Abstract
This work consists of preparation and characterization of composites produced from thermoplastic starch (TPS) and high impact polystyrene (HIPS). Due to the immiscibility of the system (TPS/HIPS), it was necessary to incorporate concentrations of 1, 2 and 3% of an organophilic montmorillonite (MMT) to improve the properties of the mixtures, in particular their rigidity. The composites thus prepared were characterized using XRD, FTIR, mechanical test, degree of swelling in water and biodegradability. The results show that the addition of MMT improves the mechanical properties of the mixtures such as the tensile strength and the Young’s modulus by 5% and 10%, respectively. In contrast, the resilience of the system has significantly decreased. Moreover, for 3% of MMT, the composites biodegradability is enhanced by 15% when compared to the TPS/HIPS mixture without MMT.
Introduction
Even though conventional plastics made from fossil fuels have attracted the market with their features such as low price and good mechanical characteristics, their resistance against biodegradation by microorganisms has caused many environmental problems. Hence, the researchers have started the discussion on their degradation methods in order to find out how to control the problems. 1 On the other side, biodegradable plastics gained a great attraction as a substitute for the conventional plastics, where the new polymer or polymer blends will provide similar properties and are convertible to non-toxic materials. To fulfil the criteria, biomaterials and their blends are most commonly used as biodegradable polymers. 2
High impact polystyrene (HIPS) is abundantly used as matrix for biodegradable polymers. HIPS presents high impact strength due to the incorporation of polybutadiene (PB) rubber particles in the polystyrene (PS) rigid matrix. HIPS has the advantages of good impact resistance, low cost and easy processing and is commonly utilized in many applications, such as electrical appliances, automotive components, electronic instruments, packaging and building materials. However, it is degraded very slowly in the environment. 3 Numerous biomaterials are blended with HIPS matrix to improve the mechanical properties and induce biodegradability.
Starch is the second largest bio-synthesized carbohydrate compound after cellulose and is the main source of energy for humans and animals. 4 Starch is a mixture of two polysaccharides which are amylose and amylopectin. It is an inexpensive and renewable product that can be mixed with synthetic polymers to confer them a biodegradable character. 5 When subjected to heat treatment and high shear in presence of suitable plasticizers, starch granules are disrupted to give thermoplastic starch (TPS) which could be transformed by conventional processes used for classic plastics such as extrusion and compression molding. 6 However, TPS has poor mechanical properties making it necessary to modify in order to extend its fields of application via blending with other natural or synthetic polymers.
Many researchers have used glycerol as a plasticizer in HIPS/PS mixture preparation and found it very effective in improving the degradability of the mixture. Schlemmer et al. 2 prepared TPS/PS composites by solvent casting technique with buriti oil and glycerol as plasticizers. The results indicate that, when compared to buriti oil, glycerol tends to degrade very fast. In a similar study, Mihai et al. 7 showed that glycerol content has a role in determining the viscosity of the composite. As the glycerol content increases, viscosity of TPS decreases and thus affects the overall TPS/PS blend viscosity and the ability to foam the material.
The overall studies suggest that blending HIPS and TPS reduces the mechanical properties when compared to the parent material. Thus, many researchers have tried different additives such as inorganic micro- or nano-particles to overcome this disagreement. 8 Previous studies by Giannelis et al. 9 suggested small amounts of clay (3–10%) can markedly increase many properties of the polymer matrix including the mechanical, thermal and barrier properties. 9 . A similar study was performed using titanium dioxide (TiO2) in starch/polyvinyl alcohol (PVA) composites by Sreekumar et al. 10
Bagcioglu et al. 11 used casting method to prepare HIPS/organophilic montmorillonite (MMT) nanocomposite and studied its properties. Morphological observations showed that clay layers were exfoliated and reduced rubber domains. Moreover, it reflected in increasing significantly the nanocomposites toughness. Furthermore, addition of MMT clay in 3 wt. % loading has produced composites with high tensile strength and toughness. Also, Abreu et al. 12 investigated the effect of adding 5% of nanoclay C30B on the rheological and physical properties of TPS mixtures with PP-g-MA according to the compositions (1:2, 1:1 and 2:1). Reactions between TPS and PP-g-MA groups allowed the synthesis at the interface of a TPS-g-PP copolymer consisting of an interpenetrating network capable of promoting the dispersion of the C30B nanofiller as confirmed from scanning electron microscopic observations which revealed remarkable homogenization of the blend.
Even though MMT has proven to increase the mechanical properties of many polymer composites, the use of MMT in TPS/HIPS blends has not been investigated yet. Thus, in the current research, we focus on the development and characterization of composites based on styrene–ethylene/butylene–styrene triblock copolymer grafted with maleic anhydride (SEBS-g-MA) compatibilized (50/50) TPS/HIPS blend composite reinforced by an organophilic nanoclay. MMT is introduced into the blend matrix in different concentrations and is expected to induce improvements on the overall properties of the system, particularly the mechanical properties. The interest of the study consists in verifying the possibility of obtaining materials with attractive properties which can access applications, in particular rigid plastic packaging. The TPS/HIPS ratio was taken as 50/50 to provide the better balance between the mechanical properties and biodegradability aptitude of the mixture. At higher TPS concentrations, the material performances could be altered due to the pronounced hydrophilic nature of TPS. However, the mixture biodegradability could be affected also, if TPS is incorporated at lower concentrations.
Materials and methods
Materials
HIPS has a melt flow index (MFI) of 2.8 g/10 min at 200°C and under 5 kg and a density of 1.04 g/cm3. Corn starch consists of a powder having medium particles size in the range of 4–8 μm. The used plasticizer is glycerol which has a density of 1.25 g/cm3. The clay is known under the trade name Nanomer 1.34TCN synthesized by Nanocor. It is an organophilic montmorillonite modified by methyl dihydroxyethyl tallow ammonium. The compatibilizing agent SEBS-g-MA, containing 1.84% of maleic anhydride, is supplied by Shell Chemical Co. (USA) under the trade name of Kraton FG 1901X.
Processing
TPS was prepared according to an experimental protocol.13,14 At first, starch is placed in an oven at 80°C for 3 h. Then, it is introduced into a mixer with an amount of glycerol equivalent to 30% and homogenized. Finally, the obtained mixture is placed into plastic bags and stored for 24 h. Afterwards, starch is mixed in a Brabender internal mixer to convert the powder into a translucent continuous mass which has been reduced into granules in Controlab pelletizer. In the next stage, the composites have been prepared by mixing HIPS/SEBS-g-MA/TPS (45/10/45) matrix with 1, 2 and 3% of MMT in a Brabender mixer at 200°C for 10 min. After processing, the formulations were pelletized for further characterization.
Characterization techniques
Fourier transform infrared (FTIR) spectroscopy
The spectral analysis was carried out using a Perkin Elmer 1000 infrared spectrophotometer with double beam. FTIR spectra were registered on thin films of 0.005 mm thickness, prepared by compression, and the transmittance was recorded for a wavelength sweep ranging between 500 and 4000 cm−1.15,16
X-Ray diffraction
X-ray diffraction measurements were performed on a XPert-Pro apparatus, using a Cu-Kα wavelength λ = 0.1540598 nm. The X-ray source is a ceramic tube fitted with a copper anode and powered by a current of 30 kV and an intensity of 25 mA. XRD studies were conducted for HIPS/TPS mixture, MMT and HIPS/TPS/MMT composites.
Melt flow index
The test was carried out using a Melt-Indexer model five which consists of a cylinder with a vertical axis placed in an oven ending with a standard die of length 8 mm and diameter 2.09 mm. The tests were carried out according to the standard ASTM D-1238, under a load of 5 kg and at a temperature of 190°C. The melt flow index was calculated using equation (1)
8
Tensile test
Tensile test was conducted to evaluate the mechanical performance of HIPS and TPS/HIPS mixtures at different weight percentages of MMT. The test was carried out on three replicates at a speed of 6 mm/min on a Zwick Roell-Z050 universal testing machine. The samples were prepared by compression according to the ASTM standard D 638.17–21 The gauge length (G), parallel length (L) and full length (L0) were 50, 57 and 165 mm, respectively. The width at grip section (W0), parallel section (Wc) and thickness (e) are 13, 19 and 7 mm, respectively. The distance between grips (D) is taken as 115 mm.
Izod impact test
Unnotched specimens with dimensions (63 × 13 × 2) mm3 were subjected to Izod impact test at room temperature on a Ceast Resil impact pendulum, equipped with a hammer of 7.5 J. The impact strength (an) is evaluated using the following equation (2)
An and b are, respectively, the energy absorbed during the impact and the width of the test specimens.
Atomic force microscopy
Atomic force microscope (AFM) MFP 3D Original is a local probe microscope, and it is utilized to view the topography of the sample surface. The surface roughness was measured for HIPS/TPS/MMT composites at different concentration of MMT.
Degree of swelling in water
To conduct test for degree of swelling in water, films of (20 × 20) mm2 were cut and weighed (m0). After drying at 40°C, the specimens were kept in cold desiccators, then directly immersed in distilled water and kept there for 24 h. Then, the specimens were taken out of the container, wiped with absorbing paper, dried in the oven, and final weights were taken (mf). Five specimens for each type were tested and average values were noted. The degree of swelling in water (W) was calculated using the equation (3)
22
Biodegradation
To carry out biodegradation test, (40 × 40) mm2 films with an average thickness of 0.05 mm and an initial mass (m0) were buried in a mixture consisting of 70% of soil, 10% of sawdust and 20% of sand; then, the residual masse (mr) was measured after 2 months. To measure the biodegradation, the variations in the residual mass percentage (Rw) were measured in terms of weight variation, using the following equation (4)
22
Results and Discussion
Structural analysis
Fourier transform infrared spectroscopy was conducted for raw materials, TPS/HIPS mixture and its 3% clay composite. Figure 1 and Table 1 show FTIR spectra of raw and plasticized starches. After starch treatment with glycerol, the FTIR spectrum shows a remarkable intensification of the hydroxyl group characteristic band due to the presence of the plasticizer. The hydroxyls of glycerol moieties could be linked to each other and/or to those present in starch by hydrogen bonds. The spectrum of TPS reveals also adsorbed water hydroxyls as detected by the valence vibration of the O-H bond of the H-O-H group observed around 1640 cm−1.23,24 FTIR spectra of raw starch and TPS. Note: TPS: thermoplastic starch; FTIR: Fourier transform infrared. FTIR results of starch and TPS compared with the data in literature. Note: FTIR: Fourier transform infrared; TPS: thermoplastic starch.

Figure 2 depicts the IR spectra of HIPS, TPS and their mixture. Table 2 compares the obtained FTIR attributions of HIPS spectra with the data in the literature. The spectrum of (50/50) (TPS/HIPS) mixture assembles the components spectra and shows a broad hydroxyl band which is exclusively induced by the higher hydroxyl groups concentration after starch plasticization by glycerol. Also, the strong bands around 753 and 690 cm−1 are due to the deformations of C-H of the monosubstituted aromatic cycle.
25
FTIR spectra of HIPS, TPS and (50/50) TPS/HIPS mixture. Note: HIPS: high impact polystyrene; TPS: thermoplastic starch; FTIR: Fourier transform infrared. FTIR attributions of HIPS spectra compared with the data in the literature. Note: FTIR: Fourier transform infrared; HIPS: high impact polystyrene.

FTIR spectra of MMT, (50/50) TPS/HIPS mixture and its 3% MMT composite are displayed in Figure 3. For MMT, the characteristic elongation band of OH is found around 3400 and 3100 cm−1, whereas the asymmetric and symmetric bands of CH3 and CH2 group of the methyl dihydroxyethyl tallow ammonium modifier are detected at 2920 and 2850 cm−1. The absorption band at 1620 cm−1 corresponds to the elongation of the O-H bond in the H-O-H group due to water adsorbed on the surface of the hydrophilic clay. At 1470 cm−1 is observed the asymmetric deformation in plane characteristic of the C-H in CH3 group.26,27 The FTIR spectrum of the TPS/HIPS/MMT composite consists essentially of the vibration bands of all the constituents of the material. FTIR spectra of MMT, (50/50) TPS/HIPS mixture and its composite with 3% MMT. Note: HIPS: high impact polystyrene; TPS: thermoplastic starch; FTIR: Fourier transform infrared; MMT: montmorillonite.
Microstructure analysis
Figure 4 shows X-ray diffractograms of the raw and plasticized starch. The diffraction diagram of the used raw starch shows that it presents an A-type crystalline structure, which is usually encountered for cereal starches like corn, with characteristic peaks for 2θ angles of 15.33°; 17.43°; 18.13°; 20.12° and 23.22°.
28
X-ray diffractograms of raw starch and thermoplastic starch.
Furthermore, the diffractogram for plasticized starch shows that significant changes have been caused to the starch crystalline structure after the plasticization process. It can be noted that due to the diffusion of glycerol and to heating, the starch granules burst to release amylose, which increases the amorphous fraction and justifies the reduced crystallinity of TPS. Also, new diffraction peaks appeared resulting from the retrogradation process which induces the reconstitution of a new crystalline form and opacity of TPS after cooling. In this context, the diffraction peaks observed at 2θ of 14.23° and 22.52° are assigned to VA-type crystalline structure attributed to the crystallization of a single amylose helix. Also, the diffraction peak noted around 2θ of 19.83° is due to a VH- crystalline structure caused by an induced crystallization of the amylose helix during the transformation of the starch in presence of a sufficient amount of plasticizer. 28 Finally, the peak noted around 2θ of 17.03° could be assigned to the residual A-type structure of the starch and confirms that the plasticization was not complete and that a fraction of raw starch granules have not been disrupted.
HIPS X-ray diffractogram, given in Figure 5, shows two diffraction peaks at 2θ values of 14.88° and 19.53°. The non-crystalline part seems quite large due to the amorphous structure of the butadiene phase. Furthermore, the diffractogram of the (50/50) TPS/HIPS mixture exhibits essentially the diffraction peaks owing to the TPS phase, thus pointing out the fact that the mixing process has not affected the crystalline structure of TPS due to the immiscibility of the blend, even though the presence of the compatibilizer. XRD patterns of TPS, HIPS and (50/50) TPS/HIPS mixture. Note: XRD: X-Ray diffraction; HIPS: high impact polystyrene; TPS: thermoplastic starch.
The X-ray diffractogram of the organophilic montmorillonite is shown in Figure 6. It exhibits the diffraction peaks characterized by the following 2θ values: 4.74°; 19.71° and 35.01°. The diffraction peak located at 4.74° is attributed to the reticular plane (001) characteristic of the basal spacing of the clay organically modified by the surfactant methyl dihydroxyethyl tallow ammonium. The XRD diagram of the TPS/HIPS mixture at 3% of MMT also reveals the diffraction peak of MMT at the same position as for organo-treated clay. This testifies to the dispersion of the nanocharge in the form of aggregates and supports the microcomposite structure of the material. XRD patterns of MMT, (50/50) TPS/HIPS blend and its 3% MMT composite. Note: XRD: X-Rray diffraction; HIPS: high impact polystyrene; TPS: thermoplastic starch; MMT: montmorillonite.
Composites rheology
Variation of melt flow index, degree of swelling and residual mass percentage for TPS/HIPS/MMT composites as a function of MMT rate.

Note: HIPS: high impact polystyrene; MMT: montmorillonite; TPS: thermoplastic starch.
Mechanical properties
Mechanical properties of HIPS and TPS/HIPS/MMT composites.

Note: HIPS: high impact polystyrene; MMT: montmorillonite; TPS: thermoplastic starch.
As expected, incorporation of TPS into HIPS resulted in substantial decrease in mechanical properties of the blend. The plastic region of (50/50) TPS/HIPS is very short, and the sample tends to fail very quickly. Also, the mixture has recorded 8.2 MPa of tensile strength, 15% less than pure HIPS. It shows that the components are immiscible and there is formation of two differentiated phases with poor interfacial adhesion.
After the incorporation of MMT, there is a marginal increase observed on the tensile strength of the blend and it tends to increase with the increase in MMT percentage. This indicates that the surface area of the nano-reinforcement has contributed to the stress transfer along the matrix by the filler at the interfaces. 34 Similar trends were observed for TM and IS of the composites. However, there is a slight reduction in strain at break observed for composite loaded with 3% MMT. This can be due to agglomeration of the clay particles at the interface, which affects chains extension ability. 35
Figure 2S (supplementary file) illustrates the variations in the tensile properties and Izod impact resistance of (50/50) TPS/HIPS mixture after the addition of increasing concentrations of MMT. It can be observed that the composites tensile strength and elastic modulus increase slightly relatively to the matrix which supports the stiffening of the mixtures after adding the clay. However, this increase in rigidity is not significant because the dispersion of the clay in the TPS/HIPS matrix resulted in a microcomposite structure. Consequently, the evolution of the strain at break as a function of the MMT rate is slight. It appears that incompatibility prevails on the compatibilizer effect. So, despite eventual interfacial interactions due to the reaction of TPS hydroxyls with SEBS-g-MA anhydride groups and the possibility of HIPS chains anchoring with styrene phase in the compatibilizer, these seem to be insufficient to promote the elongation at break. Furthermore, the Izod impact resistance of the material tends to increase with the percentage of incorporated MMT. This also can be related to the effective distribution of load by MMT into the blend matrix. These results seem also to be related to a somewhat competing effect between the stiffening effect induced by the rigid clay particles and the plasticizing effect engendered by the long chains tallow brought by the surfactant inserted into the basal spacing of the clay. Similar behaviour is observed by different nanomaterials in polymer and fibre reinforced polymer matrices.36–39
Morphology observation
To characterize the surface topography of the materials, atomic force microscopy images were taken and analysed. Figure 7 shows the AFM image of (50/50) TPS/HIPS matrix and (50/50/3) TPS/HIPS/MMT composite. It is clear from the images that the incorporation of 3% of MMT in TPS/HIPS system has significantly enhanced the roughness of the mixture from 40.46 nm to 42 nm. Moreover, in Figure 7(b), the agglomeration of the clay particles is visible which contributed to the increased surface roughness and concomitantly the stiffness of the material. The observations in the AFM micrographs are supportive to the mechanical performance of the composites. AFM micrographs of (a) (50/50) TPS/HIPS blend and (b) its 3% MMT composite. Note: AFM: atomic force microscope; HIPS: high impact polystyrene; TPS: thermoplastic starch.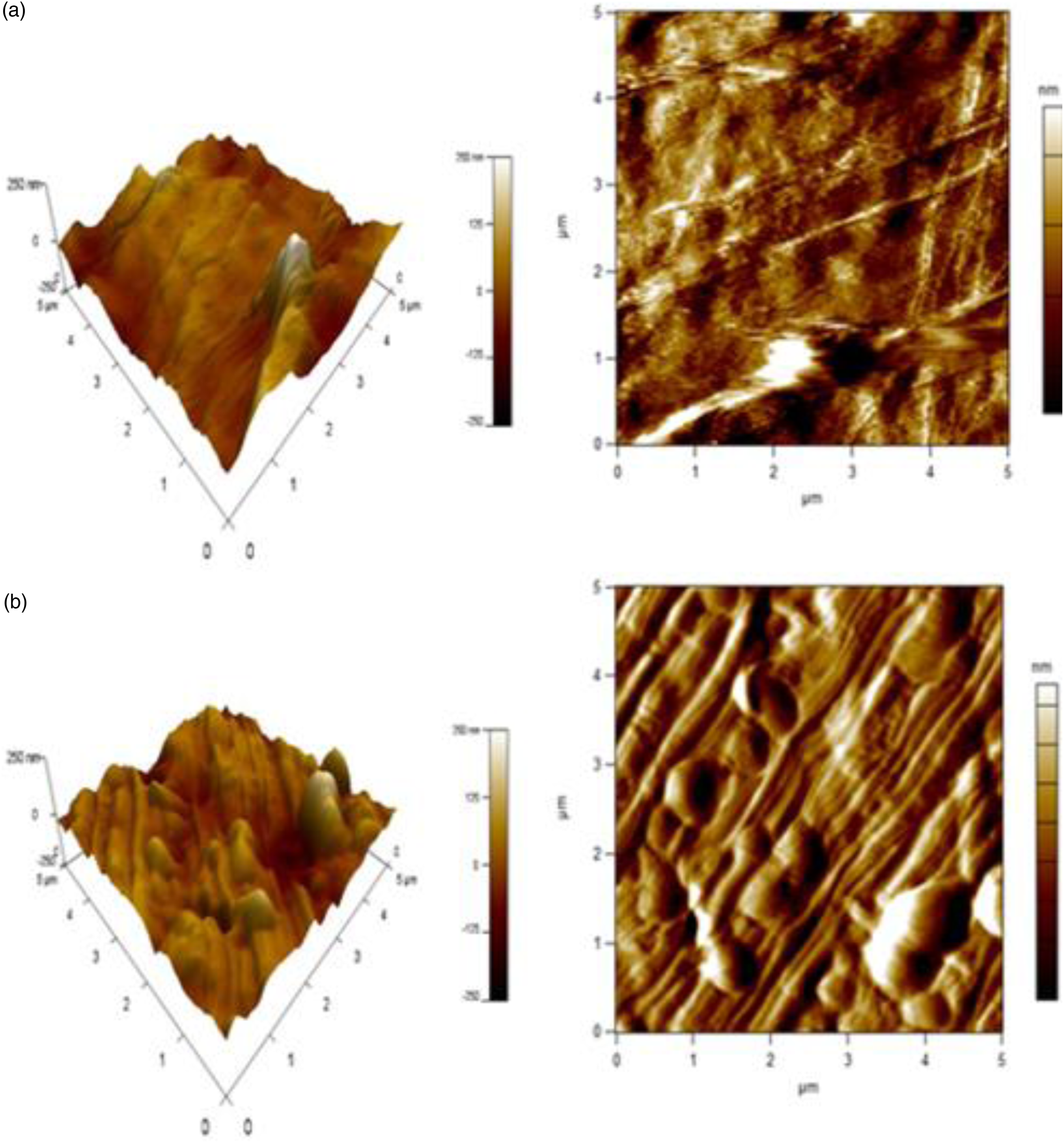
Degree of swelling in water
The degree of swelling in water is calculated using equation (3) for all composites and results are reported in Table 3. It can be observed that water content decreases with the concentration of MMT into the composites. As previously described, FTIR studies has shown that there is a reduction in the hydroxyl groups rate after the incorporation of MMT. So, the reduction in degree of swelling is expected to be due to the reduction in concentration of hydroxyl groups resulting from the interactions of TPS/HIPS system with MMT via hydrogen bonding at the interface TPS/MMT and their reaction with anhydride groups of SEBS-g-MA. Thus, the decrease in hydroxyl groups, susceptible of attracting water molecules, contributes in depressing the composites aptitude to absorb water, which is obviously an additional advantage since water uptake is considered as the major bottleneck towards the application of TPS blends and composites in various applications. Similar trend was noticed by Tang et al. 40 for nanosilica-reinforced starch/poly (vinyl alcohol) (PVA) composites and palm date flour–reinforced PP/TPS blends.
Biodegradation
Biodegradation of the blends was evaluated using the equation (4). The variations in the mass loss for TPS/HIPS/MMT composites as a function of the MMT concentration after a period of 3 months of burial in the soil is tabulated in Table 3. MMT has a well-known catalytic effect with regard to biodegradation, due to its mineral origin which promotes the bio-assimilation of composites by bacteria present in the soil. 41 However, the results show that up to 2% of MMT, mass loss of TPS/HIPS mixture decreases in the presence of MMT, undoubtedly due to the fact that water uptake prevails over biodegradation effect. However, for 3% MMT, the catalytic activity is achieved, and the biodegradability is enhanced. This could be explained by the higher concentration of the nanoclay which could have accelerated the biodegradation process; at this content, the fragmentation of the sample is promoted, thus increasing its contact area with the degrading medium containing microorganisms and enhancing its bio-assimilation aptitude.41–43 Even though the addition of HIPS affects TPS biodegradability, but on the other side, the application of TPS alone presents many disadvantages, particularly its pronounced hydrophilic character. So, even though blending decreases the mixture biodegradability, it allows surmounting TPS shortcomings and inducing many improvements on the mixture properties, thus enlarging its application possibilities.
Conclusion
In this work, biodegradable composites based on thermoplastic starch and high impact polystyrene were successfully prepared by incorporating different percentages of montmorillonite clay. XRD results confirm the formation of microcomposite structure with the incorporation of 3% MMT into the TPS/HIPS mixture. The addition of MMT improves the mechanical properties of the mixtures such as the tensile strength and the Young’s modulus. However, the resilience of the system is significantly decreased. Moreover, the degree of swelling got reduced with increasing the percentage of MMT, whereas biodegradability is enhanced when compared to the TPS/HIPS blend.
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911211046803 – Supplemental Material for Investigation of mechanical properties and biodegradability of compatibilized thermoplastic starch/ high impact polystyrene blends reinforced by organophilic montmorillonite
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911211046803 for Investigation of mechanical properties and biodegradability of compatibilized thermoplastic starch/ high impact polystyrene blends reinforced by organophilic montmorillonite by Nour El Houda Aouadi, Abdelhak Hellati, Nizamudeen Cherupurakal, Melia Guessoum and Abdel-Hamid I Mourad in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.




