Abstract
A nanogel is a cross-linked nano-sized, a three-dimensional network of hydrophilic polymers with an ability to swell by holding large amount of water while maintaining the structure due to chemical or physical cross-linking of individual polymer chains. Nanogels can be made up of synthetic and/or natural polymers resulting in a cationic, anionic, or neutral system depending on the bound groups’ charges. Currently, these materials are receiving tremendous attention in research due to their properties. They are extensively investigated as carriers in the biomedical field. At present, there is an expansion of research into dermatologic treatment due to a critical need for new treatment options to treat skin diseases. The skin itself provides a natural barrier against particle penetration for topical delivery. However, it also offers a potential approach for the delivery of therapeutics, especially in diseased skin via the openings of hair follicles. Recent innovation might be achieved in the field of dermatological treatment with improvement in the dermal localization of bio-actives into the affected skin region. This report looks at what has been done in the investigation of nanogels as drug carriers for topical therapy.
Introduction
Tremendous work has been published on hydrogels since the 1960s. 1 These materials have proven to be bioactive with remarkable biocompatibility, excellent biocompatibility, hydrophilicity, flexibility, versatility, and high-water absorptivity. For these reasons, many functional hydrogels have been prepared and studied in medical applications for drug delivery.2,3 For a very long time, the delivery of drugs has been the major short coming for various drugs. Hence, pharmaceutical nanotechnology devoted studies to formulating therapeutical and biocompatible drug carriers in nanoforms. 4 In the past years, nanotechnology has played an immense role in drug delivery since nanoparticles easily adhere to cells and are biocompatible and therapeutically active unlike macromolecules. 5 However, there are still hurdles associated with the release mechanisms of the loaded agents to specific sites due to less effective polymeric network used, and most of these drugs suddenly dissolve before reaching their destination, causing side effects. 6 The incorporation of nanoparticles and hydrogels into nanogels further improves the nanomaterials’ properties and abilities.7–10 The polymeric system of nanogels has a variety of properties of interest which includes high capacity to load drugs and the ability to deliver the loaded drug into a targeted location and increasing the permeability of a drug without harming the normal cells.11–17 Based on their properties, nanogels emerged as a new drug delivery system and received tremendous attention in the group of nanoparticulate systems as one of the most promising versatile drug carriers. 18 Their ability to carry and deliver drugs is well known.19,20 Dermal therapy has proven to be preferable route for drug delivery and localized drug action. 21 This route is preferable for treating skin disorders as the skin can be a convenient entrance for chemicals. This is mainly due to the advantages such as sustained release kinetics, controlled delivery of bioactive, ease of self-administration, bypass first-pass metabolism, elimination of GIT degradations, bowel distress, which leads to the easy selective targeting of the damaged area, limited spreading of the drug and lower dosage which will result in the decreased chances of toxic side effects. However, penetration due to the barrier caused by the stratum corneum (SC) composition has been a limiting factor for the topical application therapy. 22 When nanoparticles cannot penetrate through the skin SC therefore, they use the hair follicles as a way in. However, nanoparticles accumulate in skin furrows and hair follicles creating high local concentrations of loaded drugs that can further diffuse to the viable layers of the skin. 23 In the study where a drug loaded nanogel, it was observed that the permeability of the drug through the skin is influenced by the particle size. The release improves as the size of the particles decreases. 24 Rusu et al, conducted a research on biocompatible nanogels as carriers of Amox they noticed the embedding of these nanogels in Hyaluronic acid hydrogels to be an advantage as it improved the stability in physiological conditions and controlled the over rapid release of the loaded drug. 25 In the study conducted on “Self-assembled nanogel of Pluronic-conjugated heparin as a versatile drug nanocarrier” the ability of nanogels to solubilize hydrophobic drugs was further demonstrated, however, the amount of drug loaded is important. This study, likewise, showed that the swelling ratio of the nanogels is related to the kind of hydrophilic functional group and that the delivery of hydrophobic/hydrophilic drug highly depend on the structural properties of the polymer. 26 It is important to mention that nanogels are administered through different routes including injection, oral, pulmonary, nasal, parenteral, intra-ocular, 27 however this review focuses on the dermal therapy via topical routes of administration.
Classification of nanogels
Nanogels are generally classified into three major groups namely the linkage type, the responsive type as well as structural type (Figure 1)
Linkage type nanogels are further categorized into chemically and physically linked nanogels. Essentially, this classification is based on the method of the synthesis. Chemically cross-linked nanogels have covalent bonds linking the polymer networks. The bonding mainly involves amide-based crosslinking, disulphide-based crosslinking, photo-induced crosslinking as well as Schiff-base reactions.31,32 These nanogels are highly stable owing to the presence of covalent bonds in their structure. Physically cross-linked nanogels are formed through linkages such as hydrogen bonding, hydrophobic interactions as well as electrostatic interactions. 33 As the interactions are weak, physically crosslinked nanogels are less stable compared to chemically cross-linked. Responsive type nanogels category is divided into stimuli-responsive and non-responsive nanogels. While the nonresponsive nanogels only swell and de-swell due to water absorbance, the stimuli-responsive type swell depending on the environmental, physical, and chemical changes such as pH, temperature, ionic strength, and magnetic field.33–35 The properties of the stimuli-sensitive nanogels highly depend significantly on the polymers. Nanogels can be pH sensitive when composed of cross-linked polyelectrolytes with weakly acidic e.g., carboxylic groups or weakly basic e.g. amino groups which can be used either as proton donors or receptors or through their combination. However, other polymers are thermo-responsive and are considered adequate for delivery systems in biomedical devices where there are specific temperatures. Copolymerization leads to obtaining multi-sensitive nanogels by incorporating polymers that can respond to different stimuli.36–41 The pH-responsive gels control the drug release in an environment with certain pH through the ionization of the appropriate attached groups developing fixed charges on the polymer network, producing electrostatic repulsive forces responsible for pH-dependent swelling or de-swelling of the hydrogel. 42 Structural type Nanogels can also be classified according to their structures. These vary from simple, 43 core-shell, 44 hairy, 45 hollow, 46 multi-layered 47 and functionalized nanogels. The core-shell nanogels have been reported to exhibit dual thermo-responsive behaviour as their core would have a lower critical solution temperature, while the shell would have an upper critical solution temperature behaviour. 48 A hairy nanogel has a core-shell type of structure with the nanosized core displaying the nano-effect while the shell has linear polymeric chains that have a greater affinity with the dispersion medium.49,50 Multi-layered nanogels, prepared from linear polymers via hydrogen bonding or ionic pairing, can change their volume dramatically and can therefore accommodate different types of molecules.51,52 The functionalization on the surface of the functionalized nanogels is advantageous as it helps to control the nanogel pathway toward a specific target as well as the cargo release kinetics thus limiting the toxicity on normal cells.53,54
Synthesis of nanogels
Nanogels can be synthesized from either natural, synthetic, or both polymers. However, every design of nanogels is suitable for a specific application with carefully selected polymers. The advantage of using natural (carbohydrates) polymers is that they are biodegradable, biocompatible, and less toxic owing to their nature. However, when exposed to the external environment they give microbial contamination. Again, their dependents on the environment and different physical factors lead to uncontrollable hydration and heavy metal contaminants. On the other hand, Synthetic manufacturing is controllable which allows the attachments of all the desired branches when they are designed.35,55 Figure 2 shows the general formation of nanogels and the examples of the PH, magnetic and thermo-responsive nanogels loaded with Doxorubicin. No. Two shows the reaction between N,Nbis (acryloyl)crystamine and N-isopropylacrylamide in the presence of acrylic acid to form temperature responsive nanogels which showed speed release in the tumor cell environment.
56
No.3 shows a reaction of nanogels loaded with magnetic nanoparticle as carriers for the drug. These nanogels showed accelerated delivery when exposed to the magnetic field since the application of high-frequency magnetic field is to magnetic nanoparticles generate heat.
57

Various approaches have been utilized to formulate nanogels. These approaches can be classified into four: Physical assembly of polymers, chemical cross-linking of polymers, polymerization of monomers, and template-assisted fabrication. 58 Physical assembled polymers can form nanogels in the presence of amphiphilic copolymer and a therapeutic agent in an organic solvent through weak noncovalent interactions, such as Van der Waals, hydrogen bonding, stereo complexation, charge transfer, and polyelectrolyte complexation. 59 This self-assembly method when used shows versatility and simplicity. It has been utilized to fabricate chitosan-based nanogels that can be further functionalized in enhancement for biomedical applications. 60 Chemical synthesis of nanogels (Chemical cross-linking of polymers) in heterogeneous colloidal environments can give a wide spectrum of capabilities for various structures and properties of nanogel. This covalent cross-linking method has been extensively employed in the preparation of a wide range of functional nanogels for drug delivery as it is an approach that is likely to obtain nanogels with large pore sizes. 61 For polymerization in a homogeneous phase approach, a colloidal suspension of the polymer is made via the homogeneous nucleation of water-soluble monomers used in the synthesis of stabilized nanogels. 62 The approach is used to manipulate particle size with the particle size inversely related to surfactant concentration. For Polymerization in a heterogeneous phase, in general two methods are used to synthesize nanogels, the normal (oil/water) and reverse (water/oil) emulsions.63,64 In the oil-in-water approach, aqueous nanodroplets of water-soluble monomers and bi-functional monomers are dispersed with the aid of oil-soluble surfactant in the organic solvent. 65 On the addition of a water-soluble initiator, polymerization then takes place in aqueous droplets affording cross-linked hydrophilic nanogels. The water-in-oil emulsion methods involve emulsification of aqueous droplets of water-soluble biopolymers in a continuous oil phase with the aid of oil-soluble surfactants followed by the crosslinking of biopolymers using water-soluble crosslinkers. Multiple approaches fall under this category, and they include general approaches including inverse (mini)emulsion, membrane emulsification, and reverse micellar methods.66,67
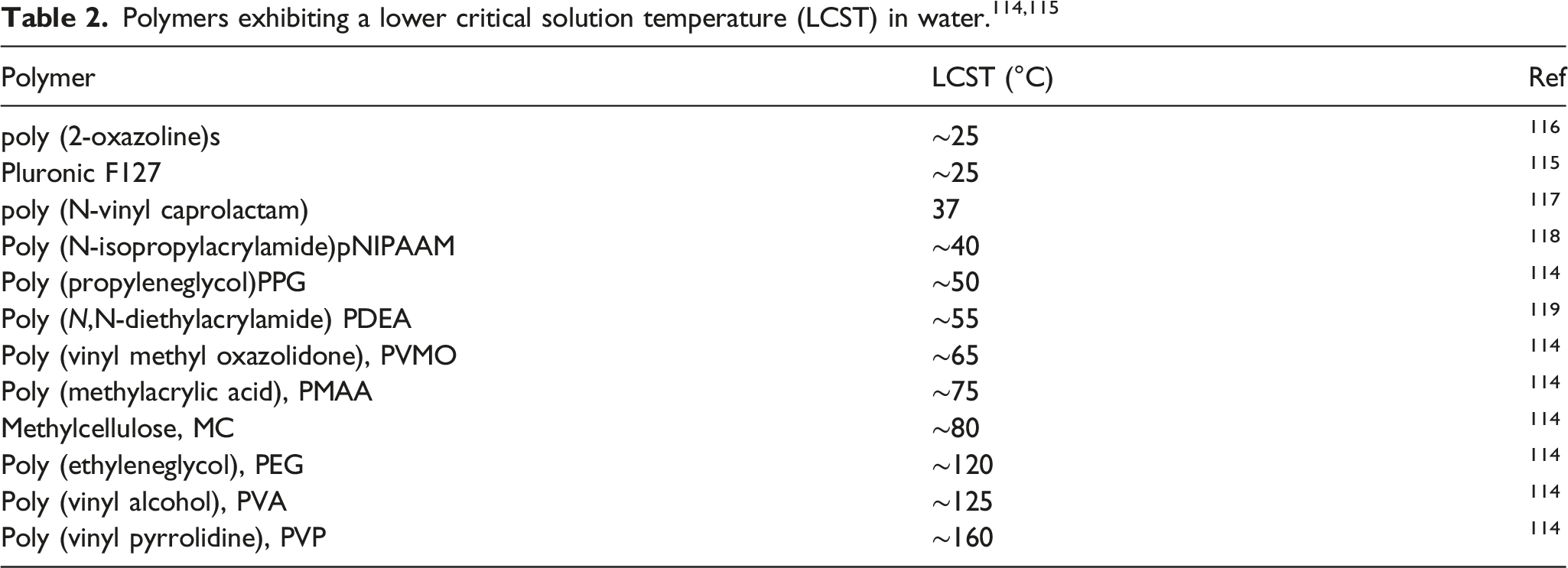
Polymers in nanogel formulation for topical treatment
The mostly studied polymers on different topical applications.

Nanogel drug release mechanisms
The release of drugs from nanogels has been reported to be dependent on the affinity of the drug to the polymer as well as the network mesh size of the nanogels. However, in the former case, polymer/drug interactions may significantly contribute to controlling and regulating the release of the drug.
103
In the latter case, nanogel swelling/de-swelling influences the likelihood of the drug going in or out of the network. Several mechanisms identified include diffusion, pH-responsive mechanism, thermosensitive transition mechanism, and nanogel degradation Figure 3. Nanogel drug release.
102

Diffusion
This is a very simple drug release mechanism expected when a drug is physically encapsulated in the nanogel. The sizes of the open spaces (meshes) between nanogel polymer networks control the steric interactions between the encased drugs and the polymer network and hence determine how drugs diffuse through the material. 104 The diffusional drug release mechanism was demonstrated by Missirlis et al. when they reported the release of doxorubicin from a nanogel based on pluronic block copolymer whose hydrophobic core provided considerable protection against hydrolytic degradation. 105 Coll Ferrer et al. also demonstrated the release of dexamethasone from dextran-lysozyme nanogels via the diffusion mechanism. 106
PH-responsive mechanism
In this mechanism, the swelling or de-swelling of nanogels containing weakly basic or acidic groups in their structure is caused by changes in pH. Thus, the mechanism exploits the changes in pH within the body for selective response in specific cellular compartments. 107 Moreover, the versatility of the pH-responsive nanogels is displayed when taking into consideration the fact that pH gradients exist between diseased and normal cells with, for example, cancerous cells. Zhou et al. have demonstrated that the Coulombic repulsions that exist between the deprotonated carboxylic groups of nanogels based on poly (methacrylic acid) (PMAA) at high pH induce the swelling of the nanogels, while under acidic conditions, the carboxylic acid groups are not ionized and the nanogels are in a collapsed state. 108
Thermosensitive mechanism
Nanogel degradation mechanism
Nanogel degradation is another mechanism that is available for the release of drugs entrapped in the polymer network. The degradation causes an increase in nanogel mesh size resulting in drugs diffusing out of the material. Nanogel degradation, which is characteristically mediated by enzyme activity or hydrolysis, can occur in the polymer backbone of the cross-links. 120
Nanogels as drug carriers in the treatment of skin diseases
Many different skin diseases affect more than 50% of the population in the world. Nanogels are currently being investigated for the treatment of different skin diseases via topical drug delivery. The available medications can help reduce and cure some skin diseases. The most common approaches include skin maintenance care techniques, topical anti-inflammatory agents, systemic antihistamine, topical or systemic antibiotics, and selectively, systemic corticosteroids, however, most skin disorders are chronic diseases that cannot be permanently removed by any drug but needs a lifetime treatment and Psoriasis is one of them.121–123 The challenge at hand is, most currently used medications are found toxic such that they result in a variety of drug-induced diseases. 124
Topical drug delivery promotes a localized effect thereby reducing the total drug to be used to reach the targeted site. The first direct evidence that nanogels can penetrate the skin and migrate across the epidermis was reported by Samah et al. using transmission electron microscopic images.125–128 Hair follicles seem to be an easy way in through the skin of the nanogels, however, the particle size range is important as the medium-sized polymeric particles in the range 300 nm to 500 nm were found to penetrate effectively unlike the smaller particles.129,130 Giulbudagian et al designed nanogels based on three thermo-responsive polymers poly (N-isopropylacrylamide), p (di(ethylene glycol) methyl ether methacrylate-co-oligo ethylene glycol methacrylate, and poly (glycidyl methyl ether-co-ethyl glycidyl ether) and studied their drug delivery properties. 131 They proposed hydration mechanism as a particular mode of skin penetration enhancement mechanism that correlated well with the chemical composition of the nanogels. Shah et al. have demonstrated that the nanogel comprising surface-modified nanoparticles enhanced skin permeation of two anti-inflammatory drugs spantide II and ketoprofen across the deeper skin layers via the improvement of skin contact time and hydration of the surface of the skin (occlusive effect). 132
Nanogels as carriers for known active drugs
The ability of nanogels as carriers has been studied by investigating their effectiveness when loaded with the currently used drugs. Drugs such as Methotrexate, Acitretin, Etretinate, etc, are known for their efficacy in the treatment of different psoriasis types. Acitretin is known to be more unique compared to other systemic therapies for psoriasis as it is not immunosuppressive. It is therefore regarded as a safe option for patients with a history of chronic infection such as HIV, hepatitis B. 133
Gels are the best carriers to transport active pharmaceutical ingredients like methotrexate (MTX) into contact with the skin. 134 When the nanogels were investigated as carriers loaded with methotrexate against the imiquimod-induced psoriasis model developed, it was concluded that MTX-NLC gel showed the gradual release with more effective eradication of psoriatic manifestations when topically applied. 135 In another study, the niosomal methotrexate gel based on chitosan was tested in clinical studies. The niosomal encapsulation was made through the lipid layer hydration method with pH 5-phosphate buffer. The gel showed better results without any side effects when compared to the gels that are already in the market. 136 Tretinoin is well known for its activity against acne as a single agent therapy, however, it is often associated with serious side effects such as erythema, dryness, peeling, and burning which are likely to discourage patients from taking and finishing the treatment. Therefore, the incorporation of nanogels is studied to minimize these side effects. A clinical trial conducted in 2015 where Tretinoin Nanogel 0.025% as compared to its Conventional Gel 0.025% proved Tretinoin nanogel to be more effective and better tolerated. 137 Tretinoin incorporated in solid lipid nanoparticles showed better-sustained release, stability, and protection from degradation. The studies showed that chitosan solid lipid nanoparticles combined with Tretinoin resulted in an efficient topical treatment for psoriasis. 138 Diclofenac sodium was also successfully tested for dermal route administration through different glycol nanogels. The gels were optimized for homogeneity, particle size, pH, drug content, in vitro drug release, skin irritation test, spread ability, extrudability, and viscosity. In the beginning, the release rate was uncontrollable. However, when the gel phase had been reached the drug release rate slowed down. This proved that the formed gels can retain diclofenac sodium for the required duration. 139 On the other hand, the combination of existing drugs with the polymers was shown to be improving the activity of the drug with more additional advantages. An example is a study where Ibuprofen and Chitosan formed an interaction through the carboxylate ion of ibuprofen and the protonated amino group of chitosan. This led to a decrease in particle size of ibuprofen from 4580 (aspect ratio) to 14.15 nm (324-folds), Ex vivo releases studies on pig skin exhibited that the chitosan–ibuprofen–gellan conjugate nanogels are more permeable than the control ibuprofen gellan hydrogel by a factor of four. 140
Even though there have been successful cis-platin based chemotherapy there are still shortfalls of the chemotherapy which are: poor bioavailability, variety of side effects, drug resistance, and high dosage, to name a few. As a result, many target improved nanocarriers are being developed. 141 Cisplatin-directed coordination crosslinking nanogels (Pt-PNA) were developed via the coordination bonds of Pt-carboxyl, the excellent antitumor efficacies of Pt-PNA nanogels were attributed to the improvement in the capacities of sustained release and long-term retention in tumor for water-soluble cisplatin. For water-soluble cisplatin, Pt-PNA nanogels showed a sustained release over 5 days at 37°C, and a pH-dependent release, in addition, Pt-PNA nanogels showed much stronger cisplatin retention abilities than free cis-platin. 142 Nanogels loaded with well-known cancer therapeutic, cis-platin, are being studied. The targeted PEG-b-PMAA nanogels for cisplatin delivery to glioma synthesized by Baklaushev et al., showed limited penetration via the endothelial barrier towards the intracranial tumours when compared to free cisplatin. To improve the penetration and efficiency of drug delivery to the glioma, the nanogels were conjugated with mAbs (antibodies) against the targeted proteins. MRI confirmed a decreased tumor growth in comparison to other investigated formulations. 143 Moreover, the use of cisplatin incorporation with other effective drugs has shown an enhanced efficacy and improved safety, and nanogels can transport many different drugs. 144
Nanogels as carriers for herbal drugs
Nanogels have been found to be suitable carriers that can transport and improve herbal drug properties as well. Owing to their flexibility and versatility these materials have many chances in herbal drug developments as drug carriers. The effectiveness of herbal medicine depends highly on the cooperation of all its active compounds. Sadly, herbal drugs have solubility challenges that lead to less bioavailability and high systemic clearance. Nanogel derivatives of herbal drugs help to overcome such limitations.145,146 When Aloe-emodin-loaded chitin-based nanogels were tested against psoriasis they were found to be effective, however, were less effective when compared to the acitretin-loaded chi-tin-based nanogels. Nevertheless, the irritation study revealed that the aloe emodin-loaded gels did not produce any histopathological changes on skin such as erythema or wrinkles and showed better effectiveness against psoriasis when combined with the acitretin loaded gels. 147 Sabitha et al., formulated curcumin-loaded chitin nanogels and studied their activity against skin cancer through topical application. The nanogels showed selective toxicity towards the skin cancer cells and with no destruction of red blood cells and toxicity on normal cells which proved that curcumin uptake through the transdermal route might be the option for the future. 148 They also showed sustained release of the drug with enhanced permeation and less toxic effects when tested for Squamous Cell Carcinoma. 149 Mangalathillam et al. have investigated the use of curcumin-loaded chitin nanogels (CCNGs) in the treatment of skin cancer. 148 They showed that the cationic nature of the chitin polymer facilitated the interaction of the drug-loaded nanogels with skin lipids thus enhancing drug delivery via both the transcellular and intercellular pathways. They further demonstrated that the CCNGs were selectively toxic to melanoma cells and not toxic to normal cells.
Potential of nanogels in different diseases
For most skin related diseases, some of challenges faced by the drugs currently in use is water insolubility, low transdermal capacity, low drug bioavailability, skin permeability that can contribute to poor therapeutic efficacy. 150 In accelerating the pharmaceutical and medical development through technology, nanogels have shown an interesting potential in a vast spectrum of disease. For dermal therapy nanogels are studied against Bacterial, Fungal, Viral, and Parasitic Skin Infections. Topical application of nanogels in bacterial infection studies are underway. In a study where the in vivo experiments done by testing efficacy of the nanogels formulated from Hyaluronan tetrabutylammonium, it was proven that these gels might be used to deliver antimicrobials against intracellular pathogens. However, they still need to be improved for better skin penetration. 151 On the other study the nanogel at a concentration of 80 µg/mL reduced the viability of the tested parasitic infections to 0%. 152 This is very promising. Nanogels are evaluated also on the treatment of non-healing chronic wounds such as chronic diabetic ulcers. A lot has been done in the attempt of improving the healing process of these wounds, the literature offers a broad spectrum of different models of treatments studied to better the experience of patients with these chronic wounds.153,154 There has been tremendous growth in the interest of developing three-dimensional bioengineered substitute’s microfibers, and nanofibers with desired functions loaded onto gels. 155 When the activity of peptide-based nanogels was studied in the treatment of wounds the nanogels showed an ability to promote the proliferation of fibroblast cells without any wound inflammation. 156 In 2009, Choi and Yoo prepared wound-adhesive and thermo-responsive hydrogels loaded with recombinant human epidermal growth factor (rhEGF) and these showed release rates that were dependent on degradation rates of the hydrogels. 157 Covalent crosslinked alginate gels are currently playing a role in wound dressing as they have an ability to absorb the wound excaudate in both gel and freeze-dried form and provide a physiologically moist environment while prohibiting the bacterial infection growth. 158 Polymers such as Chitosan 159 and alginate 160 have proven to be vital when it comes to wound dressing as individuals and as a combination. 161 Alginate and Chitosan composite after clinical tests is now commercially available for the treatment of chronic ulcers. 162 Alginate-based wound dressings exist in the form of hydrogels, films, foams, nanofibers, membrane, and sponges; However, these materials have their disadvantages low adherence of alginate is one of the challenges. 163 Currently to develop alginate-based materials, the nanoparticle related mostly silver-loaded nanoparticles studies show the composite are potentially advantageous in wound healing process. 164 On transdermal studies done nanogels were studied for ocular diseases, see for an example the eye drops and ointments used for topical therapy for ocular diseases have bioactivity of 5%. 165 Nanogels from different polymers have been and continues to be studied for application in different ocular diseases. 166 Nanogels are also investigated for central nervous system, these gels containing therapeutic T lymphocytes for localized delivery to glioblastoma cells for brain tumor immunotherapy were developed using poly (ethylene glycol)-g-chitosan They showed greater efficacy in killing glioblastoma cells compared with the Matrigel control. 167 However, one of the challenges faced by the nanogels in central nervous system application is passing through the blood–brain barrier which means that the shape, size and structure of a nanogel will have a huge impact in the development of their central nervous system application. 168
Permeability of nanogels
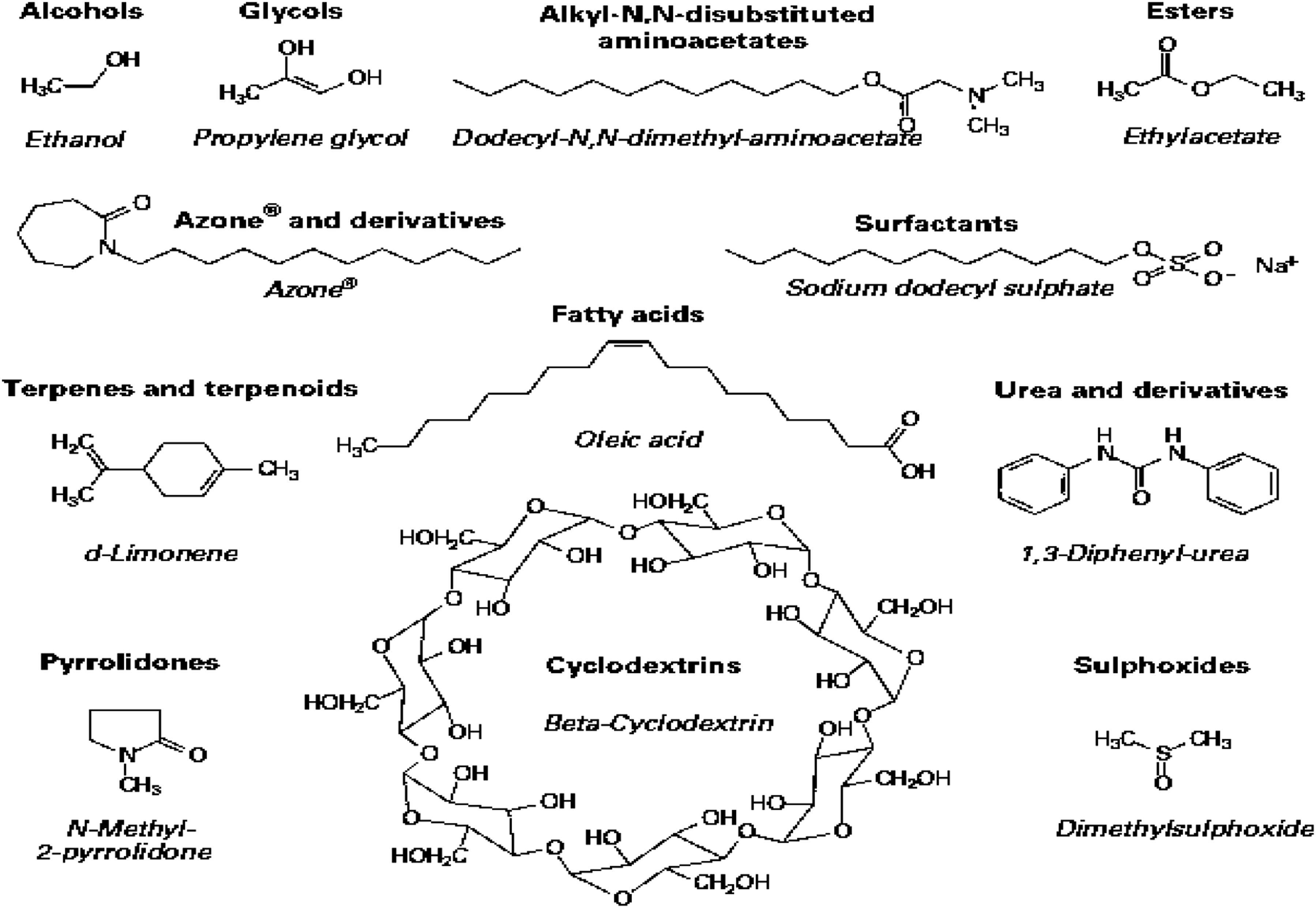
Many penetration enhancers have been investigated e.g., Hydration, Sulphoxides, Azone and derivatives, pyrrolydones, urea and derivatives, disubstituted amino acetates, propylene glycol, surfactants, terpenes and terpenoids, fatty acids, esters, and cyclodextrins as represented in Figure 4. Moreover, the aim is to find potential substances that will have both drug penetration-promoting effects and a low or no skin irritating potential.
169
Fatty acids have shown to possess both the ability to enhance skin penetration and the ability to delay or minimize penetration. Even so this capability highly depends on the structure and the formulation.
170
The combination of 95% of propylene glycol with oleic acid was tested for the ability to hold the penetration instead the results showed a significant increase in skin penetration.
171
Investigated penetration enhancers.
176
Recently it has been discovered that the use of oil-rich transportation formulas for drugs hinder the penetration of the drug through the skin as they make it difficult for the drugs to diffuse well, resulting in low drug distribution in the skin. Mostly the active drugs are not soluble in water. Therefore, to increase their water solubility polymers have a role to play, this leads the research direction to the encapsulation of hydrophobic drugs into water soluble polymers. When the hydrophobic drug was encapsulated in poly (ethylene glycol)–poly (gamma-benzyl-
Conclusions
The known shortcomings of the most marketed topical administered drugs are that they are linked with different toxicities that lead to their side effects and the disadvantage of the abnormal or affected organ. Their lack of curative treatment and poor effectiveness in topical therapy is the main force behind the design of new, safe, and more effective drug carriers. The main aim of the research in the pharmaceutical drug carriers is the discovery of a safe method to get the drug where it is needed and with less damage on other parts of the body. The researchers have focused more on improving the efficacy of the existing drugs by designing safer carriers and changing the method of administration. Nanogels have been proven to be an outstanding option for dermal therapy. This is based on their size, safety, biodegradability, and biocompatibility. However, particle size, polymers used, the charge, the density all play a role in designing the nanogels that will be suitable for this application. The degradability, surface chemistry, and size of the particles play a major role in the penetration of the drug through the skin. A clear and well understanding on these factors will surely help the design of these polymer nano-carriers. Moreover, it has been noticed that the corporation of the existing drugs with these nanogels enhances the therapeutic profile, safety, and efficacy of the drugs. Therefore, for the future, polymer nano-carriers are promising and may also help in the drug discovery search that has been continuing forever. However, to fully assess the toxic effects of nanoparticles, further complementary tests are needed to be carried out in addition to MTT method.
Footnotes
Acknowledgments
The authors would like to extend their gratitude to the South African National Research Funding, University of the Western Cape, UNESCO-University of South Africa and iThemba labs (NRF).
Author contributions
Nandipha L. Botha wrote the review Paul Moshonga and Martin Onani read and corrected the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of the Western Cape and National Research Foundation (NRF).