Abstract
A soybean oil and polydimethylsiloxane-based hybrid bioepoxy resin (HSB) has been prepared by reacting epoxidized soybean oil (ESO) and epoxy-terminated polydimethylsiloxane (EPDMS). The two components were crosslinked using citric acid in solution. The prepared hybrid (HSB) was characterized using infrared spectroscopy (FT-IR) and proton nuclear magnetic resonance spectroscopy (1H-NMR). The surface morphology was examined by scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX); thermal behavior of neat bioepoxy was explored using thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC). The blend (HSB) was examined for their anti-termite activity. The possible applications of this hybrid material were tested by coating on paper as well as wooden plates, and their coating properties were studied.
Introduction
The modern technological advances in the search of novel properties and functions of materials are driven by increasing demands of market for new materials. 1 Plant-derived chemicals and materials have captured the attention of researchers all over the world as an alternative resource for fossil fuels due to the ever-evolving concerns of global warming, depletion of its resources, and the increasing cost of petroleum-based chemicals. 2 Hence, the development of eco-friendly, low-cost, sustainable, green materials from plant-derived materials was immensely explored in several studies.3,4 Bio-based monomers and polymers are an interesting area to the chemist for the synthesis of new biomaterials due to their low carbon footprint and wide availability. 5 Vegetable oils obtained from different natural resources, namely, soybean, castor, and canola crops proved promising raw materials for making both chemicals and bio-based polymers due to their ease of availability, inexpensiveness, and sustainability. The different types of vegetable oil-based polymers, namely, polyurethane, polyester, polyether, and bio-epoxy resin have been synthesized. 6 These polymers are built through modification of triglycerides which are the main constituents of vegetable oils. 7 Bio-epoxy resins produced from vegetable oils are the most important polymer in the categories of vegetable oil-based polymers.8,9 Petroleum epoxy resins have useful properties like dimensional stability, mechanical strength, thermal stability, and chemical resistance.10,11 However, bio-based epoxies do not possess adequate thermal, mechanical, and surface properties to meet the requirements of consumer products. 12 Therefore, it is necessary to improve the properties of bioepoxy resins to meet the consumer requirement for its use in commercial products. Numerous studies have shown that blending of inorganic-organic components can improve thermal and mechanical properties of prepared materials which are rarely present in purely organic or inorganic forms of that material.13,14 Polydimethylsiloxane (PDMS) is an inorganic polymer known for its moderate thermal stability, 15 oxidation resistance, 16 and resistance to chemical attack. PDMS is also compatible with extreme weather conditions due to presence of stronger Si-O-Si bond in its chains. 17 Apart from this, it has better impact resistance, 18 biocompatibility, 22 and minimum surface energy. PDMS rubber is known for its chemical and thermal resistance, governing high elasticity and good hydrophobic properties.19–22 Hence, PDMS is a suitable material to incorporate in bioepoxy resins for improving its physical and chemical properties. The introduction of PDMS segments in bioepoxy via direct blending is difficult to achieve due to the inorganic-organic phase separation. To avoid phase separation, an epoxy, hydroxyl, amine-terminated PDMS was added in epoxy resins to enhance the compatibility and miscibility of PDMS in epoxy resins.23,24 Mohamed et al. have developed a hybrid bio-based epoxy composite from pine oil with EPDMS, amine-terminated PDMS and epoxy-terminated polyhedral oligomeric silsesquioxane chains (EPOSS). The POSS hybrid composite showed enhancement in the mechanical and thermal properties. 25 To the best of our knowledge, the hybrid products of PDMS and soybean oil are not reported in the literature. In present work, we have developed a hybrid composite using tetrahydrofuran solvent where both the epoxy monomers are dissolved and condensed through crosslinked bio-based dicarboxylic acid, that is, citric acid, in order to achieve the useful properties. The resulting material was tested against termites for protective coatings as these green materials may possibly reduce the termite attacks on cellulosic substrates.
Experiment
Materials
Epoxy-terminated polydimethylsiloxane (EPDMS, X-22-163C) equivalent weight (5400 g/mol) was procured from Shin-etsu, Japan, and was used as received. Soybean oil with an iodine value (IV) of 125 and an acid value of 0.3 mg KOH/g was purchased from a local vendor, and used as such without further purification. It was characterized by a refractive index, saponification value, acid value, and iodine value. Tetrahydrofuran (THF) and citric acid were procured from SD Fine Chem. Ltd., Mumbai, India. Other chemicals, namely, hydrogen peroxide (30%, v/v), acetic acid (85%, v/v), anhydrous sodium sulfate, and amberlyst 15-WET (strongly acidic polymeric catalyst), were procured from SD Fine Chem. Ltd and Spectrochem, Mumbai, India, and were used as received without any further purification.
Methods
Titrimetric methods (OH value, acid value, and iodine value)
Soybean oil was tested for refractive index (ASTM D1747—09), specific gravity (ASTM D5355—95), iodine value (ASTM D5768-02), hydroxyl number (ASTMD1957-86), saponification (ASTM D464—05), and acid number (ASTM D5768-02).
Synthesis of epoxidized soybean oil
Epoxidation of soybean oil was carried out by following a procedure reported in the literature. 5 The yield of epoxidized soyabean oil was found to be 93%.
Synthesis of soybean bioepoxy resins
Soybean bioepoxy was prepared by following the procedure reported in the literature.5,6 The yield of the product was 95%, and viscosity was 0.78 dL/g. The formed polymer had a glass transition temperature (Tg) of 99°C.
SB bioepoxy: [1H NMR (400 MHz, D2O) δ ppm]: 0.6 δ (t, 3H,-CH2-CH2-C
Synthesis of a soybean-silicone hybrid composite
The bioepoxy resin was synthesized by solution polymerization. A general experimental procedure is as follows: In a three-necked round-bottom flask equipped with an overhead stirrer, thermometer, nitrogen gas inlet, and reflux condenser, 1.55 g of citric acid and 20 mL of THF were added. The mixture was stirred until complete dissolution of citric acid. Further, to this solution, 10 g of ESO with 0.30 g of EPDMS (3% by weight of ESO) having an epoxy equivalent weight 340 g/mol and 2700 g/mol was added in the ratio of the epoxy’s equivalent weight of EPDMS and ESO: the acid equivalent weight of CA is 1:1. The reaction temperature was maintained at 60–80oC with continuous stirring for 0.5 h. The viscous mass thus obtained was cooled to room temperature and the content was poured onto a glass plate which was preheated in an oven at 85oC to absorb thermal shock. The glass plate was kept further in the oven at 80oC for 6 h. The film was peeled out by dipping the glass plate in water. The solvent and moisture content in the film was removed, and the film was dried in a vacuum oven. The yield of the product is 93% and viscosity is 0.68 dL/g. The polymer showed a glass transition temperature (Tg) at 99°C.
HSB bioepoxy: [1H-NMR (400 MHz, D2O) δ ppm]: 0.6 δ (t, 3H,-CH2-CH2-C
Fourier Transform Infrared Spectroscopy (FT-IR)
The Fourier transform infrared (FT-IR) spectra of SB and HSB resins (crushed film) were recorded on the Shimadzu spectrophotometer (Shimadzu, Japan, Model No. 8400) with a transmittance range of 4000-500 cm−1 at a resolution of 4 cm−1.
Proton Nuclear Magnetic Resonance Spectroscopy (1H-NMR)
1H-NMR spectra of bioepoxy resins, namely, HSB and SB were recorded on a Bruker Avance II 400 NMR spectrometer in D2O containing CaCO3.
Measurements
Solution Viscosity
The dilute solution viscosities of 0.5 wt% polymer in 1 M NaOH solution were determined using an Ubbelohde viscometer at 30° ± 0.1oC.
Chemical Resistance
The pieces of bioepoxy film (1 g of each piece) SB and HSB were placed in different solvents (25 mL of each) at room temperature in a closed jar for 7 days, and the resistance to chemicals was monitored by measuring the difference in their weights with an interval of every 24 h.
Thermogravimetric Analysis
Thermogravimetric analysis (TGA) was performed using a Perkin Elmer TGA 4000 thermogravimetric analyzer under a nitrogen atmosphere over a temperature range of 25–700°C with a heating rate of 10°C min−1. The degradation temperatures such as IDT (initial degradation temperature), Td5 (the temperature at 5% weight loss), Td30 (the temperature at 30% weight loss), heat resistance index (Ts), and char temperature were determined from TGA thermograms.
Differential Scanning Calorimeter (DSC)
The glass transition temperature (Tg) of resins were evaluated with DSC, where 0.005 and 0.01 g of resin sample was introduced into a Mettler-Toledo (Model Q100, TA Instruments, USA) at a heating rate of 10°C/min under a nitrogen atmosphere.
Pencil Hardness
The pencil hardness of coated panels was measured with the help of a pencil hardness tester (Model BYK Additive & Instruments, Germany). This test was performed by following the procedure described in the ASTM D-3363 standard.
Scratch Hardness (kg)
The scratch hardness of coated panels and hybrid PU-coated panels were measured with the help of a scratch hardness tester (Rajdhani Pvt. Ltd, India)
Contact Angle
The contact angle was determined using a lab-made setup using a tripod stand and camera.
Scanning Electron Microscopy
The surface morphology of the cured resin film surface was studied by a scanning electron microscope (SEM, Hitachi S-4200). The surface of the film was coated with gold under vacuum before observation
Energy-dispersive X-ray Analyzer
The chemical composition of SB and HSB resins was determined using an energy dispersive X-ray analyzer (EDX, EM912) coupled with a scanning electron microscope (SEM, Hitachi S-4200).
Anti-termite Activity
The labeled white papers (4 x 3.5 inches) were coated on both sides with the HSB and SB resins. The coated papers were cured for 10 h at room temperature, after that each paper was weighed properly. Then, papers were placed under a termite (Microtermes obesi) colony and left for 10 days. Moisture was constantly maintained by spraying water on the colony within the exposure period to maintain appropriate environmental conditions favorable to termites. 26 After the exposure period, the papers were cleaned to remove soil and debris and dried in a heating oven at 50°C to a constant weight. The mass of each paper was then determined, and the average weight loss was calculated.
Results and discussion
Soybean oil was found to be the most common source of vegetable oils because of its ease of availability and low price. The epoxidized soybean oil (ESO) prepared from soybean oil has a plasticizing effect and acts as a toughening agent when used as reactive diluents in petroleum-based epoxy resins.27,28 Silicones are commonly known for their anticorrosion, chemical resistance, and weatherability. In the literature, the photosensitive polysiloxane was obtained by a condensation reaction using EPDMS reacting with acrylic acid, where carboxylic acid groups (–COOH) of acrylic acid and terminal epoxy groups of EPDMS were utilized. 29 Earlier, we have reported that epoxy resin has been prepared by reacting epoxidized karanja oil/cottonseed oil/algae oil with citric acid.8,9 In the present study, ESO and EPDMS were reacted with citric acid to prepare the hybrid resin which will have special properties.
Synthesis of soybean-silicone hybrid composite
A soybean-silicone hybrid composite of ESO and EPDMS having better chemical resistance were prepared. The reaction scheme of ESO, EPDMS, and citric acid has been illustrated in Scheme 1, respectively. EPDMS was used to improve the physical properties of bioepoxy resin based on soybean oil. The epoxy group of the PDMS oligomer is important for crosslinking. Citric acid is the bio-based crosslinking agent used for obtaining the chemical bond between the EPDMS and ESO to get better compatibility of organic and inorganic phases. The different studies indicate that hybrid materials containing PDMS show microphase separation.
30
Organic and inorganic phases of both materials are immiscible with each other due to that phase separation being inevitable. Increasing the quantity of PDMS in a system will lead to more phase separation. To reduce the phase separation, we have used a PDMS oligomer (5400 g/mol) of appropriate molecular weight with a concentration of 3% for the preparation of hybrid resin. The concentration of PDMS was used to control the useful properties of combined organic and inorganic phases. Despite using lower concentration, SEM analysis revealed that minor phase separation occurred in a hybrid film, whereas when it is lower, it cannot negatively affect the properties of the hybrid copolymer. The crosslinking agent used in the present study is greener, cost-effective, and easily available bio-based dicarboxylic acid, that is, citric acid which is responsible for the joining of PDMS segments in the fatty acid backbone by a chemical bond in the THF solvent. The epoxy group of both components and the dicarboxylic acids react and yield materials with consistent properties. Synthesis of hybrid soybean bioepoxy composite
Fourier transform infrared spectroscopy
The FT-IR spectra of SB and HSB resin are shown in Figure 1. The absorption bands shown in the region 2926 cm−1 and 2854 cm−1 are due to CH2 symmetric and asymmetric stretching vibrations, respectively. The triglycerides ester bonds of the carbonyl group (>C = O) stretch are observed at band 1740 cm−1. The free hydroxyl groups of citric acid and polymer backbone showed the presence at band 3311 cm−1. The presence of PDMS-terminated glycidyl was confirmed by the strong absorption bands at 1233, 1104, and 805 cm−1 corresponding to Si-CH3 in-plane bending or scissoring the Si-O-Si stretch and out-of-plane oscillations of Si-CH3 moiety, respectively.
31
FT-IR spectra of (a) soybean bioepoxy resin (SB) and (b) soybean silicone hybrid (HSB).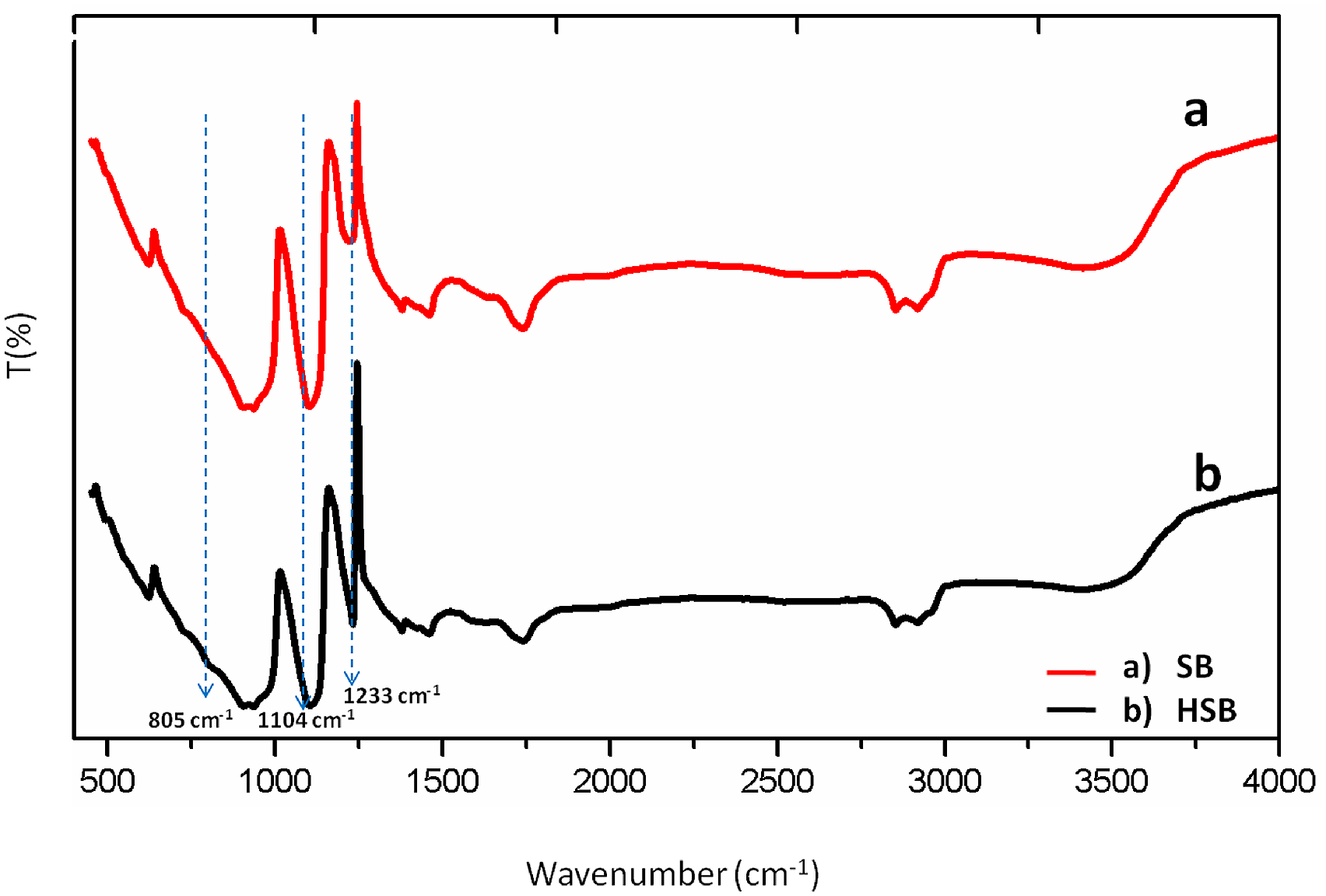
Nuclear magnetic resonance (1H-NMR)
The 1H-NMR spectra of both the resins were recorded for their comparative analysis. The spectra of soybean bio-epoxy (SB) and soybean silicon hybrid composite (HSB) are shown in Figure 2. The peak at 0.59 δ, ppm shows the proton of the terminal –C Comparative proton NMR spectra of (a) HSB and (b) SB bioepoxy.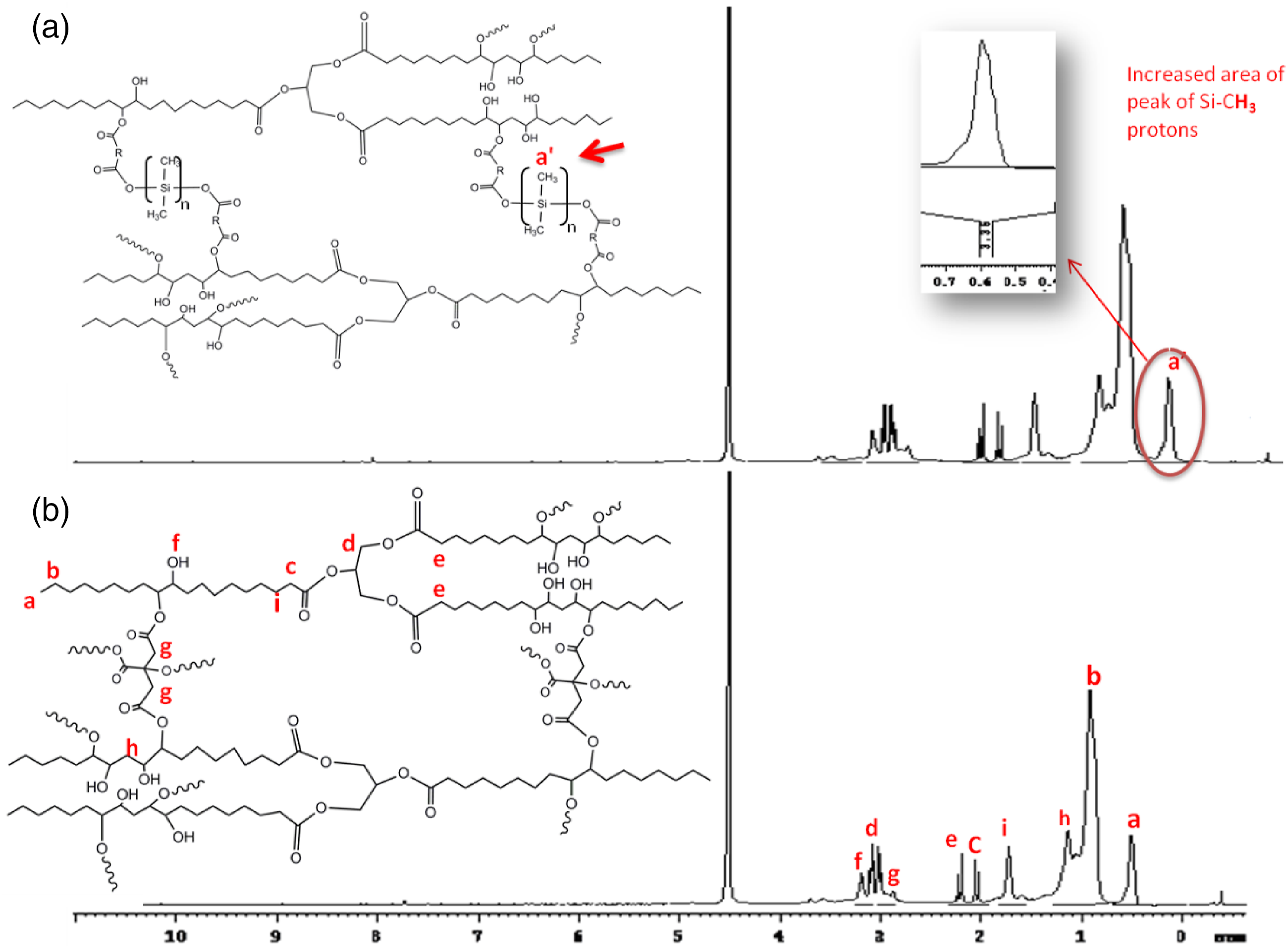
Chemical resistance and viscosity
It was observed that both resins are resistant to most of the solvents. Petroleum solvents tend to cause SB and HSB resins to swell as they contain aliphatic hydrocarbon structure of triglycerides. In NaOH, the SB resin is found to be soluble in NaOH solution. This may be due to the poor resistance to alkali solution because of the presence of a hydrolyzable ester group in bioepoxy resin, whereas HSB resin shows greater chemical resistance to alkali solution as compared to SB resin, which may be due to the PDMS chain that acts as a barrier to hydrolyzing the triglyceride structure.
Effect of different solvents on the chemical resistance of HSB and SB bioepoxy resin.

Note: “+” is soluble, “
Thermal analysis

Thermal behavior of SB and HSB bioepoxy resin.

aOnset temperature.
bTemperature of 5% weight loss.
ctemperature of 30% weight loss.
dStatistic heat-resistant index temperature calculated by equation (1).
eChar temperature at 800°C
The statistic heat-resistant index (Ts) of bioepoxy resin SB and HSB was found to be different. This is maybe due to the peculiar characteristics of PDMS blocks present in the polymer matrix; the presence of strong Si-O-Si bonds has helped to raise the heat-resistant index of HSB resin. 32 Char temperature is the temperature at which char residue, which is carbon or carbonaceous material, cannot further dissociate into smaller volatile fragments and remains there at the highest temperature of TGA analysis.
Elemental analysis and morphological studies
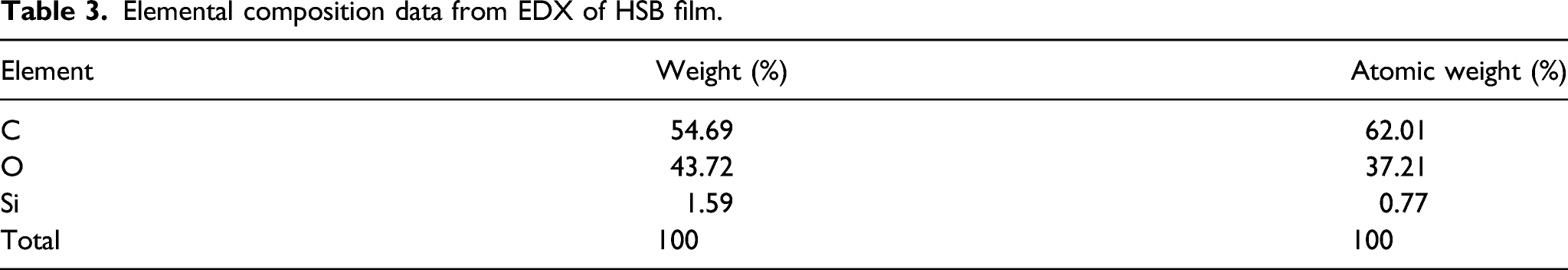
The materials were analyzed by EDX and SEM methods. The EDX analysis technique is used for the identification of elements present in the given material. Figure 3 shows the HSB films containing carbon, oxygen, and silicone elements. The EDX can also be used to analyze the semi-quantitative composition of the material (Table 3). The presence of silicone is confirmed, and its content is determined. Energy-dispersive X-ray spectroscopy (EDX) of hybrid bioepoxy. Elemental composition data from EDX of HSB film.
The SEM micrographs of neat bioepoxy and PDMS are shown in Figure 4. The fairly homogenous distribution of PDMS chain domains is observed with microphase-separated morphology in HSB film, although the inter-cross linking network is confirmed.
33
Scanning electron micrographs of soybean bioepoxy (SB) at resolution of (A) 100 µm. Hybrid soybean bioepoxy (HSB) at the resolution of (B) 100 µm.
Contact angle measurement
The angle between coated surfaces with water is defined as a contact angle. If the contact angle is above 90, then the surface is hydrophobic, and if it is less than 90, it is considered as hydrophilic. The lotus effect can be observed on hydrophobic surfaces.
34
The surface coated with bioepoxy resins (SB and HSB) shows hydrophobic properties and the lotus effect on its surface, whereas the uncoated surface is hydrophilic, having a contact angle less than 90⊝. However, the HSB resin-coated panel shows an increased contact angle compared with neat SB resin coating. The silicone present in the coating is responsible for the increase in the contact angle. The comparative images of uncoated, SB-coated, and HSB-coated wood panel showing contact angle images have been presented in Figure 5. The contact angle results are presented in Supplementary Table 3. Comparative contact angle measurements of (a) uncoated wooden plate (UC), (b) coated with soybean bioepoxy (SB), and (c) coated with a hybrid of soybean oil (HSB).
Anti-termite activity of coatings
Termites are known to be responsible for huge damage to products of wood like furniture, paper, buildings, etc. Termites are commonly found inside the ground where soil and moisture are available. They enter through small gaps from walls in search of lignocellulosic materials, generally furniture and paper. The termites cause unexpected losses as wooden materials are excessively used in furniture.
35
The different types of coatings and termite repellent are used to avoid loss and increase the survival of wooden materials. The bioepoxy coating can be a good alternative, which is a less harmful and bio-based alternative for antitermite coatings. The present study aims to find out the termite protection of bioepoxy films on paper surfaces. Termites feed on cellulose for survival and have a fast rate of consumption of wood and paper. Hence, the effect of bioepoxy resin (SB and HSB)based paper coating for anti-termite activity was studied. It was observed from these studies that uncoated paper gets damaged by the termites within 10 days. However, the bioepoxy-coated paper is less affected as shown in Figure 6; the HSB-coated paper is most effective against termites as silicone’s inherent property of water repellency was slowing down the attack of termites as shown in Table 4. This is a cost-effective approach to protect the important documents that usually fall prey to termites. Photographs showing degradation results of (a) uncoated paper, (b) SB-coated paper, and (c) HSB-coated paper. Results of antitermite activity of coated bioepoxy resins.

Application of SB and HSB resins
The prepared HSB resin can be used for coating wooden surfaces, paper lamination, and packaging films. This is demonstrated in Figure 7. The comparison is made by using coating properties like pencil hardness and scratch test. The contact angle measurement was also carried out for comparison of SB and HSB resins to check water repellence properties. Photographs of SB and HSB resin coatings on wooden panels and paper, thin-cast films (top SB and bottom HSB).
Coatings of bioepoxy resins
The coated wooden plates were tested for pencil hardness and scratch resistance. The results obtained are summarized in Supplementary Table 2. The data shows similar results of SB and HSB coatings. The concentration of silicone is very low (3%) in HSB resins which do not show a considerable effect on the coating properties.
Conclusions
The new hybrid PDMS/bioepoxy composite film was developed from soybean oil and EPDMS. Epoxidized soybean oil, citric acid, and EPDMS were used for the synthesis of hybrid materials. The prepared material was characterized using physio-chemical and spectroscopic methods. The synthesized hybrid material displayed improved chemical resistance when compared with the normal bioepoxy resin. However, their thermal properties are nearly similar. The presence of silicone in the material was confirmed by elemental analysis using EDX, and the morphology was studied by SEM showing the partially homogenous distribution of PDMS chains within bioepoxy matrix. The hybrid material and bioepoxy coatings were applied on wooden panels and the paper. The hybrid-coated plates having a contact angle higher than 90 are partially transparent and hydrophobic. The antitermite activity results indicate that hybrid-coated paper has greater protection against termites because of the hydrophobic properties of silicone in coatings.
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911221078482 – Supplemental Material for Preparation and characterization of soybean oil/polydimethylsiloxane hybrid bioepoxy resin and its application in antitermite coating
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911221078482 for Preparation and characterization of soybean oil/polydimethylsiloxane hybrid bioepoxy resin and its application in antitermite coating by Vipul Kusumkar, Pratiksha Singh and Omprakash Yemul in Polymers and Polymer Composites
Footnotes
Acknowledgments
The authors are thankful to Prof. Pandit Vidyasagar, Vice Chancellor, Swami Ramanand Teerth Marathwada University, Nanded for the moral support and encouragement during the work. Epoxy-terminated polydimethylsiloxane, a gift sample received from M/s. Supreme Silicones, Pune, is highly acknowledged. The SEM, EDX, and XRD data was provided by Dr. Kailas Tehare, and last but not least Mr. Sandesh Mutkule for his moral support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementray material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
