Abstract
The complex formation between allylamine and mineral acids (HCl, H2SO4, H3PO4) and their stabilities were investigated by Uv-Vis, DSC and TGA analyses. Free radical polymerization of allylamine salts was achieved by using 2,2′-azobis (2-methylpropanediamine)dihydrochloride at 50oC under N2 atmosphere in water. PAlAm synthesized in the presence of H2SO4 and H3PO4 were insoluble in water due to physical crosslinking of chains. They were converted to water soluble PAlAm.HCl by HCl treatment for further characterization. The structures of polymers obtained were elucidated by FT-IR, 1H NMR, 13C, and Cosy NMR spectroscopies. Thermal stability and glass transition behaviors were investigated by TGA and DSC. Tg was found to be 225°C for poly (AlAm.HCl) whereas 23°C for poly (AlAm). The poly (AlAm) showed very high heat stability with no weight loss until 440°C in nitrogen atmosphere. The molecular weight of PAlAm.HCl was determined by 1H NMR and viscosimetry to be 16,000 and 12,000 respectively.
Introduction
Synthetic or natural polymers carrying pendant primary amino groups on their backbones or side chains have been receiving growing attention due to versatile application routes the amine groups can provide.1–3 The ease of protonation of amine groups to form corresponding cations is just one of such applications due to their strong binding ability towards anions. Among various polyamines, poly (allylamine) and its HCl salt form has been the polymer most frequently studied due to the ease of commercial availability. Although one may think of using the simplest form of polyamines namely poly (vinylamine), due to unfavorable thermodynamics of precursor monomer vinylamine, poly (vinylamine) can not be prepared from vinylamine monomer. Only indirect methods such as chemical modification or conversion of some existing polymers have been used for the preparation of poly (vinylamine).4,5
Poly (allylamine) has been the polymer of choice in the preparation of polyelectrolyte thin films by layer-by-layer deposition technique. 1 The use of poly (allylamine hydrochloride) as a weak polyelectrolyte in the construction of multi-layer thin films has made the control of layer thickness and molecular organization of an adsorbed polymer relatively easy by simple adjustment of pH of dipping solutions.
The aim of this work is to revisit the free radical polymerization of allylamine (AlAm) with the anticipation of formulating synthesis procedures that would lead to relatively higher molecular weight products by understanding the effect of different inorganic acids on polymerization mechanism. The synthesis of PAlAm from its monomer was reported most thoroughly by Harada and Hasegava,6,7 where they subjected allylamine to free radical polymerization in a polar solvent by using a radical initiator having an azo group as well as a group having cationic nitrogen atom in its molecule. The well-known allylic degradative chain transfer has been claimed to be minimized by polymerization of HCl salt of allylamine.4–6,8,9 Recently free radical polymerization of allylamine has been achieved by using its HCl salt and thermal properties were investigated but no information was given on the molecular weight of the product. 8 Due to difficulty in the free radical polymerization of allyl amine plasma polymerization has been the method of choice especially for layered deposion of poly (allyl amine). 10 The radio-frequency-induced plasma polymerization of allylamine has been investigated in the pasma-gas phase and the mechanism by which allylamine plasma polymers formed are investigated. 11 Plasma polymerization/deposition of allylamine on various substrates provided a relatively easy way of modifying surfaces for antibody immobilization, 12 DNA adsorption/hybridization, 13 to improve cell adhesion and proliferation on UHMWPE surfaces. 14 In this work, we revisit the free radical polymerization of allylamine by a new thermal initiator and report our results on the polymerization of various mineral acid salts of allylamine in water. Thermal properties of poly (allylamine.hydrochloride) and poly (allylamine) were elaborated by TGA and DSC analyses.
Matherials and methods
Allylamine (98% purity) and the initiator 2,2′-azobis (2-methylpropanediamine) dihydrochloride were obtained from Aldrich company and used as received. The concentration of acids used in complexation with AlAm was 98% for H2SO4, 85% for H3PO4 and 37% for HCl. All acid solutions were purchased from Merck, Darmstadt Germany and used as received. The solvents used methanol, ethanol and some organic solvents were bought from the same company.
Preparation of allylamine salts
A 250 mL three necked flask equipped with over-head stirrer, thermometer and reflux condenser was charged with 12 N HCl. The allylamine precooled to −10 to 0°C, was added to the flask dropwise by continuous stirring while keeping the temperature at 5–10°C by external cooling. The resulting clear solution of AlAm.HCl was concentrated to a volume of about 70% under reduced pressure. Similar procedure was applied for the preparation of sulfuric acid and phosphoric acid complexes of AlAm. Although AlAm.HCl complex was water soluble, the other complexes (AlAm.H2SO4 and AlAm.H3PO4) were obtained as solid precipitates.
Synthesis of polymers
Polymerization of allylamine hydrochloride in water
A 250 mL four neck flask equipped with overhead stirrer, thermometer, reflux condenser and nitrogen inlet was charged with 6.68 g of 70% aqueous solution of allylamine hydrochloride. The solution was thoroughly deoxygenated by nitrogen purge and heated to 50°C. A solution of 0.08 g of 2,2′-azobis (2-methylpropanediamine) dihydrochloride in 0.35 mL of water was added to monomer solution.
The mixture was stirred for 10 h and the temperature was kept at 50°C by occasional mild cooling. Then stirring was stopped and the polymerization was continued at the same temperature for further 40 h.
The polymer was precipitated from the colorless and viscous liquid taken from the polymerization medium by adding it slowly to 500 mL of stirred ethanol. The precipitate was filtered, washed and extracted with ethanol in soxhlet apparaturs for 15 h to remove unpolymerized monomer complex, and dried in vacuum at 50°C to a white powder of poly (allylammonium chloride).
Polymerization of AlAm.H2SO4 and AlAm.H3PO4
The monomer complexes prepared in the presence of H2SO4 and H3PO4 as described in part 2.2 above were in solid state at room temperature. The monomer complexes prepared at different mole ratios were dispersed in water and a homogenous solution was obtained at the polymerization temperature of 50°C. Poly (AlAm.H2SO4) and poly (AlAm.H3PO4) were obtained by following the same procedure of poly (AlAm.HCl) synthesis as mentioned in 2.2. Since these two polymers were not soluble in water, they were dissolved in HCl solution (37%) and thus converted to their HCl salt forms as linear soluble poly (AlAm.HCl) for further characterization.
Deacidification of poly(AlAm.HCl)
2.9 N potassium hydroxide (KOH) in methanol was added to the suspension of poly (AlAm.HCl) in methanol at room temperature. The suspension was stirred for 1 h, and then left in a refrigerator overnight. The precipitate of KCl was removed by decanting the solution and deacidified polymers were dried by evaporation of water.
Characterization of polymers
Nicolet 520 model FTIR spectrometer was used for FTIR studies. The KBr discs were prepared to be used with the solid samples as well as liquid samples. Spectra were taken at 64 scan and 4 cm−1 resolution. Complexation of monomers with acids was followed by using Varian Cary100 model UV-Vis Spectrophotometer. 0.01 g polymer samples were dissolved in D2O (0.7 mL), and the 1H-NMR was taken by using a Bruker Ultra Shield 400 MHz NMR spectrometer at room temperature. The polymers were characterized by using Perkin Elmer Pyris model thermogravimetric analyzer (TGA) and Perkin Elmer Diamond model Differential Scanning Calorimeter (DSC). 5–10 mg powders were used for TGA analyses. The flow rate of nitrogen was adjusted as 20 mL/min and the variation of percent weight loss with temperature was recorded by heating from room temperature to 900oC at the rate of 20oC/min. Later the derivatives of dynamic thermograms were analysed by the programs present in the device. For DSC measurements, the samples of ∼8 mg were weighed in aluminium sample holders, crimped and heated at a rate of 20oC/min in nitrogen atmosphere.
Results and discussion
Poly (allylamine) (PAlAm) is a typical polycationic polymer having very high potential of application due to the amine groups present in its repeating units. Allylic monomers cannot be polymerized by free radical mechanism easily to high molecular weights due to the allylic structure and generally oligomers having low molecular weights are obtained due to the allylic degradative chain transfer taking place during the synthesis.6–9
In the first part of this work, various acid salts of allylamine were prepared and then the effect of these structures on polymerization was investigated.
Monomer/acid complex formation
Allylamine monomer was complexed and protonated in HCl, H2SO4 and H3PO4 solutions. The complex formation between allylamine and acid groups was investigated first by using UV-vis spectroscopy. UV-vis spectra of allylamine and free acid solutions and then those of allylamine-acid solution mixtures were taken. From band shifts or newly formed bands in UV spectra (Figure 1), it was observed that strong complexes were formed between allylamine and HCl, H2SO4 and H3PO4 groups (these were clearly seen in the 1st derivative of UV-vis spectra of each complex, Figure 2). The formation and strength of complexes have been verified by thermogravimetry as discussed in the latter part of this paper. UV-vis spectra of (a) AlAm.H2SO4, (b) AlAm, (c) AlAm.HCl, (d) AlAm.H3PO4 (10−3 M). UV-vis spectrum 1st derivatives of (a) AlAm.H3PO4 (max. 247 nm), (b) AlAm.H2SO4 (max. 215 nm and 310 nm new shoulder), (c) AlAm.HCl (max. 222 nm), (d) AlAm (max. 219 nm).

Strong complex formation between allylamine and three inorganic acids disturbs the allylic resonance structure thus facilitating the free-radical polymerization of allylamine complexes with thermal initiators.
Thermal characterization of complexes
The thermal behaviors of AlAm.H2SO4 and AlAm.H3PO4 monomer complexes which are solid at room temperature were investigated by using TGA and DSC. The DSC thermograms of AlAm.H2SO4 and AlAm.H3PO4 complexes both showed sharp single melting peaks at 44°C with ΔHm values of 7.62 J/g and 6.67 J/g, respectively. The melting enthalpies show that sulphuric acid forms a stronger complex with allylamine compared to phosphoric acid. It was also observed from the UV-vis spectra that AlAm.H2SO4 forms a stronger complex as supported by the interactions giving absorbance peak at 310 nm determined from the first derivative of the spectrum.
The AlAm.H2SO4 and AlAm.H3PO4 complexes were also investigated by TGA. AlAm.H3PO4 and AlAm.H2SO4 complexes began to decompose at about 177°C and 187°C respectively, qualitatively showing very similar degradation profiles. AlAm.H2SO4 shows slightly better thermal stability as compared to AlAm.H3PO4. The thermal degradation of AlAm.H2SO4 and AlAm.H3PO4 complexes resembles to each other with their three distinct weight loss steps observed in the derivative thermograms not shown here.
Polymerization kinetics of acid salts of allylamine
The kinetics of polymerization reaction of allylamine in water in the presence of HCl is shown in Figure 3. The conversion was determined by gravimetric measurement as the fraction of monomer polymerized as a function of reaction time for polymerization carried out at different monomer concentrations at 50°C. As expected with increasing concentration of allylamine higher conversions were reached at a given time. The maximum conversion was reached for AlAm.HCl complex with no added water as 50% but with no further increase in time. When the initial rate of polymerization (Rp) taken as the slope of initial part of conversion curves were plotted as a function of monomer concentration a linear relation was obtained as typically observed in a conventional free radical polymerization. Polymerization of AlAm.HCl in the presence of different amount of water (1 mol monomer +1 mol acid + different moles water).
The homopolymers obtained in the presence of two polyprotic acids were dissolved in HCl solution and converted to poly (AlAm.HCl). At 50oC and different reaction times in the presence of 0.8 mol of water it was observed that formation of poly (AlAm.H2SO4) was higher than the other two acid complexes. Under identical reaction conditions the reason for the higher rate of formation of poly (allylamine) from its H2SO4 complex is assumed to be due to its stronger complex structure rendering free radical polymerization less affected from allylic structure. Stronger the acid:allylamine complexation the less will be degradative chain transfer.
The homopolymers, obtained in the presence of various acids, were dissolved in HCl solution, were converted to poly (AlAm.HCl), and plotted as shown in Figure 4 At 50°C and different times, in N2 atmosphere and in the presence of 0.8 mol of water, it was observed that poly (AlAm.H2SO4)→poly (AlAm.HCl) conversion occurred at a high rate, poly (AlAm.HCl) conversion occurred at a little small rate, and poly (AlAm.H3PO4)→poly (AlAm.HCl) conversion occurred at a low rate. Due to the homogenous solution of the AlAm.H3PO4 complex was not obtained in the presence of 0.8 mol of water. The reason for high poly (AlAm.H2SO4)→ poly (AlAm.HCl) conversion was the high strength of the monomeric complex and the small amount of degradative chain transfer. Polymerization of AlAm.HCl, AlAm.H3PO4 and AlAm.H2SO4 converted to P(AlAm.HCl) (1 mol monomer +1 mol acid +0.8 mol water).
Insolubility of polyprotic acid salts of poly(allylamine) in water
Based on our observation of water solubility of poly (AlAm.HCl) and insolubility of the other two polymers, we assume the polymers develop the structures shown in Scheme 1 during polymerization in aqueous acidic medium. Linear and crosslinked structures of PAAm acid complexes.
From the solubility behavior of the three allylamine polymers synthesized in this work we conclude that HCl complex gives rise to the formation of linear polymer. When double protonic acid (H2SO4) or triple protonic acid (H3PO4) complexes of allylamine are used, polymers insoluble in water are synthesized indicating the formation of crosslinked structures. When these latter two polymers were treated with concentrated HCl they showed water solubility. This can be explained by the destruction of pseudo-crosslinks resulting from the double and triple amine complexes of sulfuric and phosphoric acid groups taking part in bridging the polymer chains together shown in Scheme 1. When these bridges of multi-dendate complexes were replaced by HCl due to its monoprotic structure the insoluble polymeric structures were converted into soluble linear poly (AlAm.HCl) chains.
In Figure 5 the FT-IR spectra of (a) Allylamine monomer (b) Poly (allylamine) (c) Poly (allylamine hydrochloride) are given. The main difference between AlAm and PAlAm is in the disappearance of C=C band at 1650 cm−1 in monomer. NH2 bending for PAlAm at 920 cm−1 is wider than that of AlAm monomer, and while NH2 bending at 1100 cm−1 for monomer AlAm is narrow and sharp, this peak is overlapped with 920 cm−1 peak for PAlAm. FTIR spectra of (a) allylamine, (b) PAlAm, (c) poly (allylamine hydrochloric acid).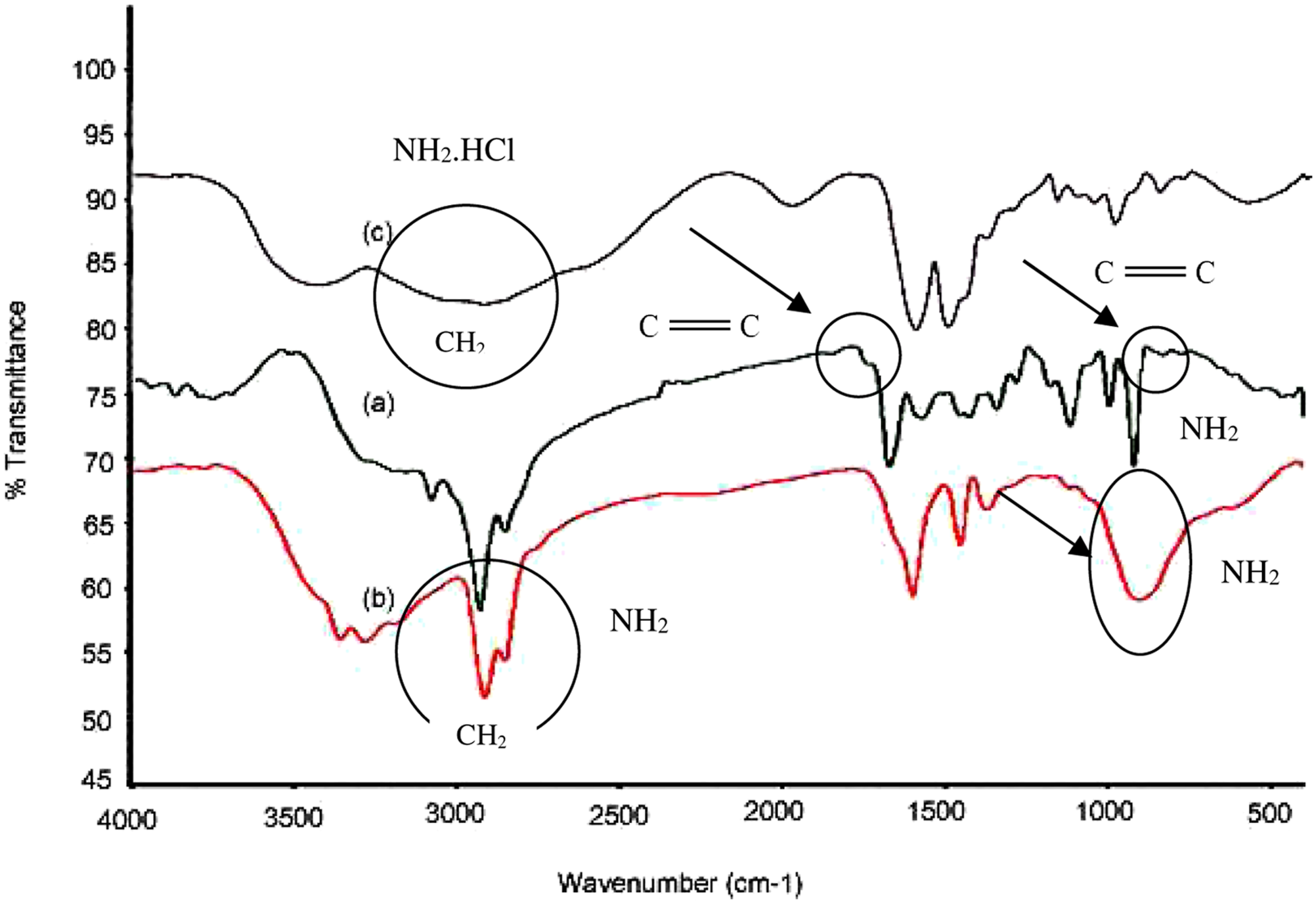
The important difference between PAlAm and PAlAm.HCl is complex formation between NH2 and HCl and it can be seen in Figure 5(c) that at 2800 cm−1and 3500 cm−1 stretching vibration of CH2 and NH2 is wider than that of PAlAm, Figure 5(b).
Characterization of homopolymer (AlAm.HCl) by NMR
For the NMR characterization of PAlAm obtained in different acidic media, corresponding 1H, 13C, 135DEPT, and Cosy-NMR spectra were taken. The characteristic spectra were given below (Figure 6 and Figure 7) 1H-NMR spectrum of Poly (AlAm) (above), 13C-NMR spectrum of poly (AlAm.HCl) (below). 1H-NMR of PAlAm.HCl (a), PAlAm.H2SO4 converted to PAlAm.HCl (b), PAlAm.H3PO4 converted to PAlAm.HCl (c).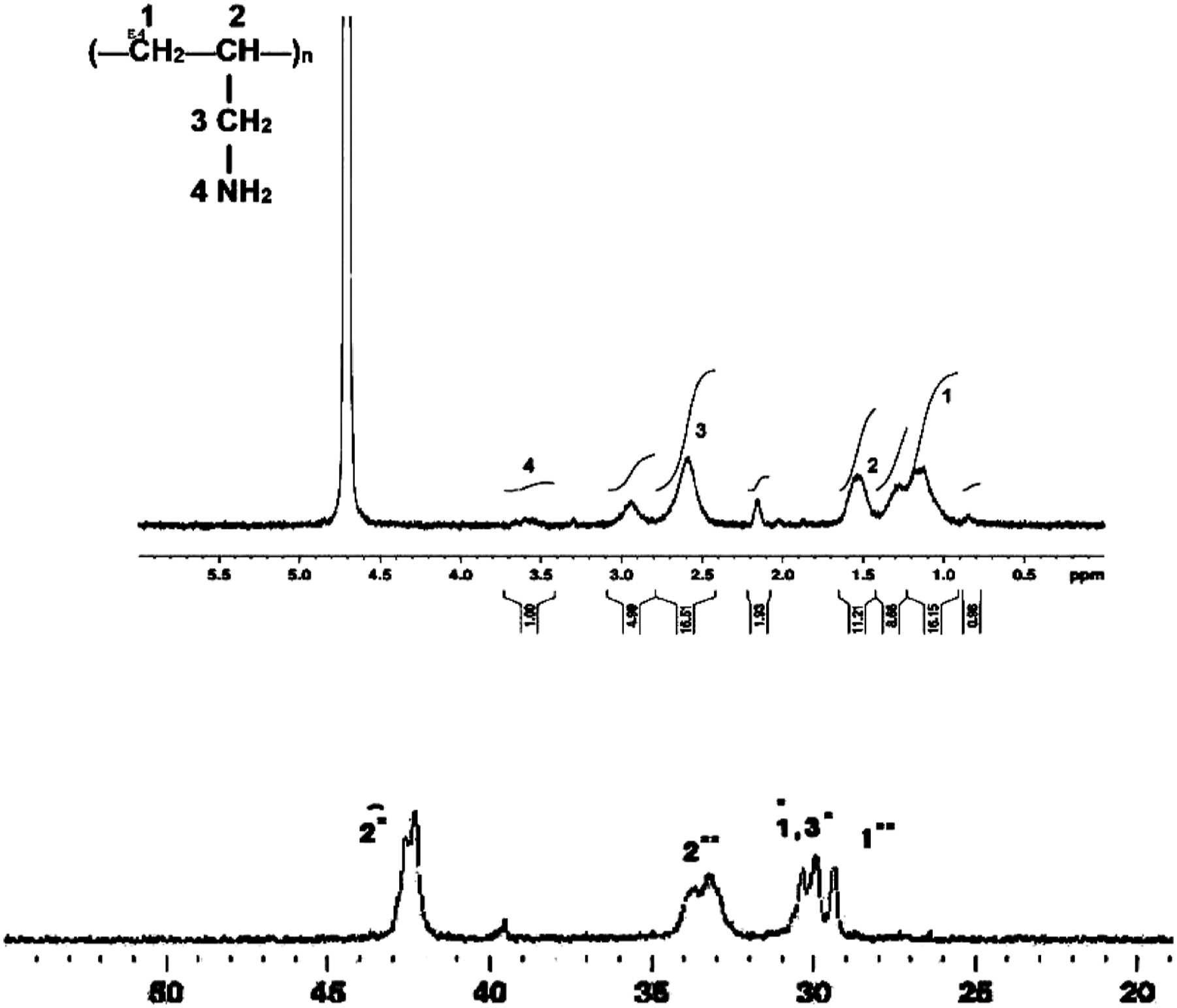

In the 1H-NMR spectrum of Poly (AlAm) in Figure 6, the CH2 peak (0.99–1.4 ppm), CH peak (1.4–1.7 ppm), CH2 peak on the side chain (2.4–2.8 ppm), NH2 peak (3.4–3.8 ppm) and CH2 peak due to the degradative chain transfer on the side chain (2.1–2.2 ppm) were shown. When the acid is removed it is observed that CH2 on the main chain shifts to 0.98 ppm, CH shifts to 1.4 ppm and CH2 on the side chain shifts to 2.4 ppm, while the free amine group shifts to 3.4 ppm. All of these observed values were indicative of removal of the acid from the poly (AlAm.HCl) complex.
In the 13C NMR spectrum of Poly (AlAm.HCl) given in Figure 4, at 29 ppm CH2 peak due to degradative chain transfer on the main chain, at 30–32 ppm CH2 peak due to normal polymerization on the main chain, at 33–34 ppm CH peak due to degradative chain transfer on the main chain, and at 43–44 ppm CH peak due to normal polymerization on the main chain can be seen.
In Figure 7 we see proton NMR of PAlAm in different three acidic media. As it is shown in Figure 7, degradative chain transfer in the presence of HCl is higher than H2SO4 and H3PO4. The reason is, complex of HCl with allylamine is more weak than complex between H2SO4 and H3PO4. We confirmed the intensity value by using Uv spectroscopy and DSC, TGA thermogravimetry.
In Figure 7 the following peaks were shown: the CH2 peak due to the degradative chain transfer on the main chain at 1 ppm, CH2 peak due to the normal polymerization on the main chain between 1.2–1.7 ppm, CH peak due to the normal polymerization between 1.8–2.2 ppm, the proton of CH2 peak due to the side chain between 2.8–3.3 ppm and CH2 peak due to the degradative chain transfer on the side chain between 3.4–3.7 ppm. (13C-NMR, 135DEPT-NMR and all of the polymers are not shown here)
In Figure 8. Cosy NMR spectrum of PAlAm the interactions of CH2 and CH on the main chain, CH and CH2 on the side chain, and NH2 and CH2 on the side chain were shown. The micro structure is found to be in head-to-tail fashion due to the dominant effect of CH and CH2 interactions on the main chain. As it is seen in the Cosy-NMR, CH2 interacts with CH and CH was in interaction with CH2 bonded to amine group. As a result it can be said that normal polymerization of allylamine to yield poly (AlAm.HCl) and the polymerization through degradative chain transfer were both taking place in the polymerization of AlAm.HCl. Cosy-NMR spectrum of PAlAm.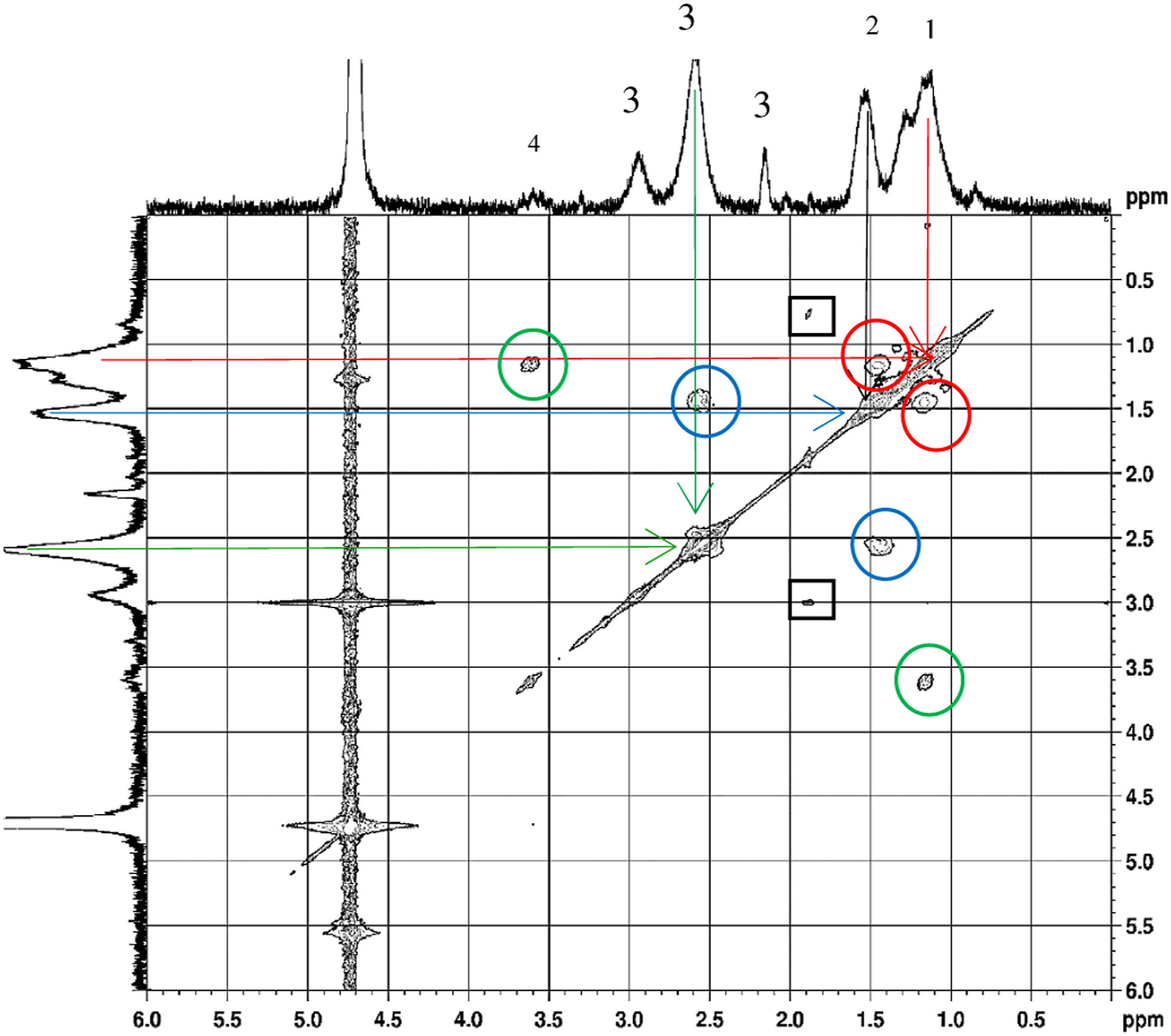
In Figure 9 the interactions of CH2 and CH on the main chain, CH and CH2 on the side chain, the degradative chain transfers due to the main and side chains, were shown. The NH2 group interacts with the CH2 group on the side chain between 7.58 ppm. The dominant effect of CH and CH2 interaction on the main chain was observed in the Cosy-NMR, which was evidence of a head-to-tail connection. In general, the behaviors of the microstructures in free radical polymerization systems were in a head-to-tail fashion. Cosy-NMR spectrum of poly (AlAm.H2SO4) converted to poly (AlAm.HCl).
For the investigation of thermal stability of PAlAm and PAlAm.HCl, TGA technique was used. As shown in Figure 10(a), poly (AlAm.HCl) thermally degrades in three distinct steps. First step starts at 300°C and finishes at around 370°C. This step involves the removal of HCl from the base polymer. Second step starts at around 380°C and finishes at 408°C which is related mainly to the loss of amine group. The percentage weight losses observed for these two stages corresponds very well to the weigth of HCl and amine groups. Third step starts at 444°C and finishes at 478°C. The polymer completely decomposes at around 480°C. Weight loss curves showing thermal behaviour of (a) poly (AlAm.HCl), (b) poly (AlAm) (red), dotted lines show the respective derivatives.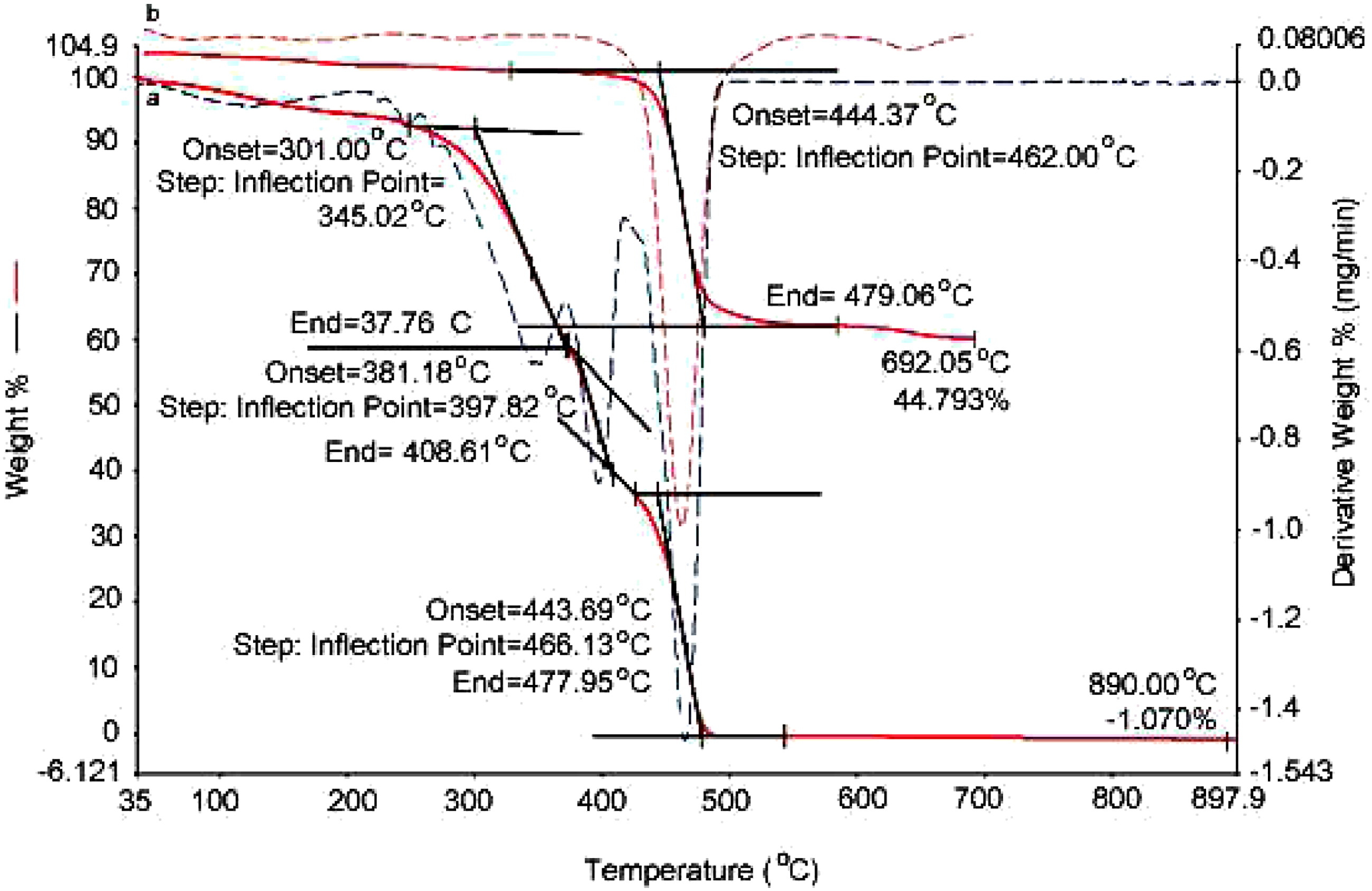
PAlAm shows very high thermal stability until around 440°C as can be seen from the thermogram in Figure 10(b). The weight loss of PAlAm starts at 444°C and finishes at 479°C and the degradation takes place in a single step. Almost 45% of this polymer remains as a thermally stable residue even until 700°C. The stability of degraded PAlAm at very high temperatures can only be explained by the formation of ring structures along the chain. Formation of cyclic hence very stable structures during thermal degradation of PAlAm under nitrogen atmosphere resembles very much the thermal behavior of poly (acrylonitrile). 15 The presence of HCl molecules attached to every amine group renders the polymer very susceptible against thermal degradation.
For poly (AlAm.HCl) and poly (AlAm) DSC method is used to find the respective glass transition temperatures (Tg). The thermograms given below show the Tg values obtained at 20ºC/min heating rates. As shown in Figure 11(a) and (b), the Tg value is about 225°C for poly (AlAm.HCl) and about 23°C for non-acidic form PAlAm, due to the polarity effect in acidic form Tg reaches to its observed very high value. Tg values of (a) poly (allylamine) (b) poly (allylamine hydrochloride) as determined by DSC.
For the determination of molecular weight of poly (AlAm.HCl) both viscosimetric and NMR methods were used. An Ubbelohde viscosimeter was used at room temperature, solvent being 0.5 M NaCl solution for poly (AlAm.HCl) and the Mark-Houwink (MH) parameters were used as a = 0.794 and K = 7.14 x 10−5. 16 When the reduced viscosity values of three PAlAms were plotted against concentration linear behaviors were observed in compliance with the Huggins equation. Both in viscosimetric and NMR methods the H2SO4 and H3PO4 insoluble salt forms of PAlAm were converted to poly (AlAm.HCl). By using the limiting viscosity numbers obtained as intercepts from the graphs mentioned above and corresponding MH equation the viscosity average molecular weights were determined to be 13,500(H2SO4), 12,000(HCl) and 11,000(H3PO4) g/mol for the three samples.
The integrated intensity of an absorption peak in the proton NMR spectrum depends only on the molar concentration of the substance times the number of nuclei per molecule responsible for the peak. The integrated intensity per nucleus per mole is the same for all substances in the sample. When a known weight of a substance (w
s
) of known molecular weight (Mw
s
) (the Standard) is added to a sample containing a known weight of a substance (w) of unknown molecular weight (Mw), the following equality holds
Therefore the molecular weight the substance (Mw) may be calculated from the above equation provided that the other parameters are known. This method can be applied only when distinct resonances are well separated from the rest of the spectrum for both unknown and the standard.
As shown in Figure 12, the 1H-NMR spectra of poly (AlAm.HCl) purchased from Aldrich company with Mw = 15,000 g/mol and poly (AlAm.HCl) synthesized in this work were taken separately. From the area integration of CH2 peak connected to amine group and other relevant information, molecular weight of poly (AlAm.HCl) was found as 16,000 g/mol by using above given equation. 1H-NMR spectrum of (a) poly (allylamine hydrochloride) with molecular weight of 15,000 obtained from Aldrich (b) poly (allylamine hydrochloride) synthesized in this work.
Conclusions
The complexes of allylamine were prepared with various mineral acids (H2SO4, H3PO4, HCl) which allowed free radical polymerization of this otherwise difficult to polymerize monomer. The polymers obtained in the presence of H2SO4 and H3PO4 were insoluble in water due to physical crosslinks formed by interchain bridges formed by polyprotic acids. They were made soluble in water by HCl treatment. PAlAm was obtained by deacidification of P(AlAm.HCl) with NaOH. Detailed structural characterization of all the polymers was made by FTIR, 1H NMR, 13C NMR, Cosy NMR spectroscopies. Thermal characterization of P(AlAm.HCl) and PAlAm was accomplished by DSC and TGA analyses. With the incorporation of HCl into the repeating units of PAlAm the glass transition temperature was found to increase from 23°C to 225°C for poly (AlAm.HCl). The molecular weight of poly (AlAm.HCl) was determined by 1H NMR and viscosimetric methods to be 16,000 and 12,000 respectively. It is shown that by proper selection of a thermal initiator and using strong acid complexes of allylamine, this monomer can be polymerized by free radiacal polymerization to relatively high molecular weights.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

