Abstract
In this paper, sodium montmorillonite was modified with gelatin of different concentrations, and various colloidal characteristics of the gelatin-treated clays were measured and analyzed in detail. First, the influence of gelatin on the interlayer space of Mt layers was investigated by X-ray diffraction analysis. Moreover, the aggregation of Mt particles was examined using a combination of electron microscopy and particle size distribution experiments, while the variation of the electrical property of Mt was measured using ζ potential test. Gelatin of different concentrations can increase the particle size of Mt in different degrees. The addition of 4% gelatin could improve the ζ potential of Mt from −30.65 to −15.55 mV. The wettability change of modified Mt induced by the adsorption of gelatin was followed by measurements of water contact angle and observations of the morphology of Mt/gelatin membrane through SEM images. 4% gelatin could improve the water contact angle of Mt to 81.3°. Finally, the rheological properties of Mt/gelatin dispersion including shear viscosity and shear stress were measured using a stress-controlled rheometer. All of the results were consistent by showing that the overall colloidal characteristics and behavior of the gelatin-treated Mt strongly varied depending on the gelatin concentration used in the modification process. These results can provide a deep and comprehensive understanding of the colloidal properties of clay/gelatin systems and give important guidance for the performance design and improvement of Mt/gelatin composite materials. Furthermore, this study can also be expanded the application of gelatin and its composites to other fields.
Introduction
Clays are typical crystalline aluminosilicates, consisting of two-dimensional arrays of the silicon–oxygen tetrahedron and that of aluminum– or magnesia–oxygen octahedron. 1 Sharing of oxygen atoms between silica and alumina sheets results in two- or three-layer minerals, such as 2:1-type montmorillonite (Mt), in which an octahedral sheet of alumina or magnesia shares oxygen atoms with two tetrahedral silica sheets. 2 Mt is one of the most popular clay minerals. Generally, Mt particles are negatively charged owing to the isomorphic substitution of the Si and Al ions by lower positive valence ions in the crystal lattice. 3 These negative charges can be compensated by cations that exist in the interlayer region. These interlayer cations are exchangeable and may trade places with other cations under appropriate conditions. 4 Clays are inexpensive, chemically and thermally stable, and have good mechanical properties. 5 They are important commercial commodities. Various clays have been extensively used in cosmetics, pharmaceutical, drilling fluids, refractories, adhesives, and plastics industries.6-7 In addition, many clay/polymer hybrid materials have been also developed and utilized as functional materials with improved performance in a wide variety of applications.8–10 For example, organoclays were synthesized through the exchange of organic cations for hydrated inorganic ones, and the surfactant-treated layered clays have attracted significant attention in the past decade. In recent years, polymer/clay nanocomposites (PCN), prepared by intercalation composite preparation technology,11–15 have become an important research focus due to their significantly improved properties, such as excellent mechanical, thermal, flame retardant, and gas barrier properties in automobiles, appliances, and packaging industries. 16 Among these composite materials, Mt, as the most swelling clay, is widely studied, showing the most extensive applied prospect.
Biopolymers are environmentally friendly and have been usually added into layered silicates for physical modification. Gelatin is a typical organic biopolymer and its raw materials are inexpensive and abundant worldwide.17–18 Gelatin is obtained by the thermal denaturation of collagen and contains a large number of adsorption groups. 19 This biopolymer is a complex polypeptide and has been widely used in food, pharmaceutical, cosmetic, photographic, and drilling fluids industries.20–22 Moreover, various gelatin/clay composites have been prepared and applied in different fields. By adjusting the physical and mechanical properties, various special film functional materials were developed.5,23–24 In addition, gelatin–Mt nanoparticles were prepared by desolvation method and used for controlled drug delivery 25 ; gelatin–Mt composites were also prepared through intercalation method and have been evaluated for adsorption of heavy metals or dyes. For example, a gelatin–Mt nanocomposite was prepared by mixing in distilled water under different temperatures, and its maximum adsorption capacity was 52.91 mg/g for an initial Cr(III) concentration of 300 mg/L. 26 Gelatin–bentonite composite gel was prepared through crosslinking using bis-acrylamide and formaldehyde, and the gel was used as adsorbent for the removal of lead (II) from aqueous solutions. 27 Besides, gelatin/bentonite composite beads were facilely prepared by compacting gelatin and bentonite, which showed excellent adsorption capacity toward congo red (CR) and methylene blue (MB). 28 Simultaneously, some other properties of gelatin/clay composites, such as swelling, 29 adsorption kinetics, 30 and thermodynamical property, 31 have been also studied and analyzed in different applications, such as structural biomaterials, bioplastics, and biological medicine.
Knowledge of clay/polymer characteristics is essential for guiding and optimizing the aforementioned applications. In particular, in the case of clay/polymer composites, the property change of the composite materials is affected by many factors, such as the dispersion of particles (intercalation and exfoliation), particle properties (particle aspect ratio), and particle-matrix interaction, 5 which are important basics in the design of materials with desired properties. Thus, the effect of gelatin on the properties of Mt was important for gelatin-Mt systems. However, many studies only focused on the macroscopic properties for production and application. The basic interaction, characteristics, and mechanisms between clay and gelatin have been seldom reported in detail.
To gain a more thorough understanding of the interactions and characteristics between clay and gelatin, we have proceeded with the study of the colloidal behavior of gelatin-treated Mt. In this article, Mt was modified by the varying concentrations of gelatin. The conformation of the gelatin molecules in the interlayer region was investigated through X-ray diffraction analysis. To study the colloidal characteristics of modified Mt, zeta potential, particle size distribution, water contact angle, and the rheological property measurements were conducted and analyzed in detail.
Materials and methods
Materials
Sodium montmorillonite (Mt, purity ≥90%) was purchased from Energy Chemical (China). Gelatin (purity ≥98%) was purchased from Qingdao Yousuo Chemical Technology (China). HCl and NaOH were purchased from Modern Oriental (Beijing) Technology Development (China).
Preparation of the gelatin-treated Mt
Firstly, Mt was purified referring to previous literatures.7,32 Raw Mt was dispersed into deionized (DI) water to form Mt suspension with a concentration of 4.0% (w/v). After stirring for 24 h, the suspension was centrifuged at 8000 r/min for 10 min using a Cence H1850 centrifuge (Hunan, China). Then, the upper colloids were siphoned off and collected while the bottom of the centrifuge tube containing miscellaneous impurities was discarded. The recovered clay colloids were then dried in a Senxin DGG-9053A drying oven (Shanghai, China) at 60°C for 48 h and finally the dried clays were ground into powder before use.
A series of gelatin-treated Mt samples modified to different degrees were prepared by the ion-exchange reaction. A suitable quantity of gelatin was dissolved in deionized water at 60°C for 2 h to obtain a gelatin aqueous solution with a different concentration between 0.25–4.0% (w/v).5,29,33 Meanwhile, the purified clays were dispersed in water at a concentration of 4.0% (w/v) and stirred for 24 h at room temperature. Then the gelatin aqueous solution was added into each clay dispersion under equal volume condition and the mixtures were stirred vigorously for another 24 h at 35°C. 26 Finally, the gelatin-treated clay precipitate was collected by centrifugation and rinsed with deionized water, until the supernatant was clear. 34
Characterization
The obtained Mt/gelatin composites were dried in a Labconco FreeZone 4.5 vacuum freeze dryer (America) at −50°C for 48 h and then ground for X-ray diffraction (XRD) measurements. The influence of the concentration of gelatin, ranging from 0.25 to 4.0%, on the structure of the modified clays was characterized. XRD analysis was performed by a Bruker D8 Advance Diffractometer (Germany) with Cu-filtered radiation (λ = 1.5406 Å). The 2θ scanning range varied from 2° to 10° at a scanning rate of 1.0°/min. The interlayer space was analyzed using Bragg’s equation. 35
As mentioned above, a certain amount of gelatin solution with different concentrations was added into an equal volume of Mt dispersion to compare the influence of gelatin on the particle size of Mt particles. The particle size distribution was determined using a Malvern Mastersizer 2000 particle size analyzer. The electron microscopy images of Mt dispersion were observed by a Leica DM4M microscope at a total magnification of ×50.
The ζ potential of the Mt dispersion containing different concentrations of gelatin was measured by a Malvern Zetasizer Nano series. The point zero charge can be exhibited. 36 The concentrations of all samples in this test were approximately 4.0 gL−1.
Water contact angle measurement of modified Mt was conducted by a JC2000 C contact angle tester. Mt/gelatin dispersions were spread to the surface of clean glass slides and dried at 60°C for 24 h. Then, a dry and smooth Mt membrane formed. DI water was dropped onto the membrane through a micro-injector and the images were captured.37–38
The rheological properties of Mt/gelatin dispersions were measured with a Haake Mars rheometer (Thermo Electron Corporation, Waltham, MA) with a cone-plate model. At each measurement, approximately 1.5 ml of the samples were carefully placed on the plate. The gap between the cone rotor and the plate is 53 μm, and the diameter of the cone rotor is 35 mm, containing a cone angle of 1.0°. The “ture” shear viscosity and shear stress were measured under a shear rate from 1000–0.1 s−1, and all of the measurements were conducted at 25°C. 39
Results and discussion
X-ray diffraction analysis
The XRD curves of various dried clay samples modified by gelatin were first presented (Figure 1). The interaction between the clay and gelatin led to a shift of the diffraction peak of Mt toward lower 2θ values, implying the increase of the (001) interlayer space (d-value) due to the intercalation of gelatin. Moreover, the changes of the d-value could reflect the proportion of the interlayer occupied by the arrangement of gelatin. XRD patterns of dried powders of natural and modified Mt with various concentrations of gelatin.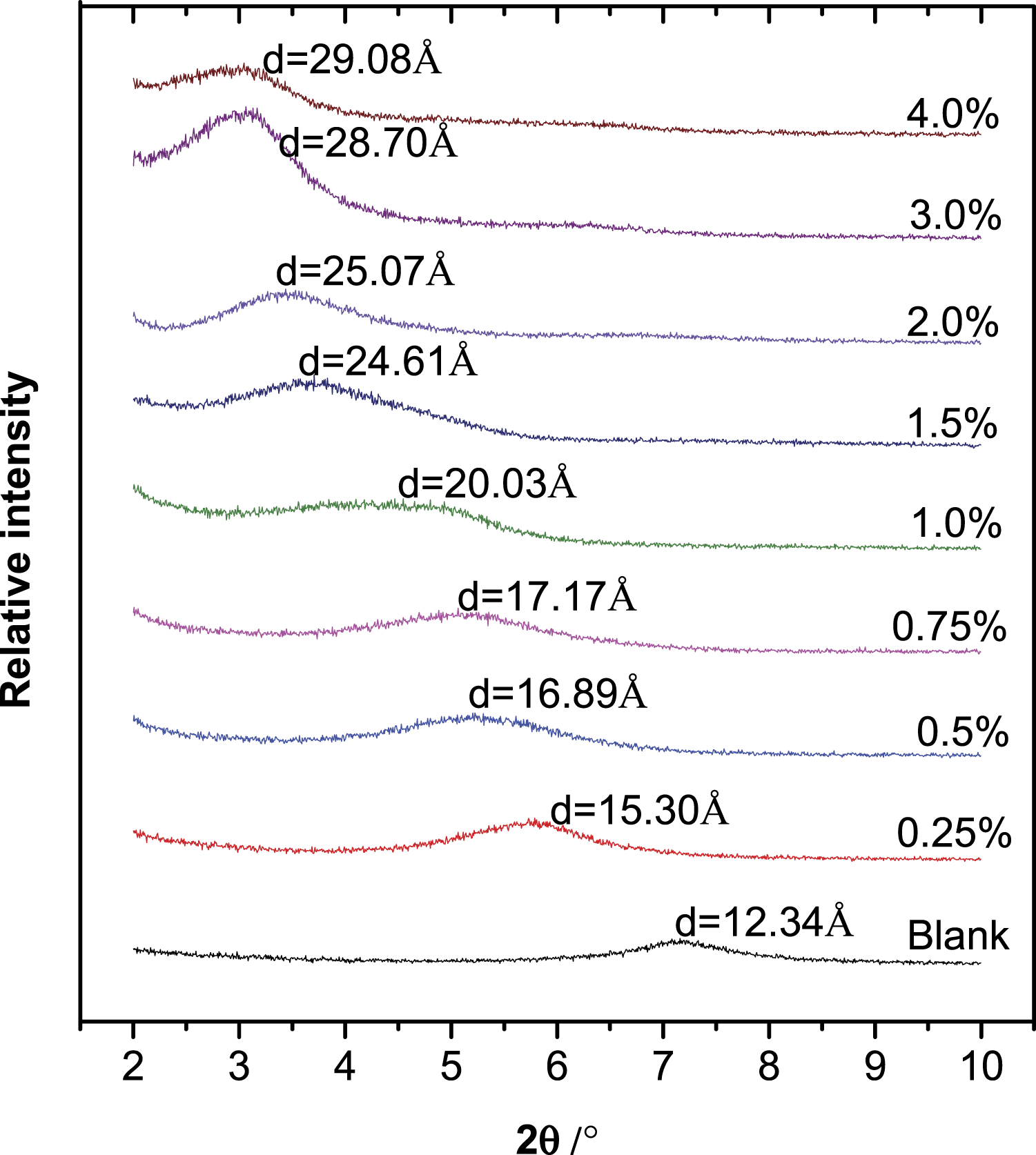
The variation law of the d-value with gelatin concentration was also exhibited (Figure 2). The d-value changed in a stepwise fashion with the increase of gelatin loading and there was a linear increase in the spacing between the steps. In detail, the three steps corresponded to three relatively constant values of the d-value: ∼17.0, ∼25.0, and ∼29.0 Å, while the d-value of pure Mt was 12.34 Å. From the results, it could be referred that the thickness of the monolayer gelatin molecule was about 4–6 Å, which was similar to previous works.40–42 When the addition of gelatin was less than 0.75%, the d-value might be mainly governed by a monolayer arrangement of the gelatin molecules. Concentration beyond 0.75% and up to 1.5% caused a linear increase of the d-value. Considering gelatin has a variable molar mass and volume, this linear increase might correspond to a mixed arrangement situation where both of monolayers and bilayers of gelatin molecules existed in the interlayer space. Furthermore, during this linear increase of the d-value, with increasing the amount of gelatin, the monolayer arrangement of gelatin gradually transformed into a bilayer arrangement. The broadening of the diffraction peak for the 1.0% concentration also proved the transition behavior, which might be caused by the superimposing effect of the two kinds of arrangements. The complete bilayer arrangement happened when the dosage of gelatin was higher than 1.5% and up to 2.0%. Further increase of the polymer concentration forced the interlayer space to expand continuously. Additional increase of gelatin to 3.0–4.0%, at the third step, exhibited a slower growth of the d-value, compared with that of 1.5–2.0%. This condition might give a more complex arrangement of the gelatin molecules inside the interlayer region, and the exfoliation of Mt layers might occur under such wide interlayer space.
43
The d-value of dried samples of natural and modified Mt with various concentrations of gelatin.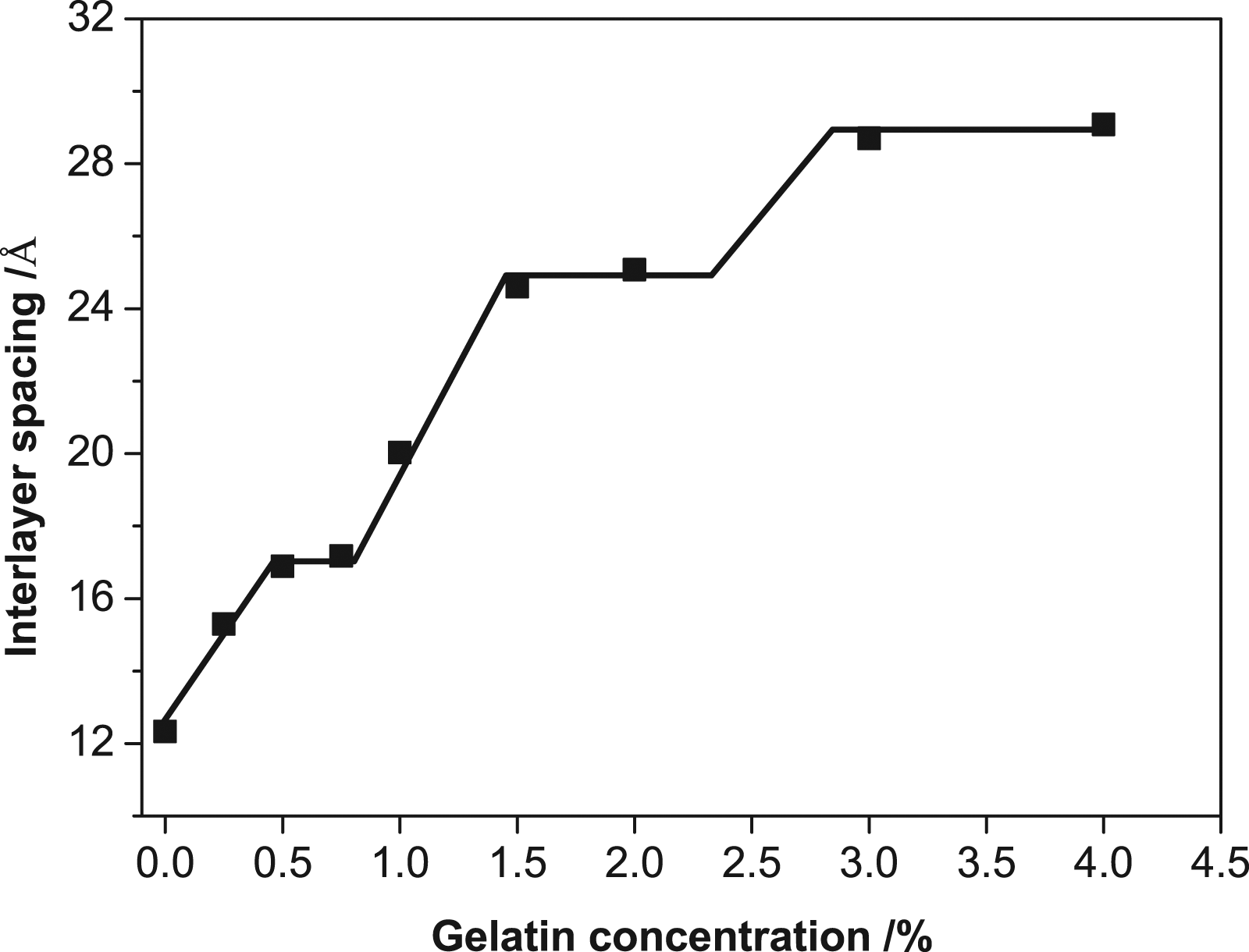
In fact, the structure of gelatin presents highly complexity and is influenced by many factors, such as gelation processing, temperature, concentration, and the origin of the raw material.
44
Generally, the gelatin films could present three different structural states: (a) amorphous coils, (b) triple helixes and coils, and (c) bundles of triple helixes and coils.
45
According to Ref. [43], the freezing/thawing method also had the advantages for gelatin molecules to renature into triple-helix and easily induced exfoliation of Mt layers. Thus, the gelatin molecule arrangement structure in the Mt layers may be more complex. However, the variation of d-value (Figure 2
Particle size distribution
The incorporation of organic polymer molecules will influence the interaction between clay particles and change the dispersion degree. Therefore, the change of particle size distribution can be used to assess the effect of gelatin on clay hydration.
The morphology of Mt particles was observed using electron microscopy (Figure 3). A stable dispersion was generated in pure water due to thorough hydration and dispersion for Mt particles. The pure Mt was dispersed equably and the particle size was quite small. By comparison, the addition of gelatin-induced aggregation of Mt particles and the Mt aggregates became larger gradually with increasing the gelatin concentration. The particle size measurement was also in accordance with the result from electron microscopy. From the whole variation trend, the particle size gradually increased (Figure 4(a)). For example, after adding 0.25, 0.5, 1.0, 2.0, and 4.0% gelatin, the volume average particle size of Mt increased from 7.687 μm to 38.407, 54.618, 73.218, 63.587, and 108.076 μm. However, after continuous stirring, the Mt particle size decreased (Figure 4(b)). In detail, compared with that of pure Mt, the particle size increased with the addition of 0.25 and 0.5% gelatin, while the Mt particle size decreased at the addition of 1.0, 2.0, and 4.0% gelatin. With the addition of 0.25, 0.5, 1.0, 2.0, and 4.0% gelatin, the volume average particle size of Mt varied from 7.687 μm to 27.958, 22.185, 13.319, 15.9, and 12.877 μm. Images of Mt dispersion before continuous stirring in different concentrations of gelatin aqueous solutions. The total magnification was 40×. Variation of particle size distribution as a function of the concentration of Mt/gelatin dispersions: (a) before continuous stirring, (b) after continuous stirring, (c) the volume average particle size.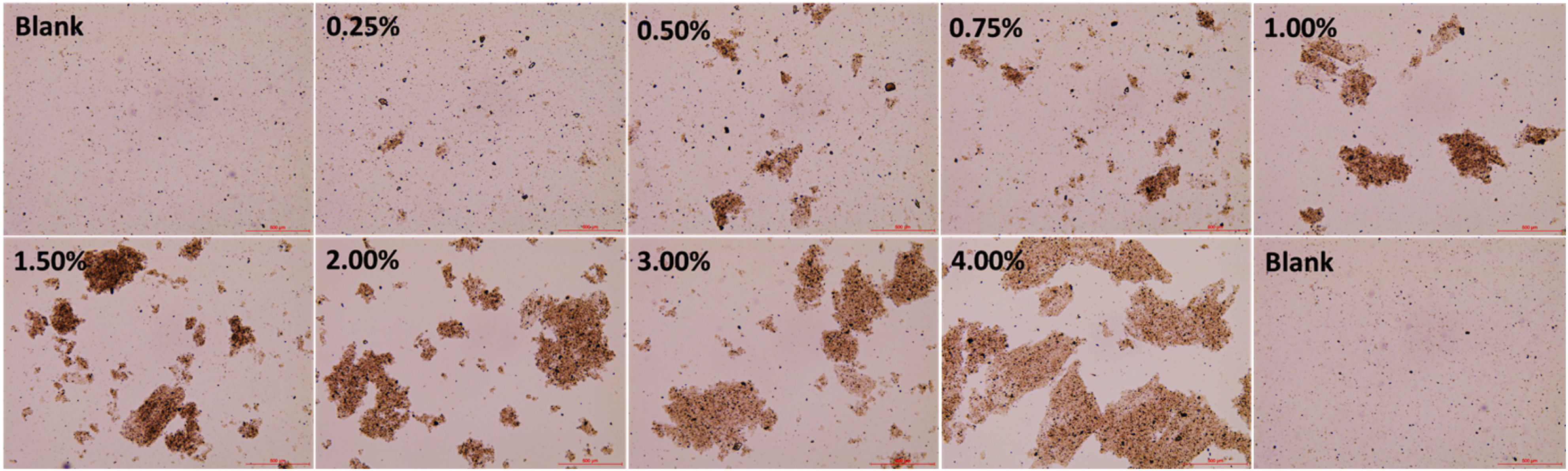
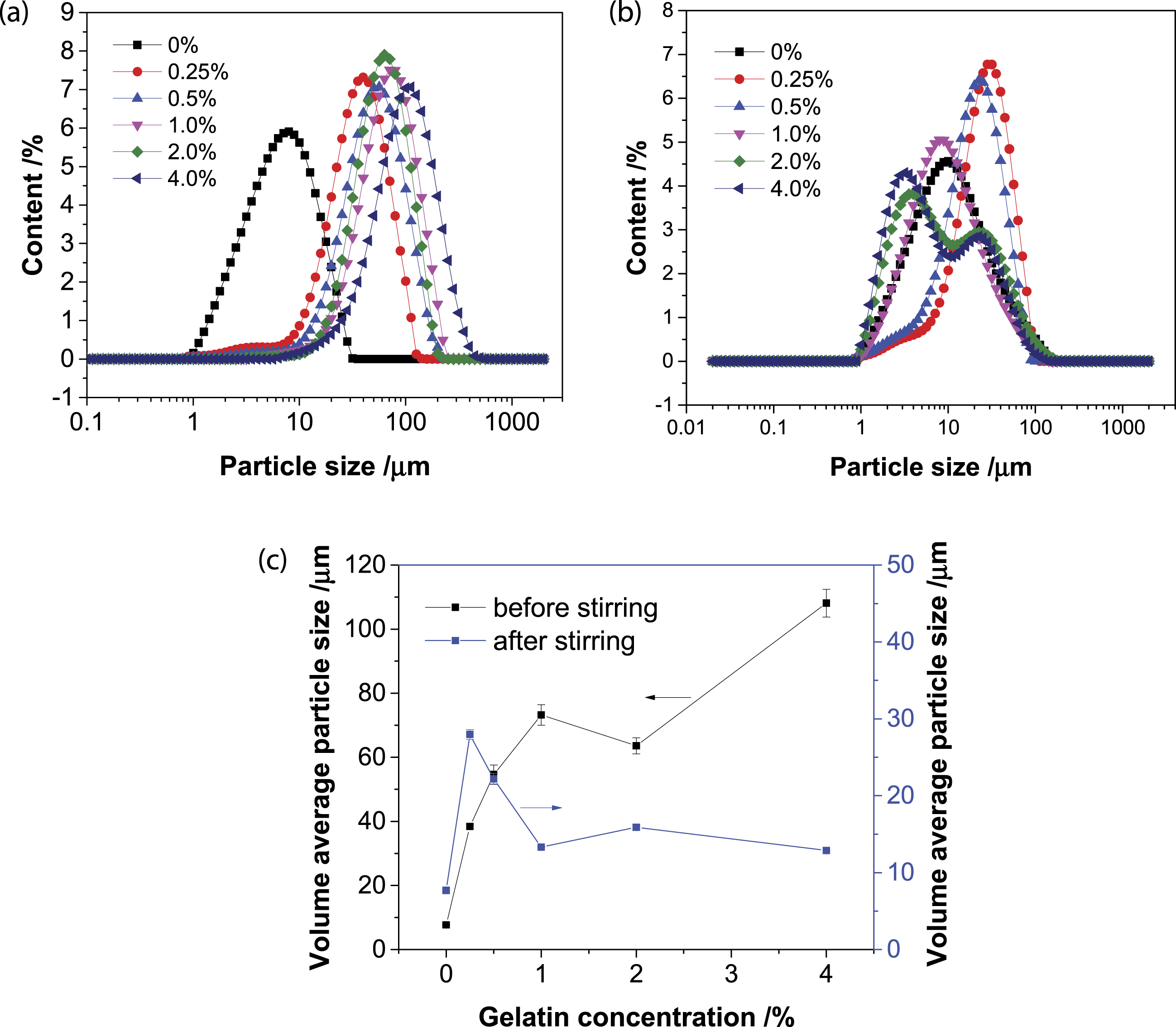
Gelatin was typical amphoteric polyelectrolytes with relatively big molecule weight and contained many adsorption groups, such as carboxyl groups, amino groups, and hydroxyl groups. 44 Amino groups were alkalescent when dissolving in water and part of amino groups would be protonated and become positively charged. 37 Thus, gelatin could be adsorbed with clay particles through the coordination of electrostatic attraction and hydrogen bonding. Based on the results of electron microscopy and particle size measurement, gelatin adsorbed onto the surface of Mt, encapsulated the particles, and induced aggregation, which was disadvantageous for the dispersion of Mt colloid. However, flocculation is a dynamic process. After continuous string vigorously, the Mt aggregates were broken up and the particle size decreased, especially at the high concentration of gelatin (Figure 4(c)). At low gelatin concentration, gelatin molecules would not interfere with each other. The gelatin adsorbed onto Mt and encapsulated them. At high gelatin concentration, adsorbed excessive gelatin molecules crossed over and crowed out each other. The adsorption of gelatin also improved the hydrophobicity of Mt (described below). These effects might separate some Mt particles and decrease their particle size. However, as shown in Figure 4(c), the average particle sizes of Mt after adding gelatin were always bigger than that of pure Mt, namely, gelatin molecules could adsorb onto Mt particles and encapsulate them, causing the increase of particle size of Mt. However, under vigorously stirring, the formed big-size Mt aggregates would be broken up and the particle size decreased. The particle size of many colloidal particles varied with stirring time. The size of clay floc in flocculation process became larger with the increase of stirring time. 46 A cationic polymer could strongly adsorb on clay and produce large aggregates after vigorously mixing with clay suspension, indicating that the interactive force between the polymer and clay was quite strong. 35 Thus, this phenomenon in this study demonstrated that the bridging and encapsulation effect of gelatin onto Mt were incompact.
Water contact angle measurement
Montmorillonite, as a kind of swelling clay mineral, is quite hydrophilic and easily adsorbs water. In our study, the water contact angle for pure Mt was 28.2°. This result was similar to the value of 25.3° measured by Zhong et al.
37
proving the hydrophilicity of Mt. The hydrophilic properties of Mt were changed after being treated by gelatin (Figure 5). The water contact angle of the Mt membrane increased gradually with increasing the concentration of gelatin. When 4.0% gelatin was added, the water contact angle reached as high as 81.3°. Meanwhile, pure gelatin membrane exhibited relatively strong hydrophobic property with a water contact angle of 85.6°. The result was consistent with previous work,
47
which indicated that the water contact angle of gelatin film was 90.1°. Gelatin film exhibited relatively certain hydrophobic properties. Thus, it can be inferred that the hydrophobic property of Mt was improved by the adsorption of gelatin. The water contact angle of Mt/gelatin membranes.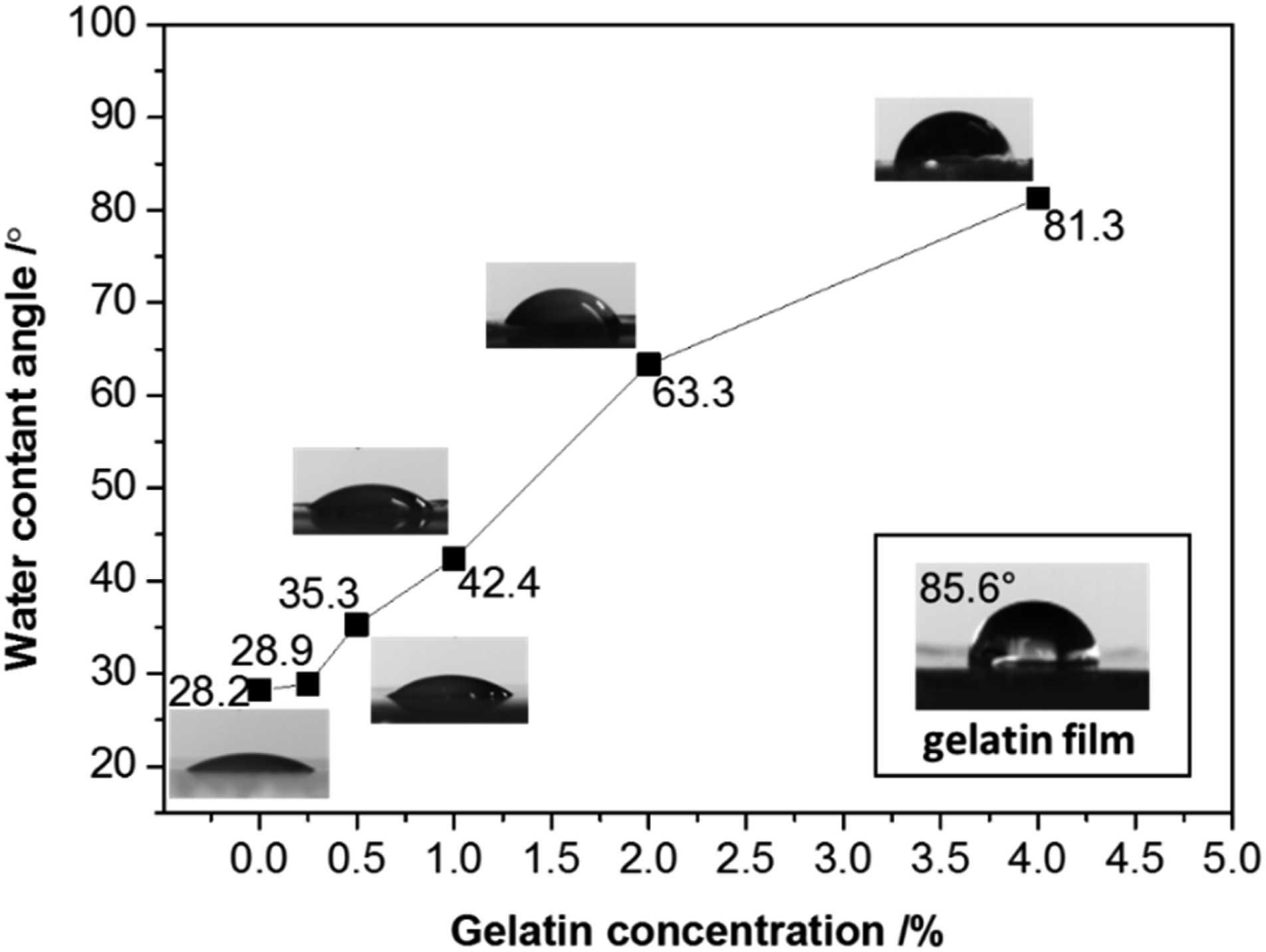
Many polar groups, such as amino groups and hydroxyl groups, are distributed throughout the whole gelatin chains. In gelatin solution, gelatin molecules will intersect into a network structure by the hydrogen bonds between the amino groups and hydroxyl groups. 20 As a result, the polar groups are closed to each other while hydrophobic hydrocarbon chains are exposed to air. Thus, the surface of gelatin film was relatively hydrophobic. 44 Lin et al. 48 also indicated that in solutions 7% of the amino acids in the gelatin structure were strongly hydrophobic and created hydrophobic interactions at the interface air/water. When Mt was composited with gelatin, the amino and hydroxyl groups in gelatin backbone were easy to form hydrogen bonds with hydrogen and oxygen in Mt. The particle size distribution was also proved that gelatin adsorbed on the Mt particles and encapsulated them. After the adsorption, the hydrophobic chains in gelatin were coated on the external surface of Mt, thereby improving the hydrophobic property of Mt particles. The result was consistent with previous study that Mt cooperated with a 3 wt % concentration of gelatin displayed a water contact angle of 74.8°. 49
To further investigate the effect of gelatin on the hydrophobic modification for Mt particles, the surface micromorphology of Mt membrane was observed by SEM images (Figure 6). The pure Mt could hydrate and swell seriously in water. In this study, the membrane surface of pure Mt was rough and porous. In comparison, as the gelatin concentration increased, and the surface of Mt membrane became more and more smooth and compacted. According to Wenzel’s equation
50
(cosθ = r (γg-s—γl-s)/γg-l), when the contact angle was less than 90°, the rough surface had better wettability and the contact angle was smaller. Therefore, the smooth surface might be also one of the reasons for the improved hydrophobic property of Mt membrane modified by gelatin. Oppositely, if the contact angle was more than 90°, the rough surface had better hydrophobicity. For example, Ni et al.
38
prepared nanofluids to increase the surface roughness of shale for improving the hydrophobicity. SEM images of Mt membrane modified by different gelatin concentration: (a) 0%, (b) 0.25%. (c) 0.5%, (d) 1.0%, (e) 2.0% and (f) 4.0% observed at a total magnification of 5000×.
ζ Potential measurement
The important role of gelatin concentration in the behavior of the modified Mt was also investigated by the ζ potential measurements. First, the pH dependence of ζ potential of pure gelatin and pure Mt was illustrated (Figure 7). Effect of the pH on the ζ potential of Mt and gelatin.
For pure Mt, it was composed of a large number of plate-like layers with permanent negative charges on the flat face and pH-dependent charges on the edges. The permanent negative charges arise from the substitution of the central Si and Al ions in the crystal lattice for lower positive valence ions. The pH-dependent charges are due to the presence of the amphoteric Al-OH and Si-OH groups, which are conditionally charged to be negative, neutral, or positive. 34 Mt particles were negatively charged over the whole pH range (Figure 7). The permanent negative charges on faces were dominant and the negative charges were always much more than the positive ones. Moreover, the negative charges of Mt increased with the increase of dispersion pH. 51 The phenomenon also proved the effect of protolytic reactions of edge OH groups in rendering the positive charge of the edge region increasingly negative and then raising the bulk negative charge of the Mt particles. 3
For pure gelatin, this biopolymer belongs to amphoteric polyelectrolyte. Gelatin molecules contain many ionizable groups, such as carboxyl groups, amino groups, and imidazole groups. 44 In neutral and alkaline solution, carboxyl groups lose H+ and become negative. In neutral and acidic solutions, amino groups obtain H+ and become positive. The gelatin in our study had an isoelectric point at around pH 5 (Figure 7). Above the isoelectric point, the ionization of the carboxyl group was dominant and gelatin was negatively charged, while the protonation of amino groups played a major role and positively charged gelatin was created below the isoelectric point.
Gelatin exhibited higher zeta potential than Mt over the whole pH range. As a result, the addition of gelatin helped to improve the ζ potential of Mt (Figure 8). The ζ potential of Mt increased with increasing the concentration of gelatin, suggesting suppression of the double electrical layers and stability reduction of the Mt colloid. When the concentration of gelatin was low, the increase of zeta potential was limited. After the concentration was higher than 0.5%, ζ potential increased quickly and maintained almost unchanged finally. On the one hand, amine groups in gelatin would be partly protonated and become positively charged, which could adsorb onto the negatively charged sites of Mt through electrostatic interaction, as well as the hydrogen bonds between the amine groups and clay particles, resulting in the compression of the double electrical layers and the particle surface less negative. On the other hand, as discussed above, gelatin encapsulated the Mt particles and formed aggregates after adsorbing onto Mt. Moreover, the surface of modified Mt particles became more hydrophobic, which would provide a shielding effect. This shielding effect would reduce the electrical repulsion between Mt particles and decrease the ζ potential.52–53 Therefore, the addition of gelatin could increase the ζ potential and decrease the stability of Mt colloid. However, these effects were insufficiently strong to reverse the charge of Mt from negative to positive. Effect of gelatin concentration on the ζ potential of Mt dispersion.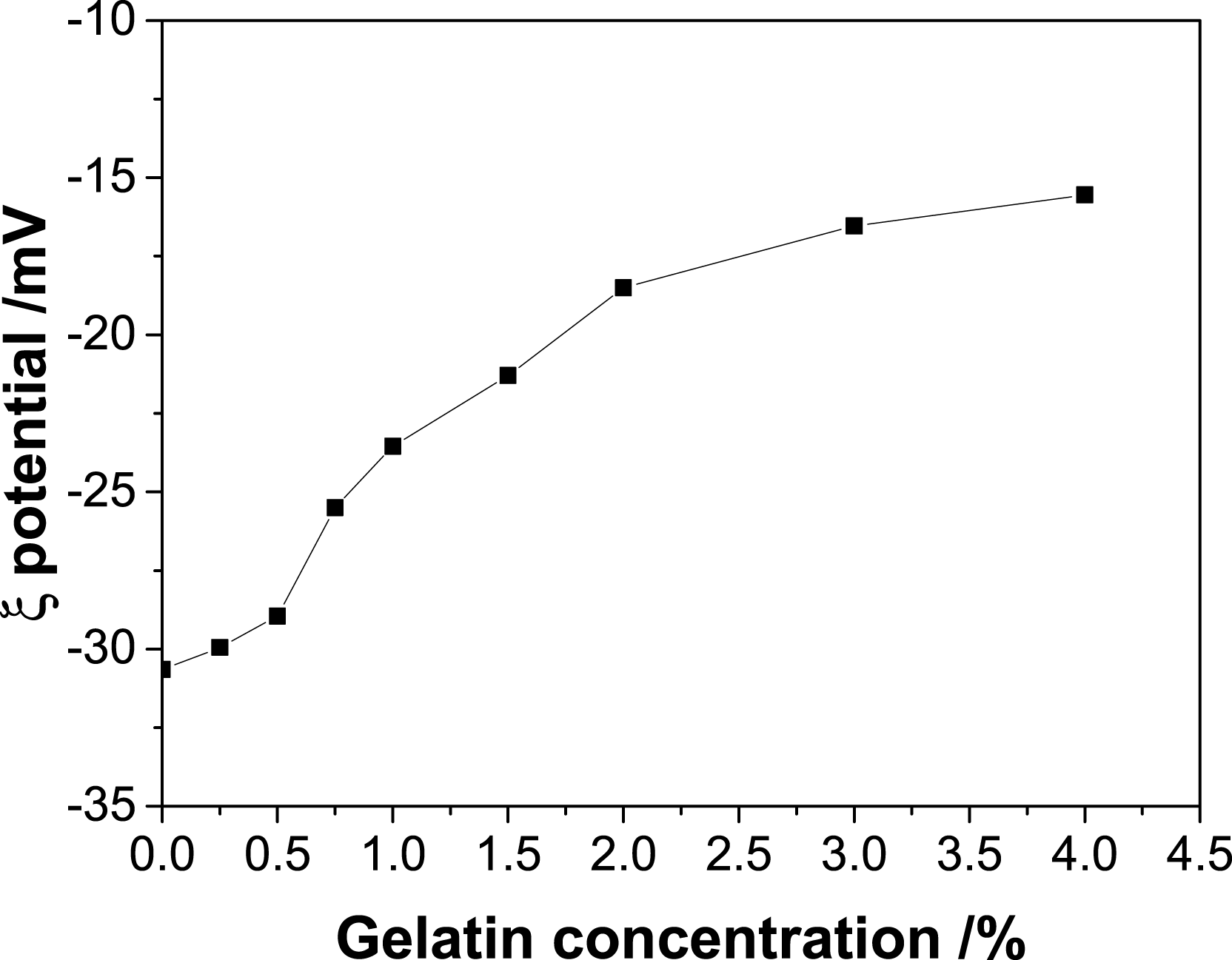
Rheological analysis
The rheological properties of Mt/gelatin dispersions were measured to investigate the interaction between Mt and gelatin. Generally, Mt plates can be linked together via face-to-face (FF), edge-to-edge (EE), and edge-to-face (EF) attractions, which can lead to stability or flocculation of Mt particles. 54 The EE and EF attractions usually form a three-dimensional network structure, which is responsible for the viscosity of Mt dispersions. The FF attraction can result in the formation of thick Mt agglomerations and reduce the dispersion of Mt particles. Different solution environments, such as pH, temperature, electrolyte concentration, and other additives, can change the attraction manners of Mt layers and lead to different colloidal properties.
Figure 9 showed the plots of “ture” shear viscosity versus shear rate for Mt/gelatin dispersions containing different concentrations of gelatin. As shown in Figure 9(a), before being vigorously mixed, there were two steps of the viscosity changes: when the addition of gelatin was below 1.0%, the viscosity of Mt dispersion decreased with the increase of gelatin concentrations. However, further increasing the gelatin concentration to 2.0–4.0%, the viscosity increased. But the viscosity of Mt/gelatin dispersions is almost always lower than that of pure Mt dispersion. The microscopic images of the flocs in Figure 3 and the particle size distribution in Figure 4(a) proved that gelatin could adsorb onto Mt particles and encapsulate them, inducing the aggregation and settlement of Mt particles. Thus, compared with that of pure Mt dispersion, the viscosity of Mt/gelatin dispersions with low concentrations of gelatin (0.25–1.0%) decreased. Whereas, when the concentration of gelatin was as high as 2.0–4.0%, excessive gelatin would provide a certain viscosity to the Mt/gelatin dispersion, resulting in a certain increase of the viscosity of Mt/gelatin dispersions. Viscosity curves of Mt/gelatin dispersions with different concentrations of gelatin before (a) and after (b) being vigorously mixed.
After being vigorously mixed (Figure 9(b)), pure Mt dispersion exhibited a typical shear-thinning rheological behavior in the whole shear rate range. When gelatin was incorporated, the viscosity of Mt dispersion increased in different degrees, and a more pronounced shear-thinning behavior was obtained. When the addition of gelatin was below 1.0%, the viscosity of Mt dispersion increased limitedly with the increase of gelatin concentration. Further increasing of gelatin concentration to 2.0–4.0%, the viscosity dramatically increased again. The particle size in Figure 4(b) showes that decreased particle size of Mt at gelatin concentration of 2.0–4.0% was also proved the variation of the rheological properties. Because of the molecular structure and a large number of hydroxyl groups, gelatin had a strong gel formation capacity. 20 A large number of water molecules could be bounded at the vicinity of gelatin via the hydrogen bond effect. With a high gelatin concentration, gelatin molecules decreased the Mt particle size. A stiff network among Mt layers, gelatin, and water molecules was created, which had a strong resistance to flow under shear force, 55 leading to a significant improvement in the rheological properties.
Conclusion
Clay/gelatin composites have been studied as functional materials in various applications. In this paper, basic colloidal characteristics of gelatin-treated Mt were investigated and analyzed in detail. The following conclusions were obtained: (1) The structural examination of Mt that was organically modified by various concentrations of gelatin, as revealed by XRD, showed that gelatin intercalated into the interlayer space of Mt particles and the conformation of gelatin inside the interlayer space strongly depended on the amount of inserted gelatin. (2) Electron microscopy and particle size distribution experiments revealed that aggregation occurred after the addition of gelatin and the particle size of Mt gradually increased with increasing the concentration of gelatin. However, after continuous string vigorously, the Mt aggregates will be broken up and the particle size decreased. (3) The zeta potential measurements demonstrated that gelatin showed higher zeta potential than Mt and had an isoelectric point at around pH 5. The addition of gelatin decreased the zeta potential of Mt. But it was insufficiently strong to reverse the charge of Mt from negative to positive. (4) Gelatin could improve the hydrophobic property of the Mt membrane through adsorbing on the surface of Mt particles and making the membrane smooth. (5) The rheological properties of Mt dispersion were influenced by the amount of gelatin. 2.0–4.0% gelatin can significantly increase the viscosity of Mt.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Foundation of China (Grant No. 51874329), and the National Natural Science Innovation Population of China (Grant No.51821092).
