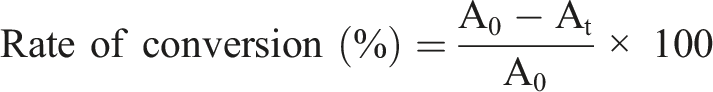Abstract
Acrylate monomer ((4-(3-(4-((4-(4-(3-(2,4-dichlorophenyl)-3-oxoprop-1-en-1-yl)phenoxy)-6-((4-nitrophenyl)amino)-1,3,5-triazin-2-yl)oxy)phenyl)-3-oxoprop-1-en-1-yl)phenyl acrylate (DTCP) is prepared and used to prepare poly (DTCP) and its copolymer poly (DTCP-co-HEA) and poly (DTCP-co-S) using 2-hydroxyethyl acrylate (HEA), styrene (S) by solution polymerization technique. Infrared spectroscopy (FT-IR), proton Nucleharmar Magnetic Resonance (1H NMR), and UV-Visible techniques are used to identify the synthesized polymers. Gel permeation chromatography method is used to determine the polymer’s weight average molecular weight, which is found to be around 10000 g/mol. According to the Thermo Gravimetric Analysis study, the synthesized polymers go through single stage decomposition centered at 340°C. The polymer reactivity ratio is found using well known Finemann-Ross (F-R), Kelen-Tudos (K-T), and extended Kelen-Tudos (ex.K-T) methods. The reactivity of the commercial monomers (HEA or S) is found to be higher than that of synthesized monomer, according to our findings. The photocrosslinking rate of the synthesized polymers is determined using a UV visible spectroscopic method and found to be high for polymer having HEA unit.
Introduction
Photosensitive polymer is used in many fields and hence many researchers are now focusing on discovering new polymers having a photosensitive group in the polymer backbone. These types of polymer have been crosslinked easily by the irradiation of UV light and used as printing inks, photoresist plates, coating materials,
1
liquid crystals,
2
tissue engineering,
3
biosensors,
4
UV-curing adhesives,
5
hole transporting materials in organic light emitting diodes
6
and optical anisotropy.
7
The polymers containing photosensitive moiety such as cinnamate,
8
chalcone,
9
stilbene
10
maleimide and anthracene
11
easily go through photo crosslinking upon irradiation
For a complete understanding about the rate of photocrosslinking in the copolymer, it is essential to know the reactivity of monomers which are used in the polymerization process. Monomer reactivity ratio exactly gives information regarding the composition of the copolymer. 17 The reactivity of monomer used for copolymer preparation is determined by the available methods like linear and many non-linear techniques.18–20 Here we synthesized an acrylate polymer based on triazine containing two photosensitive chalcone moieties in the polymer chain. The Reactivity ratio of the monomer was calculated using well known F-R, K-T and (ex. K-T) methods and found that the reactivity of the prepared monomer (DTCP) is lower than the commercially available monomer (HEA, S) used for copolymerization. The crosslinking process used was UV-visible spectrophotometric technique; the synthesized polymer undergoes photoisomerization under UV irradiation. The presence of more than one chalcone moiety in the prepared polymer will readily undergo photocrosslinking and display very high rate of crosslinking under UV irradiation. The literature shows no report on triazine based acrylate polymer of chalcone so we decided to prepare and study the photocrosslinking behavior of triazine based acrylate polymer. This article explains the preparation of acrylate polymers, monomer reactivity ratio and photocrosslinking property of polymer well in detail.
Experimental
Materials and methods
Acrylic acid, para-hydroxy benzaldehyde brought from Aldrich Chemicals. 4-nitroaniline 2,4-dichloro acetophenone (DCA), 4-hydroxyacetophenone and Triethyl amine (TEA), bought from SD Fine Chemicals. Styrene (S) Ethyl methyl ketone (MEK), hydroxy ethyl acrylate (HEA) and cyanuric chloride were received from Merck and distilled under reduced pressure before used for the reaction. Acryloyl chloride is prepared according to the standard known procedures. 21 The monomer DTCP used to prepare the polymer is reported in our previous report. 22 An IR spectrum was recorded using an ALPHA BRUKER FT-IR spectrophotometer by utilizing the KBr pellet process. The 1H NMR spectra were acquired by utilizing a Bruker FT-NMR (500 MHz and DMSO-d6, TMS) instrument. Polymer samples were dissolved in tetrahydrofuran and LABINDIA model UV 320 was used for UV absorption measurements. Thermal stability of the polymers is calculated using Mettler 3000 thermal analyzers at a heating rate of 10 °C/min in the air. The molecular weight of the polymer was figured out using a Schimadzu instrument with THF as an eluent at a flow rate of 0.3 mL/min.
Synthesis of poly (DTCP)
1.023 g of prepared monomer (DTCP) and 0.033 g of BPO taken in a polymerization tube having 10 mL of MEK, make this polymerization solution inert by passing nitrogen gas and kept in FT-IR spectrum of poly (DTCP): ◆; poly (DTCP-co-HEA): ■; and poly (DTCP-co-S): ▲. 1H NMR spectrum of poly (DTCP): ◆; poly (DTCP-co-HEA): ■; and poly (DTCP-co-S): ▲.

General procedure for the preparation of copolymer
The different mole ratio of prepared monomer (DTCP) and HEA or S has taken in a polymerization tube having 10 mL of MEK. To this 2wt.% of BPO was added, make the polymerization tube inert by passing nitrogen gas for a prolonged period and heated at 70 ± 1°C for the required time in order to control the monomer conversion rate to less than 15%. Then copolymer is obtained by pouring a polymer solution in a pure methanol with vigorous stirring, copolymer precipitate is filtered washed with methanol and weighed. The following copolymer contains a different composition of monomer was prepared using this solution polymerization method.
Preparation of poly (DTCP-co-HEA)
Scheme 1 was used to prepare Poly (DTCP-co-HEA). Under inert atmospheric conditions at 70 ± 1°C, copolymers of different composition of monomers are prepared using a solution polymerization method. FT-IR (cm−1) spectrum of equal molar composition of DTCP and HEA was given in the Figure 1, OH of HEA appeared around 3400 and CH stretching of aromatic and aliphatic appeared in 3040 and 2986 respectively. C=O of ester appears at 1748, carbonyl stretching appears at 1664 and CH stretching of vinylic bond appear at 1598 cm−1. The UV spectrum displays two distinct absorption peaks, at 250 nm and 335 nm correspond to π to π* transition of aromatic and π to π*transition of conjugated >C=C< respectively. Figure 3 shows the 1H NMR spectrum of poly (DTCP-co-HEA) (50:50). The presence of aromatic and vinylic protons appears at 6.9–8.4 ppm respectively. NH proton connected with an aryl ring appears at 4.1 and OH proton of HEA appears at 4.6 ppm. The O-CH2 proton appears at 3.6, CH2 protons connected to OH appear in 3.4 and newly formed polymer chain protons appear at 0.9–1.6 ppm. Synthesis of Poly (DTCP), poly (DTCP-co-HEA) and Poly (DTCP-co-S). Thermo gravimetric analysis spectrum of poly (DTCP): ◆; poly (DTCP-co-HEA): ■; and poly (DTCP-co-S): ▲.

Preparation of poly (DTCP-co-S)
Scheme 1 shows the synthetic route for poly (DTCP-co-S). Under inert atmospheric conditions at 70 ± 1°C, copolymers of different monomer (DTCP with S) composition are prepared using a solution polymerization method. FT-IR (cm−1) spectrum of equal molar composition of poly (DTCP-co-S) was given in the Figure 1. The NH bond stretching appears at 3300 and CH stretching of aromatic and aliphatic appears at 3053 and 2927 respectively. C=O of ester appears at 1748, carbonyl stretching appears at 1657 and CH stretching of vinylic bond appear at 1600. The UV spectrum displays two distinct absorption peaks, at 240 nm and 339 nm corresponds to π to π* transition of aromatic and π to π*transition of conjugated >C=C< respectively. 1H NMR spectrum of poly (DTCP-co-S) (50:50) given in Figure 2. The peak appears at 7.0 to 8.4 is due to the aromatic and vinylic protons respectively, peak at 4.3 is due to the Ar-NH proton and newly formed polymer chain protons appears at 0.9–1.8 ppm.
Photo irradiation studies
The prepared polymer poly (DTCP), poly (DTCP-co-HEA), poly (DTCP-co-S) dissolved in THF solvent, the solution is kept at 15 cm−1 apart from UV light and irradiated at normal temperature. UV absorption of polymer solution was measured and UV absorption changes occurred around 335 nm upon irradiation with UV light is monitored to study the photocrosslinking behavior of prepared polymer. Using the formula below, the rate of photocrosslinking of α, β-unsaturated ketone unit was determined.
Results and discussions
DTCP, the monomer, is made in a five-step process. 22 The synthetic route for poly (DTCP), poly (DTCP-co-HEA), and poly (DTCP-co-S) was shown in the Scheme 1 and was prepared in MEK solvent by adopting a free radical solution polymerization technique. The structures were confirmed using FT-IR, 1H NMR, and UV techniques. The FT-IR and 1H NMR spectra of poly (DTCP), poly (DTCP-co-HEA), and poly (DTCP-co-S) were shown in Figures 1 and 2 respectively. Intact of ester group during the polymer preparation process was confirmed by the FT-IR study. Formation of the polymer is confirmed by 1H NMR spectrum, which shows the peak of about 1–2 ppm is due to the polymer chain proton.
Molecular weights
Thermo gravimetric analysis, molecular weight data of the DTCP polymers.

Thermal analysis
Figure 3 depicts the TGA and DTG curves of poly (DTCP), poly (DTCP-co-HEA), and poly (DTCP-co-S), while Table 1 lists the decomposition temperatures. These findings indicate that the DTCP-based polymer decomposes in a single step. The temperature of poly (DTCP) decomposition was 340°C. Similar patterns were observed for other polymers containing HEA and S, and the results are shown in Table 1.
Solubility
The monomer DTCP is soluble in hexane, dimethyl sulphoxide (DMSO), ethanol, methanol, dimethyformamide (DMF) and acetone. Polymers such as poly (DTCP), poly (DTCP-co-HEA), and poly (DTCP-co-S) are freely soluble in high polar solvents such as DMSO, DMF and THF but not in methanol, water and hexane.
Determination of copolymer composition
The copolymer composition in the polymer network is calculated using the 1H NMR spectrum of poly (DTCP-co-HEA) and poly (DTCP-co-S). The copolymer chain comprising both monomeric units is confirmed in Figure 2. We used these resonances to determine the mole fraction of DTCP, HEA or S monomer units exists in the polymer chain. In poly (DTCP-co-HEA) six methylene protons along with 19 aromatic (DTCP monomer) protons are very well separated in proton NMR spectra. The mole fraction of monomeric units present in prepared polymer was found by comparing the integral values of the above said methylene and aromatic protons which were appearing in 1H-NMR spectra at 1-2 and 6–8 ppm respectively.
The used HEA mole fraction in poly (DTCP-co-HEA) was found using the following equation
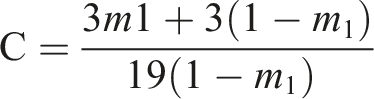
Upon simplification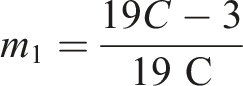
Mole fraction data for poly (DTCP-co-HEA).

[Monomer 1 + Monomer 2] = 0.5 mol/litre; Temperature: 70 ± 1°C; Initiator: BPO [1 weight % (w/w) of monomers 1 and monomer 2]; Solvent: EMK.
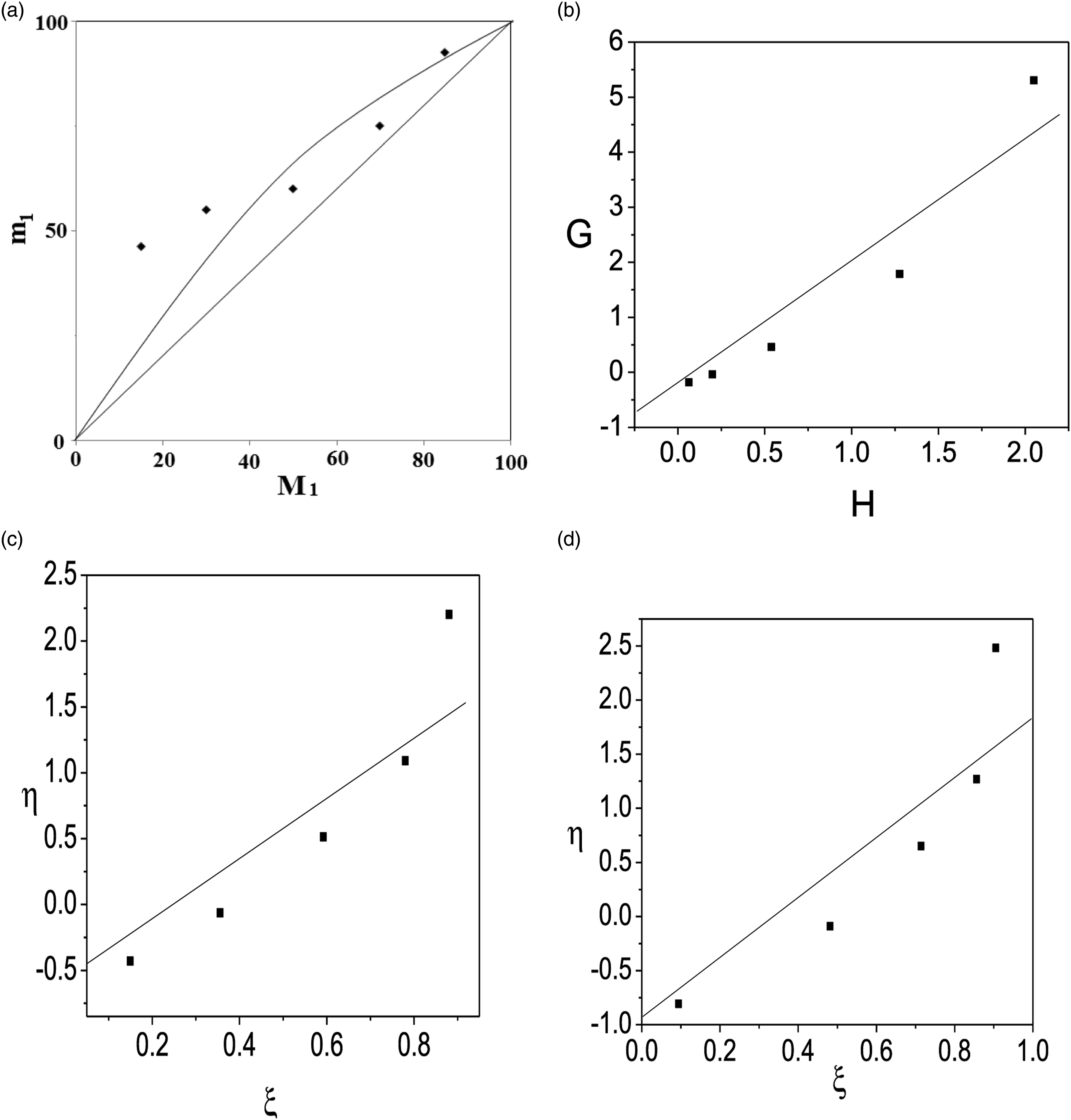
Table 2 shows the NMR data used to calculate the copolymer compositions. Figure 4 shows the mole fraction of used monomer in the copolymer (m1) versus the monomer feed mole fraction (M1). This tells us the used monomer for copolymer preparation was unevenly distributed throughout the copolymer chain. The reactivity ratios of the HEA (r1) and DTCP (r2) copolymer systems were calculated using graphical methods such as F-R, K-T, and ex. K-T. (a) Mole fraction curve (b) FR plot, (c) KT plot and (d) ex KT plot of Poly (DTCP-co-HEA).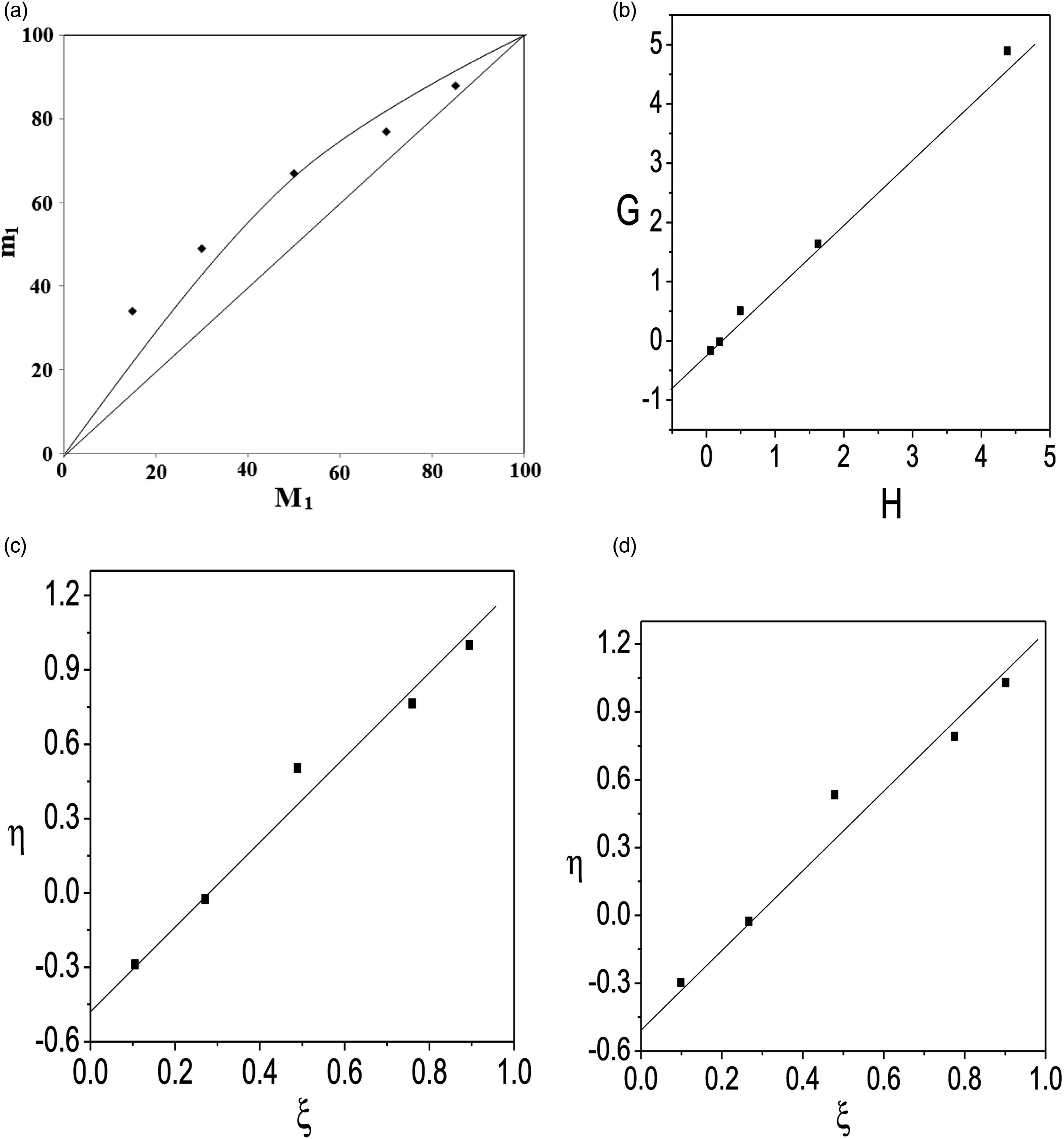
Fineman-ross method
The linear expression for calculating the monomer reactivity ratio was developed by Fineman-Ross. A straight line was drawn between G and H, with the intercept on the Y-axis equaling –r2 and the slope equaling r1 (Supplementary Table S1 and Figure 4). The reactivity ratios for prepared copolymer poly (DTCP-co-HEA): HEA (r1)= 1.11 ± 0.10 and DTCP (r2)= 0.75 ± 0.13.
Kelen-Tudos method
A straight line emerges from a plot of η Vs ξ, and we can measure r2 and r1 using the intercept value (Supplementary Table S1 and Figure 4). The reactivity ratios obtained were r1 (HEA)= 1.73 ± 0.13 and r2 (DTCP)
Extended Kelen-Tudos method
The reactivity of the monomers was found to be r1 (HEA)= 1.75 ± 0.23 and r2 (DTCP)= 0.51 ± 0.08. Since r1 is larger than r2, the likelihood of m1 joining the increasing polymer chain is far higher than that of m2. As a result, m1 is still more abundant in the prepared copolymer chain (Supplementary Table S2 and Figure 4).
Reactivity ratios Poly (DTCP-co-S)
The three graphical evaluation methods: (a) F-R, (b) K-T, and (c) ex. K-T were used to evaluate the reactivity of the commercial monomer S (r1) and prepared monomer DTCP (r2). The 1H NMR chemical shift values confirm the existence of both the monomer in the prepared poly (DTCP-co-S) polymer chain. The chemical shift values of styrene aromatic protons (5H) clearly overlap with DTCP aromatic 19 protons and the chemical shift values of methylene protons are distinguished from each other. The above said two chemical shift values were clearly distinguished, and these integral values could be used to determine the mole fraction of monomeric units present in the copolymer chain with precision. For determining the mole fraction of DTCP and S monomeric units present in the poly (DTCP-co-S) copolymer chain, the following equation was derived using 1H NMR values. Assume that m1 is the mole fraction of S in the copolymer chain and that m2 (1-m1) is the mole fraction of DTCP.
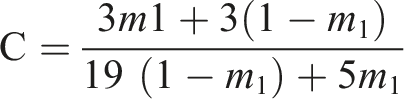
Upon simplification
Mole fraction data for poly (DTCP-co-S).

[Monomer 1 + Monomer 2] = 0.5 mol/litre; Temperature: 70 ± 1°C; Initiator: BPO [1 weight % (w/w) of monomers 1 and monomer 2]; Solvent: EMK.
(a) Mole fraction curve (b) FR plot, (c) KT plot and (d) ex KT plot of Poly (DTCP-co-S).
Fineman-Ross method
A straight line was drawn between G and H, with the intercept on the Y-axis equaling –r2 and the slope equaling r1 (Supplementary Table S3 and Figure 5). The reactivity ratios of S (r1) and DTCP (r2) were found to be 2.26 ± 0.27 and 0.70 ± 0.16 respectively.
Kelen-Tudos method
Kelen-Tudos developed a new linear equation for calculating reactivity ratios to address some of the flaws in the Fineman-Ross process. The straight line was obtained from a plot of ηverses ξ, and r2 and r1 were determined from the intercept values and slope, respectively (Supplementary Table S3 and Figure 5). The reactivity ratios of the monomers obtained by this method were r1 (S) = 2.31 ± 0.16 and r2 (DTCP) = 0.45 ± 0.23.
Extended Kelen-Tudos method
The extended Kelen-Tudos system was a slightly altered version of Kelen-Tudos. The straight line was measured using intercept and r2 and r1 slope values by utilizing a plot of η verses ξ (Supplementary Table S4 and Figure 5). The values of r1 and r2 by the ex. K-T method was found to be r1 (S) = 2.79 ± 0.11 and r2 (DTCP) = 0.93 ± 0.06.
Reactivity ratio of the monomers
r1 and r2 values of the copolymers.

Photocrosslinking studies of the polymer bearing DTCP
Figures 6–8 depicts the transition in the UV absorption spectrum of the polymer based on DTCP. There are two forms of chalcone moieties in the DTCP. Both absorb at the same wavelength and here the molar absorption is nothing but the yield of two vinylic π- π* and two carbonyl n-π*transitions of chalcone moiety present in DTCP. The above said two vinylic π- π*transition had an absorption maximum centered at 340 nm. As the polymer was exposed to UV light, the absorption maximum decreased as the UV irradiation time increased. The UV absorption maximum decreased as UV irradiation time increased due to two changes that occurred in the polymer network. One was the cyclobutane ring formation and another one was photoisomerization. The photoisomerization is nothing but the transformation of the prepared polymer structure from trans to cis form. The electron present in p-orbital on vinylic carbon destroys by two ways one was geometrical isomerization and another was a cycloaddition reaction which can take place on irradiation. Figures 6–8 shows the isobestic point around 305 nm confirms the geometrical isomerization took place in the polymer structure.
22
The prepared polymer contains DTCP unit which contains two chalcone moiety usually undergo cis-trans isomerization on irradiation
22
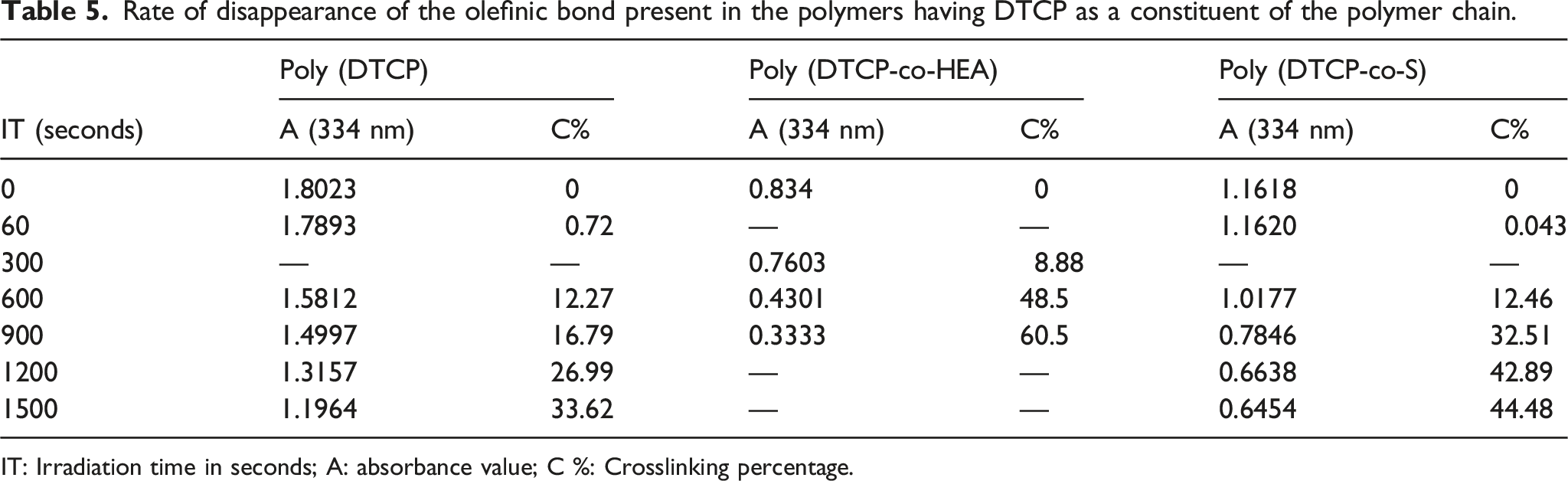
and cycloaddition reaction also took place which was confirmed by absorption maximum decreases around 340 nm. UV light was irradiated to the prepared polymer solution, hence a disruption of chromophore aggregate was not raised due to the absence of a regular arrangement of polymer in the solution. Photosensitivity of prepared polymer was calculated by considering the rate at which double bond vanishes after being exposed to light. The rate of photocrosslinking/isomerisation conversion of poly (DTCP), poly (DTCP-co-HEA), and poly (DTCP-co-S) was shown in the Table 5. Shift in the UV absorbance spectrum of Poly (DTCP) after irradiation period of 0–1500 s. Shift in the UV absorbance spectrum of Poly (DTCP-co-HEA) after irradiation period of 0–1500 s. Shift in the UV absorbance spectrum of Poly (DTCP-co-S) after irradiation period of 0–1500 s. Rate of disappearance of the olefinic bond present in the polymers having DTCP as a constituent of the polymer chain. IT: Irradiation time in seconds; A: absorbance value; C %: Crosslinking percentage.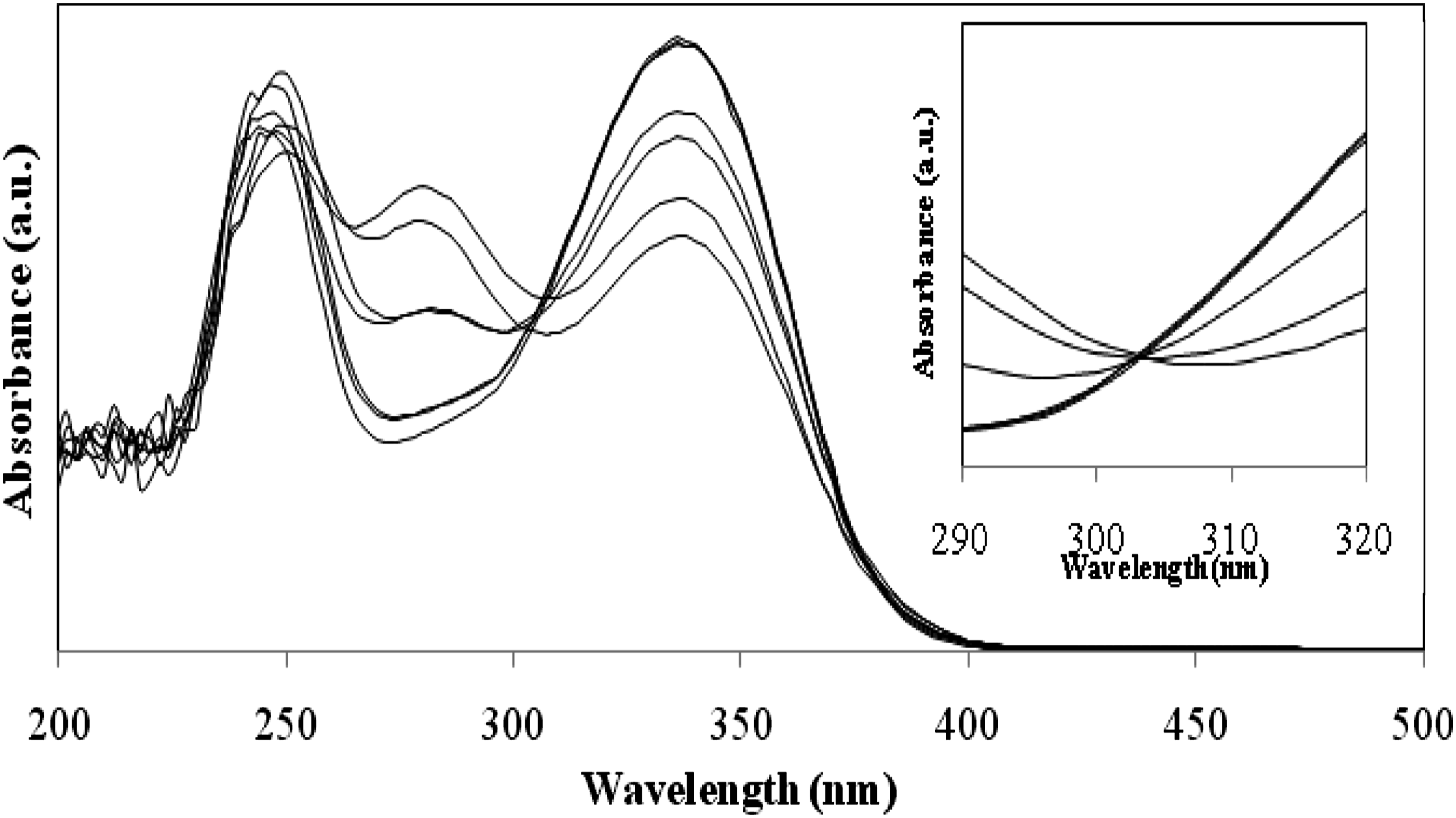


Comonomer (HEA and S) influence on the rate of photocrosslinking of DTCP polymer
Figure 9 depicts the comparative graph of the crosslinking percentage of the prepared polymer and it reveals that the rate of loss of the double bond of the copolymers was higher than that of homopolymer poly (DTCP). The arrangement of polymer molecules in solution also had a significant impact on photocrosslinking. At 600 s of UV irradiation, there was no significant difference in percent crosslinking between poly (DTCP) and poly (DTCP-co-S), but poly (DTCP-co-HEA) showed a significant difference. This may be because the inclusion of HEA in the polymer chain allows for proper photochemical reaction orientation, while styrene does not. However, after 600 s of UV irradiation, styrene does play a role in the proper photochemical reaction, but not as well as HEA did. This demonstrates that the presence of small molecules in the polymer chain causes the polymer photocrosslinking percentage to increase. Even after 1500 s of irradiation, the photoisomerisation/photocrosslinking percentage was low, implying that the large sized polymer side chain greatly disturbs the crosslinking. Rate of disappearance of >CH=CH< of poly (DTCP): ■; poly (DTCP-co-HEA): ●; poly (DTCP-co-S): ▲.
The comonomer HEA or S had not showed much support for cycloaddition reaction upon UV light irradiation, since the presence of polymer unit contain conjugated double bond adjacent to each other was necessary for the cyclobutane ring formation. Figure 9 further confirms that the rate of photocrosslinking of DTCP based polymer was majorly due to photoisomerization, i.e. cis-trans isomerization predominantly took place on irradiation. The rate of photocrosslinking was in the following order: poly (DTCP-co-HEA) > poly (DTCP-co-S) > and poly (DTCP).
Conclusion
The monomers DTCP, HEA, and S are used to make a homopolymer and two copolymers using a solution polymerization technique. FT-IR, UV-Visible and 1H NMR techniques were used to characterize the synthesized polymers. The synthesized polymers had an average molecular weight of about 10000 g/mol. The polymer heat stability was high, with a single stage decomposition temperature of around 330°C. The synthesized monomer DTCP is less reactive than the used commercial monomers HEA and S, according to the reactivity ratio data. The absorption peaks for aromatic and vinylic/carbonyl absorption were found to be around 240 and 330 nm, respectively, in the UV results. The UV visible spectrophotometric technique was used to determine photocrosslinking behavior of the polymers and it was discovered that photocrosslinking of the prepared DTCP based polymer not completed even after 25 min of UV light irradiation. Overall, using the copolymerization process, the rate of photocrosslink is increased from 33 to 60% in a simple way.
Supplemental Material
Supplemental Material - Acrylate polymers of triazine containing chalcone moiety: Synthesis, characterization, reactivity ratio and photocrosslinking studies
Supplementary Material for Acrylate polymers of triazine containing chalcone moiety: Synthesis, characterization, reactivity ratio and photocrosslinking studies by J Suresh and A Arun in Polymers and Polymer Composites.
Footnotes
Acknowledgements
The authors thank Mrs T. Deepa for correcting English in this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.