Abstract
The effect of surface roughness of electroplating chromium coated steel on bonding strength of polymer coated steel has been investigated in this paper. The experimental results have shown that the electroplating chromium coated steel with different surface roughness could be obtained through the substrate surface treatment before the electroplating process. The peeling test results have shown that the peeling force of the polymer coated steel would increase with the increase of surface roughness of the electroplating chromium coated steel, but the increase rate would become slow gradually. With the increase of surface roughness of electroplating chromium coated steel, the PET film would be damaged locally and some polymer residues would remain on the electroplating chromium coated steel; when the bonding force is large enough, the plating shedding would occur on the electroplating chromium coated steel. With the appearance of the polymer residues and the occurrence of plating shedding, the failure mode of polymer coated steel would transform from adhesion failure mode to the mixed failure mode.
Keywords
Introduction
As a new kind of plastic/metal composite material, the polymer coated steel has been widely applied in the field of can making and food packaging.1–3 The polymer coated steel is generally comprised of the polymer film as the cladding and the metal sheet as the substrate.4,5 Not only does the polymer coated steel have a high corrosion resistance and antibacterial activity from the polymer film, but it also holds a good hardness and strength from the metal sheet.6,7
The main manufacturing methods of polymer coated steel include adhesive lamination technology, hot melting coating technology, etc.8,9 Among them, the hot melting coating technology is relatively mature and effective, which is the currently main manufacturing technology for the polymer coated steel.
The polyethylene terephthalate (PET for short) film and electroplating chromium coated steel (ECCS for short) sheet (i.e. tin free steel) are commonly used as the cladding and substrate of polymer coated steel, respectively.10,11 Figure 1 shows the common structure and dimensions of polymer coated steel. The common structure and dimensions of polymer coated steel.
At present, most studies on the polymer coated steel are focus on the optimization of process and performance. Liu et al. 12 have researched the effect of lamination temperature and lamination speed on the bonding strength of polymer coated steel, the experimental results have shown that the bonding strength would increase with the increase of lamination temperature and increase with the decrease of lamination speed. Erdin et al. have investigated experimentally the deep drawing process of polymer coated steel, and they have found that the introduction of polymer could improve the surface quality and alleviate the wrinkling of the stamping part. 13 Selles et al. 14 have presented a different way to manufacture the container using a novel prelaminated two-layer polymer steel, which could reduce significantly the volatile organic compounds residues during the manufacture process. Mostofi et al. have investigated and optimized the dynamic response of polymer-coated aluminum plate under impulsive load, and the experimental results have indicated that the capacity of anti-impact of the polymer-coated aluminum plate would increase with the increase of the polymer thickness. 15 Liu et al. have researched the mechanical performance of polymer-coated sheet under different loading conditions, and they have found that applying polymer coatings to aluminum alloy sheet with weak bonding strength could improve the mechanical performance of aluminum alloy sheet. 16 Venema et al. have investigated the effect of the grain size of the steel substrate on the surface roughness during the stamping process of the polymer coated steel, the experimental results have shown that, with increase of the grain size of the steel substrate, the surface roughness would increase and the polymer thickness would decrease. 17
The main bonding mechanisms of plastic-metal composite material are the mechanical interlock and physical adsorption, which have been widely recognized by many researchers.18–20 The ECCS sheet is selected as the substrate of polymer coated steel because the layer of hydrated oxides of chromium of ECCS sheet surface could combine with the PET film through hydrogen bonding, which could improve the bonding strength between the ECCS sheet and PET film.21,22
The bonding strength between the polymer and metal has a great influence on the processing quality and service lifetime of plastic/metal composite material, which has been always the focus of relative studies.23,24 So far, the metal substrate surface treatment is a common method to improve the bonding strength between the polymer and metal.25,26 Su et al. have researched the effect of titanium surface treatments on the interface strength of Ti-C/PAEK joint, and they have found that the grit blasted titanium surface could improve the fracture toughness and bonding strength of Ti-C/PAEK joint. 27 Rodríguez-Vidal et al. 28 have produced microscopic patterns on the metal surface and then combine metal with polymer, and the experimental results have shown that the microscopic patterns could ensure the adequate mechanical interlocking at the polymer-to-metal joint interface and improve its pull-out test performance. Iqbal et al. have compared the effect of surface roughness on adhesion properties of composite bonded joint, the experimental results have shown that the joint with plasma surface treatment would have a higher shear strength than that with hand sanded surface treatment. 29 Bonpain et al. have researched the influence of the metal surface roughness on the shear strength of metal-plastic-part, and they have found that the shear strength of metal-plastic-part would increase first and then decrease with the increase of metal surface roughness. 30
Nevertheless, to our knowledge, little attention has been given to the effect of surface roughness of ECCS sheet on the bonding strength of polymer coated steel. It is because that before the manufacture of polymer coated steel, the surface treatment of ECCS sheet could not be conducted generally due to the very thin plating of ECCS sheet. 31 If the surface treatment such as sandpaper grinding is carried out directly on the ECCS sheet, the plating would fall off and lose the original function of plating. However, precisely because the plating of ECCS sheet is too thin, the plating should likely have a small effect on the surface roughness of ECCS sheet. The surface roughness of ECCS sheet could be controlled through the surface treatment of the substrate before the electroplating process. If this method could be feasible, the surface treatment of ECCS sheet could be conducted. If the change of the surface roughness of ECCS sheet has a beneficial effect on the bonding strength of polymer coated steel, the necessity, feasibility and effectiveness of the surface treatment of ECCS sheet should be reevaluated for the manufacture of polymer coated steel.
In view of this, in this paper the effect of surface roughness of ECCS sheet on the bonding strength of polymer coated steel would be researched. The ECCS sheet with different surface roughness would be prepared, and then combine the ECCS sheet with PET film through hot melting coating technology. The bonding strength between ECCS sheet and PET film would be tested through the peeling test. Moreover, the ECCS sheet surface before and after peeling test would be observed and analyzed.
Experimental
The commercial cool rolled steel sheet (Baosteel, Shanghai, China) with a thickness of 0.2 mm and the commercial PET film (HongMei, Shenzheng, China) with a thickness of 30 μm have been selected as the experiment materials. The chemical components (wt.%) of commercial cool rolled steel sheet are C 0.135, Si 0.022, Mn 0.918, P 0.015, S 0.024, Al 0.163, Cu 0.186, Ni 0.135, Cr 0.086, Mo 0.034 and Fe balance, which is characterized by the electron probe micro analyzer (EPMA-1720, Shimadzu, Kyoto, Japan). The chemical formula of commercial PET film is (C10H8O4)n, which is characterized by the Fourier transform infrared (FTIR) spectra (Nicolet 6700, Thermo Fisher Scientifie, Waltham, USA). The elastic modulus, yield strength, tensile strength and tensile strain of commercial cool rolled steel sheet are 210 GPa, 230 MPa, 320 MPa and 0.32, respectively; the elastic modulus, yield strength, tensile strength and tensile strain of PET film are 3 GPa, 110 MPa, 180 MPa and 1.5, respectively. The above tensile mechanical parameters could indicate that the commercial cool rolled steel sheet and commercial PET film would possess sufficient mechanical strength, which could be used to fabricate the polymer coated steel. The above tensile mechanical parameters are characterized by the electronic universal testing machine (UTM-1451, Jinjian, Chengde, China) according to the ASTM E345-16 standard and ASTM D882-18 standard.32,33
The fabrication process of polymer coated steel is shown in Figure 2. After being cleaned properly, the surface of commercial cool rolled steel sheet would be ground properly by sandpaper (KOVAX, Tokyo, Japan) with different average grit size (50, 80, 120, 180, 240 and 320). After being cleaned and dried properly again, the surface of commercial cool rolled steel sheet would be chromium electroplated. The electrolyte is comprised of distilled water, CrO3 (150 g/L) and H2SO4 (1.5 g/L). The electroplating current density, electroplating temperature and electroplating time are 20 A/dm3, 40°C and 10s, respectively. Before the cladding process, the PET film and ECCS sheet would be cleaned and dried properly. First, the ECCS sheet would be heated to about 250°C (the melting temperature of PET film) in a preheating furnace (B1-1200, Grieve, Round Lake, USA), then the PET film would laminated on the ECCS sheet surface through a roller mill under a proper rolling reduction rate (10%) and rolling speed (50 mm/s). After that, the polymer coated steel would be rapid water quenched at room temperature to avoid the polymer recrystallization. The fabrication process of polymer coated steel.
The bonding force between the ECCS sheet and PET film would be measured through the peeling test by using the electronic universal testing machine (UTM-1451, Jinjian, Chengde, China) with a crosshead. The peeling speed (i.e. crosshead speed) would be set as 1 mm/min. The schematic of the peeling test is shown in Figure 3. The peeling test sample would be fabricated by the wire-cut electrical discharge machining (MP1200, Mitsubishi Electric, Tokyo, Japan) under proper cooling and lubrication conditions. The length and width of peeling test sample would be 100 mm and 25 mm, respectively. The peeling test of each kind of polymer coated steel would be repeated for 5 times in order to guarantee the accuracy and repeatability of experimental results. The surface topography of ECCS sheet before and after the peeling test would be characterized by the scanning electron microscopy (Sigma-300, Zeiss, Oberkochen, Germany) equipped with energy dispersive X-ray spectroscopy (EDS). Moreover, the surface average roughness (Ra value) of the ECCS sheet before and after the peeling test would be characterized by the 3D surface profilometer (Npflex, Bruker, Billerica, USA). The Ra value of each case would be measured for 5 times, then average the measurement results. The schematic of the peeling test.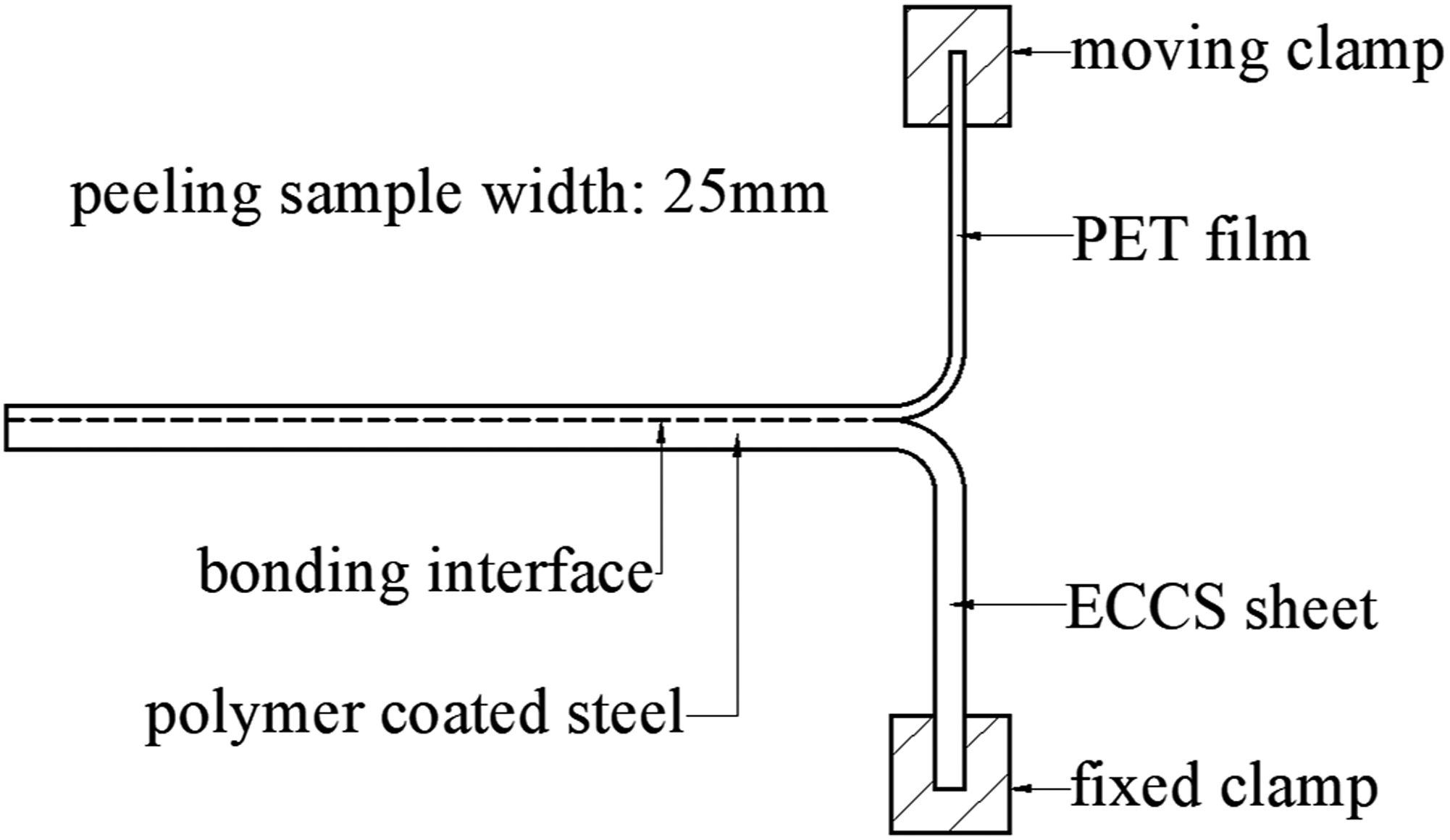
Results and discussion
Figure 4 shows the SEM image and element distribution map of ECCS sheet surface after different substrate surface grinding treatment. It could be seen that there are not obvious plating defects such as pits or voids on the ECCS sheet surface. Moreover, the element distribution maps could indicate that the ECCS sheet surface is mainly composed of Cr element and there is no Fe element on the surface basically. The above characterization results could indicate that the plating of ECCS sheet could be uniform and dense without obvious defects even after different substrate surface grinding treatment. Table 1 shows the surface roughness (Ra value) of the ECCS sheet after different substrate surface treatment. As shown in Table 1, the Ra value decreases from 3.14 μm to 0.42 μm with the increase of sandpaper grit size from 50 to 320. The above experimental results could prove that, without the plating defects, through the substrate surface treatment the control of surface roughness of ECCS sheet is feasible. The SEM image and element distribution map of ECCS sheet with different substrate surface grinding treatment: (a) sandpaper grit size 50, (b) sandpaper grit size 80, (c) sandpaper grit size 120, (d) sandpaper grit size 180, (e) sandpaper grit size 240, and (f) sandpaper grit size 320. The surface average roughness (Ra value) of the ECCS sheet with different substrate surface treatment.

Figure 5 shows the peeling force versus displacement curve of the ECCS sheet with different surface roughness. As shown in Figure 5, the changing trends of peeling force versus displacement curve are very similar. At first the peeling force would increase with the displacement increase until reach the peak, then the peeling force would decrease with the displacement increase, at last the peeling force would fluctuate between a narrow range. According to the previous research results, the average value of peeling force among the plateau region in Figure 5 could be taken as the effective peeling force of the peeling test, which could evaluate the bonding strength between the PET film and ECCS sheet.34,35 The peeling force versus displacement curve of the ECCS sheet with different surface roughness.
Figure 6 shows the effective peeling force versus the surface roughness (Ra value) of ECCS sheet. The effective peeling force would increase from 4.9 N to 11.5 N with the Ra value increase from 0.42 μm to 3.14 μm. However, the increase rate of the effective peeling force would slow down significantly with the Ra value increase. When the Ra value is relatively small, the effective peeling force would increase obviously. When the Ra value are 0.42 μm, 0.98 μm and 1.51 μm, respectively, the effective peeling force are 4.7 N, 7.6 N and 9.3 N, respectively. When the Ra value is relatively large, the effective peeling force would not increase obviously any more. When the Ra value are 2.12 μm, 2.66 μm and 3.14 μm, respectively, the effective peeling force are 10.7 N, 11.2 N and 11.5 N, respectively. The above experimental results could indicate that the bonding strength between the PET film and ECCS sheet would increase with the increase of the surface roughness (Ra value) of ECCS sheet. The effective peeling force versus the surface roughness (Ra value).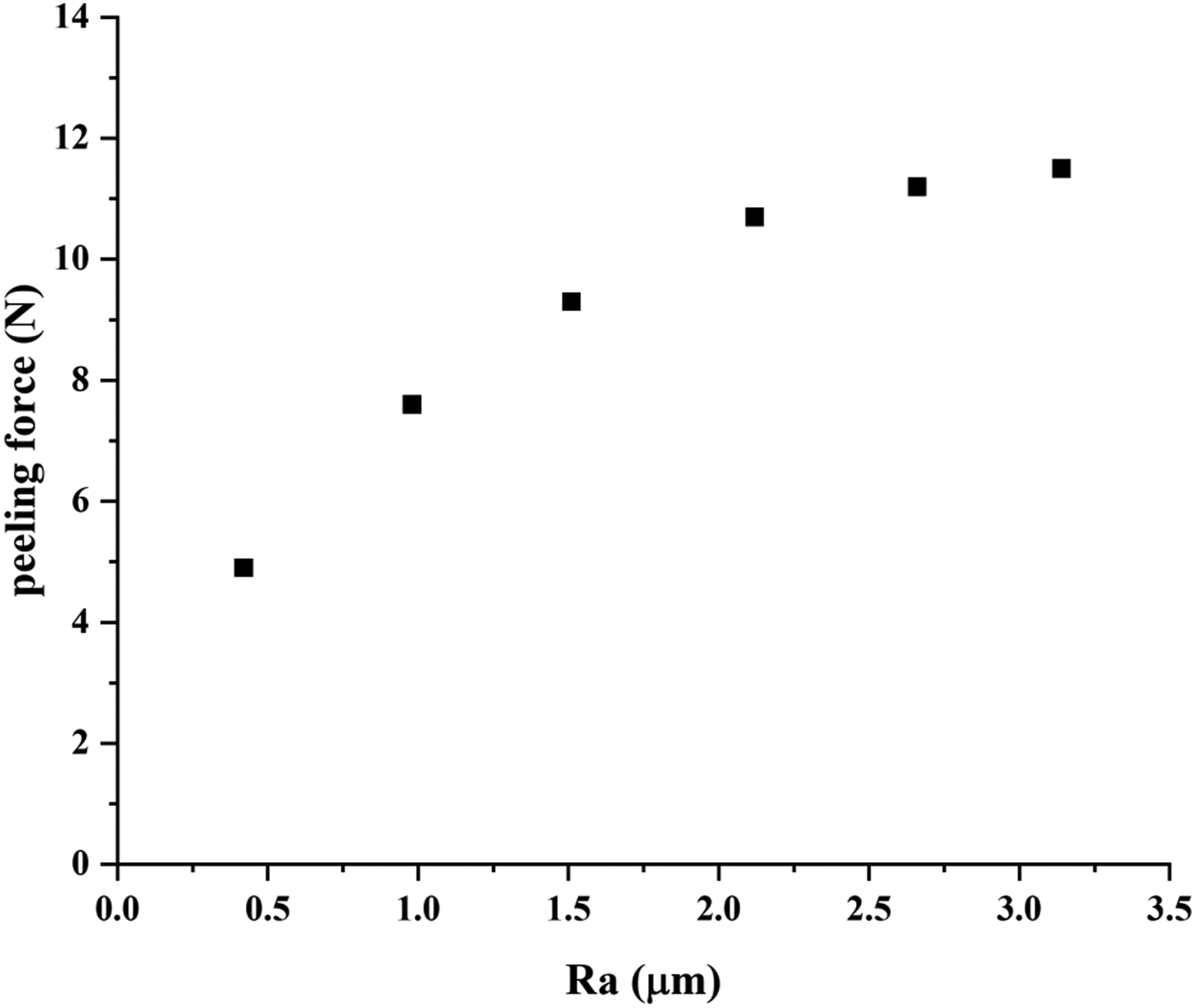
Figure 7 shows the SEM image and element distribution map of ECCS sheet surface after peeling test with different surface roughness. As shown in Figure 7(a) and (b), there are some polymer residues (i.e. the PET film) on the ECCS sheet surface, which could be also identified through the C and Cr element distribution map. Moreover, there are also some plating shedding regions on the ECCS sheet surface, which could be also identified through the Fe and Cr element distribution map. As shown in Figure 7(c) and (d), there are still some polymer residues on the ECCS sheet surface, but there are not any plating shedding regions on the ECCS sheet surface. As shown in Figure 7(e) and (f), both the polymer residues and plating shedding regions could not be found on the ECCS sheet surface. The SEM image and element distribution map (Cr and Fe) of ECCS sheet after peeling test with different surface roughness (Ra value): (a) 3.14 μm, (b) 2.66 μm, (c) 2.12 μm, (d) 1.51 μm, (e) 0.98 μm, and (f) 0.42 μm.
According to the previous research results, there are three mainly failure model of the plastic/metal composite material: adhesion failure mode, cohesive failure mode and mixed failure mode (the co-exist of the first two).36,37 The adhesion failure mode is referred to the failure occurred on the interface between the polymer (cladding) and metal (substrate) and there are not any polymer residues on the metal surface, at this time the bonding strength between the polymer and metal is smaller than that of the polymer itself; the cohesive failure mode is referred to the failure occurred in polymer and there is still a basically complete polymer cladding on the metal surface, at this time the bonding strength between the polymer and metal is larger than that of the polymer itself; the mixed failure mode is referred to both adhesion failure and cohesive failure occurred simultaneously and there are some polymer residues on the metal surface, at this time the bonding strength between the polymer and metal is between that of the first two cases.
The occurrence of the above failure mode is related to the material selection (cladding and substrate) and process parameters. In this paper, when other conditions remain unchanged, the failure model of polymer coated steel should be affected by the surface roughness of ECCS sheet.
As shown in Figure 8, when the surface roughness of ECCS sheet is relatively small, there is no polymer material on the ECCS sheet surface, and the failure mode of polymer coated steel is the adhesion failure mode. With the increase of surface roughness of ECCS sheet, some polymer residues would remain on the ECCS sheet surface, and the failure mode of polymer coated steel is the mixed failure mode. Moreover, when the surface roughness of ECCS sheet is large enough, the plating in some regions on the ECCS sheet surface would fall off and meanwhile there are still some polymer residues on the ECCS sheet surface, which could be a special failure mode of the polymer coated steel. Nevertheless, as common plating defect, the plating shedding could been considered as a kind of adhesion failure by many researchers.38,39 In view of this, this kind of failure mode of polymer coated steel could still be considered as the mixed failure mode. The interface failure model of polymer coated steel.
As previous introduced, the bonding mechanism of polymer coated steel are the mechanical interlock and physical adsorption. Some research results have indicated that, with the increase of metal surface roughness, the mechanical interlock performance and physical adsorption activity would increase, resulting in the increase of bonding strength between polymer and metal.40–42 Moreover, The relatively large substrate surface roughness would cause the interface defects and interface stress concentration easily, weakening the bonding strength in some regions between plating and substrate.43,44 When the relatively large peeling force is applied on the polymer coated steel, the plating in some regions with poor bonding strength would fall off from the substrate (just as shown in Figure 7(a) and (b)).
Conclusions
In this paper, through the surface treatment before the electroplating process, the electroplating chromium coated steel (the substrate of polymer coated steel) with different surface roughness could be produced. The peeling test results indicate that, with the increase of surface roughness of electroplating chromium coated steel, the bonding force between electroplating chromium coated steel and PET film would increase but the increase rate would become slow gradually. The results of SEM and EDS analysis show that, during the peeling test, with the surface roughness of electroplating chromium coated steel increase, the PET film would be damaged locally and some polymer residues would remain on the electroplating chromium coated steel; when the bonding force is large enough, the coating of the electroplating chromium coated steel would fall off locally. The failure mode of polymer coated steel would gradually transform from the adhesion failure mode to the mixed failure mode (the co-exist of adhesion failure mode and cohesive failure mode) with the surface roughness of electroplating chromium coated steel increase.
Footnotes
Declaration of competing interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Startup Research Fund of Zhengzhou University (No. 32212492).
