Abstract
The present study fabricated high-performance silica/epoxy resin nanocomposites having a low coefficient of linear thermal expansion (CTE) and a high glass transition temperature (Tg). This was accomplished by dispersing colloidal silica nanospheres having hydrophilic surfaces in epoxy resins, which limited the motion of the polymer chains. Nanocomposites were produced wherein isolated primary particles of colloidal silica without silane surface modification were dispersed uniformly. These particles were generated via the breakdown of loosely bound agglomerates of spherical silica particles during the agitation of a dispersion in an epoxy resin solution. Hydrogen bonding between hydroxyl groups on the hydrophilic surfaces of the dispersed silica nanoparticles and the cross-linked epoxy polymer network evidently limited thermally-induced motion of the polymer chains, resulting in a considerable reduction in the CTE and an increase in the Tg for the nanocomposite.
Keywords
Introduction
Increasing the reliability of integrated-circuit packages mounted on electronic circuit substrates will require failures at interfaces between dissimilar materials to be prevented. These failures result from mismatches in mechanical and/or thermal properties, including the coefficient of thermal expansion (CTE). Unfortunately, many electrical/electronic materials have CTEs that are particularly low, or at least lower than those of the epoxy resins commonly used to encapsulate circuits.1–3 Hence, drastically reducing the CTE of epoxy resins by adding inorganic nanoparticles could represent one approach to lowering CTE mismatches. The addition of inorganic fillers to epoxy resins has the added advantage of increasing the glass transition temperature (Tg) of the resin such that the resin’s thermal stability is improved. Silica is often added to the polymers used in microelectronics as a mean of enhancing thermo-mechanical characteristics. Silica is preferred as a filler as a consequence of its low CTE and because silica provides superior electrical insulation and mechanical properties at a reasonable cost.1–5 Even so, obtaining suitable thermo-mechanical characteristics, including an effective CTE of from 20 to 30 ppm°C-1 below the Tg of the polymer, requires the incorporation of 55–70 vol% silica particles having dimensions on the micron scale.6,7 Elevated filler levels such as these can both increase the viscosity of the molten polymer and promote delamination of the electronic device.3,8 Hence it would be desirable to develop underfill and encapsulation materials that exhibit low CTE values below their Tg at reduced filler concentrations so as to improve the processing and performance of electronic packages. Qu and Wong 1 previously investigated the relationship between the CTE of epoxy resin composites filled with micron-sized silica particles and the silica particle size, and established that smaller particles lowered the CTE at a constant silica loading. These results suggest that increases in the constriction of the matrix resulting from increases in surface area decrease the expansion of the matrix. 9 On this basis, the use of silica nanoparticles might be expected to greatly reduce the CTE values of epoxy resins. However, there have been few studies concerning the CTE of silica/epoxy resin nanocomposites. The expectation has been the work of Dittanet and Pearson 10 examining silica/epoxy resin nanocomposites fabricated via a blending method in conjunction with organosilane surface modification of the silica nanoparticles. This surface treatment was required because silica nanoparticles have a tendency to tightly aggregate to generate micron-sized agglomerates.
In the present study, anhydride-cured bisphenol-A epoxy resin, which is commonly used to insulate electric circuitry, 11 was selected as a matrix phase. We assessed silica/epoxy resin nanocomposites, in which hydrophilic colloidal silica nanospheres without silane surface modification were dispersed uniformly. The hydrophilic silica surfaces were expected to effectively lower the CTE of these blends while also increasing the Tg of the epoxy resin matrix. Previous work by Dittanet and Pearson 10 investigating silica/epoxy resin nanocomposites fabricated using a standard blending method showed that surface treatment of the silica with silane coupling agents allowed this filler to more effectively wet the epoxy matrix. This improved wetting, in turn, reduced the interfacial activity between the silica particles and the epoxy polymer networks.12,13 However, the interfaces between the silica nanoparticles and the resin in this type of nanocomposite system are so limited that interactions occurring at these interfaces cannot be easily utilized to design advanced functional nanocomposites. As an example work by Dittanet and Pearson 10 showed that the effect of volume fraction of dispersed silica nanoparticles on the CTE of a blended silica/epoxy resin nanocomposite using organosilane modified silica approximately followed the simple rule of mixture (ROM) and Kerner model 14 (see Figure 8(b) below). The results of this prior work 10 demonstrated that the addition of modified nano-silica did not have a significant effect on the Tg of the epoxy resin, which provides an indication of the thermal stability of the polymer. The results of the same study 10 also indicated that the silica volume fraction required to reduce the CTE of a resin to match those of materials such as copper wiring (approximately 20 ppm°C−1) exceeds 70 vol%.
The intent of this work reported herein was to fabricate a superior performance silica/epoxy resin nanocomposite system having a low CTE and high Tg as compared with conventional epoxy resin nanocomposites using a micron-sized silica filler or a hydrophobized silica nanofiller. This type of material could be used for electronics encapsulation while preventing failures at interfaces between dissimilar materials caused by CTE mismatches that otherwise reduce the reliability of integrated-circuit packages at high temperatures.
Silica/epoxy resin nanocomposite design and fabrication strategy
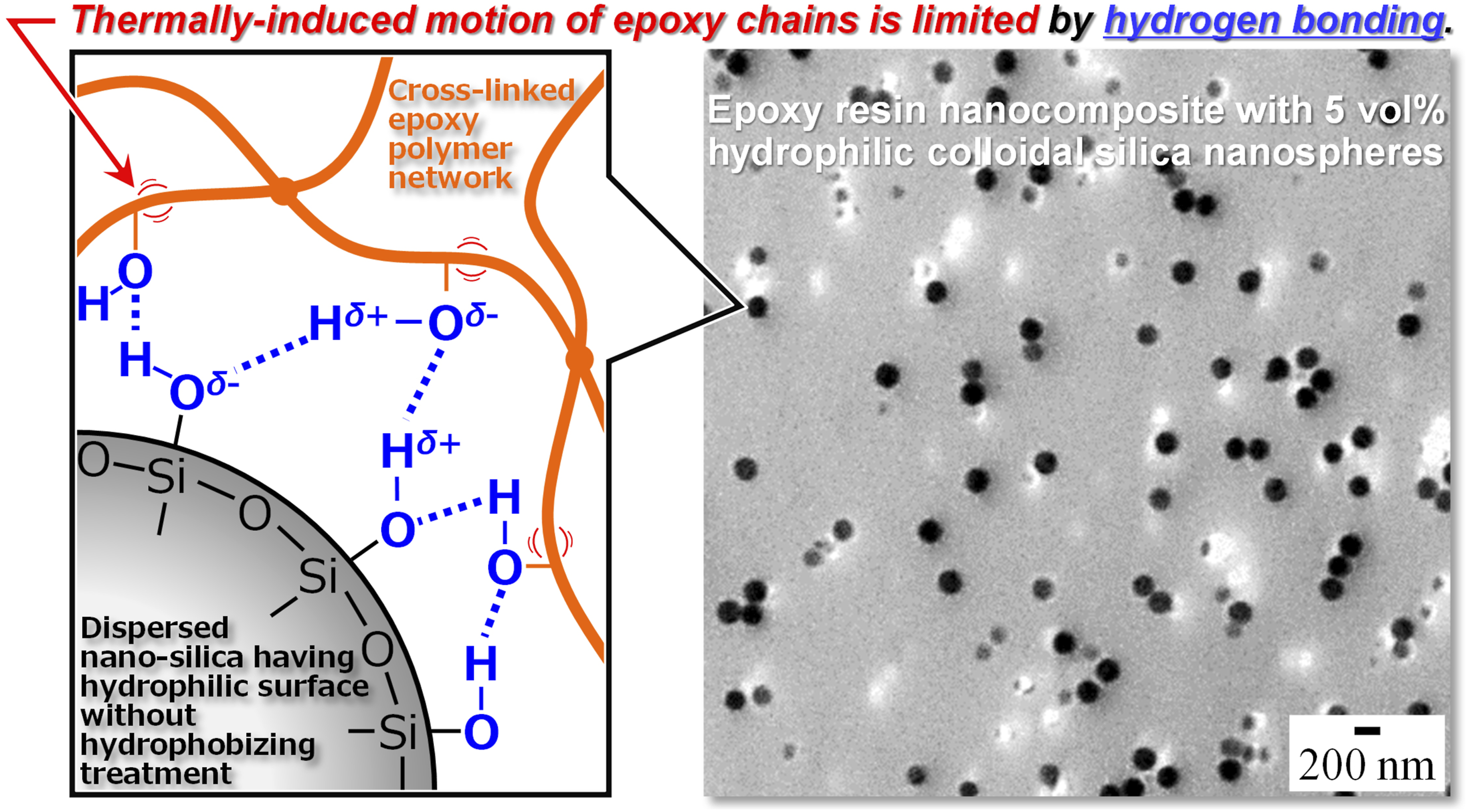
Figure 1 summarizes the present approach to obtaining a silica/epoxy resin nanocomposite exhibiting high thermal stability with a reduced CTE. In this concept, hydrogen bondings are formed over the large interfacial area between the epoxy polymer network and dispersed silica nanoparticles having hydrophilic surfaces. These interactions are expected to restrict the chain motion in the epoxy network, resulting in a drastic reduction in CTE along with an increase in Tg. A diagram showing hydrogen bonding between colloidal silica nanospheres having hydrophilic surfaces and epoxy matrix.
To take advantage of hydrogen bonding at the interfaces between silica nanoparticle having surfaces with high concentration of hydroxyl groups and epoxy matrix, these nanoparticles would, ideally, be dispersed in the epoxy resin without any chemical modification. However, it is typically extremely difficult to achieve a unform dispersion of hydrophilic silica nanoparticles in a polymeric matrix because of the poor wettability of the filler by the matrix phase. Recently, the present authors and coworkers proposed a novel approach to fabricating various types of silica/thermoplastic nanocomposites that does not require surface modification techniques.15–19 Using this process, isolated primary particles of hydrophilic colloidal silica (without surface modification using silane coupling agents) can be homogeneously dispersed throughout a polymer through mechanical breakdown of loosely bound agglomerates formed by joining spherical primary silica particles during direct melt compounding with various polymeric matrix components. A key aspect of this strategy is the preparation of the initial agglomerates by producing weakly bound primary silica particles that are readily disrupted by the shear stress imparted during blending.
The present research fabricated silica/epoxy resin nanocomposites using a solution-mixing method based on this same strategy. The fracture strength, σt, that is the tensile stress input required to break up agglomerates comprising packed spherical particles having a diameter of dp and a surface energy of γ, can be calculated using Rumpf’s equation as20-22
The fracture strength can be reduced by increasing the volume fraction occupied by the void spaces between the primary particles, εP, and by decreasing the coordination number of each primary particle, NC, meaning the number of interparticle contacts per unit primary particle. This equation indicates that the formation of a large number of nanospaces as defects in the packed structure of the silica nanospheres would be expected to greatly lower the fracture strength, promoting the homogeneous dispersion of hydrophilic silica nanospheres during mixing with the epoxy resin.
Experimental details
Materials
A commercially available aqueous colloidal suspension of spherical silica particles having a mean primary diameter of approximately 200 nm (MP-2040, Nissan Chemical Industries, Ltd) was used as the starting material for the preparation of silica fillers. A bisphenol A-type solid epoxy precursor having a number average molecular weight of 900 (jERTM 1001, epoxy equivalent weight (EEW) = 450–500 g/eq, Mitsubishi Chemical Co.) and an acid anhydride curing agent (jERCURETM YH306, Mitsubishi Chemical Co.) were used as the starting materials for the polymer. An uncured epoxy resin solution was made by dissolving the jERTM 1001 in acetone at 30 mass% along with sufficient jERCURETM YH306 to give a jERTM 1001 to jERCURETM YH306 mass ratio of 2:1, using a planetary centrifugal mixer (AR-250, THINKY Corp.). Mixing was performed for 3 min at a revolution/rotation rate of (2000 r/min)/(800 r/min).
Nanocomposite fabrication
Figure 2 summarizes the two-stage method employed to produce the silica/epoxy resin nanocomposites in the present study. The first stage of this process involved the preparation of the silica filler, while the second stage comprised mixing of the uncured epoxy resin solution described above with the silica filler prepared in the first stage. In the first stage of this process, the colloidal silica nanospheres in the MP-2040 dispersion were destabilized by adjusting the pH from the initial value of approximately 9.3 to 4 and by adding KBr at a concentration of 2.6 M. This step resulted in the formation of a random loosely packed structure composed of primary colloidal silica particles. Evaporating the water provided the silica in the form of loosely bound agglomerates. This granular material, which still contained the KBr was added to water heated to approximately 80°C to dissolve the KBr, thus generating nano-sized voids between the silica particles. The product was subsequently dried at 120°C under a vacuum of approximately 50 kPa. Herein, the product obtained from this process is referred to as the LAS filler. In this work, it needed to be examined effects of these hydrophilic groups on silica filler surface on thermal expansion characteristics of silica/epoxy resin nanocomposite fabricated using the silica filler. Therefore, two types of comparing samples of loosely bounded agglomerates of primary colloidal silica particles having different concentrations of hydroxyl groups from the LAS filler were also prepared from the same MP-2040 colloidal silica suspension by changing the drying conditions to heating at 80°C under vacuum of less than 30 Pa or heating at 600°C in air. A diagram summarizing the process used to fabricate silica/epoxy resin nanocomposites using a solution-mixing method. The SEM micrograph shown at the end of the diagram is a typical distribution of primary colloidal silica particles in an epoxy resin composite specimen fabricated with 5 vol% LAS filler via this method. The dark spots in the SEM micrograph are primary silica particles.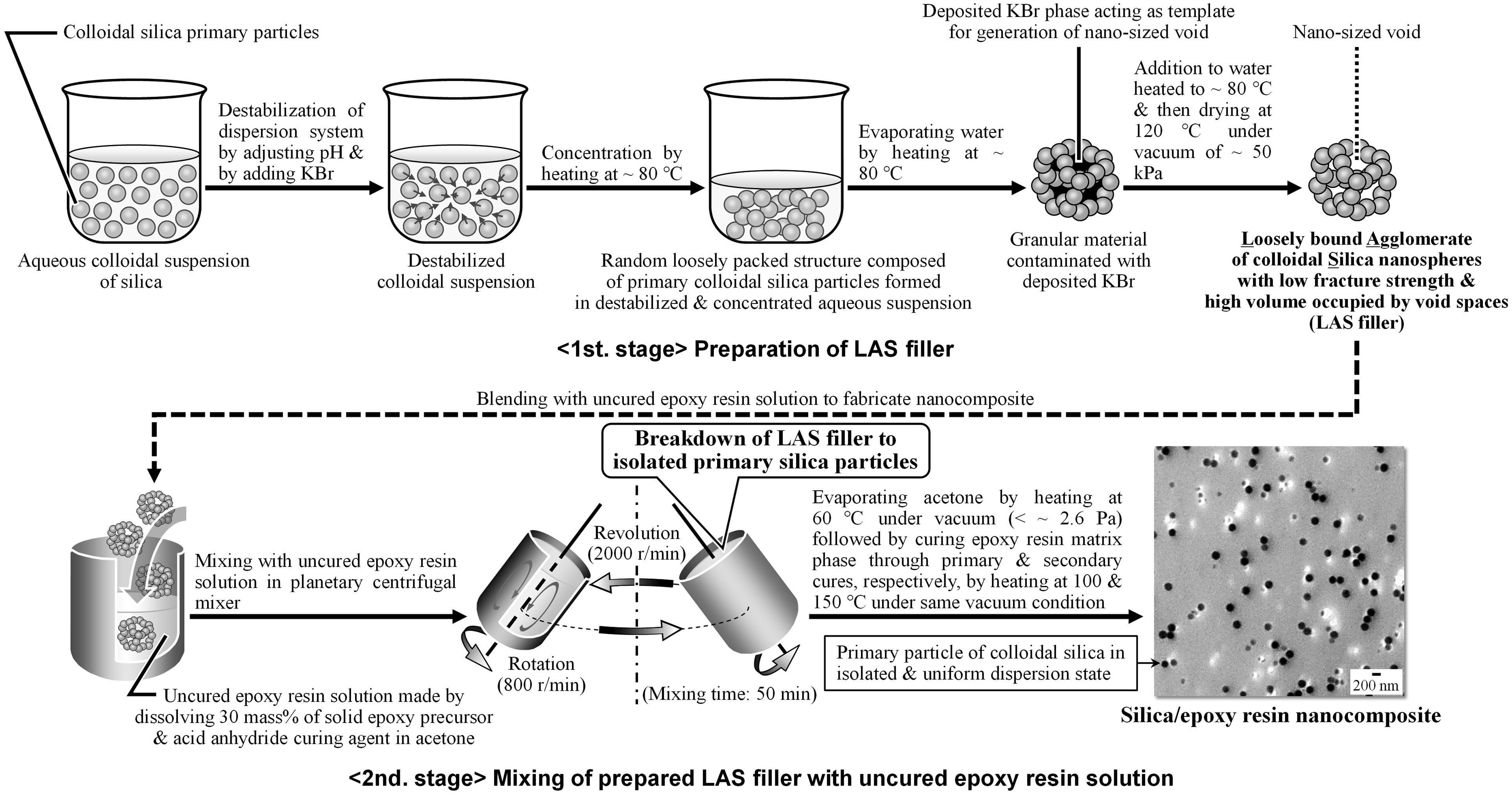
In the second stage, the epoxy resin dissolved in acetone was mixed with a specific amount of the prepared LAS filler in the same planetary centrifugal mixer previously used to prepare the uncured epoxy resin solution. This combination of materials was mixed for 50 min at a revolution/rotation rate of (2000 r/min)/(800 r/min) and then cast into mold. The acetone was subsequently evaporated by heating at 60°C for 2 h under a vacuum of less than 2.6 Pa. This was followed by a primary cure based on heating at 100°C for 2 h to yield a sample that could be released from the mold. The specimen was then subjected to a secondary cure by heating 150°C for 3 h under a vacuum less than 2.6 Pa to give the silica/epoxy resin nanocomposites. Nanocomposites were fabricated having silica volume fractions, Vf, ranging from 0 to 0.34, meaning 0 to 34 vol%. As shown in Figure 2, the scanning electron microscopy (SEM) micrograph placed at the end of the process flow diagram is of a typical specimen and indicates the distribution of primary colloidal silica particles with hydrophilic surfaces in an epoxy resin composite sample containing 5 vol% LAS filler fabricated via the two-stage method described in this section.
For fabrication of the silica/epoxy resin composites with a Vf of 0.05, planetary mixing of the LAS filler in the uncured epoxy resin solution for not only 50 min but also 5 min at the same revolution/rotation rate was employed to confirm the dispersibility of the LAS filler in the epoxy resin matrix.
Characterization
The structure and void volume of the LAS filler prepared in the first stage were characterized, respectively, by SEM and mercury intrusion porosimetry. Microcompression tests using an MCT-W500 instrument (Shimadzu Co.) were also conducted to evaluate the fracture strength of the LAS filler. In addition, solid-state silicon-29 nuclear magnetic resonance (29Si NMR) spectroscopy was employed to ascertain the concentrations of hydroxyl groups on surfaces of the LAS filler. Dipolar decoupling and magic-angle spinning (DD-MAS) NMR spectra were acquired using a pulse width of 4.50 μs and a delay time of 60 s. All chemical shifts are reported herein relative to that for hexamethylcyclotrisiloxane.
The dispersion of silica nanospheres in each silica/epoxy resin nanocomposite was ascertained using SEM while the CTE and Tg values were obtained, respectively, using thermomechanical analysis (TMA) and differential scanning calorimetry (DSC). The CTE values for each nanocomposite below the Tg of the epoxy matrix phase was determined from TMA data acquired over the temperature range from 50 to 70°C. The Tg values for the various nanocomposites were obtained from analyses using DSC. Dynamic mechanical analysis (DMA) data were also acquired from filled and unfilled epoxy resin specimens operating in tension mode at a frequency of 10 Hz under nitrogen. These data characterized the strength of interactions between the silica and resin. In these trials, the dynamic storage modulus and loss tangent were measured was −150 to 250°C at a heating rate of 0.083°Cs−1.
Results and discussion
Characterization of silica fillers
Figure 3 summarizes the relationship between the fracture strength, σt, and the void volume fraction, εP, for an LAS filler specimen and an untreated silica filler produced as a control specimen from the MP-2040 colloidal silica suspension without pH adjustment or KBr addition. From the load required for the breakdown of a test piece of the LAS filler, Ff, obtained the microcompression test, the σt value was determined using the following equation
23
Relationship between the fracture strength and void volume fraction for a LAS filler specimen prepared from a silica dispersion by adjusting the pH from 9.3 to 4 and adding KBr at 2.6 M and for an untreated silica filler produced as a control specimen from the same dispersion without pH adjustment or KBr addition. The SEM micrographs shown in the insets indicate the arrangements of primary colloidal silica particles in these two samples.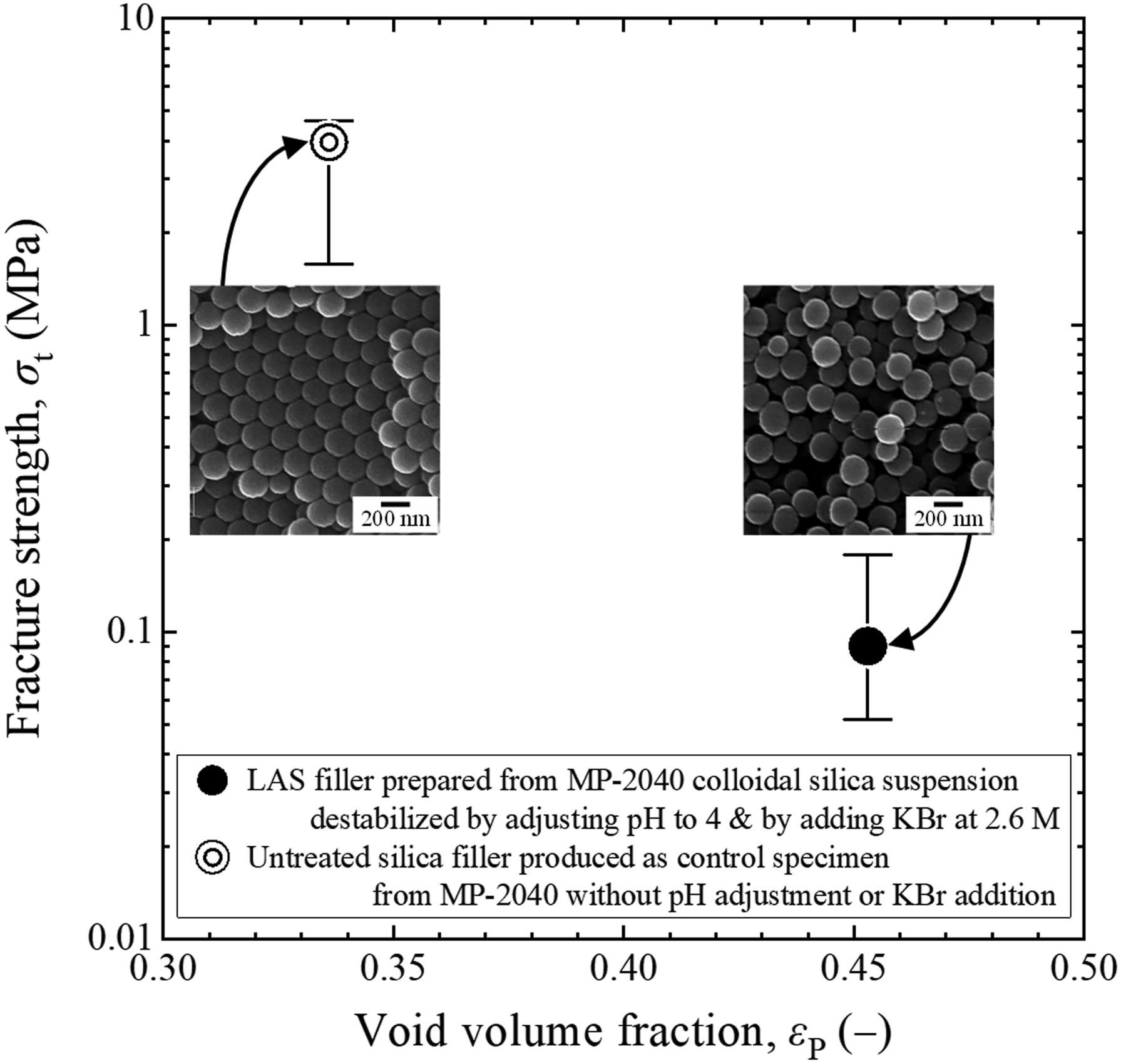
Compared with the regular close-packed structure in the untreated silica filler, numerous nano-sized void spaces were evidently generated in the LAS filler. The relationship between σt and εP values in this figure indicate that the fracture strength was reduced drastically upon increasing the void fraction, in accordance with equation (1). This result implies that the arrangement of primary silica particles and hence the fracture strength could be adjusted by destabilizing the colloidal silica suspension via pH adjustment and salt addition.
Dispersion of colloidal silica particles in silica/epoxy resin nanocomposites
Figure 4(a) and (b) present typical SEM micrographs showing the distribution of primary colloidal silica particles in the nanocomposites containing 5 vol% LAS filler and untreated silica filler, respectively. It should be noted that these two composite specimens were specially made for SEM observations by mixing either the LAS filler or the untreated silica for only 5 min so that the distinction between the two types of silica used as filler was more obvious. SEM micrographs of selected areas of epoxy resin composites fabricated with (a) 5 vol% LAS filler and (b) 5 vol% untreated silica fillers.
Figure 4(a) clearly shows that isolated primary particles of the LAS filler were dispersed uniformly throughout the cured epoxy resin matrix. The degree of dispersion of this silica is comparable to those obtained in previous works with epoxy resin composites filled with hydrophobized silica nanospheres10,24 even though the prior studies used different silica concentrations from those in the present work. In constant, the untreated silica filler had a regular close-packed structure comprising agglomerated primary silica particles densely that were not readily broken down and thus remained as micron-sized clusters, as shown in Figure 4(b). It is apparent that much better dispersion of the silica nanoparticles in the epoxy resin matrix was achieved using the LAS filler. Kang et al. 24 used spherical silica particles having a primary diameter of approximately 400 nm prepared via a sol-gel process without any hydrophobic surface treatment as a filler for as epoxy resin composite. Hence, the present study demonstrates a novel means of producing silica/epoxy resin nanocomposites without any hydrophobic surface treatment of the silica. Based on the results shown in Figure 4(a), a solution-mixing time of 50 min was, therefore, used in the following experiments to ensure uniform dispersion of isolated primary silica nanoparticles in each epoxy resin composite as a result of breakdown of the loosely bound agglomerate formed by the LAS filler.
Effects of surface hydroxyl groups on the silica on epoxy chain motion
The effects of hydroxyl groups on the surfaces of the dispersed silica nanospheres on the thermal expansion of the nanocomposites were examined. This was accomplished by ascertaining the CTE values for nanocomposite samples fabricated by mixing uncured epoxy resin solutions with silica fillers prepared as loosely bound agglomerates of silica formed by joining primary particles having different degrees of surface hydrophilicity. The hydrophilicity of the silica filler was tuned by varying the drying conditions used when processing the material. The upper part of Figure 5(b) presents a typical 29Si NMR spectrum acquired from an LAS filler prepared by applying a final drying treatment based on heating at 120°C under a vacuum, as illustrated in Figure 2. The upper part of Figure 5(b) indicate that the ratio of the Q3 signal intensity (associated with a chemical shift, δ, of approximately −108 ppm and characteristic of Si(SiO)3(OH)) to that of the Q4 intensity (associated with a δ of approximately −116 ppm and characteristic of Si(SiO)4), Q3/Q4, had a value of 0.53. This ratio reflects the concentration of hydroxyl groups on the LAS filler surfaces. The upper part of Figure 5(a) shows a typical 29Si NMR spectrum acquired from material dried at 600°C in air to accelerate the dehydrocondensation of hydroxyl groups (See Figure S1). In this case, the Q3/Q4 was reduced to 0.13. The results in the upper part of Figure 5(c) were obtained from silica dried at 80°C under a vacuum of less than 30 Pa. Under these conditions, dehydrocondensation of hydroxyl groups was suppressed and the highest Q3/Q4 ratio of 0.53 was obtained. It is apparent that the silica fillers having the ratios of 0.13 and 0.53 had the lowest and highest concentrations of surface hydroxyl groups, respectively. The former and latter materials are referred to, respectively, as the LAS-13 and LAS-53 fillers. 29Si NMR spectra of (a) LAS-13, (b) LAS, and (c) LAS-53 fillers prepared as loosely bound agglomerates formed by joining primary particles of colloidal silica having different concentrations of surface hydroxyl groups (upper part). SEM micrographs showing typical dispersion states of each of the three types of LAS fillers in epoxy resin matrices with a filler volume fraction of 25 vol% (lower part). Data are for (a) LAS-13, (b) LAS and (c) LAS-53 fillers. The circles in the SEM micrographs indicate primary silica nanoparticles in isolated and uniform dispersion states as a result of the breakdown of LAS-13, LAS and LAS-53 fillers via the solution-mixing process with the uncured epoxy resin solution.
The SEM micrographs in the lower part of Figure 5 show typical epoxy resin nanocomposite specimens containing 25 vol% of the three types of fillers. These micrographs clearly demonstrate that each nanocomposite had almost the same silica particle dispersion characteristics.
The CTE values, α, for the nanocomposites are plotted in Figure 6 against the Q3/Q4 ratios obtain from NMR spectra of the respective silica fillers. These data confirm that, in each case, α was reduced upon 25 vol% silica fillers. The extent to which the CTE was lowered relative to that of the unfilled resin by incorporating silica was approximately 37%, 49% and 47% for the LAS-13, LAS and LAS-53 fillers, respectively. It is also evident that the CTE was reduced to the least extent by incorporating the LAS-13 filler. These results confirm that fabricating silica/epoxy nanocomposites in which hydrogen bonding occurs between the hydrophilic surfaces of silica nanospheres and the cross-linked epoxy polymer can inhibit thermal expansion. It can also be seen that a Q3/Q4 ratio of 0.33 is optimal and that further increases in hydroxyl group concentration have little effect on the CTE of the nanocomposite. Relationship between the CTE values for silica/epoxy resin nanocomposites fabricated with 25% silica fillers and the NMR ratio reflecting the silica surface hydroxyl group concentration.
Figure 7 summarizes the dynamic storage modulus, E’, and the loss tangent, tan δ, data for a nanocomposite containing 25 vol% LAS filler having a Q3/Q4 ratio of 0.33 and for an unfilled epoxy resin. Data for this filled specimen were chosen because this specimen was the most different from the unfilled epoxy resin. The temperature dependencies of tan δ for the filled and unfilled epoxy resins shown here clearly establish that two relaxation processes occurred over the temperature range investigated. The first relaxation appearing at a higher temperature is attributed to so-called α relaxation while the second appearing at a lower temperature is β relaxation. Shimbo et al.
25
observed the broad low-temperature relaxation process referred to as β relaxation at approximately −70°C during a DMA study of a bisphenol-A epoxy resin cured with an acid anhydride. The small peaks between −90 and −70°C in the tan δ curves in Figure 7 can therefore be regarded as indications of β relaxation of the nanocomposite and the unfilled epoxy resin. In polymers, α relaxation involves motion of long segments of the main chains and so is related to Tg. In constant, β relaxation is associated with the rotation of side chains and therefore occurs below Tg. By filling nano-silicas, the α and the β relaxation peaks were, respectively, shifted from 99.6 to 109.4°C and from −84 to −77°C. Incorporating the silica nanoparticles shifted the α and β relaxation peaks, respectively, from 99.6 to 109.4°C and from −84 to −77°C. The latter change suggests that the mobility of the side chains in the epoxy polymer may have been restricted due to hydrogen bonding between the silica and the polymer. DMA data obtained from an epoxy resin nanocomposite containing 25 vol% LAS filler and from an unfilled epoxy resin.
Effects of silica volume fraction on CTE and Tg
The α values for the nanocomposites fabricated with the LAS filler are plotted against the silica volume fraction, Vf, in Figure 8(a). Based on the ROM, α can be calculated as Effects of the LAS filler volume fraction on the thermal expansion characteristics of epoxy resin nanocomposites with LAS filler. (a) CTE values and (b) the ratio of the CTE value for the nanocomposite to that for the unfilled epoxy resin as functions of volume fraction.
Figure 8(b) shows the effect of the silica filler volume fraction on the ratio of the CTE values for the silica/epoxy resin nanocomposite and the unfilled epoxy resin. This ratio, α/αm, in the present study is much lower than that reported for a conventional nanocomposite fabricated using nano-silica with organosilane surface modification. 10 These results established that hydrogen bonding between the hydrophilic silica and the epoxy resin limited thermally-induced motion of the epoxy polymer chains, resulting in effective suppression of thermal expansion.
In addition to TMA assessments (see Figure S2), DSC analyses were used to determine Tg values in the present study (see Figure S3). Figure 9 plots this variable as a function of Vf for two types of nanocomposites, both of which display different trends to those found in a previous study by Dittanet and Pearson.
10
The present results indicate that incorporation of the hydrophilic colloidal nano-silica increased in the Tg for the epoxy resin and so shifted the α relaxation to a higher temperature, as shown in Figure 7. It is evident from Figure 9 that the increase in Tg was greater in the case of the nanocomposite fabricated with the LAS filler having a Q3/Q4 ratio of 0.33 compared with the material containing the LAS-13 filler. Droste and Dibenedetto
26
reported that the empirical relationship between Tg and the filler volume fraction, Vf, for a polymer composite system is The Tg for epoxy resin nanocomposites incorporating LAS and LAS-13 fillers as functions of the filler volume fraction.
Conclusions
The present study fabricated silica/epoxy resin nanocomposites with CTE values lower than those for conventional systems. The aim of this work was to examine the effects of hydrophilic silica nano-fillers on the thermal expansion behavior of silica-loaded epoxy resins. The main results can be summarized as follows: • A practical technique that enables the uniform dispersion of isolated primary particles of colloidal silica with hydrophilic surfaces in an epoxy resin at volume fractions higher than 30 vol% was demonstrated. • A silica/epoxy resin nanocomposite having a much lower CTE than conventional systems was produced, taking advantage of interactions between hydroxyl groups on the polymer and on the surfaces of dispersed hydrophilic colloidal silica nano-fillers. This phenomenon increased the glass transition temperature of the resin and so improved the heat resistance of the material.
Supplemental Material
Supplemental Material - A silica/epoxy resin nanocomposite exhibiting high thermal stability and low thermal expansion based on the uniform dispersion of hydrophilic colloidal silica nanospheres
Supplemental Material for A silica/epoxy resin nanocomposite exhibiting high thermal stability and low thermal expansion based on the uniform dispersion of hydrophilic colloidal silica nanospheres by Mitsuru Tanahashi and Kazuma Hirota in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
This author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This author(s) disclosed receipt for the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for partial support given by a Grant-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (number 15K06462).
Data availability statement
All datasets supporting the conclusions are presented in the main text and supplementary materials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.





