Abstract
In this work, an insoluble three dimensional (3D) porous polymeric structure and their metal complexes were synthesised by the condensation reactions of meta(m)-phenylenediamine, para(p)-phenylenediamine and glutaraldehyde. The morphological and spectral features of the porous polymeric structures were determined using different analytical and spectroscopic methods, including field emission scanning electron microscopy, four-point probe electrical conductivity, photoluminescence spectroscopy, Fourier-transform infrared spectroscopy, surface area Brunauer–Emmett–Teller and magnetic and thermal behaviours. According to the obtained data, the shape, size and photoluminescence properties of the compounds, especially the conductivity, were clearly changed after the metalation processes.
Introduction
Technological development and an increasing human population increase energy consumption rates. Fossil fuel–based energy sources create the emission of carbon dioxide in the atmosphere, which causes global warming. The increase in green gas emissions in the atmosphere is warning us to decrease carbon dioxide emissions through more environmentally friendly solutions.1–3 One of the most likely solutions for solving this problem is the improvement of electric-based technologies.4,5 Electrical vehicles, such as electric cars and smart portable devices, need an advanced energy storage system for high efficiency. 6 Upon those demands for energy storage systems, the progression of battery technologies with power density, long life cycles, high-energy density and low costs has become one of the hottest topics for many research groups. Lithium (Li-ion) rechargeable batteries are one of the most important and likely solutions for the new generation of energy storage systems. 6 However, morphological reconstruction after cycling of a battery causes new architectural structures of morphology, such as dendrite shapes that make a Li-ion battery inactive and responsible for short circuits.7–9 To figure out the deterioration of the battery’s performance dependent on the surface fractures, the surface of the battery should be protected with solid electrodes. Transition metal oxide, porous carbon and conductive polymers have been widely investigated as solid electrodes to protect the battery’s surface from fractures.8,10,11 Conductive polymers are one of the best potential materials for protection of the Li battery surfaces.12–17 The morphological features of the polymers and their production methods, the number of donor atoms and centres, the kind of metals and their ionic charge, the hydrogen bond and the π-π stacking in the polymer and the steric hindrance are crucial for designing well-tuned and stable electrodes.14,18–22 More specifically, the type of organic linker in the polymer is also one of the most significant points to achieve a targeted, efficient porous morphology because linker ligands, having a single C–C bond (sp3) and donor group, have the ability to give flexibility to each of the polymeric units.6,23–27 Therefore, the coordination polymers can rotate around the flexible linkers. The morphology of the molecules in the polymeric structure can grow in different directions.26,28-34 The 3D structure, the porosity of the coordination polymers and the conductivity are highly effective for application, such as in a flexible electrode for new generation battery technologies.35–37 Polymers, such as polyaniline (PANI) and polypyroline (PPy), have been used as electrodes for enhancing the cycling stability and lifetime, but the swelling and shrinking of the polymeric structure after the charge and discharge of the battery deteriorates the polymer’s performance.9,38,39 For this purpose, the beneficial modification of conductive polymers with some transition metals, Ni, Co, and Cu, plays a vital role in structural-electrochemical stability and conductivity.13,17,39–41 The transition metals contribute to the capability of the battery’s capacity by providing a high number of transferred electrons as intercalation. Furthermore, the production of the coordination polymers with the transition metals enables the fast charging of a battery and improves the stable-cycling number. Our previous experiences showed that the preparation of the coordination polymers containing N and O donor atoms allows for the tuning of porous structures depending on the preparation methods. 35,36,42 In addition to the methods, the type of monomeric unit, preparation method and metalation process play a key role in the behaviour of the polymer.43–47 The monomeric units as linkers are one of the most important cases for achieving targeted compositions because of electrical conductivity and a 3D structure. If the linker has double bonds that allows the delocalisation of electron, then the conductivity rises. However, the linker, including the longer carbon chain, exhibits unconventional behaviours that mitigates the conductivity but develops the 3D structures. Therefore, we believe that this work sheds light on the effective parameters for the designing of new functionalised polymeric structures for different applications.
In this study, we present the production of imine-based polymers and their morphological, electrical and photoluminescent properties after the metalation processes. The produced compounds were characterised through elemental analysis, Fourier-transform infrared spectroscopy (FT-IR), thermogravimetric analysis (TGA), inductively coupled plasma (ICP), field emission scanning electron microscopy (FESEM), four-point probe conductivity (FPP), Brunauer–Emmett–Teller (BET), powder X-ray diffraction (PXRD) analysis, luminescent properties and magnetic susceptibility measurements. The results showed that the conductivity behaviours significantly changed after the metalation processes. Furthermore, the linkers play an important role in producing different 3D morphological and surface structures. This unique application opens new pathways for tuning a polymeric structure in the production of advanced materials.
Experiment
Materials
p- and m-phenylenediamine, glutaraldehyde (25% water solution), CoCl2.6H2O, NiCl2.6H2O, CuCl2.2H2O, ethanol, methanol and dichloromethane were commercially purchased. The pH values of the reactions were measured using the Hanna 211 pH metre. The infrared spectra were obtained on a Perkin-Elmer RX-1 (Potassium Bromide [KBr] disk; 4000–400 cm−1) FT-IR spectrometer. The surface morphologies of the compounds were determined by Carl Zeiss, SUPRA-55. The TGA was performed with a Perkin-Elmer Pyris Diamond TG/DTA N2 (30–900°C) at a heating rate of 10°C/min. A Perkin-Elmer Optima 2100 DV ICP-OES instrument was used for the ICP analysis. A Rigaku MiniFlex system with CuKα radiation (λ = 1.54,059 Å) was used for the PXRD studies. The single-photon fluorescence spectra of the compounds were collected on a Perkin-Elmer LS55 luminescence spectrometer. The magnetic susceptibility measurements were performed by using Sherwood Scientific magnetic susceptibility. The FPP electrical conductivity device and manual hydraulic pressure of 20,000.0 kg/cm2 was used. The melting point of the produced materials was measured using the melting point apparatus B450 Buchi.
Methods
Synthesis of polymeric ligands
The polymeric ligands were prepared by the refluxing of (1.081 g, 10 mmol) for p-phenylenediamine and (1.081 g, 10 mmol) for m-phenylenediamine with corresponding glutaraldehyde (4.005 g in water solution, 10 mmol) in methanol (50 mL) for 3 h. The dark brown solution was filtered, and an insoluble solid was washed three times with 20 mL methanol. The produced polymer and metal complexes were dried under a 20-mbar vacuum for 12 h. The yields were calculated as 91.5% for The structure of the polymeric ligands and the predicted metal complexes.
Synthesis of polymeric complexes
The polymeric complexes were prepared using the following template technique: p-phenylenediamine (1.081 g, 10 mmol) or m-phenylenediamine (1.081 g, 10 mmol) were dissolved in methanol (30 mL) at room temperature. After the dissociation of the monomer was completed, the metal salt was added to the solution. An appropriate amount of metal salt (0.238 g, 1 mmol) for CoCl2.6H2O; (0.237 g, 1 mmol), for NiCl2.6H2O; (0.170 g, 1 mmol) and for CuCl2.2H2O was dissolved in CH3OH (20 mL) and added to the monomers solution. These reaction mixtures were heated under reflux for 1 h; the glutaraldehyde was added (4.005 g from 25% water solution, 10 mmol) in the reaction medium; and they were boiled for 3 hours. All of the polymeric metal complexes were collected by filtration, washed with MeOH and dried under a vacuum for 12 h
Results and discussion
The ICP, elemental analysis and reaction yields of the compounds.

*Cal: Calculated.
ICP: Inductively coupled plasma.
FT-IR Spectra
FT-IR spectral bands (cm−1) and the magnetic susceptibility values of the compounds.
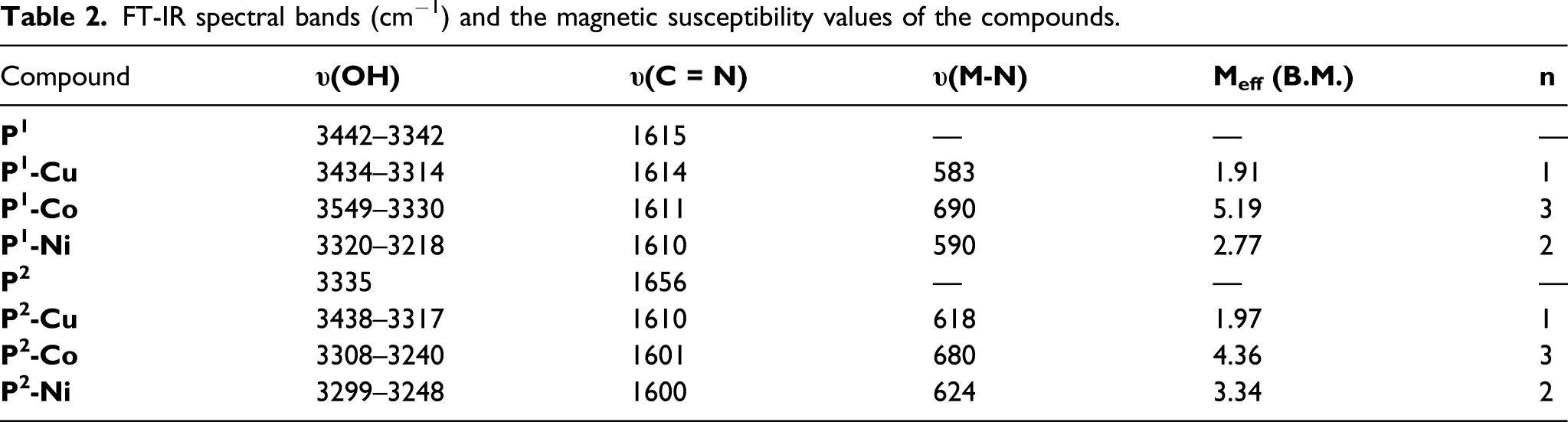
Magnetic properties of the complexes
According to the magnetic moment results (Table 2), the complex
Thermogravimetric analysis
The TG/DTA curves of the compounds and explanations are given in Supplementary Figures S5–S7 in the supplementary data. The objective of this section is to analyse the thermal behaviour of the complexes. The confirmation of the composition of the polymeric structures and the evaluation of the crystal water molecules. The TG/DTA results were investigated in three parts. The first step is described as leaving the coordinated water molecules in the polymeric structures. Interesting results were observed from
Luminescence properties
The solid state emission and excitation studies of the polymers ( Emission and excitation spectra of the ligands and complexes.
The photoluminescence properties of the ligands and their complexes were investigated in a solid state because of the insolubility of the polymers and the metalated polymers (Supplementary Table S1). The polymeric structure and its metal complexes have higher fluorescent intensive peaks. If they were compared with the previous studies, it could be explained by the polymeric structures consisting of a high number of the repeated units, such as benzene, the imine groups as a linker. The photoluminescent behaviours of the polymeric structures results from the benzene ring, the imine groups, π-π*, the ligand metal charge transfer (LMCT) and the metal ligand charge transfer (MLCT), respectively. Furthermore, the metalated polymeric structures in particular have multiple unpredictable inter-/intra-molecular interaction, which gives a unique rigidity, and the special crinkly shape of the polymers can be clearly seen in the FESEM images (Figures 3 and 4).
51
Inter-/intra-molecular interactions and the induced-strain polymeric structure restricts the ability of the C–C bond rotation and the decreased regulated benzene π-π stacking on the polymeric structure. Therefore, the radiationless vibrational relaxation channels clearly reduce, and the fluorescent intensity of the polymers slightly increases without quenching.37,52,53 Additionally, even the polymeric structure rigidity and the emission wavelength of the metalated polymers show a very small red shift towards the 5–6 nm lower energies. The photoluminescent properties of Field emission scanning electron microscopy images of P1 and their complexes. Field emission scanning electron microscopy images of P2 and their complexes.

The morphological analysis
The surface morphological structures of the compounds were inspected with FESEM.
The perspective descriptions of the polymeric ligands,
Electrical conductivity
The FPP electrical conductivity values of the polymeric compounds were measured via the four-probe technique from the sample pellets, which designed 1 mm thick and 1.0 cm in diameter under a hydraulic pressure of 10 tons. The electrical conductivity test was calculated undoped with iodine during the experimental parts.
Electrical conductivity and surface area results of the compounds.

Conclusions
The insoluble and high-temperature melting point polymer that is more than 300
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911211048287 – Supplemental Material for The effect of metalation processes on polymer morphology and conductivity properties
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911211048287 for The effect of metalation processes on polymer morphology and conductivity properties by Oğuz Yunus Sarıbıyık, İlyas Gönül, Burak Ay and Serkan Karaca in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
