Abstract
This study aimed to investigate the effect of 2,2,2-trifluoroethyl methacrylate:triethyleneglycol dimethacrylate (TFEMA: TEGDMA) ratio on various functional properties of Bis-GMA/TFEMA:TEGDMA/UDMA composites. The experimental composites were loaded with 70% (weight% of total monomer) fillers consisting of silanised fumed silica, zirconia, and strontium fluoride. The light-cured composites were analysed for degree of conversion by FTIR-ATR technique, volumetric shrinkage in according to the Archimedes' principle, and fluoride ion release and recharge capacity by ion selective electrode. Water sorption, water solubility, and flexural strength were determined in accordance with ISO 4049 standard. Results showed that the degree of conversion, fluoride release and recharge capacity, water sorption, water solubility increased, and polymerization shrinkage decreased, with increasing TFEMA:TEGDMA ratio. Besides, twofold increase in flexural strength was observed for composite with the highest TFEMA content.
Keywords
Introduction
Bisphenol A-glycidyl dimethacrylate (Bis-GMA) is one of the major dimethacrylate monomer components used in commercial dental composite restoratives, which provides the composite its core strength. However, due to its chain rigidity and overall high viscosity nature, Bis-GMA would quickly gel upon photoactivation, resulting in a low degree of conversion. 1 Hence, triethylenglycol-dimethacrylate (TEGDMA), a low-viscosity diluent monomer, was incorporated in the bis-GMA based formulation to enhance the molecular mobility, which delayed the gelation and achieved higher conversion. 2 In general, the physical properties of resin based dental composites are greatly influenced not only by the chemical structure of the monomers used but also by fillers in the matrix phase.3,4 Nonetheless, the Bis-GMA based resin composite formulations with higher amounts of TEGDMA, up to 50%, increased filler concentrations and exhibited higher degree of conversion (55 to 75%) at the expense of higher polymerization shrinkage and low mechanical properties.5-7 An ideal dental composite requires a minimal polymerization shrinkage with an optimal degree of conversion. 8 Clinically, volumetric shrinkage may cause micro leakage and recurrent caries resulting in tooth fracture. 9 TEGDMA substituents such as monomethacrylates have been introduced and found to be effective in reducing polymeric shrinkage. 10 Similar work reported recently includes the use of 2-hydroxyethyl methacrylate (HEMA) 11 and isobornyl methacrylate (IBOMA). 12 Nonetheless, to achieve low volume shrinkage and high mechanical properties, high filler content of between 60 to 87 wt% are necessary. 13 High degree of conversion (DC) has a pivotal role in ensuring the acceptable level of physical and mechanical properties as well.14,15 In addition, high degree of conversion will give less residual monomers and, thus, low risk of health issue arising from the leakages in the oral environment. Besides all these functional properties that often become the focus of optimization, equally important is the incorporation of fluoride-releasing agent in dental restoratives, which could reduce secondary caries formation.16–19 In a previous study, partial replacement of TEDGMA with TFEMA, in BisGMA/TFEMA:TEGDMA/UDMA containing maximal amount of 55% silanised nanosilica filler, has been found to have decreased water solubility as well as volumetric shrinkage significantly different from that of the control. 20 In this study, we aimed to further investigate the critical properties of BisGMA/FTEMA:TEGDMA/UDMA experimental composites loaded with mixed fillers consisting of silanised fumed silica (7 nm), Zirconia (5µm), and strontium fluoride. The fluoride release and recharge capability of dental composites, which is seldom reported, was also evaluated in this study.
Materials and methods
Materials
TFEMA, Bis-GMA, TEGDMA, UDMA, camphorquinone, tetrabutylammonium fluoride (TBAF), ethyl-4-dimethylbenzoate (4EDMB), and fumed silica (0.007 µm), zirconia oxide, and strontium fluoride were purchased from Sigma-Aldrich, St. Louis, MO, USA, and used as received. Silanised fumed silica was prepared using a reported method. 21
Methodology
Preparation of dental composites
Experimental dental composite composition.

Degree of conversion
The 2 mm by 2 mm stainless-steel molds were placed on a glass slide and filled with uncured composite paste. The infrared spectrum was recorded using diffuse reflectance fourier transform infrared spectroscopy for 32 scans, from 400–4000 cm−1 with a resolution of 4 cm−1. The composite was then irradiated with a light-cure unit (3M Elipar™ DeepCure-L; calibrated for light output of 450 mW/cm2 by Demetron radiometer) for 20 s on each side and FTIR spectrum was again recorded. The degree of conversion was calculated based on the protocol as described previously. 20
Volumetric shrinkage
Volumetric shrinkage was determined by density measurements according to Archimedes' principle. Test specimens were hand molded into spherical balls of 3 mm diameter and weighed both in water and air at room temperature (23 ± 0.1oC). The density of this uncured specimen was determined with a density weighing balance (Sartorius AX224-220M). The specimen was then molded into a stainless-steel mold of 4 mm (d) by 6 mm (h) and light cure incrementally for 40 s, 2 mm per increment. The specimen was then kept in a dark room for 15 minutes and the measurements in air and water were then obtained.
The volumetric shrinkage was then calculated using the formulas as described previously:
20
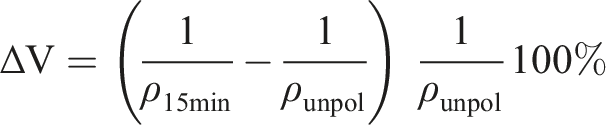
Fluoride release test
Test specimens (n = 10) were prepared in stainless-steel split mold, 4 mm diameter by 6 mm height, with the aid of a pair of glass slides. The first incremental layer of 2 mm thick was placed in the mold and light-cured for 20 seconds through the upper glass slide and the curing was repeated. The photoactivated specimen was removed and then inserted immediately into an Eppendorf plastic test tube which contained 3 mL of deionised water for fluoride ion release tests.
The fluoride ion content released by test specimen on 1st, 2nd, 10th, 20th, 30th, 44th, 58th, 72th, 86th and 100th days were analysed using Cole-Parmer Fluoride Combination Electrode and Eutech ION 2700 PH/mV/Ion/oC/oF meter (Eutech). The results were reported as cumulative fluoride release (µg/cm2) considering the test specimen surface area measured with electronic digital caliper. Before each measurement, the Eutech ION 2700 meter was calibrated with standard fluoride solutions.
Fluoride recharge capacity test
The test specimens were being kept in Eppendorf plastic test tubes containing deionised water for 30 days. Fluoride release baseline was measured daily for 3 days before the start of fluoride recharge capacity test. Each surface of a specimen was applied with ’60-seconds Taste Gel’ for one minute and then rinsed with running deionised water for one minute. The 60-seconds Taste Gel is an acidulated phosphate fluoride (containing 1.23%, w/w, fluoride ions). Fluoride release from these recharged specimens was measured daily for 7 days. The recharged cycles were repeated thrice. The results were reported as fluoride release in μg/cm2.
Water sorption and solubility test
Disk specimens of 2 mm thick by 10 mm in diameter (n = 5) were prepared in a set of split stainless-steel molds and cured for 40 s on each side. The thickness and diameter of each photoactivated specimen were measured at five and two points respectively, using dental callipers (±0.1 mm), for computing the volume of each specimen, V, in mm3. The specimens were placed in a desiccator containing freshly dried silica gel at 37°C. The specimens were weighed from time to time until a constant mass (m1) had been achieved. The test specimens were then immersed in a water bath at 37°C. At fixed time intervals, they were removed, blotted dry, weighed, and re-immersed into the water bath. The readings were taken a two-day interval interval followed by one-week interval until a constant mass (m2) was achieved. The specimens were reconditioned to constant dry mass (m3) in the desiccator as described for m1. Water sorption (Wsp) and solubility (Wsl) in μg/mm3 were calculated by the following equations: 22 Wsp = m2–m3/v; Wsl = m1–m3/v
Flexural strength
The flexural strength was determined in accordance with ISO 4049:2009 standard using a universal testing machine (Shimadzu AGX series UTM) housed in the Biomaterial Research Laboratory at University of Malaya, Kuala Lumpur. Samples of 25 mm × 2 mm × 2 mm (lxhxw), n = 10, were prepared in the same way as mentioned above. The samples were immersed in de-ionized water at 37oC for 24 hours, and then tested on the Instron testing machine with crosshead speed of 1 mm/min.
SEM
The fracture surface of experimental composite disk was sputter coated with gold/palladium film (K = 0.7, I = 10 mA, V = 1kV, t = 45 sec) and placed into Hitachi FESEM SU5000 for imaging at an accelerating voltage of 15 kV using secondary electron detection.
Statistical Analysis
The means and standard deviations were calculated. One-way ANOVA and post hoc Turkey (SPSS 13.0) were used to determine the statistically significant differences between the mean values (p ≤ 0.05).
Results
Figures 1, 2, 3 and 4 show the FTIR spectra of uncured and cured dental composites of F1-F4. Infrared spectra of the uncured (red curve) and cured (blue curve) experimental composite F1. Infrared spectra of the uncured (red curve) and cured (blue curve) experimental composite F2. Infrared spectra of the uncured (red curve) and cured (blue curve) experimental composite F3. Infrared spectra of the uncured (red curve) and cured (blue curve) experimental composite F4.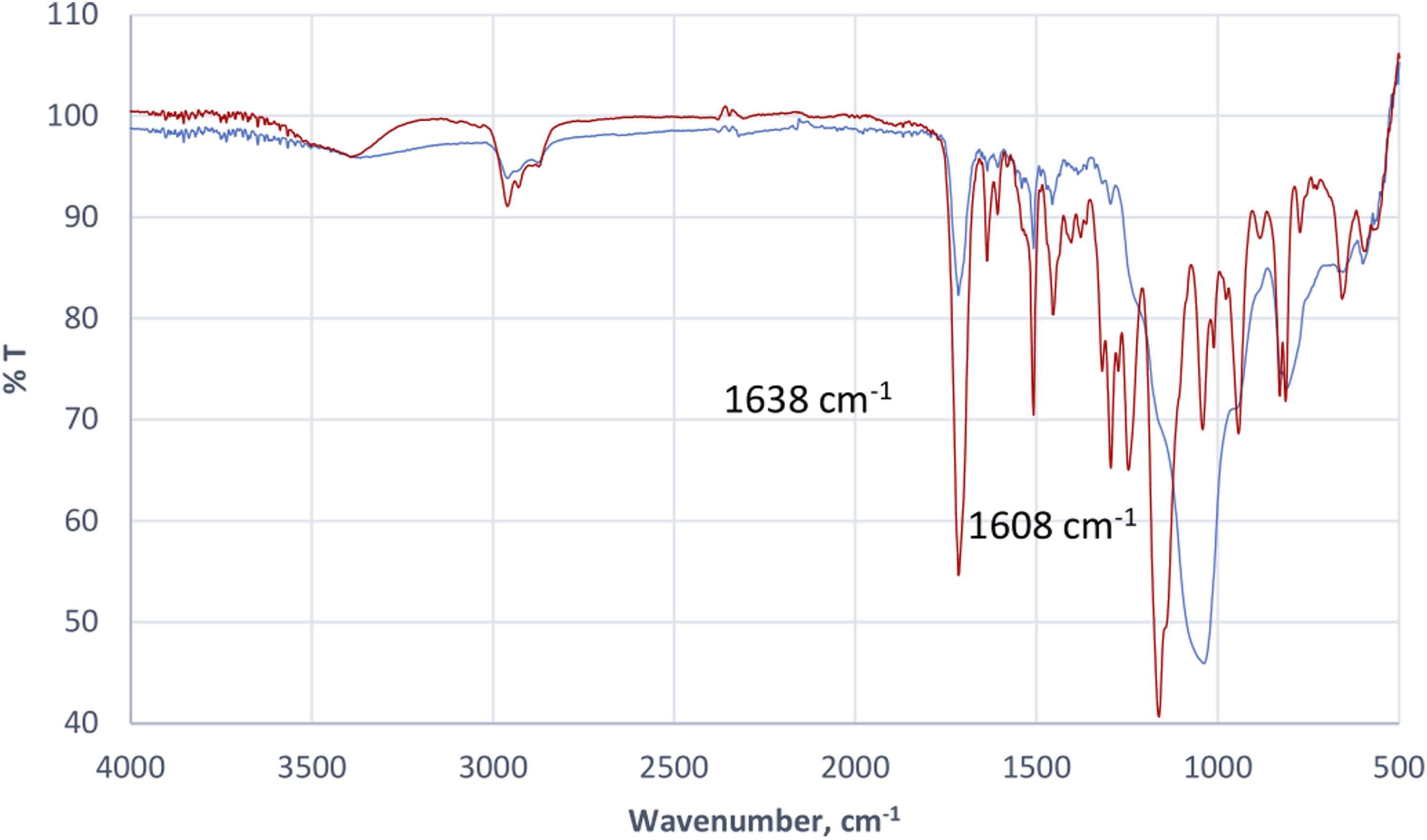

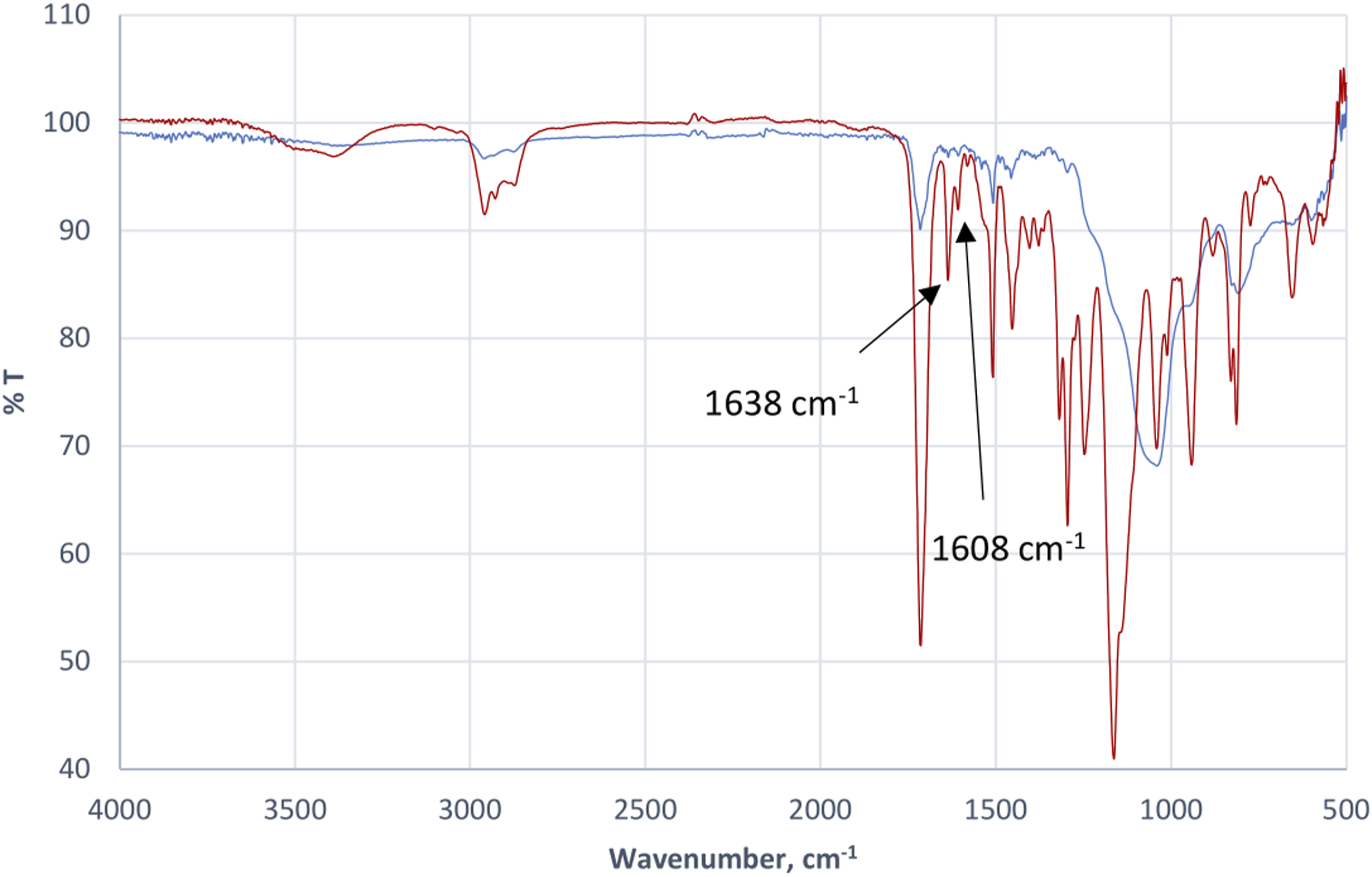

The degree of conversion was determined from the following:
Degree of conversion (%) = 100{1−[(A1638/A1608)cured/(A1638/A1608)uncured]};
whereby the ratio of absorbance peak height from baseline at wavenumbers of 1638 cm−1 and 1608 cm−1 of uncured and cured sample were computed.
Degree of conversion of experimental dental composites.
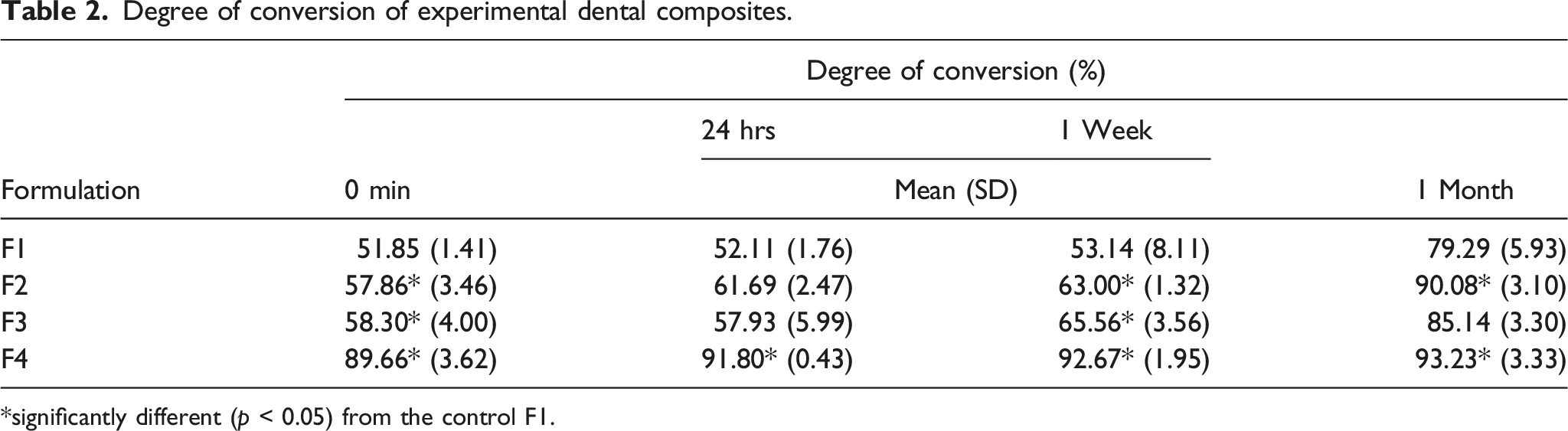
*significantly different (p < 0.05) from the control F1.
Polymerization shrinkage of experimental dental composite.

*Significantly different (p < 0.05) from the control F1.
Water sorption and water solubility of experimental dental composites.

Different letters in superscript indicate significant difference (p < 0.05).
Figure 5 shows the result of flexural strength of experimental dental composites. F4 presented the highest mean values of 81 MPa, which was significantly higher than those of F1, F2 and F3 (p < 0.01), and much higher than that of similar formulation (16.5 MPa) as reported previously.
20
Flexural strength of experimental composites.
The cumulative fluoride releases over 100 days of the control composite and experimental composites are shown in Figure 6. The fluoride release rates are significantly different from each other among all formulations throughout 100 days (p < 0.05). The fluoride ion release was highly accelerated on the first 10 days, followed by a sustained release until 100th day for all formulations. Among all formulations, F4 has the highest fluoride release rate over 100 days, followed by F3, F2 and F1. On the first 10 days, F4 has cumulative fluoride release level of 3.5 μg/cm2, indicating that it has the greatest initial burst release, and a sustained release over the next 90 days, with the highest cumulative fluoride release level of 9.2 μg/cm2. Cumulative fluoride release of experimental composites.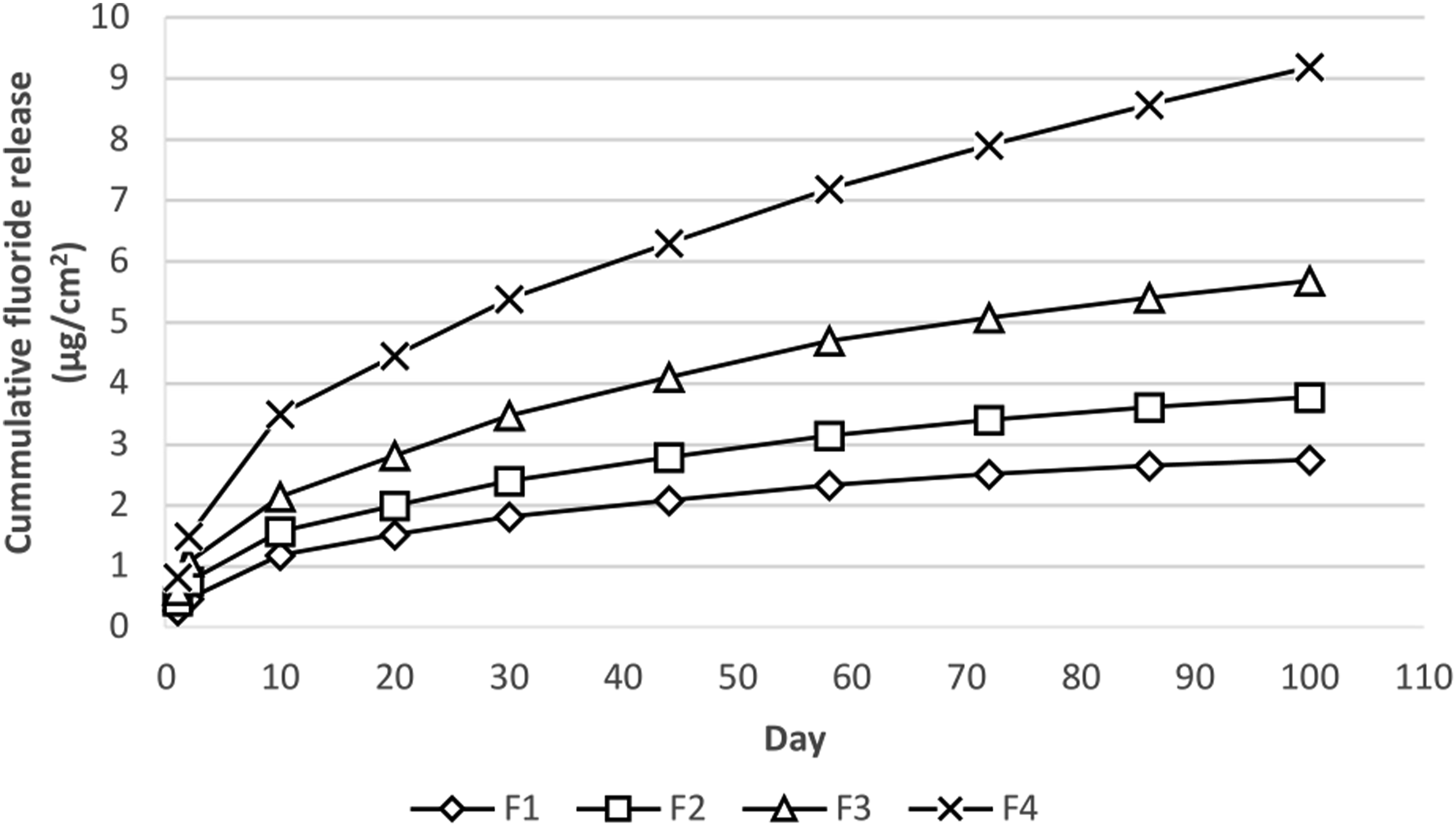
There are significant differences on fluoride recharge capabilities between the four formulations throughout 3 fluoride recharge cycles (p < 0.05) Fluoride recharge profiles of experimental composites.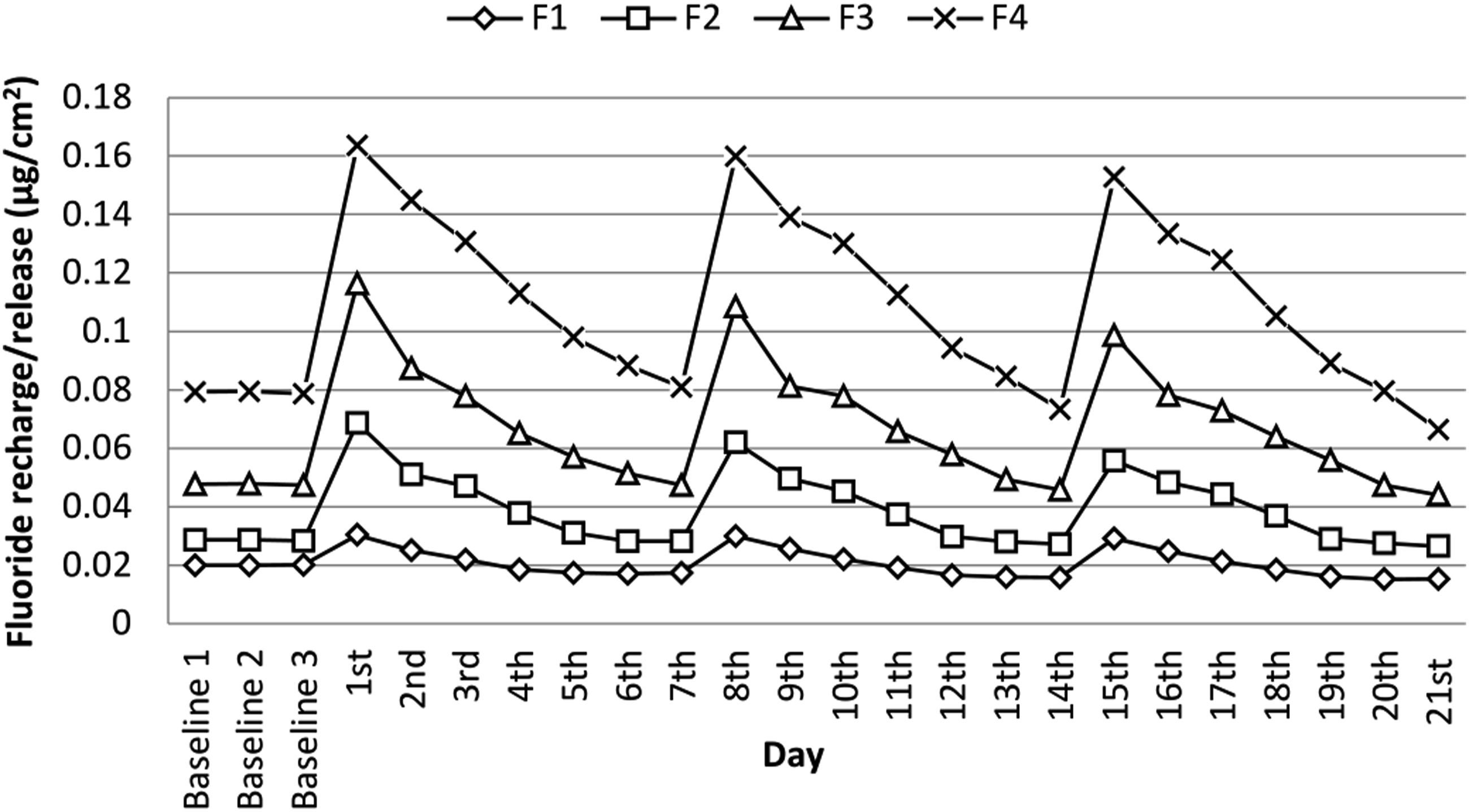
Scanning electron microscopy micrographs for the fracture surfaces of experimental dental composites, F1-F4, were recorded with accelerating voltage of 15 kV using secondary electron detection mode. Figure 8 shows the morphology of F1-F4. Small pores were apparent in F1 and F2 and voids were noticeable in F3 and F4. In general, the pore and void size increased with increasing TFEMA:TEGDMA ratio, with F1 having the smallest pores and F4 largest pore and voids. Representative FESEM images of dental composite fracture surfaces.
Discussion
In the preparation of the BisGMA/TFEMA:TEGDMA/UDMA composite test specimens, it was observed that TFEMA (0.65 mPa.s; 20°C, TOSOH F-TECH, Inc.), which is more fluid than TEDGMA (11.5 mPa.s; 20°C), 23 could be mixed with other ingredients with ease. Up to 70% of filler, from a combination of SrF, Zirconia, and silanised fumed silica, was successfully mixed to form a homogenous paste, as compared to just 55% silanised fumed nanosilica achievable in the previous study. 20 This demonstrated that nanosized filler is more difficult to be dispersed into the resin. This was attributed to particle aggregation, which increases resin viscosity at even modest filler loading. 24
As shown in Table 2, under identical curing conditions, the degree of conversion increased, from 51.9-89.7% at zero time and from 79.3-93.2% after 1 month, with increasing ratio of TFEMA:TEGDMA. This increase in degree of conversion could be attributed to much higher mobility of TFEMA than TEGDMA. This plausible explanation could be supported by a study which showed that early and highly cross-linked network arising from viscous Bis-GMA suppressed mobility of reacting species and, thus, the conversion retardation in subsequent curing stages, in the polymerization of Bis-GMA/TEGDMA multimethacrylate dental restoratives.14,25 Increased curing efficiency is essential for limiting residual monomer and thus minimising cytotoxicity effect 26 and for sustainable mechanical performance. 27
Polymerization shrinkage of dental composite resin can undermine integrity of composite-dentin bond and cause microcrack that could lead to secondary caries, which constitutes the main challenge faced by dental material scientists. Study has shown that polymerization shrinkage increased with TEGDMA concentration and monomer conversion during Bis-GMA/TEGDMA copolymerization. 28 The shrinkage reduced tremendously to 0.51% in F4 versus 6.5% in the control, and this could be explained by considering the differences in monomer molecular structures. The dimethacrylate TEGDMA has ether linkages that confer chain flexibility, 28 while the monomethacrylate TFEMA is smaller and rigid molecule. Hence, during polymerization, the flexibility of TEGDMA chains generated free volume within the lattice network, resulting in more shrinkages, in contrast to TFEMA chains which are more rigid and with much lesser free volume and, thus, less contraction. 5
Both the water sorption and water solubility increased with increasing TFEMA:TEGDMA ratio. Positive and negative correlations between water sorption and water solubility have been reported,29,30 indicating complex factors such as resin type and composition are involved in the process. Although the flexible TEGDMA chain was reported to form heterogeneous network that creates larger micropores which favours high water sorption, 7 it is unclear on how the gradual replacement of TEGDMA by TFEMA worsened the situation. Nevertheless, one possibility could be ascribed to bigger micropores generated in the cured composites, as observed in the increasing pore sizes in the order of F1, F2, F3 and F4 (Figure 8).
Flexural strength is a measurement of a material’s resistance against deformation. A dental composite with high flexural strength allows very thin restorations to be produced, which is particularly suited for minimally invasive treatment options. F4 had flexural strength of 81 MPa that, being the highest achieved in this study, is closer to ISO standard requirement of 100 MPa. However, the flexural strength of F3, F2 and the control are not significantly different, but was two folds lower than that of F4. This abrupt increase in the flexural strength could be due to the more perfect network structure developed in F4 in comparison to other formulations, possibly arising from the rigidity of polyTFEMA segments in the cross-linked network of the composites.
Fluoride ions have been known to be effective in preventing demineralization of enamel-dentin surface layers and, thus, avoiding teeth decay. 31 Many fluoride releasing dental restoratives show anti-caries effect in clinical application. Ability to release fluoride over extended period is one of the most desirable characteristics of dental composite. However, longer-term sustained release is seldom achieved and, to compensate this limitation, fluoride recharge capacity of a dental composite is essential. 32
Fixed amount of strontium fluoride and organic tetrabutylammonium fluoride (TBAF), sources of fluoride ions, were used in all the formulations. The results showed that fluoride release and recharge capability increased with the ratio of TFEMA:TEGDMA. This could be due to larger micropores formed in the cured network, which allowed more water sorption and, hence, larger area of contact with water soluble fluoride ions and ensuing release. This explanation also applied to higher recharge capacity of the experimental composites with increasing TFEMA content.
Clinical study has shown that it is impossible to remove all the microorganisms from the tooth surface after all soft dentin is removed during conventional caries removal procedure. 33 Thus, a fluoride-releasing composite like F4 which has a high level of initial “burst release” is important and essential for reducing the viability of bacteria left in the inner carious dentin, allowing enamel or dentin remineralization. 34
Conclusions
Increasing TFEMA:TEGDMA ratio in the Bis-GMA/TFEMA:TEGDMA/UDMA experimental dental composites resulted in an increase in degree of conversion, fluoride ion release and recharged capacity, water sorption, water solubility, and a decrease in polymerization shrinkage. The flexural strength of the F4 experimental composites experienced twofold increase.
Footnotes
Acknowledgements
The authors acknowledge resources and support from the Research Laboratory, International Medical University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support from the International Medical University [IJ143].