Abstract
In this work, the influence of an electric field on thermophysical properties of the PP+ZrO2 nanocomposites has been investigated. Changes in thermograms before and after applying an electric field (EFI) have been studied depending on the filler percentage in the polymer matrix. After EFI a new, weak maximum at the temperature range of 100–130°С of the thermograms for all concentrations of the filler was observed. Thermophysical parameters of the nanocomposite sample have been calculated for different values of electric field strengths that were applied are calculated depending on the effect of the ETP.
Introduction
Currently, metal oxide-based nanocomposite materials are very important materials in modern science and technology. Therefore, recent progress and advances in metal oxide-based polymeric nanocomposites, and their properties, such as physical, optical, structural and thermophysical using different methods are intensively studied by world researchers.1–5
Currently, polymer composite materials (PCM) based on polypropylene (PP) with metal oxide fillers are widely used in technology. It is known that the introduction of a filler into a polymer leads to significant changes in its supramolecular structure. Furthermore, the properties of the interphase layer of composite influence all physical-mechanical, electro-physical, and thermo-physical characteristics. Due to the set of applicable properties polymer nanocomposite based on metal and polymer are the subjects of intensive research.6–9 The widespread use of polymer micro- and nanocomposite materials in heat and power engineering requires a large amount of knowledge of their thermophysical properties. Polymeric materials are widely used in the manufacture of thermally-insulating or thermally-conductive materials. It becomes more necessary not only to bring the characteristic properties (elastic-strength, dielectric, crack resistance, etc.) of the polymer nanocomposites to the maximum possible values, but also to keep these properties in the widest possible temperature range under the influence of an electric field.
It is known that the DSC study of materials allows to obtain important information about the thermal properties of materials.10,11 Free Gibbs energy, heat capacity, enthalpy and entropy in the materials can be calculated using this method.12–15 This paper presents the results of DSC studies of the PP+ZrО2 nanocomposite exposed and not exposed to the electric field effect. The work aims to establish patterns of change in the set of thermophysical properties depending on the applied electric field. Furthermore, it is possible to calculate also the temperatures of phase and relaxation transitions. Moreover, structural studies of the obtained samples were performed by X-ray diffraction analysis, which is one of the excellent techniques to study of structural properties of the materials.16–20
Experimental
PP+ZrO2 nanocomposites were prepared through the introduction of the ZrO2 nanoparticles into the polymer matrix. 7 The size of the nanoparticles in the nanocomposites is about 20–30 nm. The nanocomposite samples were obtained by hot pressing method at the melting temperature of the PP polymer under a pressure of 15 MPa for 10 min. The thickness of the thin film is 70–100 μm. Melted nanocomposite sample was quenched into the ice-water liquid and cooled at the rate of 20deg/s. Sample was exposed to electro-thermal treatment at different values of the electric field strength. The composite is applied in a direct electric field, and polarized for 1 h in the field, after which, without removing the field, it is cooled to room temperature. A high-voltage rectifier is used as a voltage source.
Electrification is performed in the following way: samples undergo electrothermal treatment at various values of the electric field for 1 h at 100°C. For polarization, the films are heated up to this temperature, and then they stay 1 h and are cooled under the field. The electret is placed between metal electrodes. Electret characteristics have been measured with the device EFPM-1 (Electric field parameter meter).
DSC data were obtained on the NETZSCH DSC 204 F1 Phoenix heat flux calorimeter by analyzing melting and crystallization thermograms. The studies were carried out at a temperature range of 300–500 K with a heating rate of 20 deg/min. The experimentally obtained DSC curves represent the dependence of the heat flux (mJ/s) or specific heat (J/g·K) on temperature. Heating occurs in a nitrogen atmosphere at a predetermined rate according to a controlled program, and during the measurement process, the heat flux of the reference and the test material is compared.
X-ray phase studies were performed using D2 Phaser X-ray diffractometer in reflection mode (Bragg-Brentano geometry) using CuKa radiation (mean wavelength λ = 1.5406 Å, nickel β-filter). Registration was carried out in a continuous mode in the range of angles 2θ = 5–80o. Structural studies and calculations were performed using the EVA and TOPAZ programs, respectively.
Results and discussion
Figures 1(a) and (b) show thermograms of pure PP film and PP+ZrO2nanocomposites with different filler concentrations before and after EFI, respectively. The temperature interval of thermograms was chosen at 40-200°С. As can be seen from the heating thermogram, before EFI, pure PP has a single thermal effect in the range of 140–170°C, i.e., a wide endothermic maximum is observed which is characteristic for PP homopolymers (Figure 1(a), curve 1). The introduction of zirconium nanoparticles into PP and a further increase in the filler content are accompanied by a significant increase in the thermal effect (Figure 1(a), curves 3–5).
8
For pure PP the melting temperature is 150.9°C, while for the PP+7% ZrO2 nanocomposite it is 153.8°C. The energy required for the destruction of supramolecular formations (crystals) increases. It is assumed that the introduction of small additions of fillers into the polymer matrix leads to the appearance of additional nucleation centers that are located at the interface of the interphase layer of the structural elements of PP. In the process of heating, and exposure to an electric field of the nanocomposite, they contribute to a change of crystallization centers and lead to a change in the crystallization process and the formation of relatively small-spherulite structures.
9
In addition, there is a shifting of the maximum towards high temperatures. Heating thermograms of pure PP and PP + ZrO2 films with different filler concentration before (an) and after electric field:1- PP 2- PP + 10% ZrO2, 3- PP + 7% ZrO2,4- PP + 5% ZrO2, 5- PP + 3% ZrO2,6 PP + 1% ZrO2. PP: polypropylene.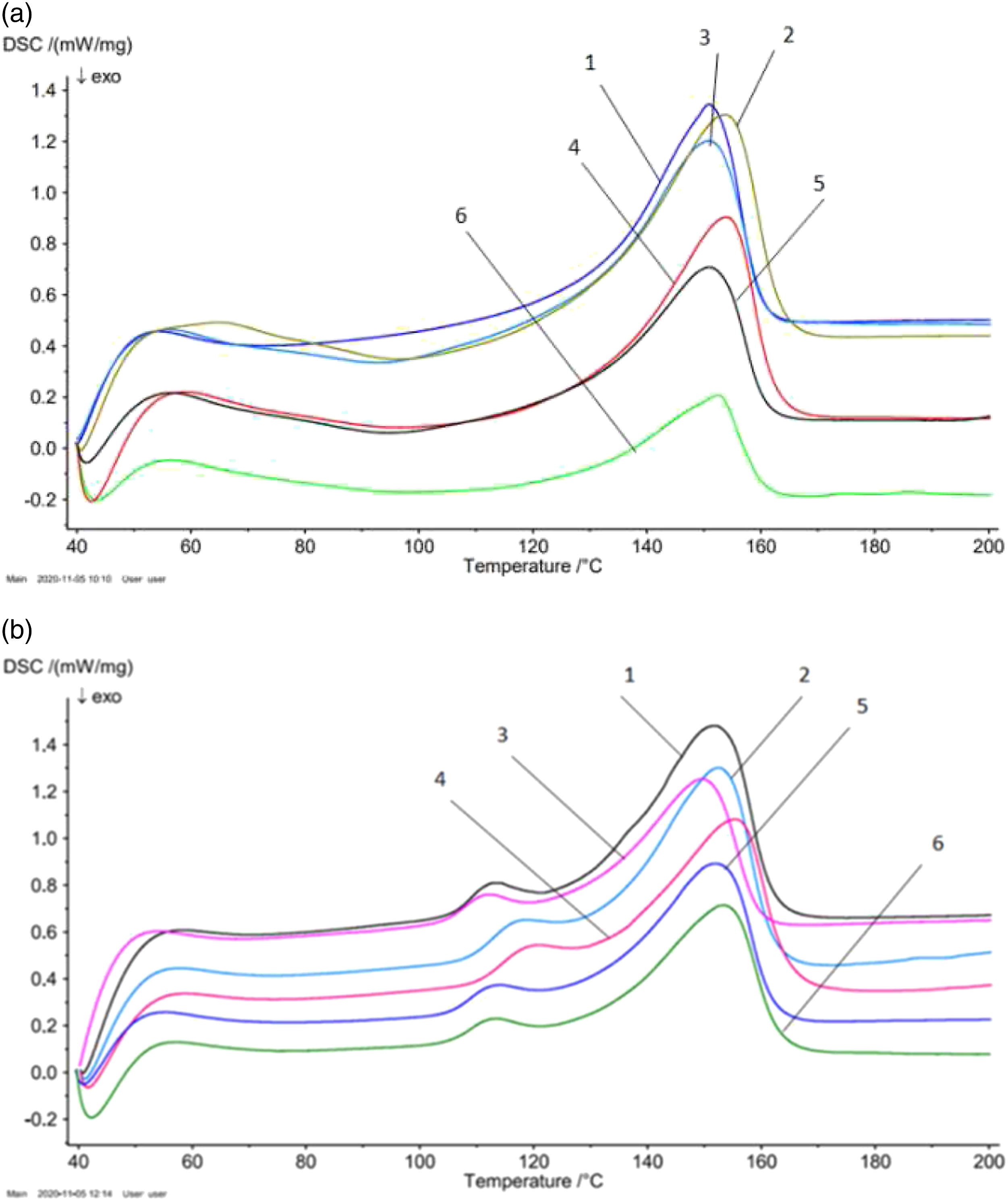
For all samples, an endothermic maximum was observed below 100°С and E = 10×106 V/m field strength. Furthermore, after EFI a new, weak maximum at the temperature range of 100–130°С of the thermograms for all concentrations of the filler was observed. It is explained by the change in the supramolecular structure of nanocomposite after EFI. It is assumed that after EFI the ratio of the crystalline and amorphous phase of nanocomposite changes. This conclusion correlates well with the XRD patterns of nanocomposites before and after EFI at a thermal treatment temperature of 100°C. By comparison of Figures 2(a) and (b), it is clear that the interlayer distance is slightly less for samples after EFI than before. In addition, the intensity of the peak at 2ϴ =18.5° increases, new peaks appear at 2ϴ = 20–23.2°. X-ray Diffraction (XRD) pattern of polypropylene + 3% ZrO2 polymer nanocomposites before (a) and after (b) electric field.
The formation of several peaks is associated with changes in the supramolecular structure of the composite. After the influence of the electric field, the intensity of the peaks changes, as well as the region of the amorphous phase. 21
An increase in the electric field strength is accompanied by a significant increase and then a decrease in the thermal effect in this region (Е = 15 × 106 V/m). Different values of the electric field strength were applied on PP + 3%ZrO2 nanocomposites to clarify the dependence of the thermophysicalparameters on the electric field strength. Figure 3 shows thermograms of the PP+3%ZrO2 nanocomposite after electric field influence with different field strength. It can be seen from the figure that thermophysical parameters of nanocomposite change after EFI even at a very small value of the electric field (E = 5 × 106 V/m). Thermograms of polypropylene + 3%ZrO2 for different values of electric field strength. Thermal treatment temperature is 100°С. 1 – E = 10 × 10−6 V/m; 2 – E = 5 × 10−6 V/m; 3 – E = 15 × 10−6 V/m
As can be seen from the experiment, the thermophysical parameters of composite materials depend on the strength of the applied external field. Furthermore, with an increase ofstrengthin the electric field to a certain value, the enthalpy of melting in the first endothermic maximum decreases, which is associated with the electric field effect. It also clear that under the electric field E = 10×10 −6 V/m, the melting temperature of the first and second endothermic maximum decreases. In our opinion, this is due to the formation of changes in the supramolecular structure at the matrix-filler interface, and this factor is decisive in the transfer of thermal energy to the filler particles through the polymer phase. It was found that the preliminary treatment of the PP + ZrO2 nanocomposite under the electric field increases the thermal conductivity of this material. Another important issue that requires serious study is the change in the thermophysical properties of a metal-polymer nanocomposite with a temperature change.
It is known that most of the polymer nanocomposites actually operate at various temperatures, but not only at room temperature. Figure 4 shows thermograms of the PP + 3%ZrO2 nanocomposite after electric thermal treatment in an electric field with an intensity of E = 107 V/m for 1 h at various temperatures. Thermograms of polypropylene + 3%ZrO2 for different values of thermal treatment temperature. The strength of applied electric field during 1 h is Е = 10∙106 V/m. 1 – T = 100°C; 2 – T = 80°C; 3 – T = 60°C
Thermal treatment of polymer nanocomposites occurred at temperatures of 60°C, 80°C, and 100°C, therefore, the thermophysical parameters were additionally determined at these temperatures
If no structural changes occur in the polymer, according to a linear law the specific heat capacity of polymers insignificantly increases with increasing temperature. 6 An increase in temperature leads to a linear increase in the melting temperature of the polymer nanocomposite at the first endothermic maximum, which is associated with the influence of an electric field. It can be explained like as the temperature rises, phase transformations - transition from the crystalline phase to the amorphous phase occur. It has been shown that with an increase in temperature, the specific heat capacity, enthalpy, and entropy of nanocomposites change both in the first and in the second endothermic maximum.
Conclusion
Effect of the electric field on thermophysical properties of the PP + ZrO2 nanocomposites has been investigated. It was clear that after the influence of the electric field another endothermic maximum appears in the temperature range 100–130°C. The appearance of this maximum is related to the change in the supramolecular structure of nanocomposite after EFI. This fact correlates with the results of X-ray studies very well. It is assumed that the introduction of small additions of fillers into the polymer matrix leads to the appearance of additional nucleation centers that are located at the interface of the interphase layer of the structural elements of PP. This contributes to the appearance of a small-spherulite structure in the polymer that is characterized by improved physical-mechanical, rheological and thermal properties of the obtained nanocomposite. Furthermore, it was investigated that an increase in temperature leads to a linear increase in the melting temperature of the polymer nanocomposite at the first endothermic maximum, which is associated with the influence of an electric field.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
