Abstract
Polyvinyl chloride/wood flour (WF)/organoclay (OMMT) ternary composites were prepared by melt blending. Chemically treated and untreated pine WF were used as filler, and organically modified montmorillonite or OMMT was added in order to get a potential synergy effect between polyvinyl chloride, untreated wood flour (UWF), or chemically treated WF. The OMMT loading in the composites was 0.5, 1, or 1.5 wt%. The chemically treated and UWF loadings were 1, 5, and 10 wt%. The composite specimens were subjected to mechanical tests (evaluation of tensile strength, elongation at break, and Shore A hardness), thermal stability (Beilstein test), processability characterizations, water resistance absorption, and morphological (optical microscopy) observations. The main results showed that the chemically treated WF were more efficient than the UWF for improving the mechanical and physical properties of polyvinyl chloride. Similar results were reported in the scientific literature. Therefore the chemically treated WF can be a cheap, eco-friendly, and renewable substitute for chalk as a filler which is normally used in the fabrication of Polyvinylchloride based cable insulators.
Introduction
Wood plastic composites (WPC) are a new kind of biomass based material that exhibits the advantages of both polymer resins and wood flour (WF). These composites have become very interesting because of their outstanding properties, such as low density, low cost, acceptable stiffness, low abrasiveness, and good processability. Biomass based polymers or biological polymers are very important and useful in the synthesis of composites for various applications in the medical fields 3 such as anticorrosive coatings.4-6 Indeed, biomass and biological polymers are based on biological components which are easy to extract from natural resource by very low cost green methods. 6 Relative to other fillers, such as inorganic fibers, organic wood fibers, or WF are receiving increasing interest as reinforcement fillers due to the satisfactory behavior of polymer/WF composites.7-8 Nevertheless, the use of WPC has been limited due to their lower ductility, lower impact resistance, and higher density relatively to the corresponding unfilled polymers. 8 Polyvinylchloride (PVC), the second-largest commodity plastic material in the world, has not been used for the production of WF/polymer composites relative to other polymers such as polyethylene and polypropylene.8-10 The main objective of this study was to test the biowaste WF as a cheap, eco-friendly, and renewable substitute of the chalk (CaCO3) used in the fabrication of PVC based cable insulators. The industrial formulation used by ENICAB (Biskra, Algeria) plant was PVC/Chalk (80/20); thus, in this study, the composite PVC/Chalk (80/20) was chosen as a reference or control sample. Furthermore, an organoclay (OMMT) was incorporated as compatibilizer in order to improve even more the performance of WF/PVC composites. The WF/PVC composites were prepared by melt blending followed by compression molding and their performances were investigated by evaluating the maximal strength, strain at break, Shore A hardness, thermal stability, processability, and water absorption. Globally, this report was divided into three parts. The first part describes the influence of untreated wood flour (UWF) on the properties of PVC. The second part concerns the influence of adding sodium hydroxide treated wood fibers (TWF) on the properties of PVC. The third part is related to the effects of adding OMMT on the properties of the PVC/UWF blend.
Experimental
Materials
A commercial grade of PVC (SHINTECH SE 1200 ITOCHU S 70, Japan) was used as the blend major constituent. It was in powder form with a 100 μm mean dry particle size and had a linear configuration. The plasticizer used was DIDP (Di-Iso-Decyl-Phtalate) (45 wt%) and the thermal stabilizer was Ca/Zn (BAEROPAN MC 9917 KA, BAERLOCHER, Germany). The OMMT used was BENTONE 38® (BENTONE SUD, France), it was used as received (Cationic Exchange Capacity = 120 mEq/100g). As mentioned by the manufacturer, the quaternary ammonium salt used for the clay organic treatment was: di-methyl di-hydrogenated tallow salt: 2M2HT (HT = C18H37). The OMMT was in powder form with a 20 μm mean dry particle size. The pinewood (Picea abies) WF was collected from local carpentry workshop in order to valorize wood biowaste or by-product. The used chalk (ARMINIUS GOEBEL GmbH, Germany) for industrial application (cable insulators fabrication) of PVC was a commercial grade.
Preparation of the samples
Before blending, the clay, WF, and PVC were dried in an oven at 80°C for 24 h. The components were first melt blended, at 175°C, in a dry blender (VEM MSH 25, Germany) operating at 3000 r/min. Afterward, the resulting blend was mixed in a two roll mill (BRABENDER POLYMIX 200P, Germany). The twin roll mill had a nip clearance of 0.5 mm and friction ratio equal to 1.3 (20/15 r/min) and the blending was carried out for approximately 15 min. The twin roll mill was operated at 145°C and 25 r/min. Finally, the blend was extruded in a single screw extruder. The extruder used was a conventional one (PLASTICORDER PLE 330, Germany); it has a ratio length/diameter equal to 21, a diameter of 20 mm, a thread thickness of 5.4 mm, and the step between two successive threads equal to 15 mm. The barrel and screw speed were set at 160°C and 30 r/min.
The WF powder was initially washed with 1% soap solution. It was followed by washing with 1% NaOH solution and finally washed with cold water. The washed WF powder was oven dried at 80°C until attainment of constant weight. This dried WF powder was sieved at about 60 mesh size and kept for subsequent use.
Weight composition of the PVC untreated wood fiber (PVC/UWF) formulations.

Weight composition of the PVC chemically treated wood fibers (PVC/TWFs).

Weight composition of the PVC/untreated wood fibers/organoclay formulations.

Alkaline treatment of the WF
The alkaline treatment (chemical processing), also called mercerization, of the pinewood flour was done in order to improve the interactions between the WF and the PVC polymeric chains.
The UWF were immersed in 2 wt% sodium hydroxide solution for 45 min with moderate magnetic stirring at ambient temperature. Afterward, the TWF were washed with distilled water containing a small amount of acetic acid in order to neutralize the excess sodium hydroxide. Finally, the TWF were washed with distilled water until the pH was equal to 7.11-12 Then, the fibers were placed in an oven at 80°C for 24 h (Figure 1). Next, the TWF were subjected to Fourier Transform Infrared (FTIR) analysis in order to ensure that the chemical processing had modified the chemical structure of the fibers (Figure 1). This treatment mainly modifies the distribution of hydrogen bonds in the network structure of the fiber. The schematic reaction of NaOH with the WF is as follows
11
Main steps used for wood flour alkaline processing: (a) decantation after NaOH treatment; (b) filtering; (c) spread out before oven drying; (d) pellets of wood flour/KBr for Fourier Transform Infrared analysis.

Characterizations
Tensile tests were done at ambient temperature (25 ± 2°C) according to ASTM D-638 standard with a ZWICK ROELL Z100 (Zwick Roell, Germany) testing machine. The dumb-bell shaped specimens were extended at 100 mm/min cross head speed. The reported values of the tensile strength and strain at break were averages of five runs. The standard deviations were 2% and 5% for the tensile strength and the strain at break, respectively. The dumb-bell shaped samples were cut from 2 mm thick sheets that were obtained by compression molding 35 g of each sample at 165°C in a SCHWABENTHAN POLYSTAT 300S (Schwabenthan, Germany) press according to the following sequence: preheating for 4 min, 100 Bars pressure exerted for 2 min, 200 Bars exerted for 2 min, 300 Bars exerted for 4 min, and finally, minutes by running cold water through the plates while under pressure.
Shore A hardness tests were carried out by Zwick Roell HPE (Zwick Roell, Germany) apparatus according to the ISO 868 standard. Reported results are averages of six measurements on each side of a 6 mm thick plate obtained by compression molding. The standard deviations for the Shore A hardness tests were 3%.
The thermal stability of the blends and neat PVC were investigated by a dehydrochlorination apparatus at 200°C according to the well-known Beilstein test. The dehydrochlorination apparatus is mainly consisting of an electric resistance in which test tubes, containing samples, were placed vertically and then heated. The thermal stability was evaluated by measuring the exact time for the occurrence of the sample degradation caused by the dehydrochlorination monitored by the released HCl which results in the red coloration of the pH paper which was placed with the sample at the top of the tube test. The obtained results were averaged for three successive measurements.
The water uptake measurements were done according to the ASTM D 2765-95 standard. The samples, having 1 mm thickness obtained by compression molding, were immersed in distilled water for 5 days at ambient temperature. The results were averaged for three measurements. The water uptake degree was calculated as follows: water uptake degree = (W2 − W1)/W1 × 100%, where W2 is the weight of the wet sample and W1 is the weight of the dried sample.
The rheological analyses were done with a Brabender (Brabender, Germany) R100/4308 model Rheometer, in order to obtain the plots of the applied torque versus time. For each formulation, samples of 4.5 g weight were analyzed at 160°C. PVC/Chalk (80/20), PVC/UWF (95/5), and PVC/TWF (95/5) formulations were tested.
The FTIR analysis was done with a SHIMADZU FTIR 8400S (Shimadzu, Japan) spectrophotometer. The WF were ground with KBr in equal weights and then pressed at 567 kBar in order to get pellets. The FTIR analysis was done from 4000 to 400 cm−1, using 20 scans.
Optical microscopy (OM) was done with a BRESSER (Bresser, Germany) microscope in a transmission light mode. The observed samples were either WF particles or PVC/WF films having a 1 mm thickness obtained by compression molding.
Results and discussion
Mechanical properties
Mechanical properties values of neat PVC, PVC/UWF, PVC/TWF, and PVC/UWF/OMMT.

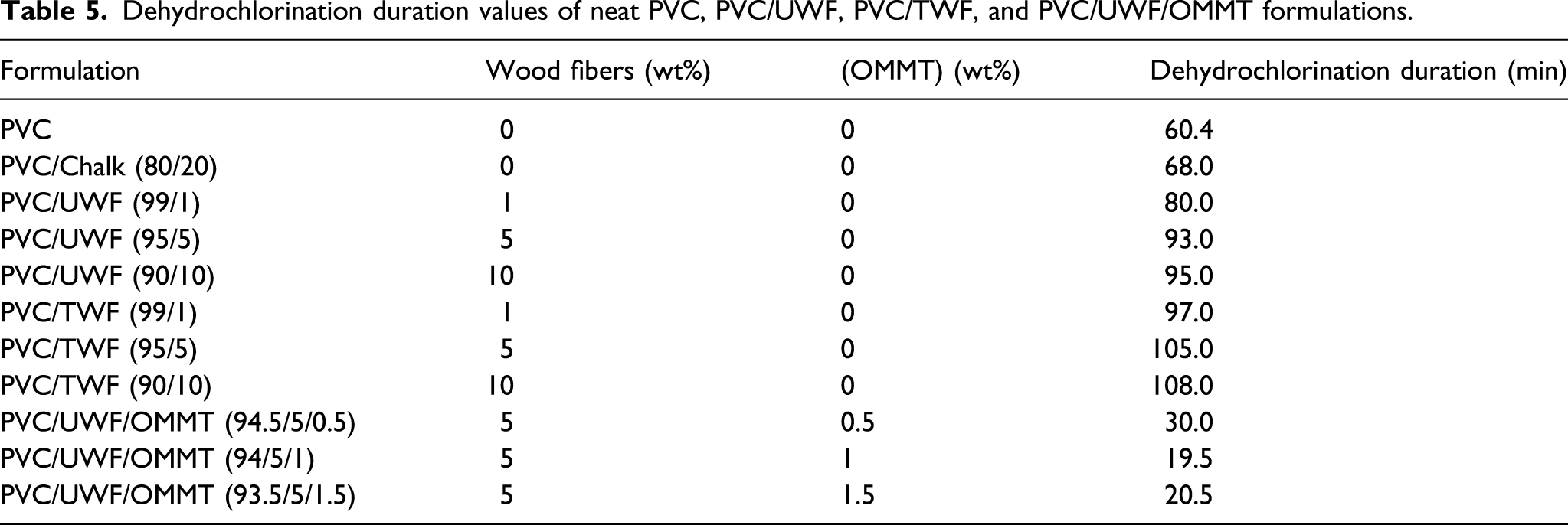
The simultaneous increase of the tensile strength values and the Shore A hardness of the PVC/TWF can be explained by the favorable interactions between the WF and the PVC macromolecular chains due to the chemical processing sustained by the wood fibers. 10 Indeed, the hydroxide processing is one of the most used treatment when natural fibers are used to reinforce thermoplastic and thermoset polymers.12-13 The major change provided by alkali treatment is a decrease of the hydrogen bonds between cellulose and the two other wood fiber components, namely, the hemicellulose and the lignin.12-13 The alkaline processing modifies the topography of the surface wood fibers, deleting wax, pectin, and one part of lignin and hemicellulose, which is favorable to the subsequent use of the TWF as a reinforcement for polymer materials.13-18
Figure 2 shows the FTIR spectra of UWF and TWF. In this figure, it can be noticed that the 1730, 1500 cm−1, and 1465 cm−1 peaks decreased after the chemical processing of the wood fibers. The 1730 cm−1 peak is the carbonyl characteristic peak and it is the main characteristic peak of the hemicellulose. This peak is related to the carbonyl stretching vibration in hemicellulose.19-23 The decrease of 1730 cm−1 peak is due to the fact that a great amount of uranic acid, an important constituent of hemicellulose, was removed from the wood fiber.
22
The 1500 cm−1 peak is attributed to phenyl ring vibration.
23
The phenyl rings being the major structural constituent of lignin, so, the decrease of 1500 cm−1 peak is due to the fact that a great amount of lignin was removed from the fiber after alkaline processing. The 1465 cm−1 peak is attributed to OH group in plane bending.
23
The OH group being a major structural constituent of hemicellulose, so the decrease of 1465 cm−1 peak is also due to the fact that a great amount of hemicellulose was removed from the wood fibers. The dissolution of wax from the wood fibers surface, due to the alkaline treatment, may also cause increased physical interlocking between the wood fibers and polymeric matrix chains resulting in a stronger material.24-27 Fourier Transform Infrared absorption spectra of (a) untreated and (b) alkaline treated pinewood wood flour.
Beilstein test
Dehydrochlorination duration values of neat PVC, PVC/UWF, PVC/TWF, and PVC/UWF/OMMT formulations.
Rheological study
It has been reported that Brabender torque–time curves can be used to analyze the processing characteristics of polymer melt mixing.
30
The steady state or equilibrium torque reached at the end of melt mixing in a Brabender Rheometer is used to measure the processability of the polymer mixture which is related directly to the melt viscosity of the blends or composite.30-31 Furthermore, the maximal torque (MH) can be considered as a measure of the blend stiffness or Young’s modulus in the solid state,12, and the minimal torque (ML) is related to the melt viscosity of the blend. 30,31 Figure 3 shows the plastograms of PVC/Chalk, the PVC/TWF, and PVC/UWF blends and their related results are summarized in Table 6. It can be deduced from Figure 3 and Table 6 that the incorporation of the TWF increased the initial, maximal and steady state torque relative to PVC/Chalk blend. This observation means that, after the adding of TWF to neat PVC, the composite solid state stiffness and the melt viscosity of the resulting composite increased. The increase of the solid state stiffness is in good agreement with the increase of the tensile strength and Shore A hardness of PVC/TWF (95/5) composite described above. However, despite the increase of the melt viscosity, the processability of the resulting material was not too affected or impacted because the equilibrium or steady state torque of the PVC/Chalk and the PVC/TWF blends were close. The increase of melt viscosity of the PVC/TWF can be explained by the decrease of lignin and hemicelluloses concentrations after chemical processing due to the increase of hydrogen bonds between the hydroxyl groups on the surfaces of the wood fibers and the hydrogen atoms of the PVC chains because the lignin acted as a shield between these hydroxyl groups and the PVC chains in the PVC/UWF composites and prevented the occurrence of hydrogen bonds between them.
13
Moreover, it can be noticed that the PVC/UWF exhibited low initial, maximal and steady state torques relative to the PVC/Chalk. This observation means that, after the adding of UWF to the neat PVC, the solid state stiffness and melt viscosity of the resulting composites decreased. The decrease of the solid state stiffness is in good agreement with the decrease of the tensile strength and Shore A hardness of the PVC/UWF (95/5) blend previously observed. This observation can be explained by the fact that there were few interactions between the PVC chains and the UWF due to the lack of hydrogen bonds between the hydroxyl groups of the wood fibers and the hydrogen atoms of the PVC chains.13-14 Torque versus time of Polyvinylchloride/Chalk (80/20), Polyvinylchloride/treated wood fibers(95/5), and Polyvinylchloride/untreated wood flour(95/5) composites. Maximum torque (MH), torque minimum (ML), and equilibrium torque (ET) for PVC/Chalk(80/20), PVC/TWF (95/5), and/PVC/UWF (95/5) blends.

Water uptake properties
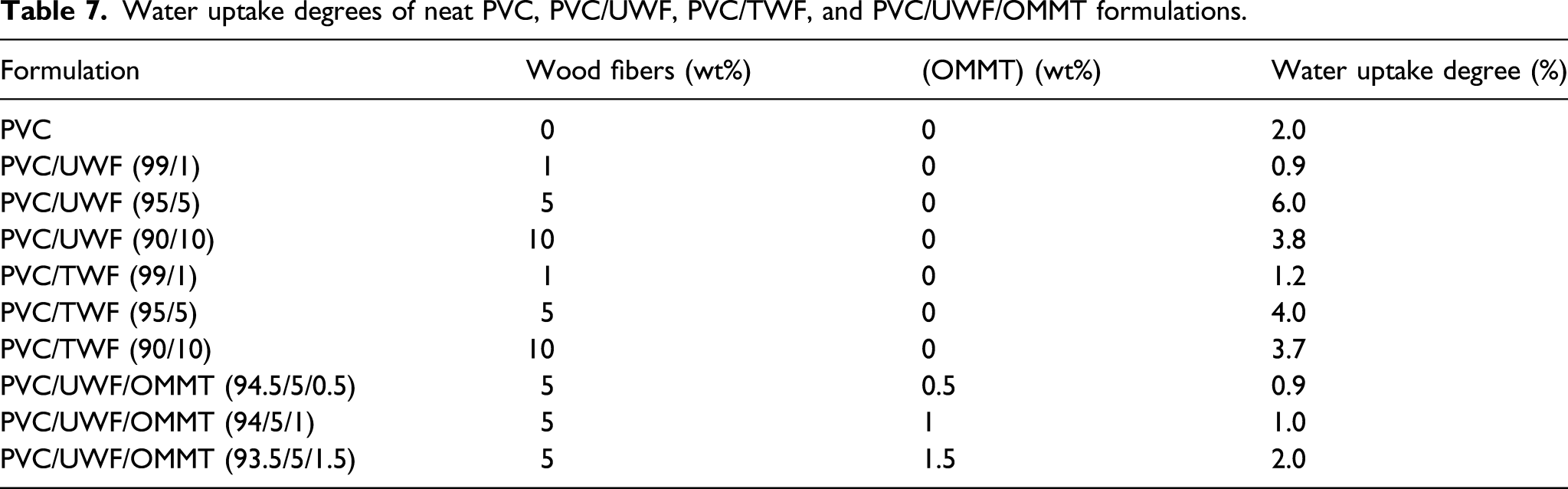
Water uptake degrees of neat PVC, PVC/UWF, PVC/TWF, and PVC/UWF/OMMT formulations.
Morphological characterization (optical examinations)
Figure 4 shows two optical micrographs taken at the same high magnification of WF particle surfaces before and after the alkali treatment. It can be seen from this figure that the surfaces of the UWF seemed to be more regular relative to the surface of TWF which seems to be rough. The roughness aspect of TWF is likely due to the effect of the chemical processing of the wood fibers.
38
Optical micrographs of wood flour particle showing (a) untreated wood fiber surface and (b) alkaline treated wood fiber surface.
Figure 5 shows two optical micrographs taken at the same low magnification showing the surface of the composite before and after the WF alkaline treatment. It can be observed from Figure 5 that after alkaline treatment, there was the appearance of voids on the surface of the composites. These voids were probably induced by the effects of the alkaline processing on the PVC macromolecular matrix. It can also be noticed from Figure 5 that after chemical processing, the interfaces between the TWF and PVC matrix seemed to be more fine and clear. On the other hand, it can be seen from Figure 5 that after the alkaline treatment, the TWF had a tendency to create voluminous agglomerates which constitute a continuous interconnected network acting as an efficient reinforcing framework. The creation of a reinforcing filler network is commonly reported in the fiber reinforcement of matrix polymer,38-41 and this fact can explain the improvement of the interfacial adhesion between the TWF and the PVC matrix induced by the alkaline processing leading thereby to better mechanical properties, especially strain at break since it was reported that the strain at break is directly related to the adhesion or the compatibility between two immiscible phases in a binary macromolecular composite.
40
Optical micrographs images showing (a) the untreated Polyvinylchloride/wood flour (90/10) and (b) the alkaline treated Polyvinylchloride/wood flour (90/10).
Furthermore, it can be seen from Figure 6 that as the OMMT concentration increased, its dispersion state became non uniform and the OMMT particles tended to agglomerate, forming voluminous agglomerates, due to the weak interfacial interactions between them, the WF, and the PVC chains. Optical micrographs showing (a) Polyvinylchloride/untreated wood flour/organoclay (94/5/1) and (b) Polyvinylchloride/untreated wood flour/organoclay (93.5/5/1.5) composites.
Conclusions
The results obtained in this study showed that the mechanical properties were greatly improved when sodium hydroxyl treated WF were included in a PVC matrix. Moreover, it was found that the adding of 0.5–1 wt% of OMMT to the PVC/UWF (95/5) composite improves slightly its tensile strength and elongation at break. The thermal stability measured by Beilstein test showed that the adding of 10 wt% of UWF to PVC improves the thermal stability of the resulting composite. On the other hand, it was found that the PVC/TWF (95/5) composite is the most thermally stable relative to PVC/UWF. The torque analysis showed that the melt viscosities of the studied formulations were close in the steady state and the addition of chemically treated WF to the PVC matrix did not damage the processability of the resulting composite relatively to PVC/Chalk blend. Furthermore, as the torque, expressed in Nm or Joule; represents the energy consumed during the fabrication of the composite (by melt mixing of the components blend), and as the steady state torque values of PVC/chalk and PVC/TWF are very close, so the fabrication of the PVC/TWF composite did not consume an excess of energy relatively to the case of the fabrication of PVC/Chalk blend used in the industry. The water absorption showed that the alkaline processing of the WF increased the resistance of the resulting blend against the water uptake. The optical microscopy examinations showed the appearance of WF agglomerates’ continuous interconnected network which explained the better interfacial interaction between TWF and the PVC matrix. Moreover, the adding of the OMMT to the PVC/UWF decreased the water uptake, this fact showed the barrier effect of the OMMT particles inside the PVC matrix. The alkaline processing of the WF was efficient for improving the properties of the PVC resin and consequently, the chemically processed pinewood WF constitute a good alternative for substituting the chalk as cheap, eco-friendly, non-irritant, and renewable biomass filler in the fabrication of PVC based cable insulators.
Footnotes
Acknowledgments
The authors are indebted to ENICAB Biskra plant, specialized in PVC based cable manufacturing, for skillful technical assistance, helpful discussions, materials supplying, laboratory access, and facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
