Abstract
Polymer-supported reagents have become popular in synthetic organic chemistry over the past decades. But the kinetics of polymer-supported reactions is slow compared to solution phase synthesis because of the poor diffusion of the reactants through the macromolecular polymer matrix. This difficulty can be reduced to a great extent by performing polymer-supported reactions under microwave (MW) conditions. The present work is focussed on the design and development of an innovative, powerful, MW stable and recyclable polymeric reagent prepared by attaching bromoderivative of 2-oxazolidone into the macromolecular matrix of polystyrene. 3% cross-linked polystyrene was prepared by free radical aqueous suspension polymerization technique using tetra ethylene glycol diacrylate as the cross-linking agent and the resulting beads were functionalized by chloromethylation followed by reaction with 2-oxazolidone. Bromine functionality is introduced into the polymer by treating with bromine in carbon tetrachloride. The synthetic utility of the prepared polymeric reagent was demonstrated by the oxidative coupling of thiols to disulfides under MW irradiation. No over oxidation was observed in this protocol and the utilization of polystyrene support simplifies work up and product isolation. The synthesised polymeric reagent displayed good cyclic stability up to five cycles without any substantial decrease in bromine content and satisfactory storage stability under normal laboratory condition. Moreover this may be the first report that uses MW energy for the oxidation of thiols to disulfides using polymer-supported reagents.
Keywords
Introduction
Compounds having sulphur–sulphur (S–S) bonds are one of the most valued organic compounds in synthetic organic chemistry. The disulfide bonds have a critical role in biology and chemistry.1,2 In peptides and bioactive molecules, these disulfide bond formation is very important as they stabilize the folded peptides and the secondary and tertiary structure of proteins.3,4,5 In various biologically important molecules namely peptides, enzymes, oligonucleotides, amino acids and nucleic acids, the S–S bonds act as structural backbones. Jiang et al. reported the presence of metabolites having disulfide as well as multisulfide bonds in marine organisms. 6 Glutathione disulfide and cystine are two familiar examples of molecules having S–S bonds that are significant from a biological viewpoint. 7 Ellman’s reagent, 5, 5′-dithio-bis (2-nitrobenzoic acid) used to estimate the free thiol groups in proteins is another example of a biologically valuable molecule containing disulfide bonds. 8 Diallyl disulfide present in garlic has excellent antimicrobial, larvicidal and fungicidal properties. 9 Studies also show that it can prevent the growth of leukaemia cells. 10 Ayodele et al. reported the fungicidal activity of benzyl disulfides. 11 In industry, the disulfides have wide applications such as vulcanizing agents for rubbers 12 and in the design of lithium-ion batteries.13,14 These compounds have a key role in synthetic organic chemistry as they are used for the synthesis of heterocycles.15,16 Since compounds having disulfide bonds show a greater stability to organic reactions compared to the corresponding thiols, the thiol groups can powerfully be protected as disulfides. 17
Because of the various uses of disulphides, scientists are in quest of new methods to synthesise these compounds. Although the synthesis of disulfides from thiols may become challenging due to over oxidation of thiols to sulphoxides and sometimes to sulfones, still the oxidative coupling of thiols is the best and modest way for the synthesis of organic disulfides. Various research groups reported the synthesis of disulphides from thiols using different reagents such as CoSalen, 18 ammonium persulphate, 19 monochloropoly (styrenehydantoin) beads, 20 cetyltrimethylammonium dichromate, 21 solid anhydrous potassium phosphate, 22 silica , 23 potassium permanganate, 24 dicarboxy pyridinium chlorochromate, 25 nanocatalysts,26,27,28 nano organometal catalysts, 29 thiolate-bridged iron–ruthenium complex, 30 1.3‐diisopropylcarbodiimide 31 and polymer-supported reagents.32,33,34
The microwave-assisted (MW) synthesis of disulfides was also reported by several research groups. Ghammamy S and Tajbakhsh M described the synthesis of disulphides using tripropylammonium chlorochromate under MW irradiation. 35 Mohammadi et al. reported the MW-assisted synthesis of symmetrical disulfides using tripropylammonium fluorochromate. 36 Another work described the solvent free synthesis of disulfides with tributylammonium halochromates. 37 Solvent- and metal-free selective oxidation of thiols to disulfides was developed by Bettanin L and co-workers using I2/DMSO catalytic system. 38 However, the existing literature in the field of oxidative coupling of thiols using polymer-supported reagents under MW condition is rare.
Since MW couples directly with the molecules in the reaction mixture, the rate of the reaction enhances rapidly. The distinctive features of MW-assisted organic synthesis are smaller reaction time, high pureness and yield of the product, dry reaction condition and ease of separation of the product. 39 While the capacity of a substance to transfer MW energy into heat is given by the dielectric loss factor (ε’’), the dielectric constant (ε′) determines its capability to absorb MW energy. Those materials with high dissipation factor (tanδ = ε’’/ε′) are more susceptible to MW energy. 40 Out of the various polymers, polyethylene, polytetrafluoroethylene and polystyrene are having high dielectric properties.41,42 and polystyrene is the best in terms of mechanical property such as rigidity.
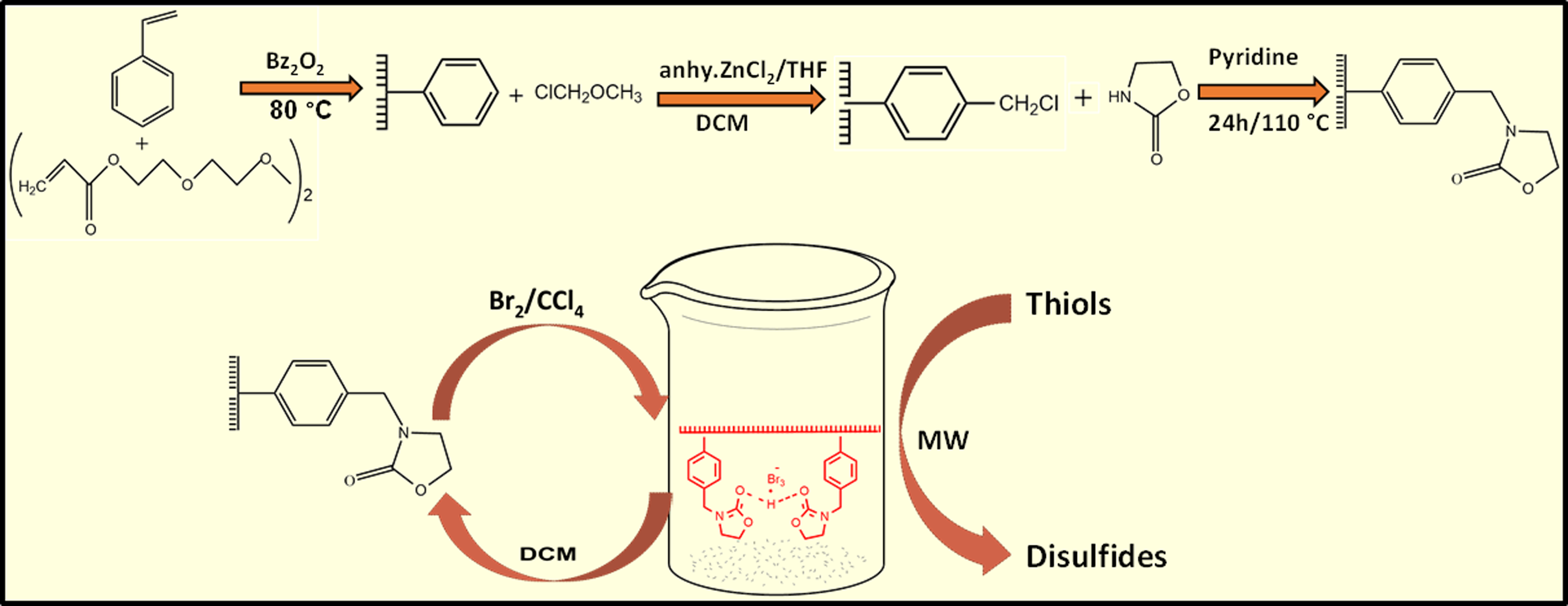
With this background, we wish to disclose the synthesis and full characterization of a new polymer-supported reagent, 3% tetra ethylene glycol diacrylate (TTEGDA) cross-linked polystyrene supported bromo derivatives of 2-oxazolidone (TEGDA-PS-OX-Br) and its efficiency towards the oxidative coupling of thiols to disulfides under MW irradiation.
Materials and methods
The chemicals TTEGDA (C14H22O7: 302.32 g/mol), styrene (C8H8: 104.15 g/mol) and 2-oxazolidone (C3H5NO2: 87.07 g/mol) were purchased from Sigma Aldrich, Germany. The monomer styrene was washed with a 1% NaOH solution (5 mL × 3) and with distilled water (5 mL × 3) to remove the inhibitor. TTEGDA and 2-oxazolidone were used without any additional purification. Fourier transform infrared spectrophotometer (FTIR) spectra of the resins were scanned on a Perkin Elmer (USA) and the measurements were done in the range 4000–4450 cm−1. The size and the morphology of the synthesized resin were examined by scanning electron microscope (SEM) JEOL JSM-6390 (SEM) (Japan). The thermal stability of the synthesized resin was found out by thermogravimetric analysis using a Perkin Elmer, Diamond TG/DTA instrument (USA) heating from 30 to 600°C at a rate of 10°C/min in nitrogen atmosphere. The presence of nitrogen and bromine in the sample was confirmed by Energy Dispersive X-ray (EDX) spectroscopy using JEOL JSM-6390 SEM with EDX attachment (Japan). CHN analysis was carried out using Elementar Vario EL III analyser (Germany). The progress of the chemical reaction was observed by thin-layer chromatography (TLC) with Merck precoated silica plates. The products obtained by the oxidative coupling of thiols were characterized by Thermoscientific Trace GC 1300 equipped with mass ISQLT Single Quadrupole Mass Spectrometer (USA). The spectrum was recorded in EI mode. A single-mode MW synthesis reactor – Anton Paar Monowave 300 (USA) is used for carrying out MW-assisted chemical reaction.
Preparation of polystyrene supported 2-oxazolidone resin (TTEGDA-PS-OX)
Three% TTEGDA cross-linked polystyrene (TTEGDA-PS), its chloromethylated derivative (TTEGDA-PS-Cl), and chloromethyl methyl ether (CMME) were prepared using the reported procedures.43,44,45 To a suspension of chloromethylated resin (2 g), 1 g oxazolidone and 1 mL pyridine were added. The mixture was refluxed for 24 h at 110°C. The product was filtered and washed with acetone and methanol. The resin was again washed in a soxhlet apparatus with methanol as solvent and dried at 80°C and designated as TTEGDA-PS-OX. Yield: 2.4 g.
Preparation of bromo derivatives of polystyrene supported 2-oxazolidone (TTEGDA-PS-OX-Br)
To a suspension of the resin (TTEGDA-PS-OX, 2 g) in CCl4 (10 mL), Bromine (2 mL) was added and stirred at 0°C for 4 h. 45 The resultant resin was filtered and washed with CCl4 to get a stable dark orange non-hygroscopic product that was dried in vacuum at 60°C and designated as TTEGDA-PS-OX-Br. Yield: 3 g.
Estimation of bromine capacity of TTEGDA-PS-OX-Br
Approximately 100 mg of polymer TTEGDA-PS-OX-Br was weighed and dispersed in 20 mL ice-cold dimethylformamide taken in an iodine flask. To this suspension, 1g potassium iodide was added and kept in dark with occasional shaking. The liberated iodine was titrated against std. (0.05 M) Na2S2O3 solution until the disappearance of the brown colour of the solution. This was repeated two or three times until all the complexed halogen has reacted. From the titre value, the bromine capacity was calculated and found to be 2.60 mmol/g of the polymeric reagent. 46
Determination of the stability of TTEGDA-PS-OX-Br under Microwave condition
The stability of TTEGDA-PS-OX-Br resin towards MW irradiation was investigated by heating 1 g of the resin at 100°C in an Anton Parr Monowave 300 MW synthesizer at a MW power level of 300 W for 5 min. After each minute, 0.1 g of the resin was taken out, and its bromine efficiency was determined by iodometric titration. 47
Microwave-assisted oxidative coupling of thiols using TTEGDA-PS-OX-Br
A fivefold molar excess of TTEGDA-PS-OX-Br (1 g) was impregnated with a solution of the thiol (0.2 g) in DCM (1 mL). The solvent was evaporated and the polymeric reagent with the adsorbed substrate was MW irradiated for 5 min at an MW power of 300 W in an Anton Parr Monowave 300 MW synthesizer. Impregnation of the polymeric reagent with low molecular weight substrate was repeated after every 1 min of MW irradiation, by adding dichloromethane followed by evaporation of the solvent. The extent of conversion of the substrate to the product was followed by TLC with Merck precoated silica plates. After 5 min, the insoluble spent reagent was filtered and washed with more solvent. The combined filtrate and washings were dried over anhydrous sodium sulphate. The conversion and selectivity were found out by GC-MS analysis. The above procedure was repeated with different thiols.
Regeneration of the spent reagent
The used resin was washed with dichloromethane (5 × 2 mL) to eliminate the organic substrate or product. The washed resin was dried and the bromine functionality was introduced into the polymer by following the original procedure. The recycled bromo resin was used for the coupling of various organic substrates. 47
Results and discussion
Preparation of TTEGDA-PS-OX-Br
2-oxazolidone was anchored on to TTEGDA cross-linked chloromethylated polystyrene by heating under reflux in the presence of pyridine for 24 h at 110°C. Polystyrene-supported bromo derivatives of 2-oxazolidone resin were synthesized by stirring a suspension of the resin TTEGDA-PS-OX in CCl4 with Br2 on a magnetic stirrer for 4 h at room temperature. According to Koshy et al. in polyvinylpyrrolidone–bromine complex, the bromine functionality exists in the form of a tribromide complex.
48
They suggested that the absorbed water may facilitate the formation of bromide ion which can also complex with Br2 to form tribromide complex. Another group of scientists described the structure of the PVP–iodine complex as a triiodide complex based on X-ray analysis and infrared spectroscopic investigation.
47
Based on these, we propose a tribromide structure for our reagent. Schemes 1–3 illustrate the different steps involved in the synthesis of TTEGDA-PS-OX-Br. Functionalization of TTEGDA cross-linked polystyrene with chloromethyl group. Functionalization of chloromethylated polystyrene support with 2-oxazolidone. Preparation of polystyrene supported bromo derivatives of 2-oxazolidone.


Fourier transform infrared spectrophotometer studies
TTEGDA-PS-Cl and TTEGDA-PS-OX were characterized by IR spectroscopy. The FTIR spectra of TTEGDA-PS-Cl (Figure 1(a)) shows the characteristic absorptions at 1731 cm−1 and 1111 cm−1 due to ester carbonyls and ether linkages of the cross-linking agent. Another band at 2930 cm−1 corresponds to aromatic hydrogen. In addition to these spectral features, the spectrum also shows a band at 685 cm−1 corresponding to C–Cl stretching. The chemical modification of TTEGDA-PS-Cl with 2-oxazolidone has been confirmed by the splitting of the peak at 1737 cm−1 in the IR spectrum of TTEGDA-PS-OX due to merging of amide carbonyl from the oxazolidone unit with ester carbonyl from TTEGDA (Figure 1(b)). The spectrum also shows bands at 1495 cm−1 due to the C–N stretching of the oxazolidone unit. Fourier transform infrared spectrophotometer spectra of (a) TTEGDA-PS-Cl and (b) TTEGDA-PS-OX.
CHN analysis
CHN data of TTEGDA-PS-Cl and TTEGDA-PS-OX.

MERIC REAGENTS IN SOLID PHASE.
Thermogravimetric analysis
The thermal stability of TTEGDA-PS-Cl and TTEGDA-PS-OX was found out by thermogravimetric analysis. Thermal analysis of TTEGDA-PS-Cl reveals that the resin is stable up to 250°C. Above this temperature the elimination of chloromethyl group occurs TGA curves of (a) TTEGDA-PS-Cl and (b) TTEGDA-PS-OX.
Scanning electron microscope analysis
Scanning electron microscope analysis is a potent tool for studying the surface morphology of particles. The scanning electron micrograph of TTEGDA-PS-OX resin is shown in Figure 3. Scanning electron microscope images of TTEGDA-PS-OX under different magnification (a, b).
Energy dispersive x-ray spectroscopy
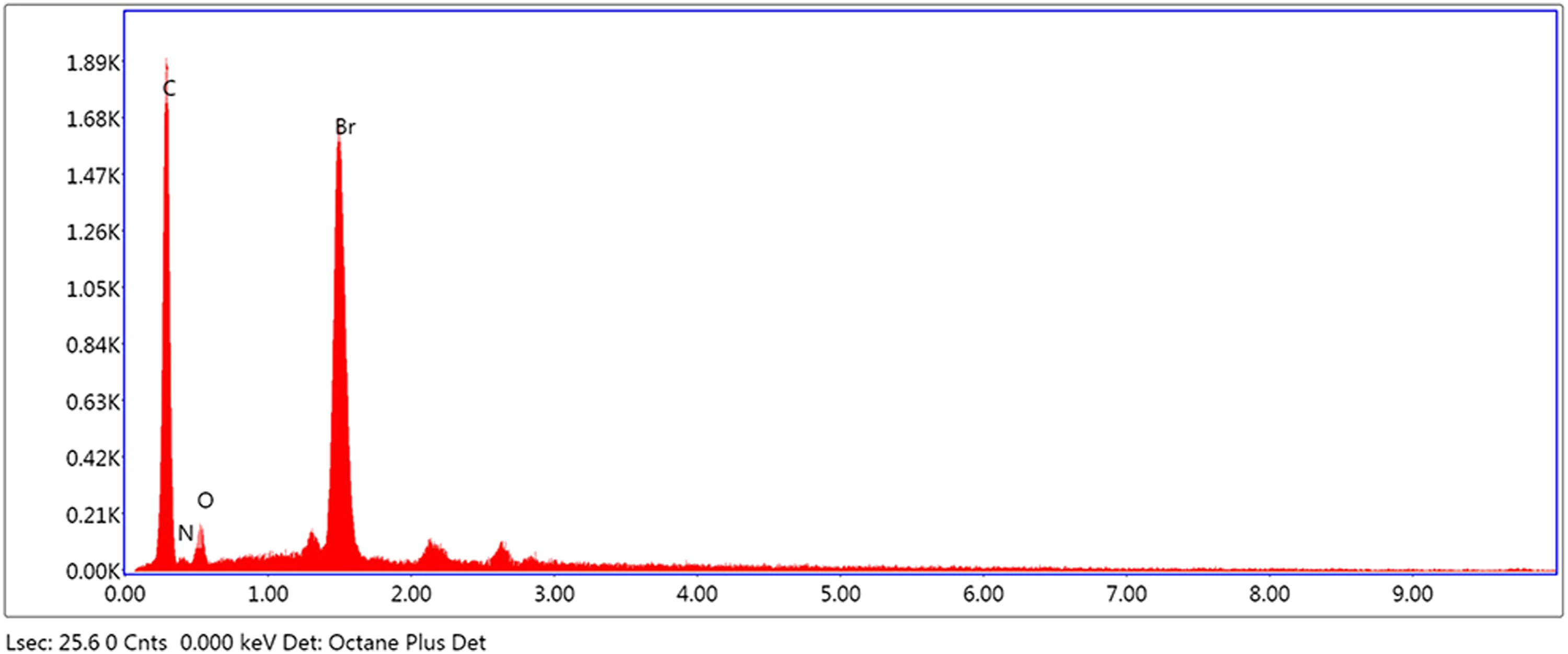
Energy Dispersive X-ray spectroscopy was also used for the characterization of TTEGDA-PS-Cl and TTEGDA-PS-OX-Br. The EDX profile of TTEGDA-PS-Cl displays peaks of carbon, oxygen and chorine at 0.2 keV, 0.5 keV and 2.6 keV respectively (Figure 4). Grafting of bromine and 2-oxazolidone to the chloromethylated resin is established by the appearance of new peaks at 0.4 keV and 1.4 keV corresponding to nitrogen and bromine, respectively, in the EDX spectrum of TTEGDA-PS-OX-Br (Figure 5). Energy dispersive x-ray spectrum of TTEGDA cross-linked polystyrene-chloromethylated. Energy dispersive x-ray spectrum of TTEGDA-PS-OX.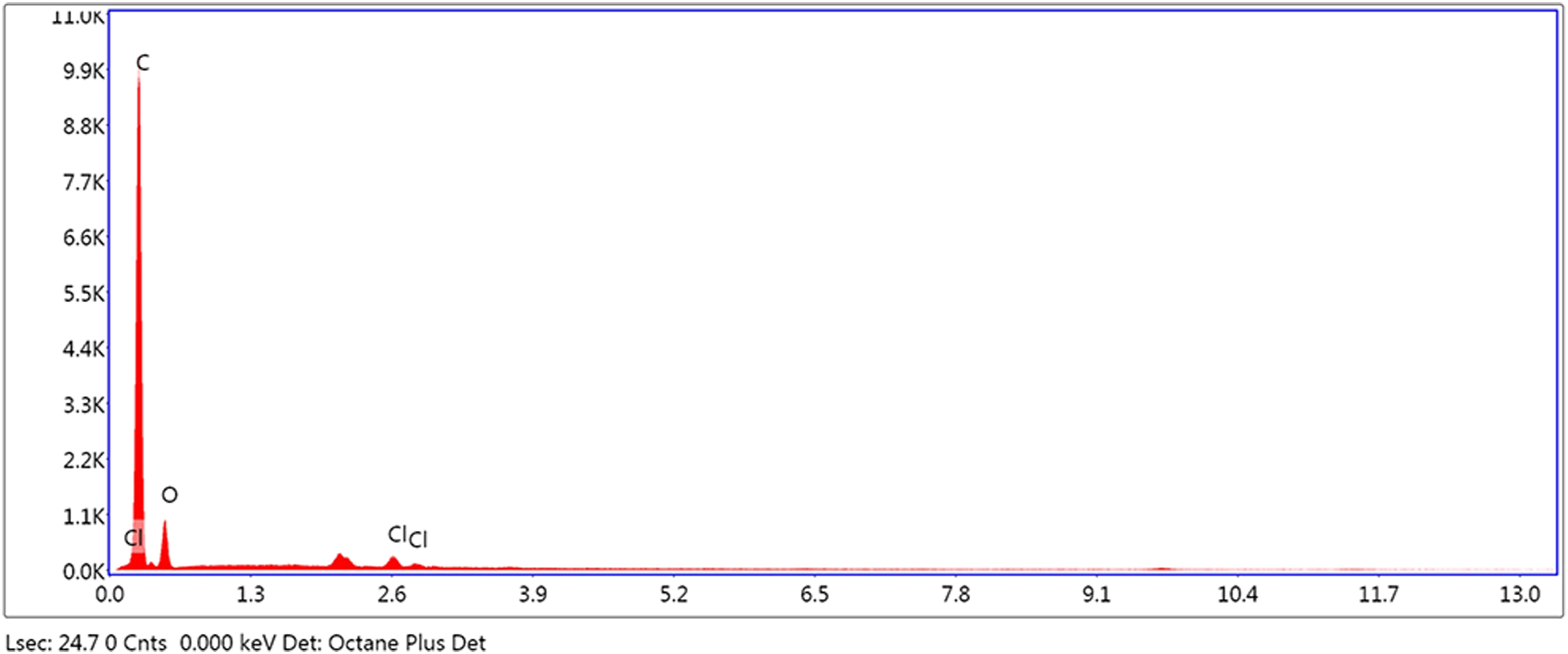
Microwave stability of TTEGDA-PS-OX-Br
Microwave stability of TTEGDA-PS-OX-Br is very crucial for performing synthetic reactions under MW conditions. To investigate the MW stability, 1 g of the bromo resin was irradiated at a MW power level of 300 W for about 5min in an Anton Parr Monowave 300 MW synthesizer at 100°C. After 1,2,3,4 and 5 min, 0.1 g of the resin was taken out and its bromine capacity was estimated by iodometric titration. The bromine capacity was found to be 2.48 mmol/g after 1 min of MW irradiation and there is a decrease of 0.8 mmol/g in bromine efficiency after 5 min of MW irradiation.
Microwave-assisted oxidative coupling of thiols using TTEGDA-PS-OX-Br
Oxidative coupling of thiols to disulfides using TTEGDA-PS-OX-Br.
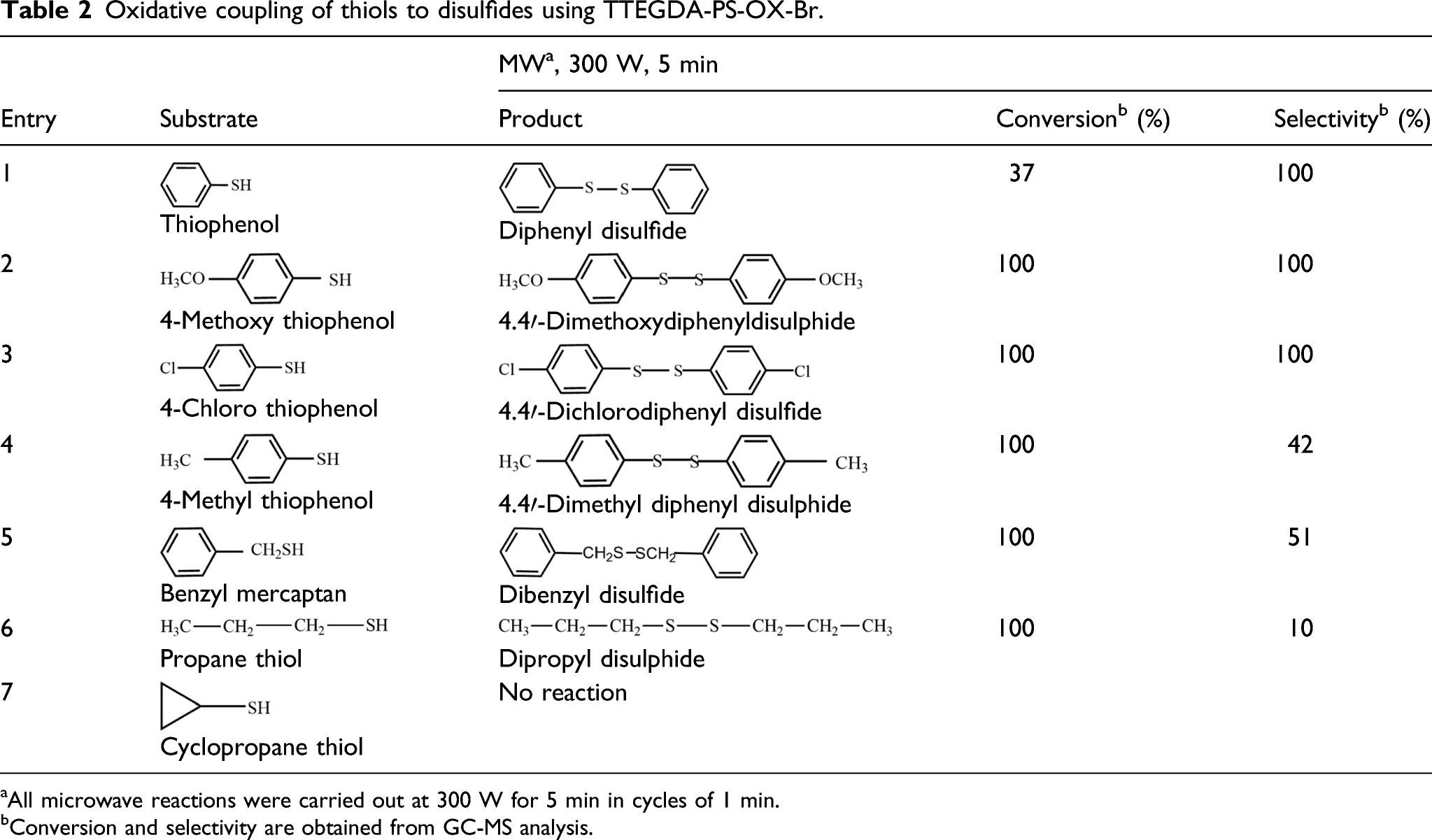
aAll microwave reactions were carried out at 300 W for 5 min in cycles of 1 min.
bConversion and selectivity are obtained from GC-MS analysis.
The proposed mechanism for the oxidative coupling of thiols based on the tribromide structure is given in Scheme 4. Bromine that is released from TTEGDA-PS-OX-Br under MW condition is responsible for the coupling reaction and the mechanism is given below. The absence of aromaticity may be the reason for the inactivity of cyclopropane thiol. Plausible mechanism of coupling of thiols by TTEGDA-PS-OX-Br.
Recyclability is one of the main advantages of polymer-supported reagents. The spent TTEGDA-PS-OX-Br complex obtained after various coupling reactions can be recycled back to the original reagent by washing with dichloromethane followed by treatment with Br2 in CCl4. In the current study, the spent TTEGDA-PS-OX-Br obtained from MW-assisted reactions can be regenerated up to five cycles and the filterability and mechanical stability of this reagent was retained even after five cycles of regeneration and reuse.
Conclusion
Here, we have explored the efficacy of the polymeric reagent 3% TTEGDA cross-linked polystyrene-supported bromo derivatives of 2-oxazolidone for organic functional group transformation. The new polymeric reagent acts as an efficient oxidizing agent for the coupling of thiols to disulfides under MW irradiation. One of the striking features of this protocol is that the only product obtained was disulphide and no over oxidation product was observed. Some advantages of this method include: moderate to good product yield, mild reaction conditions, easy workup procedure and shorter reaction time. The filterability and mechanical stability of this reagent was retained even after five cycles of regeneration and reuse.
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911211048607 – Supplemental Material for Microwave-assisted oxidative coupling of thiols using polystyrene supported bromoderivatives of 2-oxazolidone
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911211048607 for Microwave-assisted oxidative coupling of thiols using polystyrene supported bromoderivatives of 2-oxazolidone by Anjaly Mathew, Beena Mathew and Ebey P Koshy in Polymers and Polymer Composites
Footnotes
Acknowledgements
The financial assistance to Anjaly Mathew from University Grants Commission, Government of India under minor research project, is gratefully acknowledged. The authors would like to thank SAIF STIC, CUSAT, Kerala, India for characterization facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University Grants Commission Government of India, under the Minor Research Project. [No. 2265-MRP /15–16/KLCA029/UGC-SWRO dated 25 April 2016.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
