Abstract
Thermal management plays an important role in electrical and electronic systems. Owing to both excellent thermal conduction and electrical insulation, boron nitride nanosheets (BNNSs) are particularly attractive as fillers in polymer composites. While the thermal properties rely on the connection of BNNSs in polymer matrices significantly. Herein, BNNSs absorbed with silver acetate and 2-ethyl-4-methylimidazole (Ag (2E4MI)2Ac) complex were prepared as thermal conductive fillers for epoxy resin. During the cure of matrix, nano silver ions were in-situ reduced, sintered and bridged individual BNNSs together. Therefore, thermal contact resistance between BNNSs decreased and thermal conducting networks were effectively constructed. The thermal conductivity increased from 1.26 W/mK for the composites only with BNNSs to 2.35 W/mK for the composites with BNNS/[Ag (2E4MI)2Ac] hybrids at 20 vol% BNNSs content. Fitting the measured thermal conductivity results indicated that the thermal contact resistance between fillers decreased with the connections by sintered silver. In addition, the electrically insulating properties of the composites were well preserved and the tensile strength of the composites containing sintered silver interconnects was obviously improved.
Introduction
Electrical insulation and thermal management are of importance to electrical and electronic equipment. During the work of equipment, the heat generated by the thermal effect of electric current must be transmitted or emitted timely, or it will influence the reliability and even cause accidents.1–4 The main development trend of modern electrical and electronic equipment is high power, small volume and more compact, thus more heat is generated in the limited volume, which put forward higher demands on thermal conductivity of insulating materials.5–8 Epoxy resins and their composites have been widely used in electrical insulation, but the pure epoxy is a thermal insulating material with very low thermal conductivity (about 0.20 W/mK).9,10 It is difficult to meet the requirements for efficient heat transfer, becoming a bottleneck in the development of electrical and electronic industry.
In order to meet the requirements for efficient heat transfer in electrical and electronic equipment with high performance used in new energy, rail transit and intelligent manufacturing, the development and applications of epoxy insulation materials with highly thermal conductivity have attracted great attention recently. According to preparation technology, intrinsic and filled type are brought out. However, preparation of the former is complex, and the product demonstrates high melting point and viscosity.11–13 Usually, special molding equipment is needed, which is difficult to meet the requirement of insulation impregnating and encapsulating treatment for electrical equipment. On the contrary, filler type is the main method for preparing epoxy insulating materials with high thermal conductivity, possessing the advantages of simple operation and easy industrialized production. Several ceramic materials such as boron nitride (BN), aluminum oxide (Al2O3), magnesium oxide (MgO), aluminum nitride (AlN), silicon carbide (SiC) and beryllium oxide (BeO) are widely used as fillers due to their high thermal conductivities and electrical insulating performances.14–18 Among them, BN flakes have drawn more and more attention thanks to their superior chemical stability, high electrical insulating performance and thermal conductivity, whereas low dielectric loss and thermal expansion.19–21 Multilayer BN flakes could be exfoliated into few-layered BN nanosheets (BNNSs) which possess the similar two dimensional structure and thermal conductivity with graphene.22–24 However, BNNSs exhibit electrical insulation, thus having great potential as high thermal conductive fillers to enhance the thermal conductivity of polymeric insulating materials.25,26 Many researchers tried to develop BNNSs/epoxy composites with high thermal conductivity. Unfortunately, the thermal conductivity of the composites is still lower than the expected value in most cases. This is mainly ascribed to the absence of thermal conducting paths and high thermal contact resistance among BNNSs’ interfaces. 27 Gu et al.28–30 synthesized the hetero-structured thermally conductive fillers including SiC-BNNS and BNN-30@BNNS (spherical BN and BNNS) by sol-gel and electrostatic self-assembly methods. It is believed that the hetero-structured fillers could fully utilize the advantages of multielement fillers for synergistic improvement effects, thereby forming more efficient thermal conduction pathways. 30
In this work, novel hybrids composed of BNNSs absorbed with silver-imidazole complex was prepared as fillers for epoxy matrix composites to achieve a high thermal conductivity. The silver-imidazole complex was synthesized with silver acetate (AgAc) and 2-ethyl-4-methyl imidazole (2E4MI). We believed that [Ag (2E4MI)2]Ac complex absorbed on BNNSs surface could release 2E4MI and then initiate the curing reaction of epoxy at a suitable temperature, as shown in Figure 1.
31
Simultaneously, with the aid of the released 2E4MI as reducing agents, silver ions on BNNSs surface could be in-situ reduced to elemental silver, which could be further sintered together and play a bridge role to connect the individual BNNSs. Therefore, the thermal contact resistance between BNNSs decreased and the thermal conducting paths were effectively constructed. Figure 2 presents an overall schematic diagram of the composites. BNNSs/[Ag (2E4MI)2]Ac filled epoxy composites with high thermal conductivity of 2.35 W/mK at 20 vol% BNNSs content was obtained, whereas the electrically insulating property was not compromised. It was useful for the future application as an electrical insulation material. Schematic diagram of [Ag (2E4MI)2]Ac complex and the curing reaction of epoxy initiated by the released 2E4MI. Schematic illustration of preparation for BNNSs/[Ag (2E4MI)2]Ac hybrids (a) and BNNSs/[Ag (2E4MI)2]Ac filled epoxy composites (b).
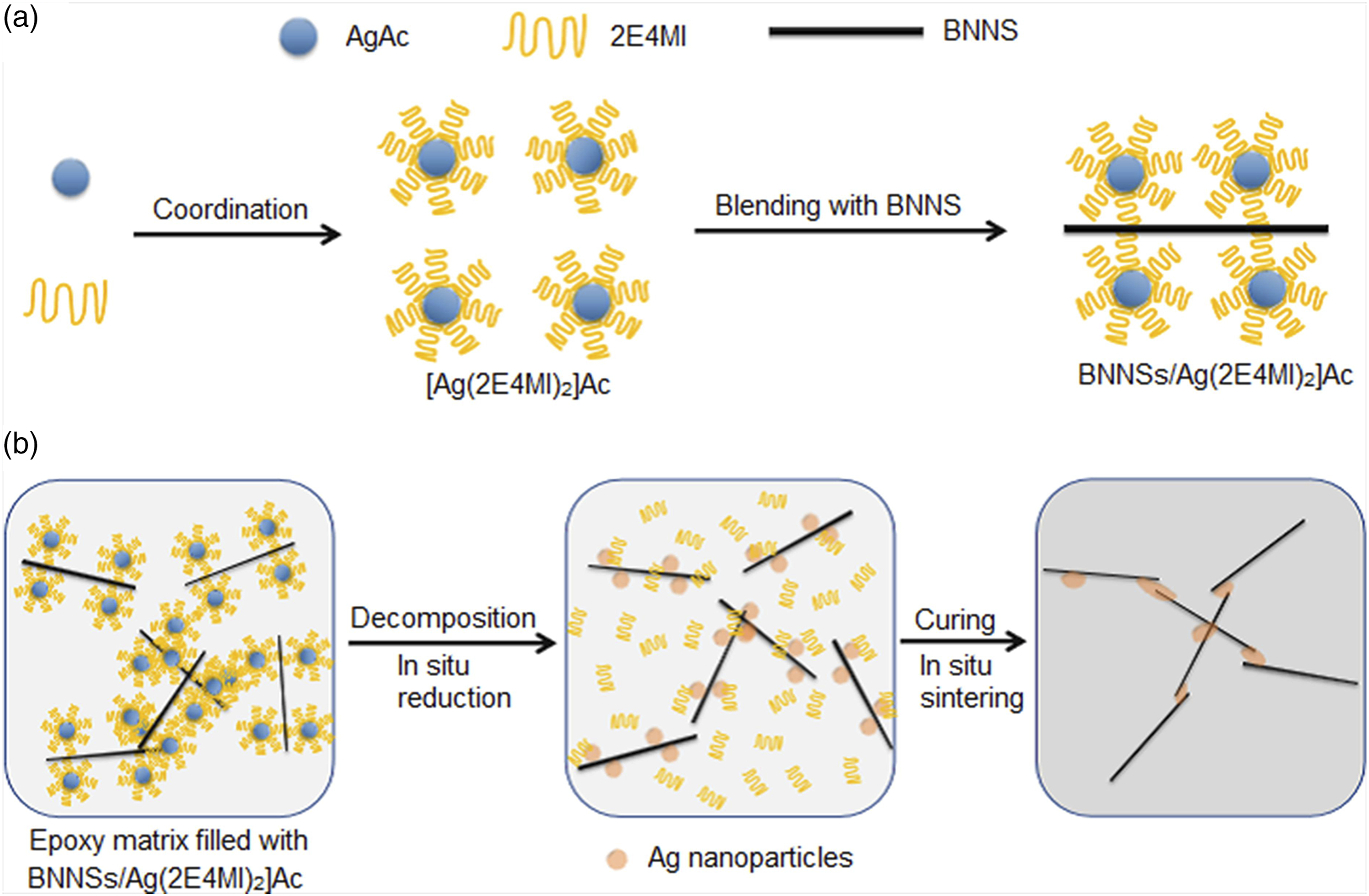
Experimental
Materials
Boron nitride (BN) was purchased from Dandong Chemical Engineering Institute. Diglycidyl ether of bisphenol A epoxy resin (DGEBA, epoxy equivalent 185) and 2-ethyl-4-methyl imidazole (2E4MI) was commercially available from Jiangsu Sanmu Group Corporation. Silver acetate (AgAc, 99.5%), N,N-dimethylformamide (DMF) and CH2Cl2 was purchased from Sinopharm Chemical Reagent Co., Ltd All chemicals were used as received.
Exfoliation of multilayer boron nitride
Few layer BNNSs were exfoliated from multilayer BN in liquid phase according to the method reported in literature.32,33 Briefly, 2 g BN micropowder was dispersed in 300 mL DMF. The dispersion was sonicated for 48 h in a sonic bath and then centrifuged at 1000 r/min for 20 min. After centrifugation, the supernatant was decanted and then vacuum dried at 150°C for 24 h.
Preparation of BNNSs/[Ag(2E4MI)2]Ac
The silver-imidazole complex was synthesized following a previous study with modifications. 34 Silver acetate (AgAc, 5 mmol) and 2E4MI (10 mmol) was added to 100 mL CH2Cl2. The mixture was stirred for 2 h until all the AgAc solid disappeared and the suspension became a light yellow solution of [Ag (2E4MI)2]Ac, which exhibits obvious Tyndall effects. Then the different amount of BNNSs was dispersed in the formed [Ag (2E4MI)2]Ac solution. After sonicated for 24 h, the dispersion was kept at room temperature and then vacuum dried at 40°C for complete CH2Cl2 evaporation.
Fabrication of epoxy composites filled with various boron nitride nanosheets contents
The prepared BNNSs/[Ag (2E4MI)2]Ac hybrids with different BNNSs content in 2.3 and DGEBA epoxy resin were mixed together. The mass ratio of [Ag (2E4MI)2]Ac to DGEBA was kept at 12.5:100 and the composites with different BNNSs content were obtained by altering the content of BNNSs in BNNSs/[Ag (2E4MI)2]Ac hybrids. The mixtures were placed in a vacuum oven at 80°C to remove the air and then cured at 150°C for 8 h. For comparison, the composites contain BNNSs, 2E4MI and DEGBA were fabricated in the same way described above.
Characterization
X-Ray photoelectron spectroscopy (XPS) was performed on a PHI-5000C ESCA system (Perkin-Elmer, USA) with Al Kα radiation (hv = 1486.6 eV). The survey spectra (from 0 to 1200 eV) with a normal resolution of 1 eV and the core-lever spectra with much high resolution (0.1 eV) were both recorded. The morphology of materials was characterized by field-emission scanning electron microscope (SEM, JSM-5600LV), transmission electron microscopy (TEM, JEOL JEM-2100) and atomic force microscopy (AFM, Nanoscope Multimode IIIa, Veeco Instruments). The thermal conductivity of the composites was measured through the laser flash technique (LFA467 HyperFlash, Netzsch) following ASTM E1461-13 standards. The electric resistance was measured by Model 6517B electrometer in accordance with ASTM D257-2014. The tensile strength was measured using computerized Tinius Olsen universal testing machine in accordance with ASTM D638-2014. Except as otherwise specified, all the measurements were carried out at room temperature.
Results and discussion
Morphology and chemical change of boron nitride nanosheets and BNNSs/[Ag(2E4MI)2]Ac
Figure 3 displays TEM and high-resolution TEM (HRTEM) micrographs of BNNSs and BNNSs/[Ag (2E4MI)2]Ac hybrids with 25 wt% of [Ag (2E4MI)2]Ac. All samples are ultrathin and transparent as revealed by TEM (Figure 3(a) and Figure 3(b)). The TEM micrograph demonstrates that the size of [Ag (2E4MI)2]Ac complex deposited on BNNSs/[Ag (2E4MI)2]Ac surface is about 5–20 nm. The HRTEM images (Figure 3(c) and Figure 3(d) exhibit that the thickness of samples is about 5 nm (15 layers). Furthermore, a typical lateral size of about 3 μm and thickness of about 5 nm is demonstrated in AFM images (Figure 4), and the size of [Ag (2E4MI)2]Ac deposited on BNNSs/[Ag (2E4MI)2]Ac surface is 5–20 nm, which are in agreement with TEM. Transmission electron microscopy (a,b) and High resolution transmission electron microscopy (c,d) micrographs of boron nitride nanosheets (a,c) and BNNSs/[Ag (2E4MI)2]Ac hybrids (b,d). Atomic force microscopy micrographs of BNNSs (a) and BNNSs/[Ag (2E4MI)2]Ac hybrids (b).

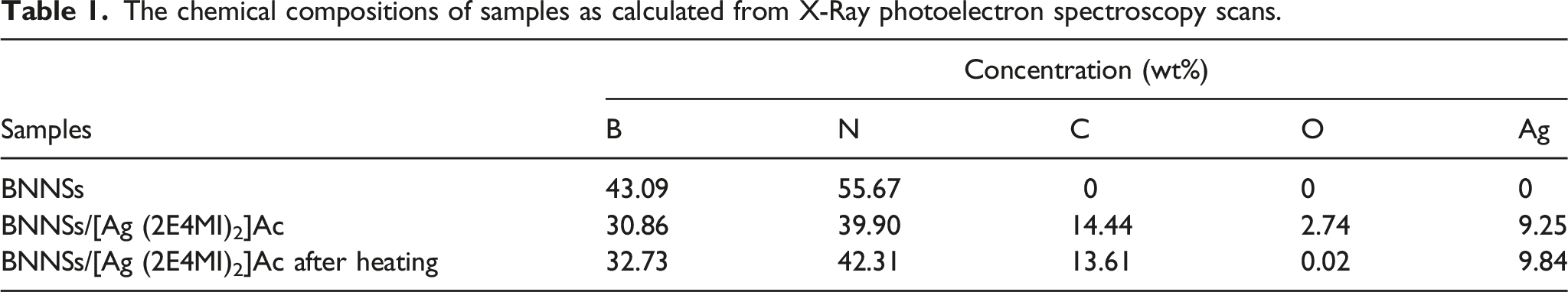
Figure 5 presents the XPS survey spectra (A) and Ag 3d high resolution spectra (B) of BNNSs, BNNSs/[Ag (2E4MI)2]Ac hybrids and BNNSs/[Ag (2E4MI)2]Ac after treatment in 150°C for 8 h. [Ag (2E4MI)2]Ac contents in BNNSs/[Ag (2E4MI)2]Ac hybrids were 25 wt%. The peaks at 190.8 eV, 285.0 eV, 368.2 eV, 374.2 eV, 398.4 eV, 533.5 eV and 573.6 eV can be as-signed to B 1s, C 1s, Ag 3d5/2, Ag 3d3/2, N 1s, O 1s and Ag 3p respectively. The elemental compositions calculated from the spectra are listed in Table 1. C, O and Ag elements are presented in BNNSs/[Ag (2E4MI)2]Ac hybrids. After heating, the oxygen content of the hybrids decreases significantly and the content of other elements correspondingly changes because of the reduction reaction of Ag+ ions. X-Ray photoelectron spectroscopy survey spectra (A) and Ag 3d high resolution spectra (B) of boron nitride nanosheets (a), BNNSs/[Ag (2E4MI)2]Ac hybrids (b) and BNNSs/[Ag (2E4MI)2]Ac after thermal treatment (c). The chemical compositions of samples as calculated from X-Ray photoelectron spectroscopy scans.
Thermal conductivity and morphology of epoxy composites filled with boron nitride nanosheets and BNNSs/[Ag(2E4MI)2]Ac
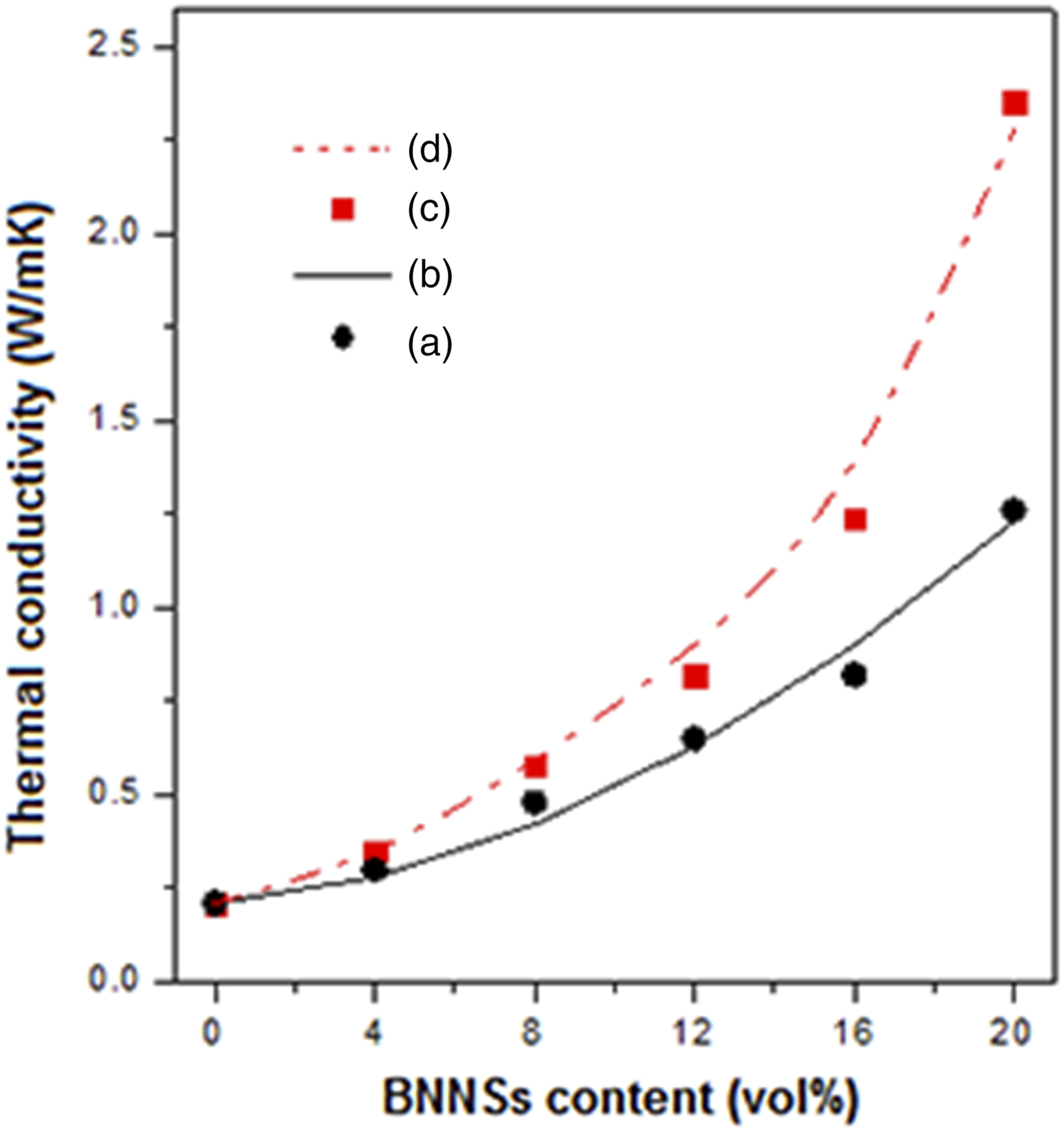
Figure 6 shows the thermal conductivity ( Effects of BNNSs content on thermal conductivity of epoxy composites filled with boron nitride nanosheets and BNNSs/[Ag (2E4MI)2]Ac hybrids.
To understand the high Scanning electron microscope cross section micrographs of the composites filled with boron nitride nanosheets (a) and BNNSs/[Ag (2E4MI)2]Ac hybrids (b) at 20 vol% BNNSs content.
Simulation on thermal conductivity of boron nitride nanosheets and BNNSs/[Ag(2E4MI)2]Ac filled epoxy composites
Many research groups have computationally simulated the thermal boundaries within nanofillers in order to decrease the thermal contact resistance and Umklapp phonon scatterings.35–37 Here, we apply a physical model proposed by Foygel et al. to analyze the experimental results. According to the Foygel model, the Foygel simulation on thermal conductivity of the composites: experimental values (a) and simulation curve (b) for boron nitride nanosheets filled composites; experimental values (c) and simulation curve (d) for BNNSs/[Ag (2E4MI)2]Ac filled composites.


Figure 9 presents the modified Foygel simulation for the Modified Foygel simulation on thermal conductivity of the composites: experimental values (a) and simulation curve (b) for BNNSs filled composites; experimental values (c) and simulation curve (d) for BNNSs/[Ag (2E4MI)2]Ac filled composites.
The
Electrical resistivity and tensile strength of epoxy composites filled with boron nitride nanosheets and BNNSs/[Ag(2E4MI)2]Ac
It is well known that BNNSs is an electrical insulator and considered as the promising dielectric material in the most applications of electrical equipment and electronic devices. Therefore, we anticipate that the electrical insulation performance of the epoxy composites will be well preserved after the addition of BNNSs/[Ag (2E4MI)2]Ac fillers. Figure 10 shows the effects of BNNSs content on the volume resistivity of the epoxy composites filled with BNNSs and BNNSs/[Ag (2E4MI)2]Ac hybrids. All the composites present a characteristic of the insulator (volume resistivity > 1011 Ω•m). Usually, the formation of the electrical percolation network promotes a decrease in electrical resistivity of the composite with the electrically insulating matrix. However, in our experiment, the sintered silver in the composites is not enough to reach the percolation threshold. The heat can be dissipated through phonon transport in BNNSs, whereas the electronic transmission through sintered silver cannot fully occur. On the other hand, the electrically insulating matrix and BNNSs may cause the tunneling barrier for the electrons and hinder the electronic transmission. Effects of BNNSs content on electrical resistivity of epoxy composites filled with BNNSs and BNNSs/[Ag (2E4MI)2]Ac hybrids.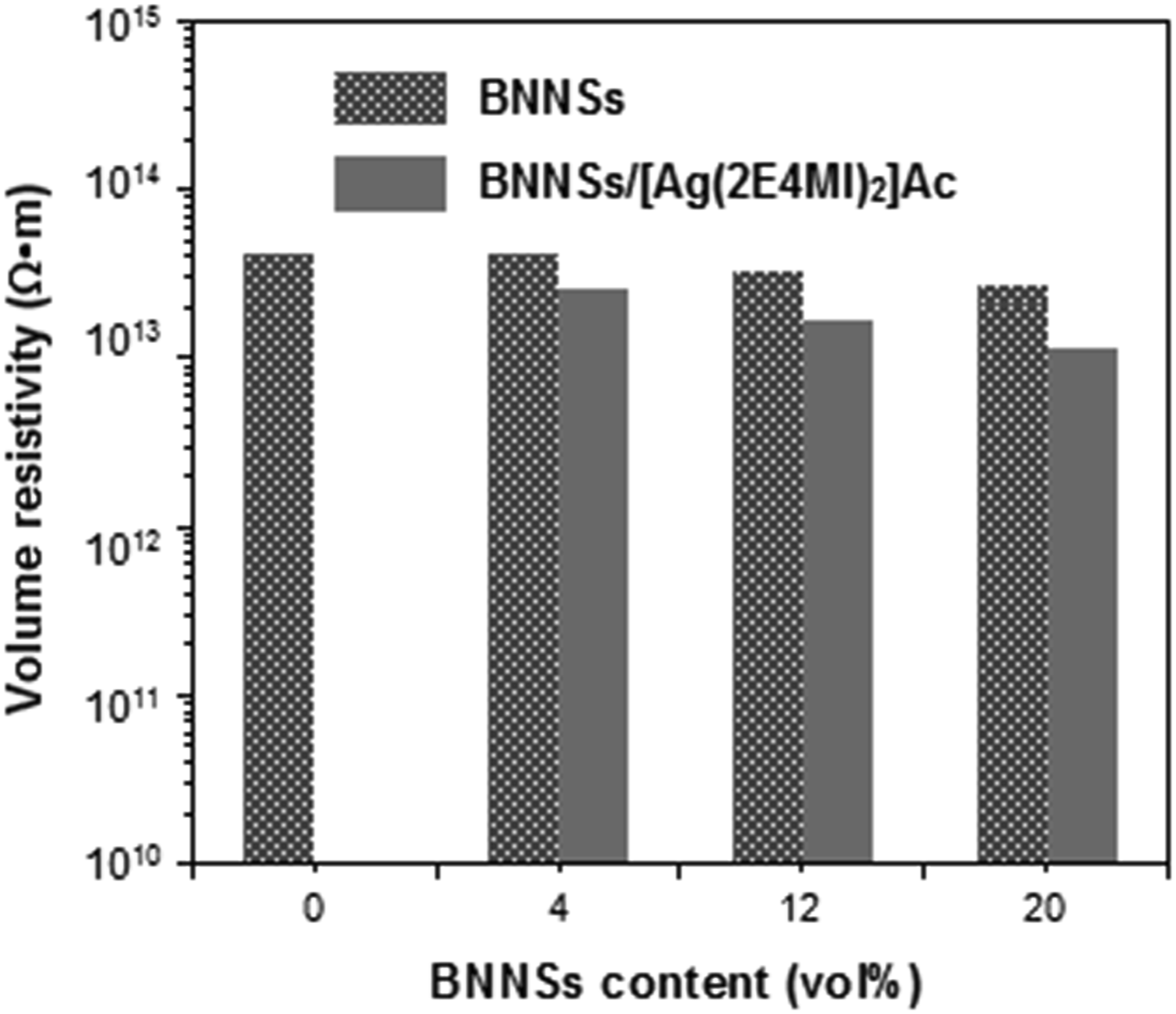
Figure 11 shows the effects of BNNSs content on the tensile strength of epoxy composites filled with BNNSs and BNNSs/[Ag (2E4MI)2]Ac hybrids. It can be seen that BNNSs content has a negligible effect on the tensile strength of epoxy composites only filled with BNNSs. However, the tensile strength of BNNSs/[Ag (2E4MI)2]Ac hybrids filled composites increases with BNNSs content obviously, due to the bridging connections with sintered silver between BNNSs. The network structure formed by BNNSs and sintered silver particles as revealed in Figure 7(b) can transfer stress and avoid the expanding of cracks, thus enhancing the tensile strength of the composites. Effects of BNNSs content on tensile strength of epoxy composites filled with BNNSs and BNNSs/[Ag (2E4MI)2]Ac hybrids.
Conclusions
In this study, BNNSs/[Ag (2E4MI)2]Ac hybrids and BNNSs/[Ag (2E4MI)2]Ac/epoxy composites with high thermal conductivity were successfully fabricated. During the curing process of BNNSs/[Ag (2E4MI)2]Ac hybrids filled epoxy resin, silver ions were simultaneously in-situ reduced, sintered and bridged individual BNNSs together. Thanks to the bridging connections between BNNSs with sintered silver rodlike particles, the thermal conductivity was increased from 1.26 W/mK for the composites only filled with BNNSs to 2.35 W/mK for the composites filled with BNNSs/[Ag (2E4MI)2]Ac hybrids at 20 vol% BNNSs content. Fitting the experimental results with a modified Foygel model demonstrated that the remarkable improvement in thermal conductivity of BNNSs/[Ag (2E4MI)2]Ac filled epoxy composites could be attributed to the decreased thermal contact resistance between BNNSs. Moreover, the tensile strength of BNNSs/[Ag (2E4MI)2]Ac filled composites was obviously improved and the electrically insulating properties of the composites were not compromised. Our findings contribute to a better understanding on the formation of thermal pathway in epoxy composites. The BNNSs/[Ag (2E4MI)2]Ac hybrids filled epoxy composites are promisingly applied in the thermal management materials for advanced electrical machinery, power systems, communication equipment and 5G electronic devices.
Supplemental Material
Supplemental Material - Improved thermal conductivity of epoxy composites via linked boron nitride nanosheets with in-situ generated silver nanoparticles
Supplemental Material for Improved thermal conductivity of epoxy composites via linked boron nitride nanosheets with in-situ generated silver nanoparticles by Wei Wang, Liyi Yang, Minmin Zheng, Fan Ge and Hui Ma in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Zhejiang Province Public Welfare Technology Application Research Project (Grant Nos. LGG19E030006) and the Applied Basic Research Projects of Jiaxing City of China (Grant Nos. 2019AD32004).
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
