Abstract
The objective of this study was the preparation of ionically crosslinked 6-mercaptopurine (6-MP) monohydrate microparticles through preparing polyelectrolyte complexes of drug and polymers. Polymers such as chitosan, casein, and carrageenan were used to prepare crosslinked microparticles, and sodium tripolyphosphate was used as crosslinker. Microparticles were characterized for their flow behavior, compressibility, percentage yield, micromeritic, and entrapment efficiency. Scanning electron microscopy was conducted to understand the surface morphology of the microparticles, and the result was correlated with the swelling index and percentage drug release. Mathematical modeling of drug release in order to determine the drug release kinetics was also determined to understand the mechanism involving the release of 6-mercaptopurine from the microparticles. The ionic crosslinked microparticles were in the range of 664 μm–798 μm particle size having good flow and compressibility properties with percentage yield were found to be from 77.5% to 87.5% in range. The entrapment efficiency for the formulations were found to be from 63.5% to 83.5%, with MCP-5 gave maximum entrapment efficiency of 83.5%. In vitro swelling and drug release studies were in accordance with the polymer properties following zero-order model with super-case transport II.
Introduction
Colorectal cancer is the one of the most common cancers accounting for 10–11% of cancer cases in both men and women. Generally, it is the third leading cause of death in cancer patients, predominantly a malignant disease of elderly people.1,2 Patients suffering from ulcerative colitis and Crohn’s disease have high risk of colon cancer. 3 Targeted drug delivery to colon should ideally be capable of protecting the drug delivered to the colon, that is, drug release and absorption should not occur in the stomach as well as the small intestine and neither the bioactive agent should be degraded in either of the dissolution sites but only released and absorbed once the system reaches the colon. Colon-targeted drug delivery systems have gained a great deal of attention as potential carriers for the local treatment of colonic diseases with reduced systemic side effects and also for the enhanced oral delivery of various therapeutics vulnerable to acidic and enzymatic degradation in the upper gastrointestinal tract. 4 Colon-targeted drug delivery systems for macromolecules can provide therapeutic benefits including better patient and lower costs. To achieve more efficient colonic drug delivery for local or systemic drug effects, various strategies have been explored including pH-dependent systems,5,6 enzyme-triggered systems, 7 and polymeric nano/microparticles.4,8 Previously, polymeric microparticles for the purpose of controlled drug delivery were produced by chemically crosslinking with glutaraldehyde, 9 sodium hydroxide, and ethylene glycoldiglycidyl ether. 10 However, these chemical crosslinking agents have possibility of inducing undesirable effects. 9 To overcome this disadvantage of chemical crosslinking, ionic crosslinking interaction has been favored.11,12 The ionic crosslinking of polymers with sodium tripolyphosphate (TPP), leads to formation of biocompatible crosslinked nano or microparticles, which can be efficiently employed in drug delivery and even delivery of vaccines and proteins.12,13 The crosslinking of polymers can allow modulation of drug release and extend its range of potential applications in drug delivery.
The aim of this study was to develop pH-responsive, ionically crosslinked drug-polymeric microparticles, colon-targeted drug delivery system. For this purpose, ionically crosslinked 6-mercaptopurine monohydrate microparticles were prepared. 6-mercaptopurine monohydrate belongs to antimetabolites that can prevent the biosynthesis of normal cellular metabolites. 14 It has been used with other chemotherapy drugs for the treatment of different types of acute leukemias. 15 It is also effective in the treatment of inflammatory bowel disease (IBD) including Crohn’s disease and ulcerative colitis and arthritis.16–18 6-mercaptopurine also helps to decrease the dose of steroids in patients with IBD and also reduce their dependence on steroids.17,19 Although 6-mercaptopurine is known for its toxicity, 20 attempts have been made to control the toxicity via polymeric-controlled drug delivery systems.16,18 Microparticles of 6-mercaptopurine were prepared by ionic gelation and emulsification techniques using natural polymers due to their biocompatible nature. In this study, chitosan (CS), casein (CAS), and carrageenan (CRG) polymers were used along with sodium tripolyphosphate (TPP) as a crosslinker. The microparticles were evaluated for surface morphology and shape by scanning electron microscopy (SEM), percentage yield, micromeritic properties, encapsulation efficiency, in vitro swelling index, and in vitro drug release studies by applying kinetic models.
Materials and methods
Materials
6-mercaptopurine monohydrate (Pharmedic Laboratories (Pvt.) Ltd. Pakistan), chitosan, sodium caseinate, sodium tripolyphosphate, ethanol, glacial acetic acid, dichloromethane, and tween 80 were commercially obtained from Sigma-Aldrich (United States). Sodium hydroxide, lactic acid, monobasic potassium phosphate, and hydrochloric acid were commercially obtained from Merck (Pakistan) and carrageenan from Flukachemika (Malaysia). All the purchased chemicals were of analytical grade.
Methods
Preparation of ionically crosslinked particles
Composition of formulation of ionically crosslinked particles.

Preparation of MCP-1 and MCP-2 ionically crosslinked particles
Solution of chitosan (2–3%w/v) was prepared by addition of CS to glacial acetic acid solution (1%w/w). Above prepared drug solution was added to CS solution, and after that, flask was tightly closed. Both solutions were mixed for 15 min by magnetic stirrer. Aqueous casein solution (0.5–1% w/v) was adjusted to pH 2.0 with 1 N hydrochloric acid. Tween 80 (2% v/v) was added as a surfactant to CAS solution under magnetic stirring and added to previously prepared CS solution. Solution of CRG (0.5–1%) prepared in distilled water was added in previously prepared CS-CAS solution. TTP Solution (5–7%w/v) was prepared in distilled water,11,21 and then, CS solution was added dropwise by hypodermic syringe having needle size of 20G into solution of TPP. The ionically crosslinked particles were prepared and named as MCP-1 and MCP-2, respectively. The crosslinking time was 30 min, and ionic crosslinked particles were filtered, washed with distilled water, and dried at room temperature.
Preparation of MCP-3 and MCP-4 ionically crosslinked particles
MCP-3 and MCP-4 were prepared by emulsification followed by polyelectrolyte complexation method. 22 Aqueous CAS solution (0.5–1% w/v) was adjusted to pH 2.0 with 1 N hydrochloric acid. Tween 80 (2% v/v) was added as a surfactant to CAS solution under magnetic stirring. Methylene chloride solution of 6-mercaptopurine as the oil phase was mixed with aqueous CAS solution phase by homogenization at a speed of 1000 rpm for 1 h to obtain an oil-in-water emulsion. The ratio of oil and aqueous phase was 1:10 v/v, and prepared emulsion was after sonication mixed with (2–3%) CS solution. CS-CAS solution was added with the help of syringe in (5–7%) TPP solution under gentle magnetic stirring. The prepared crosslinked particles were named as MCP-3 and MCP-4, respectively, and after 2 h of cross linking, nanoparticles were isolated and subsequently washed several times with Tween 80 solution (0.1% v/v) and water and then dried at room temperature.
Preparation of MCP-5 and MCP-6 ionically crosslinked particles
CS (2–3%w/v in 1% glacial acetic acid) solution and drug solution (as prepared above) were mixed for 15 min by magnetic stirrer. Solution of CRG (0.5–1%) prepared in distilled water was added in previously prepared chitosan solution, and solution was homogenized for 2 h. 21 Then CS-CRG solution was added dropwise by hypodermic syringe having needle size of 20G into solution of (5–7%) TPP solution. The prepared ionic crosslinked particles were named as MCP-5 and MCP-6, respectively. The crosslinking time was 30 min, and ionically crosslinked particles were filtered, washed with distilled water, and dried at room temperature.
Micromeritics



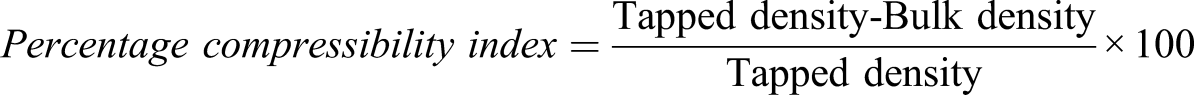
Micromeritic properties of ionic crosslinked microparticles and percentage yield and entrapment efficiency.
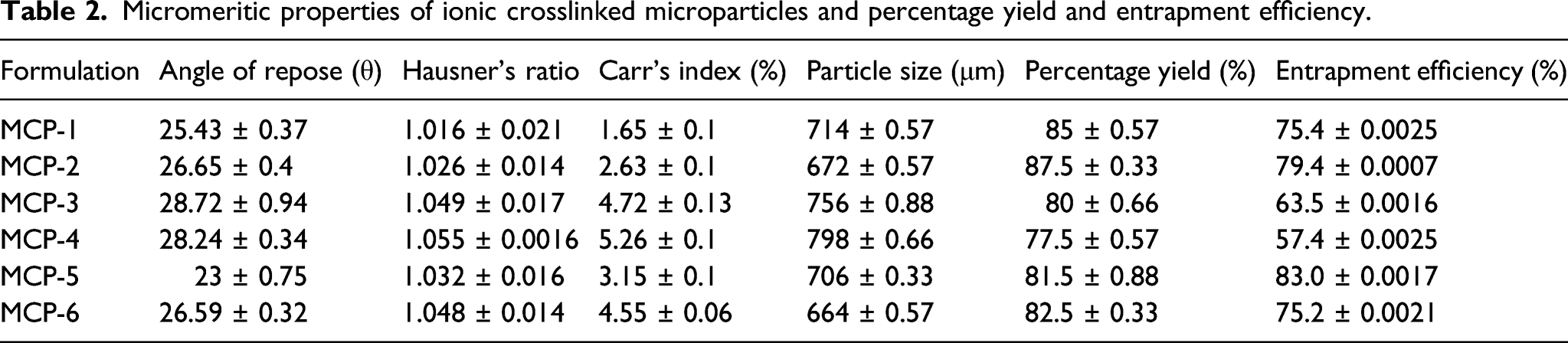
Percentage yield
Percentage yield was calculated as the weight of final product with respect to the total weight of starting materials. The total amount of drug and polymer used was noted or wet microspheres were weighed; then, microspheres were dried and weighed again; percentage (w/w) was determined by using the given formula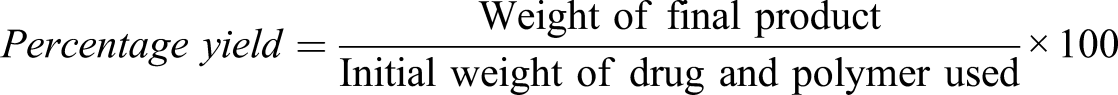
Determination of percentage entrapment efficiency
Percentage entrapment efficiency is percentage of drug entrapped in the microparticles regarding to original quantity used. Measured amount of microparticles MCP-1 to MCP-6, that is, 10 mg were taken one by one; after crushing, they were added to 100 mL of phosphate buffer pH 7.2, by using an ultrasonic bath for complete removal of 6-mercaptopurine monohydrate from microparticles, respectively. Then, solution was filtered with the help of 0.45 μm filter paper. 0.5 mL of filtered solution was taken with pipette and diluted to 5 mL with phosphate buffer solution of pH 7.2. After dilution, absorbance of 6-mercaptopurine was measured at 316 nm by using a UV–Vis spectrophotometer. And that absorbance was then converted to the quantity of 6-mercaptopurine monohydrate by applying data in standard calibration curve. Entrapment efficiency was measured by using following formula
Scanning electron microscopy
The surface morphology of formulated ionic crosslinked particles was evaluated by scanning electron microscope (SEM). For SEM, samples were arranged by sprinkling microspheres powder on an aluminum stub with the help of double adhesive tape. The stubs were coated with gold–palladium alloy to a thickness of (150–200 Å) using coat sputter JFC 1100 (JEOL-Japan). The microparticles were observed and examined under scanning electron microscope (JEOL, JSM-6100 SEM, and JAPAN), and photograph was taken.
In vitro swelling studies
Swelling studies were performed with the help of empty tea bags that contained 50 mg of each formulation; the empty bag was placed in a closed flat bottom tube with 100 mL of a buffer solution that was maintained at 37°C up to 24 h. It was carried out for MCP-1 to MCP-6 formulation in two different solutions with different pH values, that is, of pH 2.0 in first beaker for first 2 h; then, tea bags were shifted to beakers for conforming small intestine condition (pH = 5.5) up to 6 h, and finally to confirm large intestine conditions, all previously used tea bags at pH 5.5 were shifted to beakers having phosphate buffer (pH = 7.4) and studied up to 24 h. It was done to analyze swelling in stomach, intestinal, and colon region. At specific time intervals, sample bags were removed from the swelling medium, blotted with a piece of paper for 5 s to absorb excess water on surface, and then, change in the weight was determined to calculate the swelling index using following equation26,27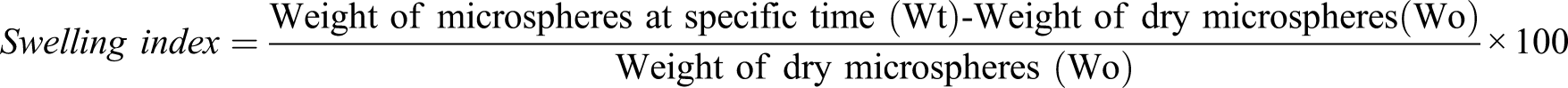
In vitro drug release studies
In vitro drug release or dissolution profiles of microparticles of 6-mercaptopurine monohydrate were found out by enclosing micro-beads in hard gelatin capsule in a quantity equivalent to 50 mg dose of drug. (Pharma max. TEST) dissolution apparatus was used to carry out the study to check the release behavior of microparticles. The dissolution was carried out for 2 h in 900 mL of 0.1 N HCl solution and then in phosphate buffer having pH 5.5, 7.2, and 7.4 for 12 h at 37°C
Drug release kinetic analysis
Drug release kinetic analysis regarding all formulations MCP-1 to MCP-6 was applied in excel by using add-in DDSolver software to calculate the highest values of regression coefficient that tells us about best fit model either zero order, first order, Highuchi, Hixson–Crowell, Korsmeyer–Peppas, and also value of n was seen by applying Korsmeyer–Peppas model to check out the pattern of drug release.
The ideal kinetic models for drug release and permeation were estimated using the different mathematical equation plots: cumulative amount of drug permeated versus time (zero-order model), log cumulative percentage of drug remaining versus time (first-order model), cumulative percentage drug permeated versus square root of time (Higuchi model), cube root of drug percentage remaining in matrix versus time (Hixson–Crowell model), and log cumulative percentage drug permeated versus log time (Korsmeyer–Peppas model). 29
Following equations were used for the calculation of each kinetic model.
Zero-order equation: Qt = Q0 + K0t
First-order equation:
Higuchi equation: Q = K3√ t.
Hixson–Crowell equation: Q01/3 − Qt1/3 = κ t.
Korsmeyer–Peppas model equation: Q/Q0= κ tn
Results and discussion
Micromeritics
Micromeritic properties of microparticles were evaluated in terms of particle size, flowability, and compressibility. Particle size is a simple concept, but it is a most critical process parameter in preformulation and manufacturing. It is also true that the reduction of particle size impacts directly on the bioavailability of drug. Particle size of the formulations ranged from 664 to 798 μm. The ionic crosslinked particles prepared from emulsification method produced microparticles of larger size as compared to other two methods. MCP-6 formulation exhibited smallest particle size of 664 ± 0.57 μm, whereas MCP-4 formulation displayed the largest particle size of 798 ± 0.66 μm. The concentration of casein polymer could also be contributing factor in larger particle size. All the formulations displayed excellent flowability with angle of repose values between 23 and 28o angles. The Hausner’s ratio may be related to the compressibility of powder and values of <1.25 is indicative of good compressibility. The Carr’s index may also be indicative of flowability and degree of packing of thematerial, which are relevant properties when filling the matrices of the tablet press. CI of <15% indicates an adequate flow of powders and stable packing, while values of >25% are characteristic of poor flow properties. All the microparticles showed excellent compressibility and flow properties,30,31 and the results of Hausner’s ratio, Carr’s index, and angle of repose for each formulation are given in Table 2.
Percentage yield
The percentage yield varies from 77.5 to 87.5% in different formulations. Formulations that were prepared from emulsification process (MCP-3 and MCP-4) showed lower percentage yield as compared to other formulations. The factors that could have been contributed in low yield are the drying of ionic crosslinked particles would be less efficient through emulsions as compared to the aqueous solutions of other formulations and emulsification process required more crosslinking time as compared to other methods. The results for percentage yield of each formulation are given Table 2.
Percentage entrapment efficiency of ionic crosslinked particles
Percentage entrapment efficiencies of ionic crosslinked microparticles were in the range of 57.4%–83%, whereas the highest percentage entrapment was 83% of formulation MCP-5. The entrapment efficiency was increased with the increase in polymeric concentration except in case of MCP-4 as shown in Table 2. The ionic crosslinked particles prepared from emulsification method produced showed lowest entrapment efficiency thus proving that emulsification might not be a suitable method for 6-mercaptopurine entrapment. It was also observed that lower concentration of casein or complete absence of casein resulted into better entrapment efficiency.
Scanning electron microscopy
To determine the surface morphology of the formulations, microparticles were evaluated by scanning electron microscope (SEM). The microparticles prepared with chitosan and TPP were smooth and spherical in shape as shown in the Figure 1. Microparticles prepared with chitosan, casein, and TPP were appeared to be porous, and these pores could affect the release rate of drug from polymer.
32
Microparticles prepared with chitosan, carrageenan, and TPP were also smooth and spherical. The spherical particles with smooth even surface indicated the absence of any drug crystal on the surface and confirmed the even distribution of drug in the polymeric matrix. The spherical microparticles that were smooth in nature resulted into better entrapment of the drug that could also influence release of drug in a control manner.33,34 Surface morphology also indicated that the emulsification method for preparing the microparticles lacked proper drying that resulted into porous morphology of the microparticles prepared by emulsification. Scanning electron microscopy of microparticles prepared from different polymer combinations. 
In vitro swelling ratio
The in vitro swelling studies of microparticles were performed at different pH values. The swelling behavior was in accordance with the increasing affinity of polymers for the buffer as the pH increased. Thus, the systems MCP-1 to MCP-6 were swelled highly at pH 7.2, while in acidic environment pH 2, the swelling was considerably smaller. The formulation in pH 2 never surpassed the 8%. MCP-1 to MCP-6 microparticles swelling increased as the pH increased. At pH 2.0 and 5.5, the swelling index never reached the 10%. At pH 7.4, swelling was constantly increased reaching more than 22% after 24 h and showing a tendency to still increase as shown in the Figure 2. This could be a useful property as it might result into a controlled manner of drug release in large intestine, thus achieving a pH-responsive microparticulate drug delivery system. The swelling and pH-responsiveness are important for the targeted intestinal drug delivery systems. The principle sites of drug absorption are the stomach and proximal part of small intestine, but for the purpose of drug to be delivered into colon region, drug delivery systems should offer restraint toward releasing the drug in lower pH or stomach and small intestine environment. Our results indicated the control of swelling at lower pH which could also result into controlled drug release. Furthermore, there was a relationship observed between the concentration of the TPP crosslinker, pH, and the swelling properties.26,27 Formulations containing lower concentrations of TPP showed lower swelling index at pH 2 and PH 5.5 but higher swelling index at colonic environment of pH 7.2. The influence of TPP crosslinker on the swelling properties has been previously reported and in accordance with our findings. Thus, it could be suggested that the concentration of TPP crosslinker could influence the controlled drug delivery and targeted drug release in the colon region. Swelling index of ionically crosslinked particles for all the formulations.
In vitro drug release studies
The mechanism of drug release from the prepared ionically crosslinked microparticles depends on various factors such as composition of polymers, crosslinker, geometry (size and shape), preparation technique, and environmental conditions during drug release. The amount of drug release was measured by UV–Vis spectroscopy and is plotted as a cumulative release with time in Figure 3. All the formulations were analyzed in acidic and basic medium to determine their drug release in gastrointestinal environment. All formulations were subjected to in vitro dug release studies; the in vitro drug release of MCP-1 to MCP-6 was carried out for first 2 h in 0.1 N HCl at 37 ± 0.5°C at 100 rpm to mimic conditions of stomach; then, each formulation was shifted and analyzed under small intestinal environment for next 2 h (phosphate buffer of 5.5 pH), and then, that same formulations were shifted to phosphate buffer of 7.2 pH to analyze its release in large intestinal and colon environment. The formulations MCP-3 and MCP-4, which were prepared by emulsification process, had lower drug entrapment efficiency that was evident through their SEM images. Therefore, these formulations resulted into lower percentage drug release (Figure 3). All the other formulations had smooth and spherical morphology and had very good drug entrapment efficiency resulted into controlled drug release in an effective manner and at colonic region thus proving them to be excellent colon-specific, pH-responsive, drug delivery vehicle systems (Figure 3). The maximum drug release was offered by MCP-5 and MCP-6 combination formulations releasing 99.3% and 99.6% of drug, respectively (as shown in Figure 3). The results clearly displayed the pH-dependent drug delivery mechanism from all the formulations. Less than 10% drug was released at pH 1.2 which highlighted a successful control and delay of release of active ingredient from the microparticles. Around 20–40% drug was released at pH 5.5, whereas maximum drug was released at pH 7.2 which represents the colon pH. The drug entrapped in the microparticles released gradually over the period of 12 h. There was almost no initial drug release from the microparticles in an acidic environment, but as formulations came into contact with increased pH simulating the small intestine and colon environment (pH 5.5 and pH 7.2 respectively), the microparticles gradually started to release the drug in response to the changes in the dissolution medium’s pH thus making these formulations an ideal candidate for inflammatory bowel diseases and colon cancer. In vitro percentage drug release for all the formulations.
Drug release kinetics
Regression coefficient and constants values of microparticles found out by kinetic equation models.

Conclusion
Controlled release pH-responsive microparticles of 6-mercaptopurine monohydrate were successfully prepared by polyelectrolyte complexation and emulsification method that will improve patient compliance by decreasing side effects regarding to anti-cancer drugs due to controlled effect. Ionic crosslinked microparticles were prepared by polyelectrolyte complexation, and emulsification method produced microspheres of regular and spherical shape except for emulsification method as found in SEM analysis. The porous morphology of microparticles prepared from emulsification method (MCP-3 and MCP-4) was resulted into low entrapment efficient and least percentage drug release. The analytical determinations of entrapment efficiency, percentage yield, and mean particle size and drug release behavior have seen among all formulations due to different polymer combinations and ratio. In vitro swelling and drug release studies were in accordance with the polymer properties, showing the highest swelling ratio and drug release at pH 7.2 where microparticles were able to deliver more than 85% of 6-MP monohydrate during 12 h of experiments. A significant relationship was observed between TPP crosslinker concentration, polymer, and the pH-responsiveness. The MCP-5 was considered the good encapsulating option amongst the six formulations to make sustained release formulation. In vitro release studies pointed out that there was pH-responsive controlled drug release from MCP-1 to MCP-6 and considered to be the best in order to achieve the objective of the study regarding to side effects of drug for patient suffering from ulcerative colitis. Drug release was super transport case II which followed zero-order release model that indicates patient compliance because of decrease in dosing frequency by decreasing side effects regarding to anti-cancer drugs due to controlled, sustained, and targeted effect.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
