Abstract
This study presents functionalized carbon nanotubes (CNTs) decorated with functionalized barium titanate (BT) (denoted as BT@CNTs) via hydrothermal and self-assembly methods to improve dispersion in polypropylene (PP) matrix with enhanced thermal, mechanical, and thermomechanical properties. The CNTs, BT, and BT@CNTs with the addition of polypropylene maleic anhydride (PPMA) compatibilizer were used in the fabrication of the PP based nanocomposites for this study via solution mixing and melt compounding. FTIR, TEM, and SEM analyses, respectively revealed that the nanoparticles were successfully functionalized, CNTs decorated with BT, and well dispersion in the PP matrix. PP/BT@CNTs-based nanocomposite showed optimal tensile strength, modulus, heat deflection, and thermal stability of about 16.8%, 26.5%, 20.1%, and 30oC higher than that obtained for the pure PP. These properties were also respectively 10.6%, 17.6%, 19.0%, and 5.0oC higher than PP/3CNTs nanocomposite when compared to PP/3BT@CNTs. The PP/BT@CNTs based nanocomposites also revealed enhanced dynamic mechanical properties. The better performance in the measured properties of PP/BT@CNTs compared to pure PP and PP/CNTs were attributed to their uniform microstructures, effective interlocking of the PP matrix due to the presence of BT on CNTs surfaces.
Introduction
Recently, research attention has been drawn to carbon nanotubes (CNTs) due to their high thermal, electrical, mechanical, structure properties, etc. The size and nature of CNTs enhance transfer of load from the matrix to the CNTs and bridging of thermal barriers with improvement in mechanical, thermal, and other properties for various engineering applications. It is believed that the high surface area of CNTs provides good adhesion with the polymer matrix in which it is incorporated. 1 However, CNTs often show significant agglomeration in polymer matrix due to the force of attraction between individual CNTs and the large difference in surface tension when compared with polymer matrices. Good dispersion of CNTs and adhesion with the polymer matrix in which it is introduced are very vital for high performance of such nanocomposites. 2 Agglomeration of CNTs in the polymer matrix serves as localized stress concentration for microcracks initiation and propagation3,4 and reduces effective load transfer from the matrix to CNTs. 5 Therefore, various studies have tried to improve the dispersion and adhesion of CNTs with polymer matrix via surface functionalization with enhancement in various properties.6,7
One of the commonly used polymers for various engineering applications is polypropylene (PP) due to its distinct properties such as industrial suitability, thermal stability, mouldability into various shapes, colourability, and resistance to crack initiation.8,9 However, PP faces some challenges which limit its wide application such as low mechanical strength and low crack propagation once a crack is initiated, leading to early mechanical failure. 9 The low crack propagation resistance of PP on the application of load and its low working temperature9,10 also result in low thermomechanical properties, which limits its uses where simultaneous application of load and heat are required. However, incorporation of reinforcing particles and/or fibres in polymer matrix has shown good potential in restricting the mobility of their molecular chains on the application of load and/or heat with enhancement in mechanical, thermal, thermomechanical properties, etc.11,12 On that note, various degrees of improvement in tensile strength, flexural strength, modulus, impact strength, thermal, and thermomechanical properties of PP matrix using CNTs have been reported.2,5,13
However, the endless need for further improvement in the dispersion and adhesion of CNTs with PP matrix to significantly enhance mechanical, thermal, and thermomechanical properties of PP for various advanced engineering applications formed the focus of this study. These were achieved by decorating the surfaces of CNTs with barium titanate (BT) via hydrothermal and self-assembly techniques. The decoration of CNTs with BT (BT@CNTs) produced CNTs with dot-like surfaces, which promoted mechanical interlocking of the PP chains. This also promoted adhesion of the BT@CNTs with the matrix, which resulted in dense microstructure and uniform morphology since BT with small size and spherical shape could close potential micro-voids in the polymer nanocomposites and prevent wall to wall interactions of the CNTs in the PP matrix. The PP/BT@CNTs nanocomposites showed better thermal, mechanical, and thermomechanical properties compared to pure PP and their PP/CNTs counterparts. This was basically due to the obtained uniform microstructure, efficient load transfer from the matrix to the BT@CNTs, interlocking, and reduction in the mobility of the PP chains by the presence of the BT@CNTs on the application of load/heat. The developed nanocomposites can find advanced applications where high thermal, mechanical and thermomechanical properties are required.
Materials and Method
Materials
Polypropylene grafted maleic anhydride (PPMA) (0.5%, density – 0.92 g/mL), barium titanate (BT) (assay >99%, particles size <100 nm, density 6.08 g/mL at 25°C), xylene plus ethylbenzene basis (ACS reagent, essay >98.5%), dopamine hydrochloride (PDA) (essay >98%, MW - 189.64, BP - 248–250oC), 3-glycidoxypropyltrimethoxysilane (GPTMS) (BP - 120oC/2 mmHg) and polypropylene (PP) (melt index – 4 g/10min, 230oC/2.16 kg, density - 0.9 g/mL) were all purchased from Sigma-Aldrich, South Africa. Hongwu International Group, China supplied multi walled carbon nanotubes (CNTs) (purity – >98%, diameter – 10–30 nm, length – 5–20 μm).
Nanoparticles Functionalization and Nanocomposites Fabrication
The surface functionalization of the CNTs and BT were respectively done using PDA and GPTMS. 14 The nanoparticles were dispersed and ultrasonicated at 80oC for 4 h in different beakers containing distilled water (for CNTs) and xylene (for BT). The PDA was introduced in the suspension of CNTs with dropwise addition of ammonia solution under ultrasonication. On the other hand, GPTMS was introduced into the suspension of BT under ultrasonication. Both resultant nanoparticles were washed several times after 10 h under ultrasonication and reaction to obtain functionalized CNTs and BT. To decorate the surfaces of CNTs with BT, the nanoparticles were then mixed and dispersed in distilled water at a ratio of 1 wt% to 5 wt% respectively and kept under ultrasonication for 4 h at 80oC. The mixture was later sealed with aluminium foil and kept in an oven at 140oC for 10 h to obtain BT decorated CNTs (BT@CNTs). The nanocomposites were fabricated by solution mixing of the PPMA (dissolved in xylene at 140oC) and the nanoparticles under continuous stirring for 1 h to produce masterbatches of different concentrations of the nanoparticles. The PPMA was maintained at 8 wt% for each concentration of the nanoparticles. The mixture was transferred to a clean ceramic plate and dried in an oven at 80oC. The use of the PPMA was to enhance compatibility between the matrix and the reinforcing nanoparticles. The prepared PPMA/nanoparticles masterbatch with pure PP were used in the preparation of the nanocomposites via melt compounding using HAAK rheomixer 600 OS at a temperature of 190oC, screw rotating speed of 100 r/min, and mixing time of 10 min. The nanocomposites were later granulated and pressed using a Carver presser at 200oC and 10 MPa for 10 min. Nanocomposites developed for this study include PP/1CNTs (1 wt%), PP/3CNTs (3 wt%), PP/15BT (15 wt%), PP/1BT@CNTs (1 wt%) and PP/3BT@CNTs (3 wt%).
Characterization and Properties measurement
To determine the successful functionalization of the nanoparticles, Fourier transfer infrared (FTIR) (Perkin Elmer Precise Spectrum 100) was used. The X-ray diffraction analysis was carried out on the nanocomposites using an X-ray diffractometer (XRD) (X’pert PRO PANalytical) with Cu Kα radiation and a scanning speed of 5o/min. A transmission electron microscope (TEM) (JEM-2100) was used to examine the structure and dimensions of the nanoparticles and decoration of CNTs with BT. Morphological and microstructural study of the nanocomposites was carried out using a scanning electron microscope (SEM) (VEGA three TESCAN). A thermogravimetry analyzer (TGA) (TA instrument Q500) was used to determine the thermal stability of the nanocomposites in an inert atmosphere using nitrogen gas. The tensile properties of the nanocomposites were obtained using a universal testing machine (LLOYD EZ50) at a crosshead of 5 mm/min. An average of five tests per sample are presented in the study in accordance with ASTM D638 (Type V) standard. The heat deflection temperature (HDT) and Vicat softening temperature (VST) of the nanocomposites were obtained using HDT/Vicat tester (CEAST HV6) by applying a load of 0.45 MPa and a heating rate of 2oC/min in accordance with ASTM D648 standard. The HDT was recorded at the temperature where the nanocomposites deflected by 0.25 mm. On the other hand, the VST was recorded when a flat-ended needle of 1 mm2 circular cross-section penetrated the samples by 1 mm under an applied load of 10 N in accordance with ASTM D1525 standard. An average of three tests per sample are presented in this study. A dynamic mechanical analyser (DMA) (DMA 8000, PerkinElmer) was used in the determination of the dynamic mechanical properties of the nanocomposites in accordance with ASTM D-4065 standard. This was carried out by increasing temperature from −50 to 50oC at a heating rate of 2oC/min and vibration frequency of 1 Hz.
Results and Discussion
FTIR and XRD Analyses
Figure 1 presents the FTIR analysis of the nanoparticles. It shows that the nanoparticles were successfully functionalized. Typical functional groups corresponding to different vibration bands of CNTs were detected at various wavenumbers as indicated in Figure 1(a), which include C=O, C-H, C=C, and O-H. However, the functionalized CNTs (f-CNTs) revealed an additional peak at 1048.50 cm−1, corresponding to the vibration band of C-O functional group. On the other hand, Figure 1(b) shows typical FTIR spectra of the BT and their corresponding vibration bands as labelled in the figure. For both pristine BT and functionalized BT (f-BT), at 459.2 cm−1 and 653.8 cm−1, bending and stretching vibrations corresponding to Ti-O were respectively detected.15,16 A stretching vibration of CO32- corresponding to 1533.6 cm−1 was also noted.15,17 However, new vibration bands were observed (as shown in Figure 1(b)), which include Si-O-Si and Si-O-BTparticles as a result of GPTMs used in the functionalization process,17,18 and H-O-H due to absorbed water by the BT.17,19 The new vibration peaks corresponding to different functional groups observed for the functionalized nanoparticles compared to their pristine counterparts, confirmed that the nanoparticles were successfully functionalized. X-Ray diffraction patterns of the nanocomposites are presented in Figure 2. Typical diffraction peaks at various 2θ are labelled with their corresponding crystal planes in figure.20,21 Notably, the addition of the nanoparticles in the PP matrix decreased the broad diffraction peaks of pure PP at 2θ = 27.3o and 41.7o, which a indicates reduction in amorphous regions of the nanocomposites with possible increase in the crystalline domains. This could have also contributed to the improved thermal, mechanical, and thermomechanical properties of the nanocomposites in this study. The nanocomposites also showed various diffraction peaks at different 2θ, corresponding to the two nanoparticles used in this study as labelled in Figure 2, which are not present for the pure PP. FTIR spectra of (a) CNTs, f-CNTs and (b) BT, f-BT nanoparticles. XRD patterns of the nanocomposites.

Microstructural Analyses
The structure and dimensions of the nanoparticles used in this study were revealed by the TEM micrographs as presented in Figure 3. The CNTs show entangled one-dimensional structure with relatively smooth surfaces as shown in Figure 3(a). While the BT@CNTs shown in Figure 3(b) reveals rough and dot-like surfaces due to the decoration of the CNTs with the BT. The dot-like surfaces of BT@CNTs aided in the effective interlocking of the polymer chains, dispersion, and uniform microstructure of the nanocomposites with improved properties. Morphological structures of the developed nanocomposites are presented in Figure 4 as investigated using SEM. TEM micrographs of (a) CNTs and (b) BT@CNTs nanoparticles. SEM micrographs of (a) pure PP (b) PP/15BT (c) PP/1CNTs (d) PP/3CNTs (e) PP/1BT@CNTs and (f) PP/3BT@CNTs nanocomposites.

The pure PP revealed smooth microstructure as shown in Figure 4(a). The addition of the nanoparticles in the matrix changed the microstructure of the PP matrix. The nanocomposites containing 15 wt% BT showed uniform and dense Microstructure as presented in Figure 4(b). Microstructures of PP/1CNTs and PP/3CNTs respectively shown in Figure 4(c) and Figure 4(d) revealed the presence of the CNTs in the PP matrix but were not dense as that of PP/15BT. PP/3CNTs nanocomposite showed clustering of CNTs in the matrix due to a high concentration of the CNTs since the clustering was not noticeable for PP/1CNTs nanocomposite. The wall to wall interaction of the CNTs in the PP matrix was reduced by decorating the CNTs with the BT. Hence, PP/1BT@CNTs (Figure 4(e)) and PP/3BT@CNTs (Figure 4(f)) showed uniform microstructure with no significant agglomeration of nanoparticles in the matrix. The presence of BT on the surfaces of CNTs aided in filling micro-voids in the nanocomposites, thereby making them denser compared to PP/CNTs based nanocomposites. The uniform and dense microstructures of PP/BT@CNTs based nanocomposites, which indicate that the PP matrix was more efficiently locked compared to PP/CNTs based nanocomposites contributed to their enhanced properties in this study. This was also contributed by the surface functionalization of the nanoparticles and the use of PPMA during nanocomposites preparation, which promoted dispersion and compatibility of the matrix with the nanoparticles.
TGA Analysis
The nanocomposites showed higher thermal stability compared to the pure PP as presented in Figure 5. This is due to the high thermal stability of the incorporated nanoparticles, which increased the thermal barrier of the PP matrix and enhanced its decomposition temperature.
22
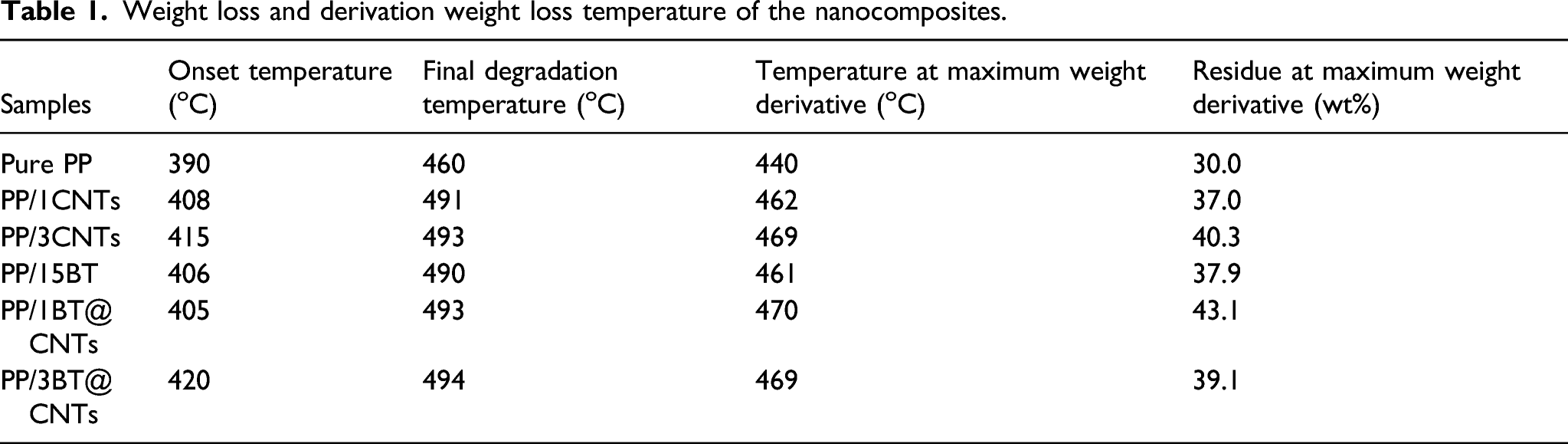
The onset of decomposition temperature of all the nanocomposites shifted to the higher temperature as depicted in Figure 5(a) and summarized in Table 1. The onset of decomposition and final degradation temperature of the pure PP were recorded as 390oC and 460oC, respectively. Which increased to about 420oC and 494oC for PP/3BT@CNTs and 415oC and 493oC for PP/3CNTs respectively. Therefore, optimal increase in the onset and final decomposition of about 30oC and 34oC can be deduced for PP/3BT@CNTs nanocomposite when matched with the pure PP. This confirms that higher thermal energy was required to melt, causing the PP chains to flow and finally decompose for the nanocomposites compared to that required for the pure PP. This was contributed by the good synergy and relationships among the nanoparticles and the PP matrix.
22
The pure PP also showed lower temperature and residues at maximum weight loss compared to all the nanocomposites as represented in Figure 5(b). This was more pronounced for PP/BT@CNTs nanocomposites, which can be credited to the high thermal energy required to decompose the bond between the BT@CNTs and the PP Matrix. TGA curves of (a) Weight loss and (b) derivative weight loss of the nanocomposites. Weight loss and derivation weight loss temperature of the nanocomposites.
Tensile and Young Modulus
The tensile strength increased from 33.6 MPa for the pure PP to an optimal of about 39.2 MPa for PP/1BT@CNTs as shown in Figure 6(a), which is about 16.8% increment. The increased tensile strength can be attributed to the attachment of the BT on the CNTs surfaces, which gave them dot-like surfaces (TEM micrographs in Figure 3(b)) and promoted interlocking and restriction of the PP chains against mobility on the application of load. However, increase in the BT@CNTs concentration to 3 wt% reduced the tensile strength to about 34.3 MPa, but was still about 10.6% higher than that of PP/3CNTs. The reduction can be ascribed to the formation of nanoparticles aggregates in the PP matrix as its content increased, which resulted in more nanoparticles interactions than nanoparticles to matrix interactions since CNTs have a high aspect ratio and are capable of nanotubes entanglement. This often leads to poor load transfer from the matrix to the nanoparticles due to reduction in adhesion between the nanocomposites’ constituents. The percentage elongation of all the nanocomposites decreased compared to the pure PP. The introduction of hard nanoparticles in the PP matrix often causes the resultant nanocomposites to be stiff with a reduction in percentage elongation.23,24 This was confirmed by the higher Young Modulus of the nanocomposites compared to the pure PP as shown in Figure 6(b). This was more pronounced for PP/3BT@CNTs with a Young Modulus of about 1558.3 MPa, which is about 26.5% (1232 MPa) and 17.6% (1325.2 MPa) higher than pure PP and PP/3CNTs respectively. Efficient load transmission, good dispersion, and filling of micro-voids contributed to the enhanced Young Modulus. (a) Tensile strength and (b) tensile modulus of the nanocomposites.
Heat Deflection and Vicat Softening Temperature
The nanocomposites showed significant improvement in the heat deflection temperature (HDT) when compared to the pure PP as represented in Figure 7(a). The formation of network structures in the PP matrix through the interpenetration of the nanoparticles supported the reason for the enhanced HDT. Optimal HDT was obtained for PP/3BT@CNTs nanocomposite, which is about 98.1oC versus 81.7oC measured for the pure PP. This is about 20.1% increase compared to the pure PP, while it is about 19.0% increase when matched with PP/3CNTs nanocomposite that has HDT of about 82.5oC. Also, the Vicat softening temperature (VST) of the nanocomposites is presented in Figure 7(b), which is also enhanced. This implies that it took a higher temperature for the nanocomposites to be softened and allowed the penetration of the flat-ended needle. Hence, the nanoparticles in the matrix resisted the movement of the flat-ended needle into the nanocomposites. It can also be mentioned that the formation of network configurations by the CNTs in the matrix promoted quick heat dissipation,
25
which improved the nanocomposites’ softening temperature and resistance to deformation. The VST increased from about 148.7oC for the pure PP to approximately 155.7oC for PP/3CNTs and 156.9oC for PP/3BT@CNTs. In general, good interfacial adhesion between the nanocomposites’ constituents and well-dispersed nanoparticles in the PP matrix are also contributing factors towards the enhanced HDT and VST. (a) Heat deflection and (b) vicat softening temperature of the nanocomposites.
Dynamic Mechanical Properties
Figure 8(a) shows reduction in storage modulus (Gl) of the nanocomposites with increasing temperature due to the increase in mobility of the PP chains on the application of heat.
26
However, the Gl of the nanocomposites was higher compared to that of the pure PP. This shows that the movement of PP chains was controlled and resisted by the presence of the nanoparticles in the matrix. Although carbon-based percolative nanoparticles such as CNTs can improve the Gl of a polymer matrix,27,28 it can be more enhanced when hybridized with ceramic nanoparticles as noted in the study. The BT nanoparticles on the surfaces of the CNTs acted as intermediaries for effective load transmission from the matrix to CNTs. In addition, it assisted in the mechanical interlocking of the polymer chains, which causes the restricted movement of the PP chains with enhanced Gl.
29
Since the BT assisted in the uniform dispersion of the CNTs in conjunction with their good adhesion with the PP matrix, it was possible to record increase in Gl. The dense network configurations of the nanocomposites (as revealed by SEM micrographs in Figure 4) through the interpenetration of the CNTs and BT nanoparticles (as revealed by TEM micrograph in Figure 3(b)) resulted in the improved Gl.
30
Therefore, the Gl of the PP/BT@CNT group was higher than that of the PP/CNTs nanocomposites. Also, all the nanocomposites showed higher loss modulus (Gll) compared to the pure PP as represented in Figure 8(b). This is because of the internal friction between the nanoparticles and the matrix interfaces during the application of periodic stress and increasing temperature. The interfacial friction generates heat, which is dissipated in the form of energy and causes high Gll.
31
Therefore, the addition of foreign bodies into a polymer matrix often causes high Gll due to sliding movement at the interfaces of the composite’s constituents.30,32 Nanocomposites containing 3CNTs, 1BT@CNTs, and 3BT@CNTs have higher Gll when compared with other nanocomposites and the pure PP. This is an indicative that there was significant frictional heat generated at the nanoparticles-nanoparticles and nanoparticles-matrix interfaces. The generated heat was quickly dissipated through the interconnected CNTs in the matrix, producing higher Gll of the nanocomposites compared to the pure PP. (a) Storage modulus and (b) loss modulus of the nanocomposites.
Figure 9(a) represents the loss factor (tanδ) of the nanocomposites. Apart from PP/1BT@CNTs, all other nanocomposites showed a lower tanδ peak compared to the pure PP. The high tanδ of the pure PP can be attributed to low stiffness and high slipping of the PP chains.
32
However, the low tanδ of other nanocomposites revealed their good resistance to PP chains’ mobility offered by the presence of the nanoparticles. The glass transition temperature (Tg) of the nanocomposites obtained from the peak temperature of the tanδ plot is shown in Figure 9(b). Notably, all the nanocomposites have higher Tg than the pure PP, which indicates an enhancement in the rigid to elastic or glassy to rubbery transition temperature. This implies that by the introduction of the nanoparticles in the PP matrix, its chains could not flow at a lower temperature like the pure PP. The achieved increase in Tg can also be considered from the angle of uniform dispersion of the nanoparticles in the matrix and their compatibility with the matrix due to the presence of PPMA compatibilizer.
33
(a) Loss factor and (b) glass transition temperature of the nanocomposites.
Conclusion
In this study, CNTs surfaces were decorated with BT nanoparticles via hydrothermal and self-assembly methods to promote dispersion, adhesion, interlocking of PP chains, and enhancement of thermal, mechanical, and thermomechanical properties of the PP based nanocomposites. Prior to the nanocomposites’ development, the nanoparticles were surface-functionalized to enhance wettability with each other and dispersion in the PP matrix, which was later promoted using PPMA in the fabrication process. TEM and SEM analyses confirmed successful decorating of the CNTs surfaces with BT nanoparticles and uniform microstructures, while PP/BT@CNTs based nanocomposites showed denser microstructures compared to PP/CNTs group. The nanocomposites showed significant improvement in the thermal stability, where about 30oC increase was obtained compared to the pure PP. Further, tensile strength, tensile modulus, and HDT of the nanocomposites were also enhanced, where optimal of about 16.8%, 26.5%, and 20.1% increments were recorded relative to the pure PP respectively. In the same way, dynamic mechanical properties of the nanocomposites were also improved with significant enhancement in the glass transition temperature, indicating increase in rigid to rubbery transition temperature. The enhanced properties of the nanocomposites were more pronounced for the PP/BT@CNTs based nanocomposites, which were attributed to efficient locking of the PP matrix, load transfer from the matrix to the nanoparticles, dense microstructure, well-dispersed nanoparticles, and formation of network structures in the PP matrix.
Footnotes
Acknowledgements
We appreciate the Faculty of Engineering and the Built Environment and the Centre for Energy and Electric Power, Tshwane University of Technology, South Africa for their supports.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
