Abstract
Unmodified natural rubber is not suitable for any elstomeric applications. Therefore, it is appropriate to modify natural rubber chemically to enhance the stability, which can be termed as vulcanization. Incorporation of fibers/fabrics is a common method to increase the stability of natural rubber along with chemical modification. Natural rubber-based composites have been prepared by the addition of silk fabric into natural rubber. The matrix material for the composite is glutaraldehyde cured natural rubber. Silk is an ecofriendly and biodegradable material with excellent tensile strength. When such kind of fabric is introduced into the vulcanized rubber as the matrix, all the physical properties were found to be enhanced considerably. Tensile properties in terms of ultimate tensile strength, elongation at break, and modulus of elasticity are measured for the composites of natural rubber/silk fabric at various glutaraldehyde concentrations. Thermogravimetric analysis and temperature scanning stress relaxation techniques are employed to evaluate the thermal stability of the resulting composites. Effects of glutaraldehyde addition on the physical properties of the composite were studied in detail. Considerable enhancement in the stability of natural rubber in terms of tensile properties, thermal stability, and solvent resistance is noticed up on the incorporation of silk fabric as well as glutaraldehyde curing.
Introduction
Natural rubber (NR) is an elastomeric material collected from natural rubber trees. Uncured natural rubber cannot be used for any applications due to its low thermal stability and it must be chemically treated to improve the stability. The processes of chemical modification using different ingredients have been reported.1,2 Generally, natural rubber is vulcanized by using sulfur, peroxides and radiation. Sulfur and peroxide vulcanization were found to be toxic and radiation vulcanization is expensive. A small amount of glutaraldehyde (GA) effectively cross-links the rubber and it immobilizes allergenic proteins present in natural rubber latex. 3 The formation of linkages between natural rubber molecules occurs during vulcanization.
Recently, it was reported that, glutaraldehyde can be used to cure NR as an effective cross-linking agent for NR in latex stage at low temperature.4,5 The cross-linking reaction of NR with glutaraldehyde was accomplished in two steps. The first step involved the creation of pentane-1,5-deylidenediamine by allowing glutaraldehyde to react with the ammonia. The second step was the formation of cross-links via an “ene” reaction between molecules of NR with pentane-1,5-deylidenediamine. Considerable enhancement in the stability of rubber in terms of physical and chemical properties was also noticed more than for other cross-linkers. The thermal stability of glutaraldehyde cured natural rubber was found to be better compared to sulfur cured rubber. 6
Short silk fibers were used as reinforcements for elastomeric rubber composites. 7 Silk fiber–reinforced polymers were found to be weak in terms of stiffness, flexural, and tensile strength, which was comparable to plant fiber reinforced polymers, but much below that of glass fiber–reinforced polymers.
Some natural fibers have relatively high mechanical properties with modulus close to that of high-performance synthetic fibers. Pineapple leaf fiber has modulus almost like aramid fiber, which can be used to reinforce for natural rubber effectively than aramid fiber. 8 Modified silk has been blended with natural rubber and it exhibited better physical properties than that of unmodified silk composites. 9 Due to chemical modifications, the silk composites showed changes in chemical and dominant secondary structures.
Hybrid composites of jute, sisal, and E-glass fabrics reinforced natural rubber/epoxy with particulate filler tungsten carbide powder enhanced the mechanical and tribological properties. 10 Composites of silk fabric–reinforced poly butylene succinate were fabricated using the hot-press technique. Tensile, flexural, and dynamic mechanical properties of poly butylene succinate composite with various silk contents were increased considerably. 11 Carbonized waste cotton fabric was reinforced into natural rubber for flexible sensor applications. 12
Green composites of natural rubber with short jute fiber as reinforcement were produced by twin roll mill method to study the dynamic mechanical properties and swelling properties. 13 It was noted that addition of fibrous fillers resulted in increasing of tensile moduli, stiffness, and elongation along with hardness.
Composites were prepared by sandwiching a layer of sisal fabric between two layers of rubber sheets were subjected to various chemical modifications like mercerization, silanation, and heat treatment. 14 Thermally treated composites exhibited superior mechanical properties because of increased crystallinity and chemically modified composites exhibited high hardness values.
The reinforcements like reused sugarcane bagasse fiber and untreated/alkaline treated filler were used in natural rubber to produce composites that were used for manufacture of sandals. 15 The alkali treatment decreased the hardness and increased the tensile strength by maintaining the flexibility of the rubber composites.
Silk fiber contains many reactive functional groups like NH2, -NHCO, and –CH2OH -COOH on its surface. Addition of silk fiber to natural rubber causes increase in hardness, reduction in resilience, elongation, increase in heat buildup and set, and increase in tear resistance. 16 Woven and nonwoven silk fibers were reinforced with natural rubber with considerable flexural and tensile strength. 7 Higher fracture strain capacities were seen in woven silk natural rubber composites than in flax and glass composites. Further, these composites show deficient physical, mechanical, and soaring moisture sensitivity. 17 Tensile strength, tensile modulus, bending strength, bending modulus, impact strength and hardness of the natural silk fiber–reinforced polypropylene composites prepared composite were studied. 18 Studies revealed that the mechanical properties of the composite decrease with increasing natural rubber–reinforced polypropylene composites. The composites were prepared by sandwiching a single layer of silk textile between layers of natural rubber. Silk reinforced natural rubber composites exhibited higher static and dynamic mechanical properties than nylon-reinforced natural rubber composites. 18 The silk fabric had a higher areal density than the nylon fabric–reinforced natural rubber.
Composites of natural rubber reinforced with untreated coir fiber chopped to different lengths were prepared by vulcanization method.19–21 The rubber coir interface bonding was improved by the addition of a resorcinol-hexamethylenetetraminedry-bonding system. Short silk fibers were incorporated as reinforcements for (a) thermoplastic polymers or (b) elastomeric rubbers. Further, woven composites showed performances better than nonwoven composites.
Development of elastomeric composites is very essential in the field of rubber technology to fulfill the flexible material applications effectively. Natural fibers are attractive choice towards the development of composites by incorporating in to natural rubber. The resulting composite material can be termed as green composites due to its natural origin and biodegradability. In the present investigation, silk fabric has been selected as reinforcement material and incorporated for cross-linked rubber Proposed vulcanization mechanism of natural rubber via ene reaction.
3

Materials and methods
Materials
Concentrated medium ammonia (0.47%) natural rubber latex with approximately 60% dry rubber content was purchased from Shilpa Latex products Ltd. Bengaluru, India. 25% Glutaraldehyde solution (C5H8O2) in H2O with molecular weight 100.12 and benzene to carry out swelling studies were supplied by MERCK India. Silk fabric with density of 1.38 g/cm3 was obtained from Central Silk Board of India, Bengaluru. The silkworm cocoon having single fiber consists of two filaments or brins of fibroin strands surrounded by a glue-like protein called sericin, which fixes the fibroin fibers to each other. Single continuous silk strand of length ranging from 1000 to 1400 m contributes 78–80% and the rest is removed as waste. This silk strands (protein layer) resists oxidation, is antibacterial, ultra-violet (UV) resistant, absorbs, and releases moisture easily. 22 Since protein layer can be cross-linked, copolymerized and blended with other macromolecular materials (especially artificial polymers) can be used to produce materials with improved properties.
Methods
Preparation of vulcanized natural rubber
A 10% glutaraldehyde solution was prepared by diluting 25% glutaraldehyde solution in distilled water. Separate specimens of silk fabric (1.5 g) are made by stretching them to a circular ring casing (Figure 1) and the setup is placed in a petri dish. Known quantities of glutaraldehyde (GA) and natural rubber latex were mixed physically in a beaker and the mixture was then poured into the petri dish to obtain cured composite specimens of silk fabric reinforced vulcanized natural rubber. The composites of silk fabric reinforced with respect to various amounts of glutaraldehyde into 100 mL natural rubber (60% dry rubber content) have been prepared separately to optimize the composition and conditions of curing. Optimization had been done by varying the amount of glutaraldehyde (0 ml–14 mL) as the cross-linking agent for rubber. This setup was placed under electric bulb for 24 hours for curing. These casted samples were then peeled out from the casing as shown in Figure 1. Composites prepared using glutaraldehyde with silk fabrics in natural rubber.
Attenuated total reflectance - fourier transform infrared spectroscopy (ATR - FTIR)
The formation of cross-linking was confirmed by using a Bruker (Model Tensor 207, Germany) FTIR. The spectra were recorded on transmittance mode up to 4000 cm−1 with 32 scans at a resolution of 4 cm−1. ATR-FTIR was performed only for the composites with 0 mL and 8 mL GA to confirm the cross-linking reaction.
Mechanical properties
Tensile testing of the vulcanizates was performed according to the ASTM D412 using universal testing machine (Model H10KS, Hounsfield, England). The samples were cut into dumbbell-shaped specimens and the test was carried out with a crosshead speed of 500 mm/min at room temperature. The mechanical properties in terms of modulus, tensile strength, and elongation at break were reported.
Theromogravimetric analysis (TGA)
Thermogravimetric analyzer was adopted to evaluate the thermal stability of the samples. Thermal analysis was performed using a Perkin Elmer (USA) TGA. A small amount (1–10 mg) of sample was taken in a crucible and placed inside the machine from 40oC to 800oC at a rate of 10oC/min in oxygen atmosphere with flow rate of 10 mL/min.
Thermo-mechanical properties
Thermo-mechanical properties and cross-link density of the silk fabric–reinforced NR were analyzed by using a novel temperature scanning stress relaxation technique (TSSR) (Brabender GmbH Duisburg, Germany). Cured composites were prepared according to the ISO527 type 5B. The specimen was placed in the electrically heated test chamber and pre-conditioned at 100% strain at room temperature for 2 hours. Further, the non-isothermal tests were performed by raising the temperature with a constant rate of 2°C/min until the sample ruptures. The initial forces from TSSR results were obtained from the relation of normalized force-against temperature. It is noted that the Tx indicates the temperature at which the force has been reduced to x%, compared with initial force. T90 describes the thermal resistance of the material. 23
Solvent transport properties
Circular specimens of diameter 20 mm were used for swelling studies. Specimens of known weight were immersed in benzene as the solvent in diffusion test bottles and kept at room temperature. Samples were removed from the bottles at periodic intervals and the wet surfaces were quickly dried using tissue paper and weight of the specimens after swelling were determined at regular intervals until no further increase in solvent uptake. The solvent uptake Qt (%) of the samples was computed using the equation
Results and discussion
Vulcanization mechanism of natural rubber in the presence of glutaraldehyde is as shown in Scheme 1.
3
Ammonia present in the latex initially reacts with the glutaraldehyde and further the reaction product Pentane-1, 5-diylidenediamine reacts with natural rubber molecules. It forms linkages between the rubber molecules and the reaction can be termed as ene reaction. FTIR spectra of pristine and cured natural rubbers are depicted in Figure 2 Fourier transform infrared spectrosco spectrum of pure and vulcanized natural rubber.
Stress–strain curve provides the information about the nature of deformation of any material. Figure 3 shows the stress–strain plots of silk fabric–reinforced natural rubber composites with various amounts of curing agent. Uncured composite exhibits higher elastic properties than that of other cured composites in the presence of GA. As increasing the amount of GA, the formation of three-dimensional networks increases the stability of the composite. Thus, the tensile strength increases and the elongation at break decreases by increasing the modulus of elasticity. 12 mL GA cured composite shows a considerable tensile property among the series of the sample prepared. Stress-Strain plots for (a) 0 mL glutaraldehyde (GA), (b) 2 mL GA, (c) 4 mL GA, (d) 6 mL GA, (e) 8 mL GA, (f) 10 mL GA, (g) 12 mL GA, (h) 14 mL GA cured composites of Silk reinforced Natural rubber (NR).
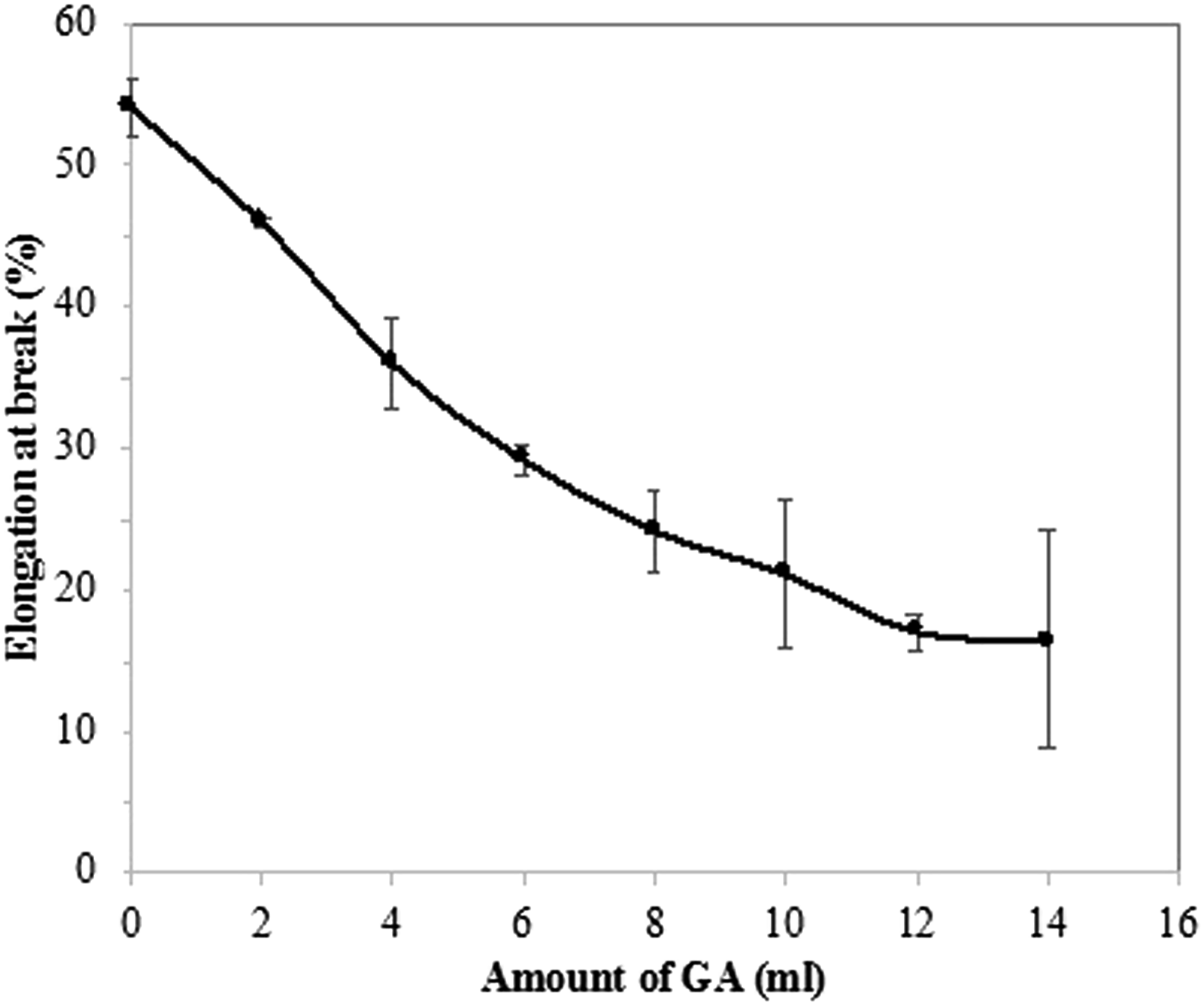
The values of ultimate tensile strength of the composites are depicted in Figure 4. A gradual increase in tensile strength is observed on increasing the amount of GA in the composite. Uncured natural rubber has poor tensile strength and higher elongation at break. Incorporation of curing agent enhances the cross-linking density, which causes the increased ultimate tensile strength of natural rubber. Similarly, silk fabric incorporated in natural rubber provides better tensile strength to NR. Vulcanized natural rubber interpenetrated into the silk fabric and the fabric could be tightly bounded within the cured rubber. Hence, the resulting composite exhibits better stability when compared to unmodified composite. As a result, the tensile strength increases upon the addition of GA up to 12 mL. An increase of 170% in the tensile strength can be seen on the addition of 12 mL GA when compared to the uncured one. Further, the reduction in the strength can be seen, which is attributed by the reduction in cross-linking density of the matrix phase. Figure 5shows elongation at break of pure and cured composites for various amounts of GA. Cured natural rubber cannot be easily stretched under stress. Uncured natural rubber has higher entropy and shows more elongation as its poor interaction within their molecules. Cross-linking increases with the addition of GA and reduces the elongation of the NR composites due to interfacial interaction between the two phases. A decrease of 65% in the elongation is observed up on the addition of GA in the composites. The composite with GA above 12 mL shows no further change in the elongation. Variation of ultimate tensile strength with the amount of GA for composites of Silk reinforced NR. Variation of elongation at break with the amount of GA for composites of Silk reinforced NR.
Thermogravimetry is an effective technique to evaluate the thermal stability of the elastomeric composites. Thermogravimetric curve shows the weight loss with temperature and it provides the information about the decomposition mechanism for different amounts of GA with silk fabric–reinforced natural rubber as depicted in Figure 6. The weight loss occurs in the composites in three stages. The first stage of weight loss ranges between 295oC and 350oC, which is attributed to the evaporation of volatile compounds present in the material. The decomposition in this stage mainly involved dehydration of hydroxyl groups, followed by the formation of volatile organic compounds. The major weight loss observed in the second stage is due to the decomposition of rubber molecules. Thermal cross-linking takes place in rubber in the initial stage of weight loss and further, these cross-linked rubber molecules undergo scission. A drastic reduction in weight can be seen in the TGA thermogram during the second stage of weight loss (350oC–395oC). In this stage rubber gives 39% isoprene, 13.2% dipentene and a small amount of p-methane.
24
Dipentene and isoprene are the major decomposition products in the third stage of weight loss ranges in between 395oC to 600oC. It has been reported that decomposition of silk starts at 280oC and ends at 308oC.
24
Theromogravimetric analysis curves for silk fabric reinforced rubber with different amounts of GA.
Temperature at various weight losses for different amount of glutaraldehyde.

Recently, TSSR test method has been developed by Vennemann et al.
25
to characterize the thermal–mechanical properties of elastomer by evaluating the stress relaxation as a function of temperature with a constant heating rate. In order to verify the cross-link density of the silk fabric–reinforced NR, the test was performed by isothermal and non-isothermal conditions. At constant stretching ratio (λ = L/L
0
), the cross-link density of elastomeric polymer can be evaluated from the slope of the normalized force-temperature curves from TSSR measurement. The cross-link density (
The cross-link density is related to average molar mass of the network chains as
Figure 7 shows the normalized force-temperature curves which evaluate the initial stress, thermal stability, and cross-link density of the silk fabric–reinforced NR samples as summarized in Table 2. It was found that the silk fabric–reinforced NR without GA shows the lowest cross-link density. This might be due to no chemical cross-linking of the NR molecules in the system at the same time occurred the physical cross-linking or the interfacial interaction between NR molecules and silk fabrics. In addition, cross-link density increases with the addition of GA content from 87.13 mol/m3 for uncured sample to 530.10 mol/m3 for 14 mL GA cured sample. Increase in cross-link density of silk fabric reinforced NR can be verified by the formation of cross-links between pentane-1, 5-diylidenediamene and NR molecules. The thermal stability of silk fabric–reinforced NR can be evaluated from TSSR measurement. The measurement provides T10, T50, and T90 values which indicate the force has been reduced to 10, 50, and 90% compared with the initial force.
27
T90 describes the thermal resistance of the silk fabric–reinforced NR as summarized in Table 2. It is observed that the silk fabric–reinforced NR vulcanizates using GA exhibited an increased T90 compared to the uncured composite. According to the highest tensile strength of the 12 mL GA sample, the highest T90 of 12 mL GA is observed. This correlates well with the increased cross-link density upon the addition GA. The normalized force as a function of temperature of silk fabric reinforced NR composites using GA as curing agent with different amounts of GA. The degradation temperature and cross-link density obtained from the TSSR of silk fabric reinforced natural rubber with different amounts of GA. GA: glutaraldehyde; TSSR: temperature scanning stress relaxation technique.

Furthermore, the relaxation phenomenon of the silk fabric reinforced NR can be studied using the relaxation spectrum (H(T)) obtained from TSSR measurement. The relaxation spectrum can be calculated using the equation (4), where the relaxation modulus (E (T)) as a function of temperature at constant heating rate
26

The relaxation spectrum as a function of temperature in the composites with different amounts of GA is as shown in Figure 8. Two peaks are clearly visible in the relaxation spectrum. The first peak at 25–100°C indicates the physical interaction of silk fabric with NR molecules, including molecular chain relaxation and physical interaction of non-rubber components.
27
The second peak at 125–200°C is associated with thermo-oxidative degradation of GA-rubber chains. For 12 mL and 14 mL GA cured samples, the peak with higher intensity are observed in the first period when compared to others. This might be due to increased interfacial interaction between NR molecules and silk fabrics on increasing GA content. Considering the second peak, the 12 mL GA sample shows highest intensity. This correlates well with the tensile strength and cross-link density results, which clearly clarifies the superior linkage formation of GA-rubber cross-links and the also condensation reaction between carbonyl groups present in silk fabrics and GA molecules.
28
Above 12 mL of GA, the excess intermediate compound which vulcanizes natural rubber aggregates and the cross-link density is found to be reduced. Consequently, the thermal stability of the composite reduces on exceeding 12 mL. The relaxation spectrum as a function of temperature of silk reinforced NR composites using GA as curing agent with different amounts of GA.
Solvent transport studies have been carried out in benzene as a solvent. The transport of small molecules through the elastomers depends on several factors such as the cross-link density, degree of unsaturation, presence of fillers, time, etc. The variation in solvent uptake as a function of time for different composites with various GA is shown in Figure 9. Swelling experiments cannot be conducted in case of uncured NR composites due to its dissolution nature in the solvent. Solvent sorption in the cured composites occurs in four stages. The existence of concentration gradient of the solvent molecules resulted in severe solvent stress on the matrix phase occurs in the first stage of absorption. In the second stage, an increase in the rate of transport is noticed due to the relaxed elastomeric molecular chains and further the rate is found to be reduced. An equilibrium uptake is observed in the fourth stage attributed to the penetrant uptake reaches a saturation state. Upon increasing the amount of curing agent, there is a gradual decrease in the absorption of solvent. Formation of cross-links between the NR molecules on the addition of GA reduces the solvent transport and the swelling resistance enhances as the amount of GA increased in the composite. As a natural fiber, silk does not interact with hydrocarbon solvents like benzene. Therefore, the presence of silk fabric minimizes the solvent sorption through the cured NR composites. Solvent transport curves for silk fabric reinforced rubber with varying amount of GA.
The diffusion rate of a solvent molecule is associated with the penetrant size and the mobility of the elastomeric segments. The diffusion coefficient “D” of our elastomeric samples immersed in the benzene solvent was determined using the following equation
29

Diffusion coefficient values for various amounts of GA.

GA: glutaraldehyde.

Conclusion
In the present paper, the possibility of reinforcing an ecofriendly silk fabric for cured natural rubber using glutaraldehyde as cross-linking agent has been studied. The preparation of the composite is a novel method and cost-effective compared to other conventional methods. The study reveals that addition of glutaraldehyde into natural rubber/silk fabric composite is found to be worth in view of developing material with excellent stability. Enhancement in tensile strength, thermal stability, and solvent resistance of the resulting composites suggests the material can be used in high-performance elastomeric applications.
Footnotes
Acknowledgments
Authors express thanks to the Principal and Management, RajaRajeswari College of Engineering for the encouragement and moral support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

