flexibility of the cured film reached 1 mm.
In the future, we will study the crosslinking density of the resin-cured film by the dynamic thermomechanical analysis (DMA) tests and the mechanical properties of the resin-cured film by the Instron tests. We will make the new non-ionic water-based epoxy resin curing agent and epoxy resin into a water-based epoxy coating that is used in actual production and daily life and is expected to make a certain contribution to the protection of the environment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Sichuan Province Key Research and Development Projects (2019YFG0252 and 2023YFG0237).
