Abstract
The bisphenol A diglycidyl ether-polyaminoamide (epoxy resin)–graphene oxide (GO) nanocomposites have been prepared by chemical methods. The aim of this study is to produce epoxy –GO coatings on Aluminum Alloy 7075 and study the anticorrosion properties. Thin films have been characterized by scanning electron microscope – Energy Dispersive X-rays Spectroscopy (EDX) and Raman spectra. Raman spectra show the successful bonding of GO functional groups with epoxy. The composition of epoxy and GO is characterized by EDX. The Tafel plots were undertaken for analysis of corrosion of coating material. Therefore, the reduction of corrosion current of epoxy–GO nanocomposites coated Al alloy 7075 show its superior corrosion resistance properties. This is further confirmed by electrochemical impedance spectroscopy and potentiodynamic polarization. The very high value of low frequency impedance modulus (i.e. 1010 Ω cm2) shows surprising anticorrosive effect on coating. The epoxy–GO spin coating in Al Alloy 7075 is significantly reduces the corrosion current density (Icorr) and corrosion rate due to anticorrosion properties of GO.
Keywords
Introduction
The nanoscale engineering produces nanostructured material, especially in polymers, the extraordinary properties could be achievable depending on selection of nanomaterials and its dispersion throughout the polymer matrix.1–6 The presence of functional groups in polymeric materials can easily adsorb at the surface of metals. This property of polymers makes them efficient corrosion inhibitors. 7 Epoxy resins are very important class of thermosetting polymers that often exhibit high tensile strength and modulus, good corrosion resistance and good dimensional stability. The epoxy resins are the most important thermosetting materials used in various industrial applications such as in automobile, aerospace, and defense due to their excellent adhesion and mechanical properties.8–10 The main drawback of nanoparticles in epoxy chain is the poor dispersibility, which restricted the potential application of nanocomposites in several cases. 11 Recently, it is reported that hardener cured epoxy with nano or micro structure enhances adhesion, anti-corrosion, viscoelastic and rheological properties.12–25 The metallic nanofillers in epoxy matrix may hinder cross-linking reaction into epoxy chain reduces the corrosion resistance. Also metallic nanoparticles in epoxy matrix revealed the difference in adsorption potential due to change in curing characteristics26–28, however, carbonaceous nanofiller like GO and Carbon Nano Tube (CNT) in epoxy are most promising candidate to enhance the mechanical properties, electrical properties and corrosion resistance is due to high surface area, high mechanical strength, low density and high thermal resistance.29,30
Chen et al. reported the sandwich-like structures of polydopamine and 8-hydroxyquinoline coated graphene oxide for excellent corrosion resistance of epoxy coatings. 31 Yankun Xie Chen et al. reported novel approach to fabricate polyacrylate modified graphene oxide for improving the corrosion resistance of epoxy coatings. 32 Mohammad et al reported the development of metal-organic framework (MOF) decorated graphene oxide nano-plateforms for anti-corrosion epoxy coatings. 33 These efforts develop a scientific knowledge to develop anticorrosion material. Ali et al reported that epoxy-polyamide nanocomposites coating with graphene oxide as cerium nanocontainer generating effective dual active/barrier corrosion protection. 34 The several publications have explored more attempts for the organic coating materials; however, these methods still have much room for improvement for the complex process steps, chemical modifiers, and therefore fail to satisfy the requirements of high efficiency, low cost and environment friendliness in industrial production. The novelty of this work is to present a method to coat uniform thin film of epoxy–GO nanocomposites on Al substrate to enhance anticorrosion properties as compare to other method. The spin coating method is most suitable for nanocomposites coatings as compare to other method. There are several advantages of spin coating over other methods such as (i) uniform coating thickness could be maintained, (ii) thickness of coating can be varied by varying the speed of spin coater, (iii) the solvent can be removed during the spinning process due to evaporation, (iii) strong adhesive coating and (iv) layer by layer coating. The main target of present work is to develop corrosion resistant novel coating method and material for aircraft aluminum. This objective could be achieved by applying Electrochemical impedance spectroscopy (EIS) and potentio-dynamic polarization techniques. Further, structural morphology of epoxy–GO thin film was studied by SEM and Raman to justify the compatibility of coating.
Experimental details
Materials
Epoxy resin, graphite flakes, curing agent (hardener) KMnO4, H2SO4, H3PO4, HCl, H2O2, acetone, ethanol, and dimethylformamide were procured from Sigma Aldrich, India. The commercially available Aluminum Alloy 7075 (99% pure) sheet were procured from Mallinath Metals, Mumbai (India).
Preparation of graphene oxide
GO was prepared by modified Hummers method. In this method, a mixture of graphite powder (2 g), H2SO4 (50 mL) and H3PO4 (5.5 mL) were mixed and stirred for 2 h. During stirring, 5.5 g of KMnO4 was slowly added into the solution. This mixture was stirred for 6 h until the solution became dark green. The more details of this method is reported in literature. 35 To reduce the excess of KMnO4, 12 mL H2O2 and 80 mL of H2O was dropped slowly and stirred for 15 min. The physical or chemical changes are occurred in exothermic reaction, they are generally accompanied by transfer of heat. 10 mL of hydrochloric acid (HCl) and 30 mL of deionized water (DIW) was added and centrifuged for 5 min at 4000 rpm for 30 min. The precipitate was washed with DI water, HCl and ethanol. The final precipitate was dissolved in DI water and sonicated for 1 h. The washed GO solution was dried using oven at 90 °C for 24 h to produce the powder of GO. The 80 mL of H2O and 12 mL H2O2 was added to remove other impurity.
Preparation of epoxy/graphene oxide nanocomposites
The graphene oxide/epoxy nanocomposites thin films were prepared by sol gel method. The amounts of epoxy resins and curing agent were 70 and 30 parts by weight/weight ratio have been taken and stirred at room temperature for 20 min. For preparation of epoxy –GO solution, the GO of 0.4 wt.% were mixed slowly in previous solution and again stirred for the period of 20 minutes. The uniform viscous solution of epoxy and epoxy –GO have been prepared. The optically plane glass plates were immersed in epoxy solution and epoxy solution of different GO concentration at room temperature for about 10 minutes. The glass plates were gradually drawn out from solution and kept in vacuum oven at 50 °C overnight to remove the residual traces of solvents. The thin films were peeled off from the plates and again kept in vacuum over the period of 24 h to prevent them from impurity and moisture. Figure 1 shows the mechanism of chemical interaction/bonding of GO with epoxy resin. Mostly epoxy-based polymers are cross-structured polymers. The cross-linking of polymers is significantly affected the mechanical properties. The covalent bond based schematic epoxy network structure is shown in Figure 1 (represented by a solid black line in Figure 1(c)). The two active sites of epoxy resin are represented by rhombus E (Figure 1(a)) and the pentagon H (Figure 1(b)). Figure 1(b) shows the five active sites of curing agent. The formations of covalent bond are based on the sharing of electron between active sites and stabilize their potential energy. In this process four dots of hardener generates four covalent bond by cross-linking, but when GO was mixed, the last dot of pentagon connected with GO sheet functional group and make very strong cross-linking or very complex matrix, which is shown in Figure1(d). The mechanical strength of epoxy matrix could be increased in presence of GO. Further, Al 7075 of dimension 1×1 cm2 is rotated at high speed in order to spread the coating materials by centrifugal force. The solvent from coating is removed during spinning process due to evaporation and keeping the coated samples at 50 °C in vacuum oven. The spin coating method is presented in Figure 2. Mechanism of chemical interaction/bonding of GO with epoxy. Spin coating of epoxy–GO in Al alloy 7075 sample.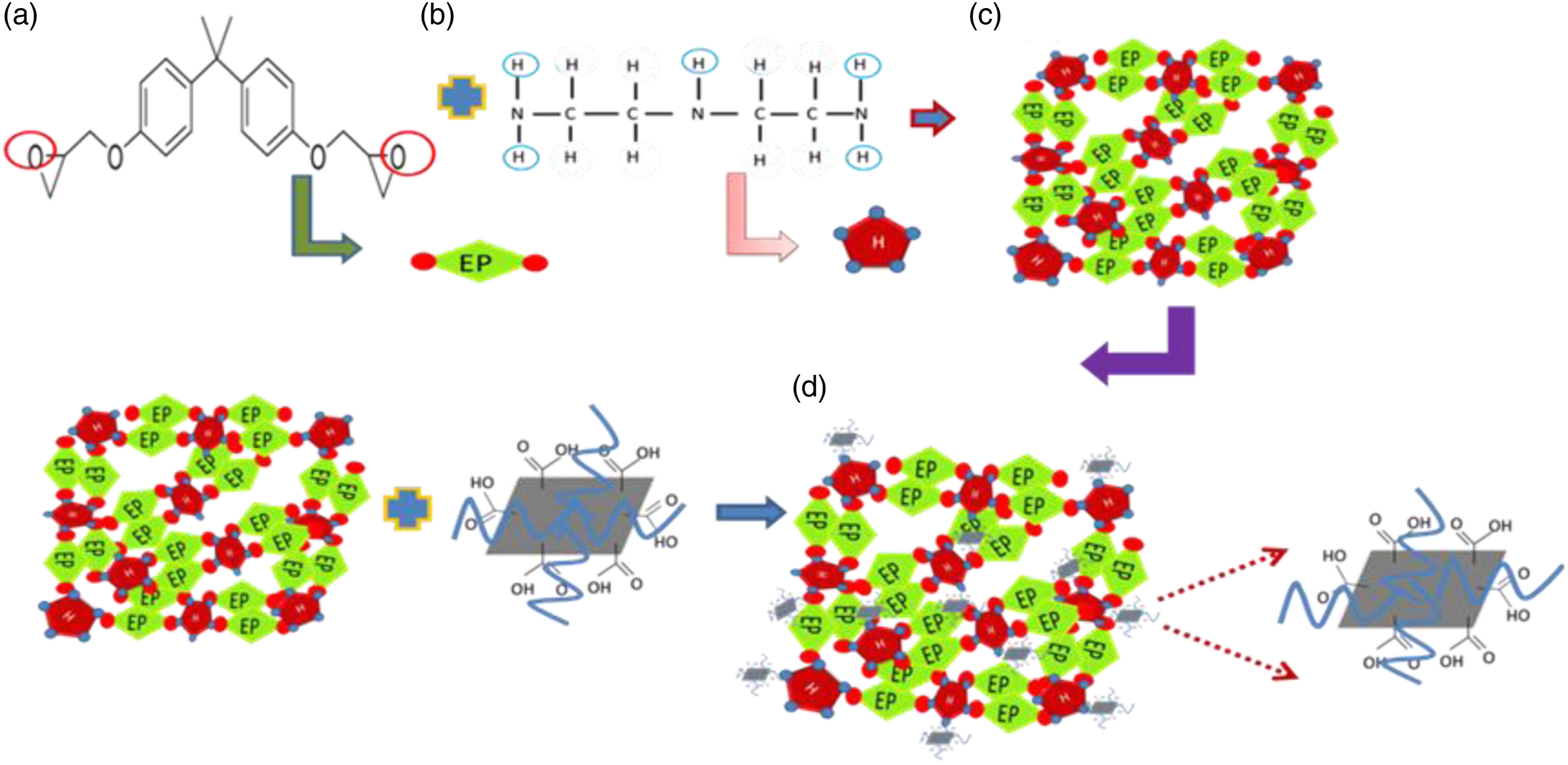

Results and discussion
Scanning electron microscope–Energy Dispersive X-rays Spectroscopy analysis of epoxy/GO nanocomposites
The microstructure and their compositions of epoxy–GO nanocomposites were characterized by Scanning electron microscope (SEM)–Energy Dispersive X-rays Spectroscopy (EDX) (MIRA II LMH form TESCAN). Figures 3 and 4 show the SEM –EDX images of epoxy–GO nanocomposites. It has been observed that when concentration of GO increases, the dispersion of GO multilayers also increased, which is shown in Figure 3(b) and (c). It is well known that epoxy exhibits typically brittle fracture, however, epoxy –GO have gliding fracture. This shows that fault age was rough for the epoxy–GO nanocomposites. Thus, GO in epoxy matrix increases the toughness of the epoxy by preventing crack propagation. The strong interfacial bonding of GO with epoxy is due to influence of GO owned oxygen-containing groups. The oxidation potency of GO is high due to presence of the oxygen-containing (i.e., epoxide, C=O, hydroxyl) groups. The phytochemicals can be oxidized into quinone forms and, in this way, eliminate the oxygen-containing groups of GO.36,37 This is an evidence that epoxy resin molecules are not only reduced the oxygen-containing groups, but also increase the GO interlayer d-spacing.
38
This suggests that strong adhesion exists between the epoxy and GO. At the same time, the orientation movement of molecular chain of polar groups in the epoxy–GO nanocomposites is restricted. EDX spectra of pure epoxy and epoxy –GO nanocomposites are shown in Figure 4. The EDX shows the presence of carbon, oxygen, chlorine, calcium, and palladium. The change in value of C, O and Cl indicates the occurrence of chemical reaction in pure epoxy and GO. This data attributed the successful preparation of nanocomposites. EDX presented the 66% carbon and 22% oxygen because of the fine grain graphite is oxidized from the surface and propagates gradually to the inner side. The graphene oxide in porous structure of epoxy reduces porosity. Thus, epoxy–GO coating in metal inhibited the effects between the protected metal and corrosive media.
39
SEM images of nanocomposites with the varying amounts of GO (a) pure epoxy, (b) epoxy –0.2 wt.% GO and (c) epoxy–0.4 wt.% GO. EDX analysis of nanocomposites with the varying amounts of GO (a) pure epoxy, (b) epoxy –0.2 wt.% GO and (c) epoxy–0.4 wt.% GO.

Raman spectra of epoxy resin- GO nanocomposites
Raman spectroscopy (Senterra, Bruker) is important technique to evaluate the structural changes in the prepared samples. Figure 5 shows the Raman spectra of pure epoxy and epoxy–GO (0.2 and 0.4 wt.%). As expected, the spectra of pure epoxy and epoxy–GO (0.2 & 0.4 wt. %) exhibits some characteristics peaks, which represents the groups present in GO, epoxy and epoxy –GO (0.2 and 0.4 wt.%) nanocomposites. Figure 5 shows the peaks at 75, 392, 606, 804, 997, 1097, 1304, 1378, 1545, 1592, 2065, 2528, 2852, 2874, 2922, and 3067 cm-1.40,41 The peak at 75 cm-1 could be ascribed to the lattice vibration or LA mode and 392 cm-1describing the aliphatic chains (250-400 cm-1) group and similarly at 606 cm-1. This is corresponding to (CC) alicyclic aliphatic chain vibrations (600-1300 cm-1). Other peak or band likes 804 cm-1 is indicating C-O-C (800-970 cm-1) and 997 & 1097 cm-1 represents (CC) aromatic ring chain vibrations and (C-O-C) asymmetric group respectively.
42
The peak at 1304 cm-1 and 1408 cm-1 is corresponding to CH3 group and N=N aromatic compounds. The peaks at 1615 and 1656 cm-1 is showing successful functionalization of GO.
43
The Raman spectra of pure GO displays two main peaks; the G band at 1592 cm-1 and D band at 1378 cm-1, corresponding to the vibration of sp2-bonded carbon atoms and sp3 defects of disordered ones in the hexagonal graphitic layers.44,45 The intensity ratio of D band against G band (ID/IG) represents the structural disorder degree of carbon material. The peaks at 2852, 2874, 2922 cm-1are indicating C-H group (i.e. ranging from 2800 to 3000 cm-1). However, peak at 3067 cm-1 is responsible for =C-H group. It has been observed that the some disturbance in the actual position of peaks (i. e. Table 1) attributed the presence of GO in epoxy resin composites. Raman spectra of nanocomposites with the varying amounts of GO in pure epoxy. Analysis of different peaks/bands in Raman spectra.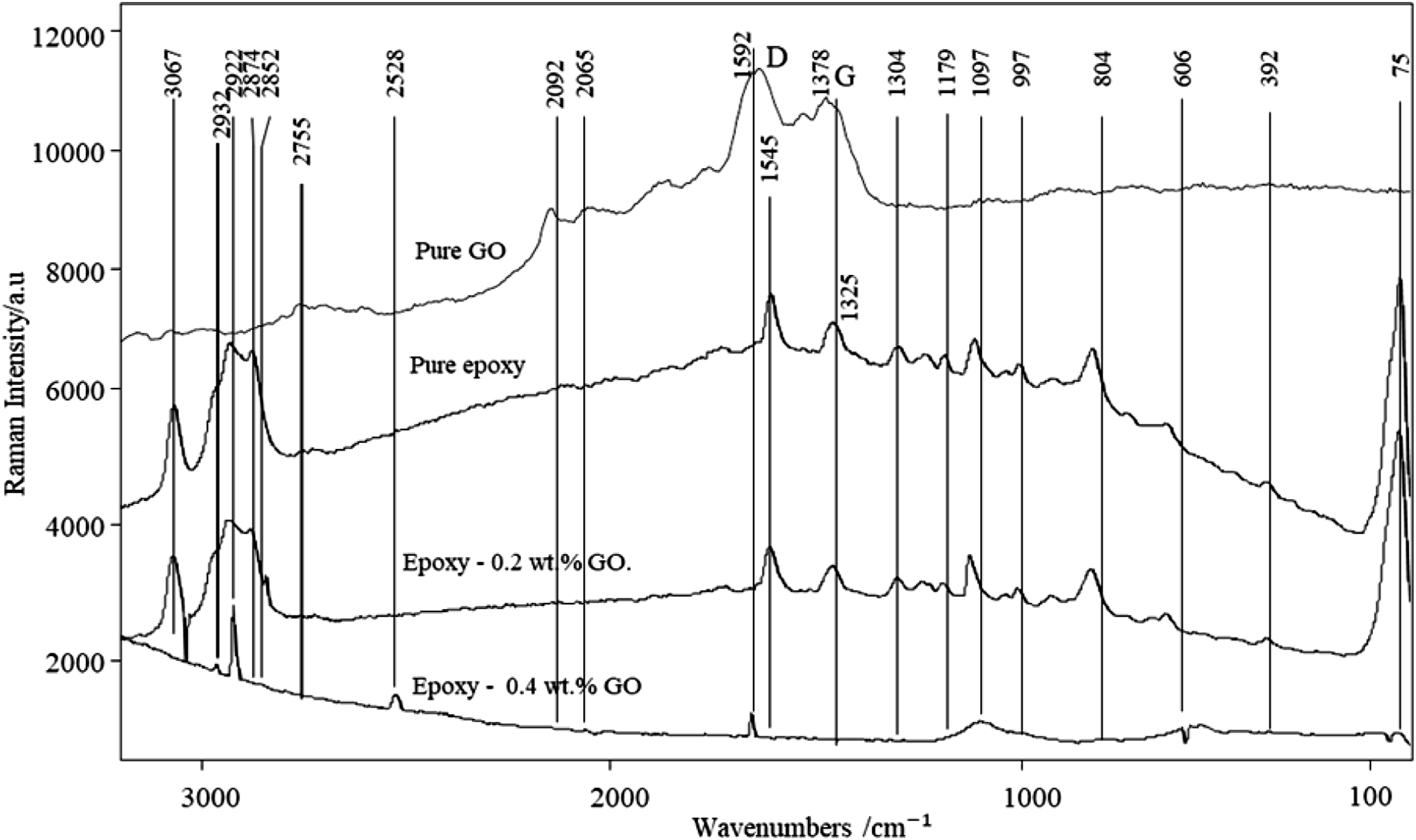

Study of corrosion behavior
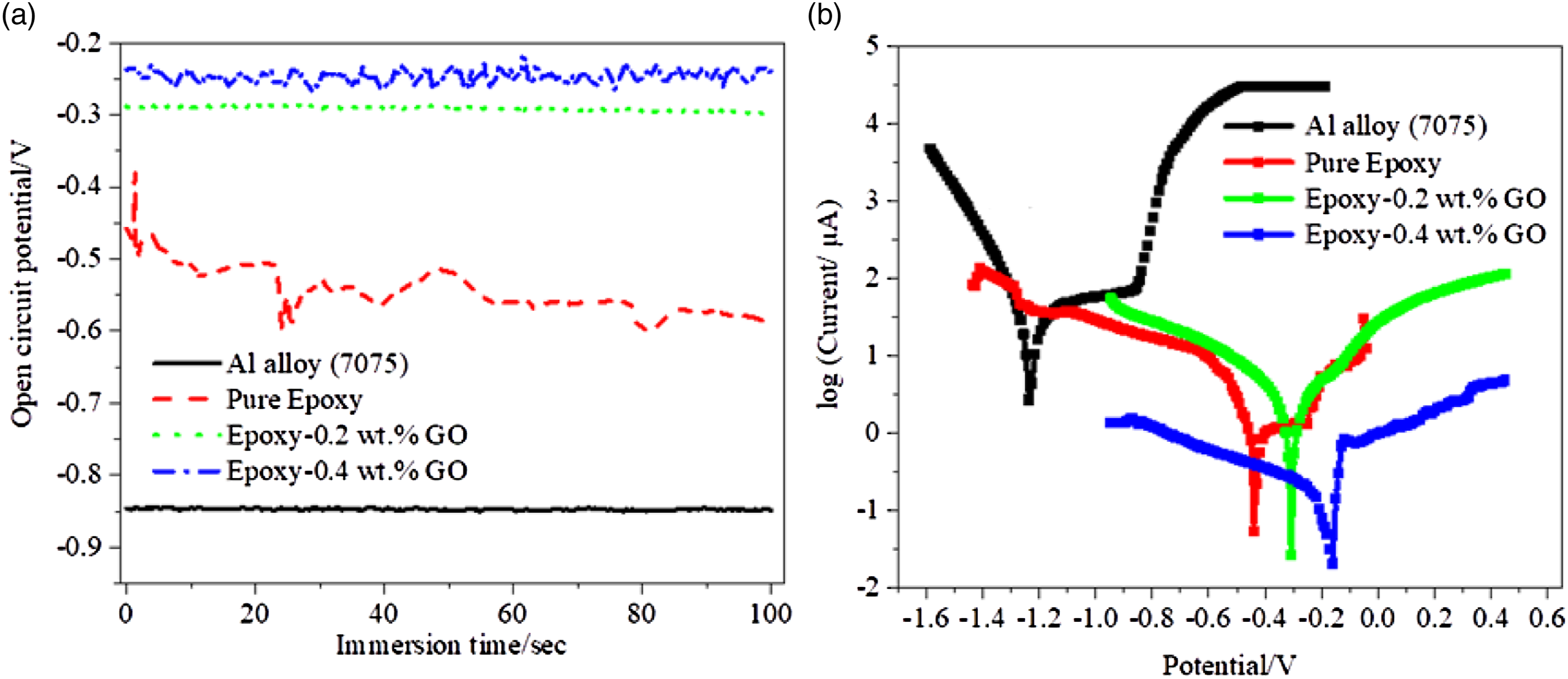
The corrosion properties were recorded by using Palmsan3 potentiostat system with microcode version 1.8. The corrosion resistance and barrier properties of epoxy–GO coatings were demonstrated in a solution of 3.0 wt.% NaCl at pH 5. The corrosion resistance of coating was performed in a salt cabinet. The epoxy/GO coated Al alloy (7075) samples of dimension 1x1cm2 were immersed in sodium chloride solution of 3.5 wt.% for corrosion study. Throughout the test process, the three-electrode system, Ag/AgCl electrode as a reference, epoxy/epoxy nanocomposites coated Al alloy (7075) as active electrode and platinum as a counter electrode were used. The open circuit potential is an demonstration of polarization study of epoxy/GO coated Al alloy samples at pH 5. The OCP values were obtained for Al alloy (7075), pure epoxy coated Al alloy, epoxy–0.2 wt.% GO and epoxy–0.4 wt.% GO nanocomposites coatings as shown in Figure 6(a). It has been observed that epoxy–0.4 wt.% GO nanocomposites are more positive than other coatings. This shows that 0.4 wt.% of GO in epoxy favours the high corrosion resistance and suitability of material for commercial coating. The positive shift in OCP shows less corrosion, while negative shift of OCP suggests high corrosion of material.
46
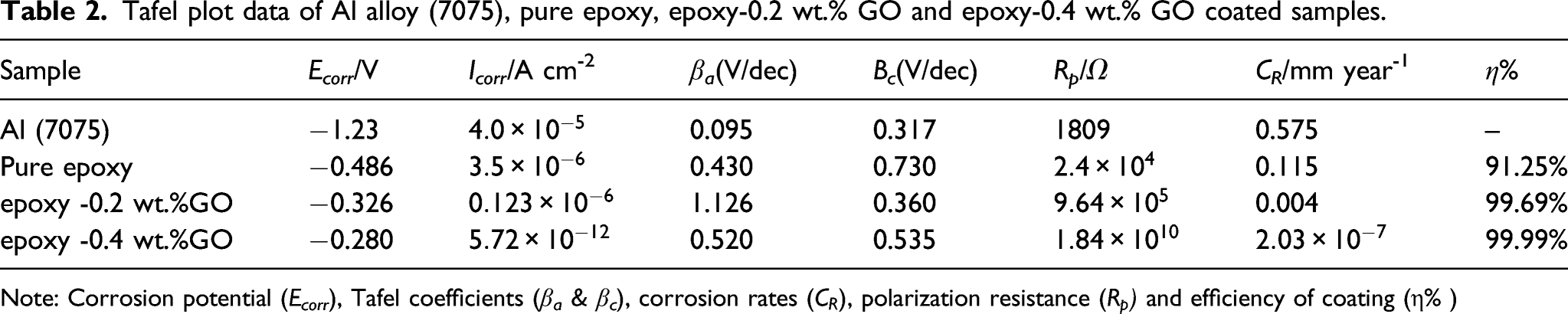
The corrosion current density was determined by the method of Tafel extrapolation or by fitting the Butler-Volmer equation on a linear polarization measurement. (a) Analysis of open circuit potential in uncoated and coated Al alloy with different concentration of GO in 3.5 wt% NaCl solution and (b) Tafel plots of Al alloy (7075), pure epoxy, epoxy–0.2 wt.% GO and epoxy–0.4 wt.% GO coated samples in 3.5 wt.% NaCl solution at pH 5. Tafel plot data of Al alloy (7075), pure epoxy, epoxy-0.2 wt.% GO and epoxy-0.4 wt.% GO coated samples. Note: Corrosion potential (E
corr
), Tafel coefficients (β
a
& β
c
), corrosion rates ( Comparative studies of the current corrosion parameters with other work.



Electrochemical impedance measurement
Electrochemical impedance spectroscopy is an effective approach to decide the corrosion safety performance of coatings. Sinusoidal amplitude of 20 mV among the frequency varies from 0.01 to 50 kHz. The Bode and Nyquist plots for corrosion study are shown in Figure 7(a) and (b). It has been observed that impedance value of epoxy–0.4 wt.% GO coated sample is rather high (0.86× 1010 Ω. cm2) as compare to other coating concentration of GO with epoxy. This indicates the improvement in low frequency impedance modulus as compare to previously published data.
32
The Bode plots are an evidence for anticorrosion performance of epoxy–GO coating in Al Alloy 7075 by spin coating method at the level of lower frequency. The corrosion cell in surface of metal alloy will stop the corrosion reaction in presence of GO due to good dispersion and proper alignment in the epoxy matrix. The high aspect ratio and large surface area make longer diffusion pathway for permeation of corrosion agent. It has been observed that the lower concentration (0.2 wt.%) of GO do not block the pores and the defects in matrix are remains the same. The value of phase angle at higher frequency (0.01 Hz) in Bode Plot (Figure 7) also represents the comparison of coating material. In principle, corrosion resistance is high for higher value of phase angle. The pattern of phase angle plot is similar to Bode plot. It is observed that epoxy coating exhibit small value of phase angle as compare to epoxy–GO coating. Its value decreases with time in a particular salt. The barrier properties of epoxy–GO (0.4 wt.%) of coated sample was carried out in NaCl solution. It has been observed that barrier properties are maintained without much degradation of coating. Figure 8 shows the appropriate equivalent circuit for EIS study. The equivalent circuit is well agreed with EIS data. (a) and (b) Bode plots and, (c) Nyquist plots for Al alloy (7075), pure epoxy, epoxy–0.2 wt.% GO and epoxy–0.4 wt.% GO coated samples in 3.5 wt.% NaCl solution at pH 5. Equivalent circuit diagram of Al alloy (7075), pure epoxy, epoxy–0.2 wt.% GO and epoxy–0.4 wt.% GO coated samples in 3.5 wt.% NaCl solution at pH 5.

Conclusions
The structural morphology of epoxy are significantly changed by cross-linking of GO with side groups of epoxy as demonstrated by Raman spectra. The SEM–EDX demonstrate the influence of GO in epoxy resin and its compositional analysis. The epoxy–GO coating is also acted as good barrier for the defects and microspores in the coating and provides excellent barrier property for oxygen, water and other corrosive medium to diffuse into the coating. Tafel results indicates the lower corrosion current density (i.e. 5.72×10−12 A cm2) and higher corrosion protection efficiency (i.e. 99.99%) with corrosion rate of 2.03×10−7 mm/year for epoxy–0.4 wt.% GO nanocomposites coating as compare to epoxy and epoxy −0.2 wt.% GO coating. The low frequency impedance modulus of GO in epoxy coating of Al alloy 7075 was 1010 Ω cm2. The anticorrosion properties of epoxy increases due to enhancement of hydrophobicity and adhesion strength by incorporation of GO in epoxy matrix. This study provided a new coating material for corrosion prevention of aluminum alloy. Successful alignment of the composite cover on the aluminum alloy surface has been critical in improving the aluminum structures and extending its service life in the aviation area and these nanocomposites can be used in other applications.
Supplemental Material
Supplemental Material - Investigation of anticorrosion properties of epoxy GO nanocomposites spin coated Aluminum Alloy 7075
Supplemental Material for Investigation of anticorrosion properties of epoxy GO nanocomposites spin coated Aluminum Alloy 7075 by Mulayam S Gaur, Rajesh K Raghav, Rohan Sagar and R.K. Tiwari in Polymers and Polymer Composites
Footnotes
Acknowledgements
Acknowledgements Financial assistance from Aeronautics Research & Development Board, DRDO Bhawan, New Delhi-110011, India (Letter no. ARDB/01 l1051902l/M/l), is gratefully acknowledged). We are also thankful to Director IUAC, New Delhi for providing characterization facilities.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Aeronautics Research and Development Board and grand id ARDB/01 l1051902l/M/l.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
